By Marcello Cherchi, MD PhD
Overview
The auditory system has both afferent and efferent pathways. In the efferent auditory system, the vibration of incoming sound waves moves the tympanic membrane, and the ossicular chain in the middle ear mechanically focuses this movement into a piston-like action at the oval window, which generates fluid waves that propagate through the cochlea and stimulate type 1 cochlear hair cells in a frequency-specific fashion, triggering action potentials that eventually reach the auditory cortex and are perceived as specific tones. In short, the afferent pathway transduces kinetic energy from the vibration of sound waves into the electrochemical activity that constitutes a neural signal, and functions as a frequency analyzer. In the efferent pathway, signals from the inferior colliculus synapse on type 2 cochlear hair cells which serve to modulate (amplify or dampen) the activity of type 1 cochlear hair cells; thus, the efferent auditory system functions as an equalizer. The efferent auditory pathway also controls the activity of the tensor tympani and the stapedius, mediating a protective reflex against overly loud acoustic stimuli. A range of neurotransmitters are found throughout the auditory system.
Introduction
The auditory system has both afferent and efferent pathways. The afferent auditory pathways detect and interpret acoustic stimuli. The efferent auditory pathways protect the cochleae and serve as an “equalizer” in filtering incoming acoustic stimuli. We briefly review the anatomy and physiology of these systems.
Afferent auditory system
The main task of the afferent auditory system is to transduce the kinetic energy from the vibration of sound waves into the electrochemical activity that constitutes a neural signal. Acoustic stimuli can present with a range of frequencies and amplitudes, and the auditory system can process this by functioning as a frequency analyzer.
The middle ear, the labyrinth, and the interfaces between them
The bony labyrinth is a convoluted space embedded within the temporal bone. A very small amount of fluid can pass very slowly in or out of the labyrinth via various channels (cochlear aqueduct, endolymphatic duct, vestibular aqueduct), but otherwise the total amount of perilymphatic and endolymphatic fluid remains approximately constant. This fluid is effectively incompressible. There round window and the oval window are two “holes,” each covered with a thin membrane; these comprise an interface between the inner ear and the middle ear.
The tympanic membrane and ossicular chain
As sound enters the external auditory canal, the oscillating pressure waves get transduced into the kinetic energy of the eardrum moving inwards and outwards, and this motion is transmitted through the ossicular chain. At the end of the ossicular chain, the stapes footplate is attached to the membrane covering the oval window. As sounds make the eardrum vibrate, the stapes footplate will push the membrane of the oval window inwards and outwards in a piston-like motion. Because endolymphatic and perilymphatic fluid is incompressible, any inward movement of the membrane at the round window will move fluid of a certain volume, and that volume must be exactly offset by outward motion somewhere else, which is usually the round window.
The cochlea
The alternating inward and outward motion of the membrane of the oval window, and the corresponding alternating outward and inward motion of the membrane of the round window, create fluid waves that mechanically stimulate the cochlea. This is depicted schematically in the Figure below, from Schunke and colleagues (Schünke et al. 2007).

The Figures below, from Schunke and colleagues (Schünke et al. 2007), provide a progressive “zooming in” on the architecture from a cross section of the cochlea.
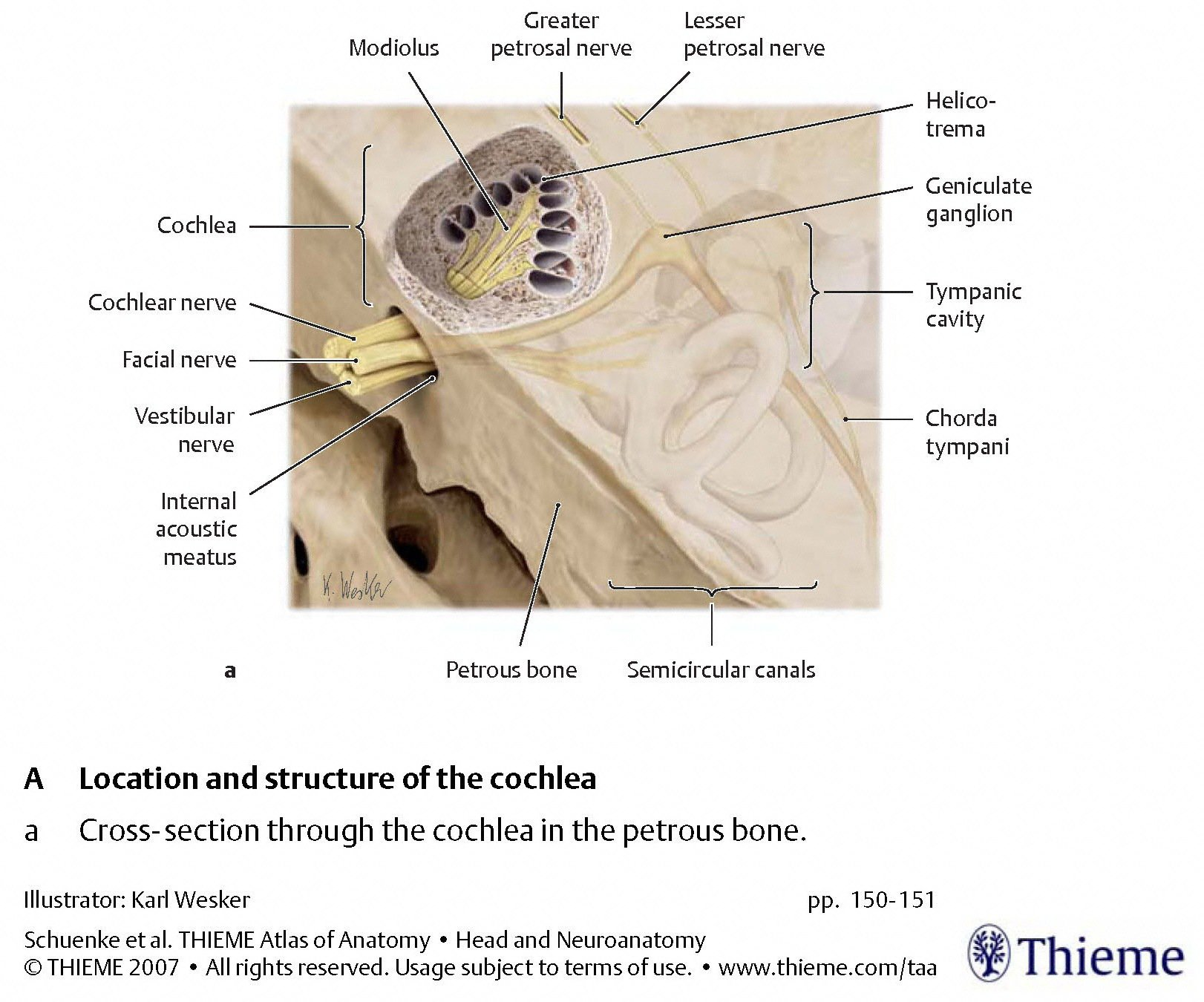 |
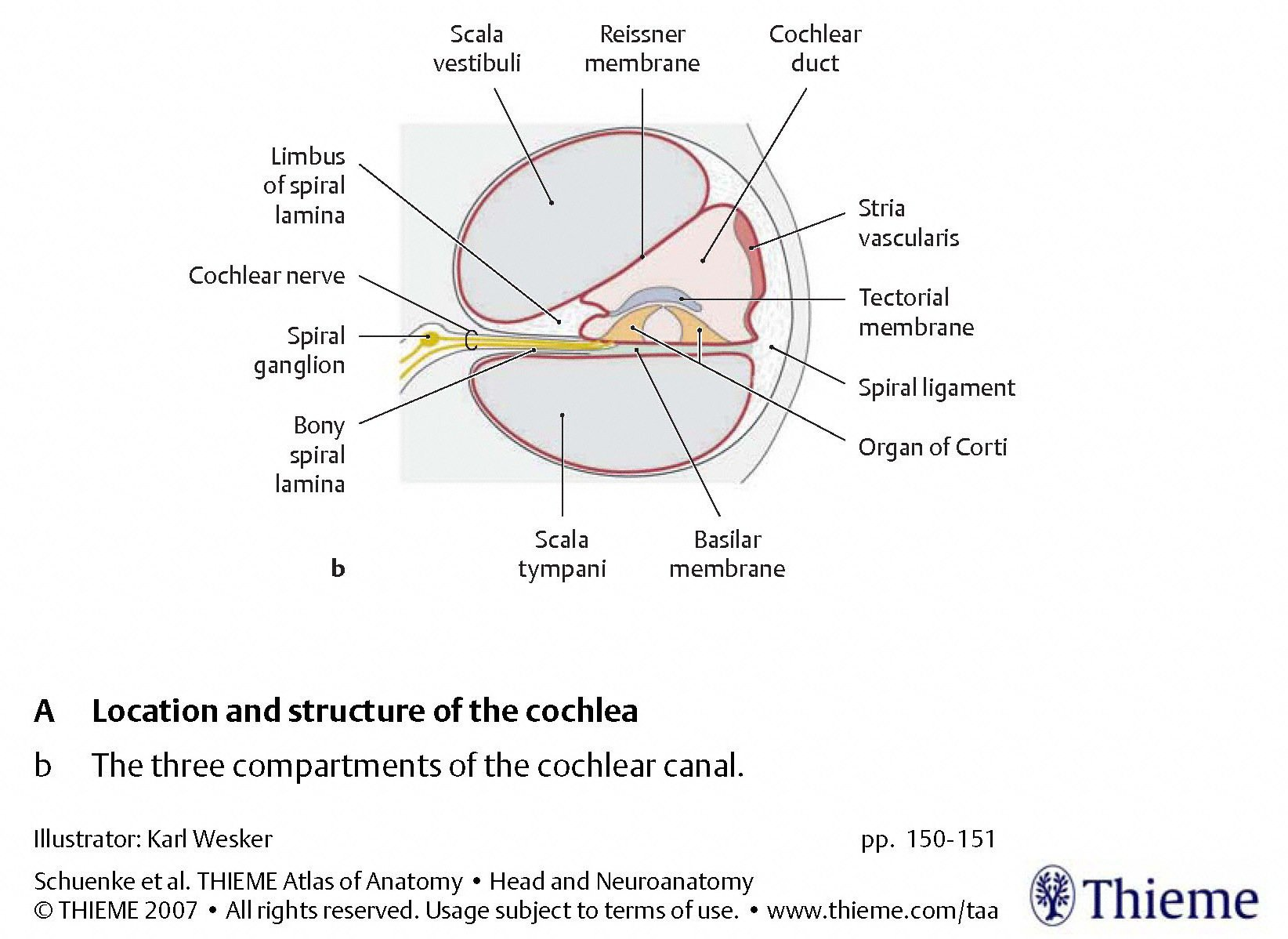 |
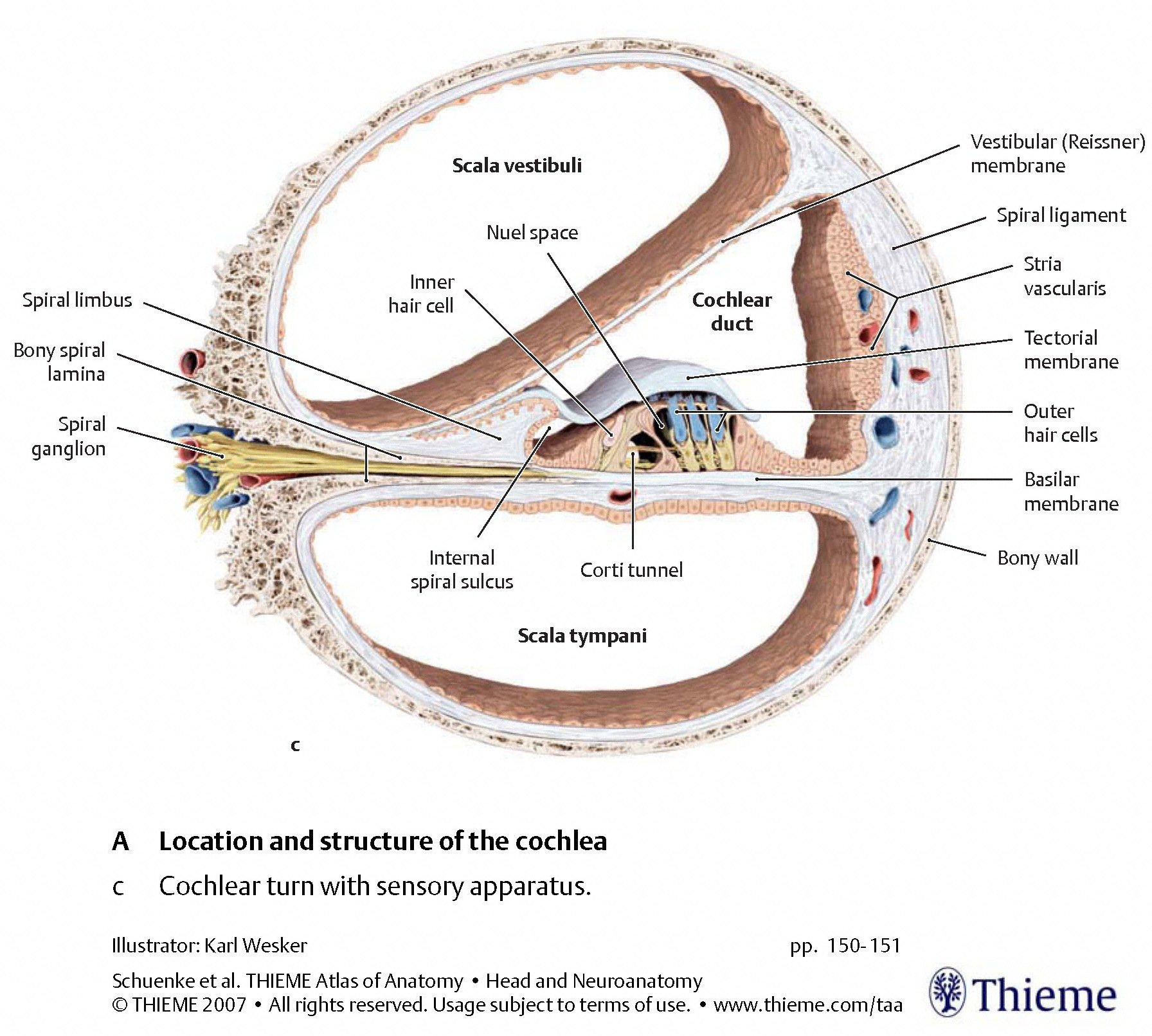 |
The Figure below, from Haines (Haines 2006), shows a detailed view of a cross section of the cochlea.
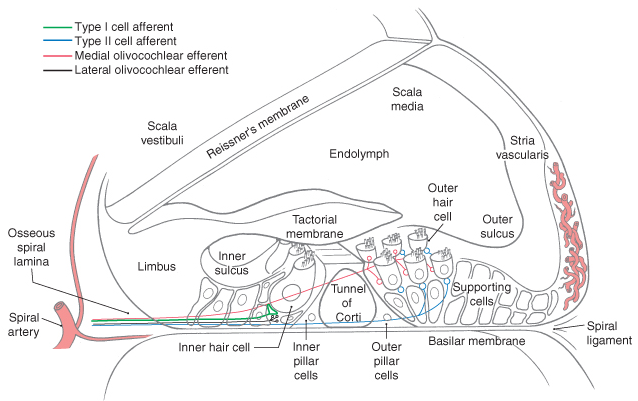
Cochlear hair cells
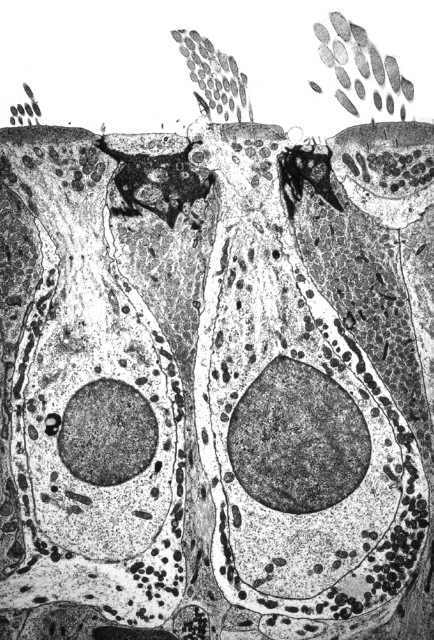
The Figures below show the arrangement of type 1 (inner) and type 2 (outer) cochlear hair cells. The picture on the left is an electron micrograph; the one on the right is a schematic.
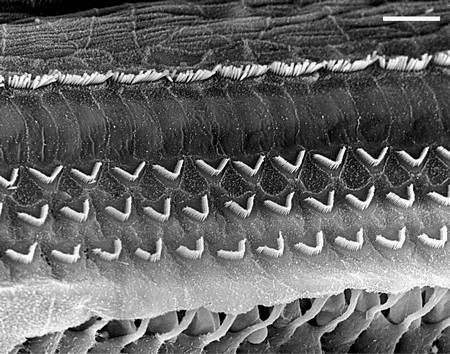 |
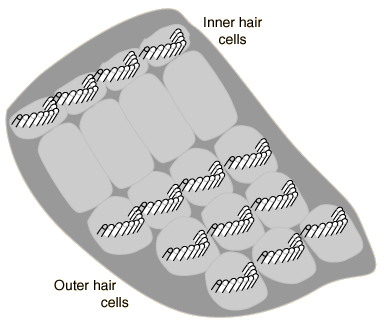 |
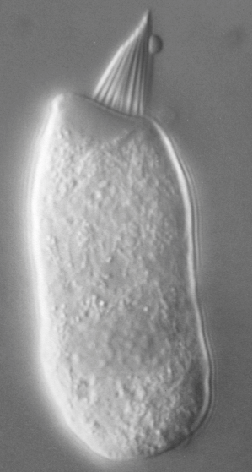
The Figures below show an isolated type 1 (inner) hair cell under light microscopy, and an electron micrograph of several hair cells.
|
Figure : Isolated type 1 cochlear hair cell under light microscopy. From http://umech.mit.edu/hearing/intro/intro.html (accessed 2/25/2016). |
Figure : Isolated type 1 cochlear hair cell under light microscopy. From http://umech.mit.edu/hearing/intro/intro.html (accessed 2/25/2016). |
The Figures below show (1) an electron micrograph of several hair cells embedded among the sustentacular cells of the basilar membrane; (2) a single such cell; (3) a schematic of a single hair cell.
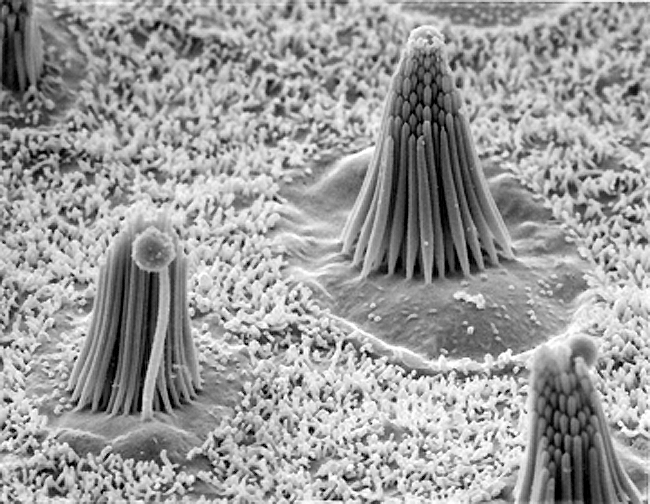 |
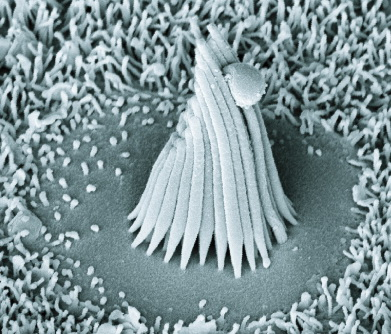 |
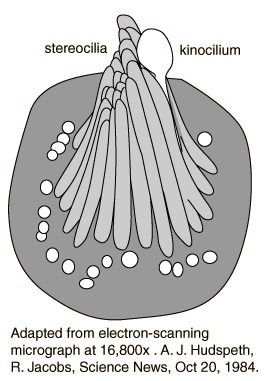 |
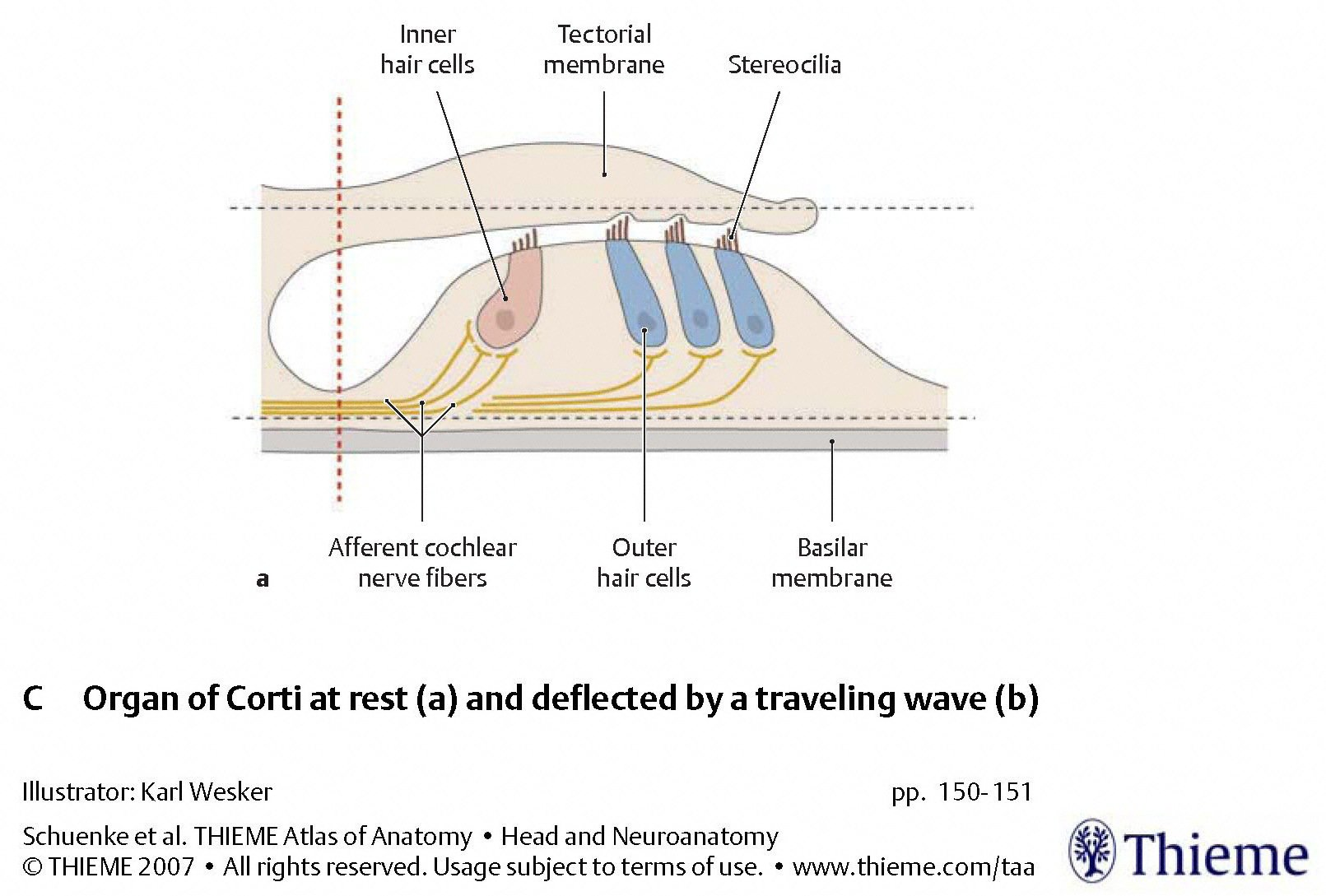
Deflection of the basal and tectorial membranes
The Figures below, from Schunke and colleagues (Schünke et al. 2007), display the basilar and tectorial membrane at rest, and when deflected by a traveling fluid wave. In the deflected phase, the tectorial membrane exerts a shearing force on the stereocilia of the type 1 (inner) cochlear hair cells.
 |
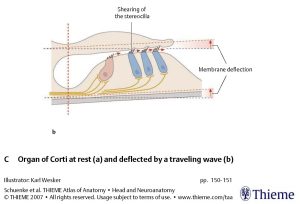 |
During vibration of the basilar membrane, its upward displacement causes the stereocilia of the type 1 (inner) cochlear hair cells to move against the tectorial membrane; as the stereocilia move towards the modiolus, mechanically gated ion channels open and current flows in. Conversely, during downward movement of the basilar membrane, the stereocilia to move away from the modiolus, thereby closing the ion channels.
Ion channel activity in type 1 cochlear hair cells
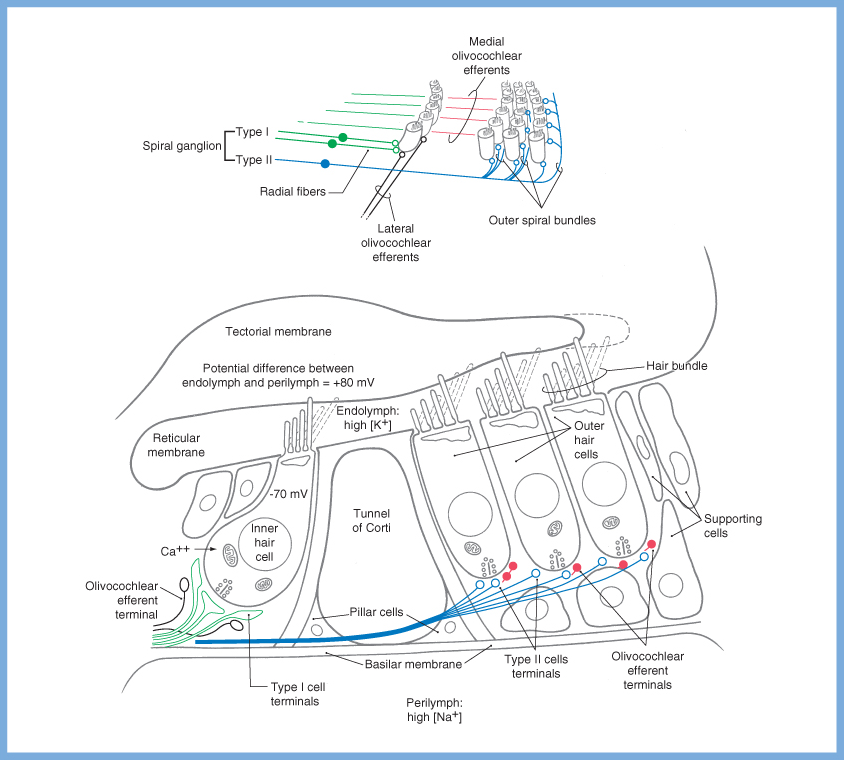
The specific ion channel activity is as follows. When the shearing force deflects the stereocilia towards the kinocilia, mechanically gated ion channels permit influx of K+ at the cell apex from the endolymph, depolarizing the cell membrane. In turn that depolarization triggers voltage gated channels at the base of the cell to permit influx of Ca2+ into the cell, which in turn causes release of synaptic vesicles into the synaptic cleft. When the shearing force reverses direction, the ion channels close and the cell repolarizes. These are depicted in the Figure below, from Haines (Haines 2006)
The cochlea’s tonotopic mapping makes it a frequency analyzer
The spectrum of vibrations in the fluid causes corresponding vibrations in the tonotopically mapped areas of the basilar membrane. Higher frequencies are mediated towards the proximal (stiffer) end of the basilar membrane, while lower frequencies are mediated towards the (floppier) distal end (apex) of the basilar membrane, as shown in the Figures below from Li and colleagues (Li et al. 2021) and Helpard and colleagues (Helpard et al. 2021)
 |
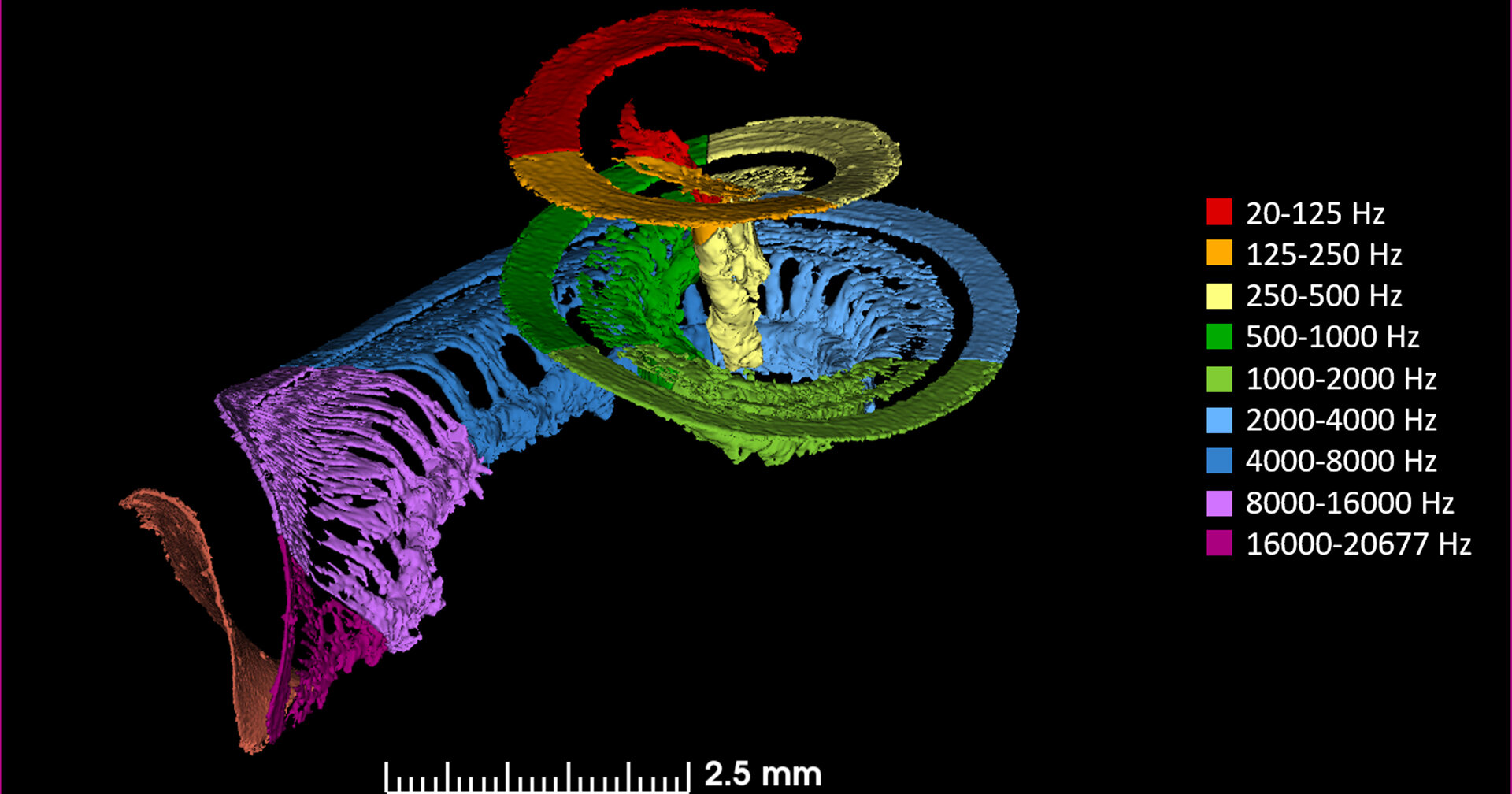 |
Through this process the cochlea transduces the mechanical energy of vibration (from acoustic stimuli) into electrical voltage. By doing this over a range of pitches, it functions (in engineering terms) as a frequency analyzer. The resulting electrical signals correspond to the spectrum of auditory stimulus originally received.
Cochlear microphonic, summation potential and action potential
As mentioned above, the oscillation of the basilar and tectorial membranes causes an alternating ionic activity of type 1 cochlear hair cells whose frequency parallels that of the auditory stimulus. This alternating current is called the cochlear microphonic (CM).
That same activity (of the inner cochlear hair cells) also induces a direct current that reflects the “time-displacement pattern of the cochlear partition in response to the stimulus envelope.” This (unidirectional) direct current induces a shift in the cochlear microphonic’s electrical baseline, manifesting as the summation potential (SP).
The electrical potential derived from the cochlear microphonic triggers release of neurotransmitters (from the end-organ) into the synaptic cleft between the hair cells and the neurons of the spiral ganglion (the first-order neuron). The post-synaptic potential accumulates and, when it exceeds a specific threshold, triggers depolarization of the post-synaptic neuron. The summed responses of the synchronous firing of several thousand auditory nerve fibers results in an alternating current potential that is termed the action potential (AP). The use of the term “action potential” is this audiologic context has a specific meaning here that differs somewhat from what is familiar to neurologists, in that it refers to the aggregate activity of thousands of axons, rather than a single axon.
Afferent auditory pathways ascending from the spiral ganglion
These signals are transmitted from the primary neurons in the spiral ganglion through the auditory component of the cochleovestibular nerve, and then synapse in the dorsal and ventral cochlear nuclei in the brainstem. From there, some fibers decussate immediately via commissural pathways through the trapezoid body, while other fibers ascend. There are multiple other decussations during this ascent through the afferent auditory system, including at the levels of the nuclei of the lateral lemniscus (via the commissure of Probst), the inferior colliculus (via the commissure of the inferior colliculus) and the auditory cortex (via the corpus callosum). Thus, by the time auditory signals have reached primary auditory cortex, there is significant bilaterality of encoding. (For this reason, a lesion of one primary auditory cortex does not cause deafness, though it can cause a variety of other acoustic perceptual abnormalities.)
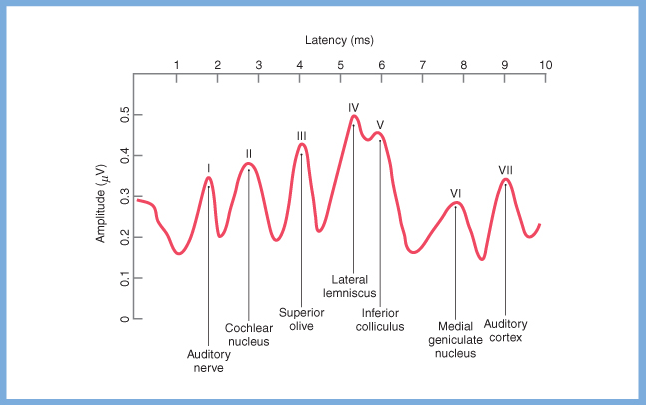
A far field electrical potential can be detected for each point in the auditory pathway where a synapse occurs. The Figures below, from Haines (Haines 2006), depict these synapses, and the corresponding electrical potentials as detected on an idealized brainstem auditory response.
 |
 |
Schematics of the afferent auditory pathways are shows in the Figures below.
 |
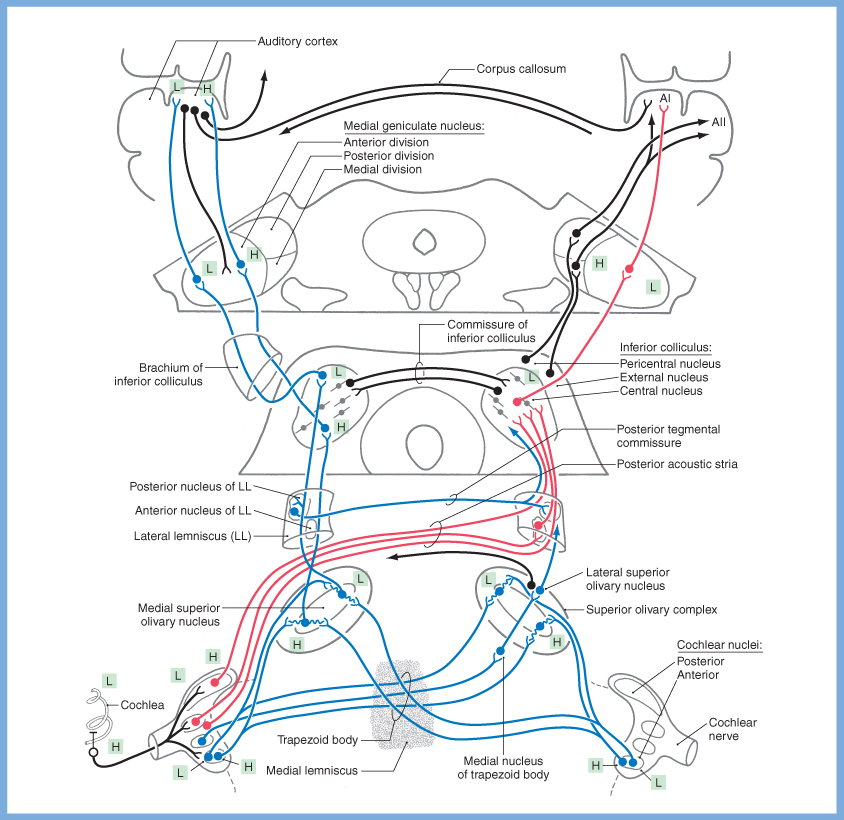 |
The tonotopic arrangement that began in the cochlea is preserved all the way up to the auditory cortex, as shown in the Figure below, from Saenz and Langers (Saenz and Langers 2014).
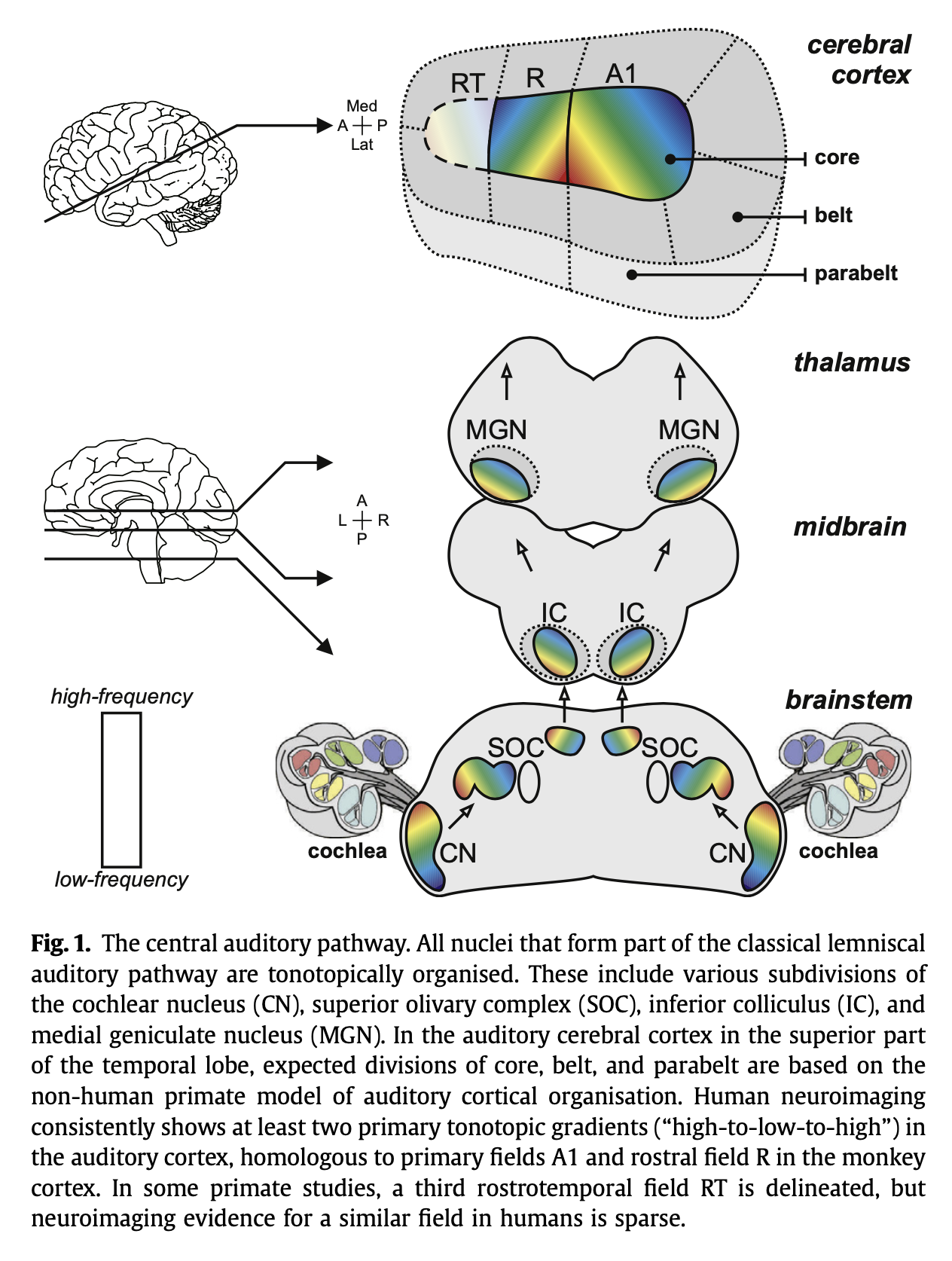
Ascending auditory pathways terminate in the primary auditory cortex
When these signals reach primary auditory cortex we perceive them as sound. From there, neurons in primary auditory cortex send projections via the arcuate fasciculus to the auditory association cortex. From the auditory association cortex there are projections to many other cortical areas that subserve higher-order functions such as language (e.g., Wernicke’s area) and parietal areas that interpret the significance of perceived sounds. The Figure below, from Haines (Haines 2006), depicts several such areas.
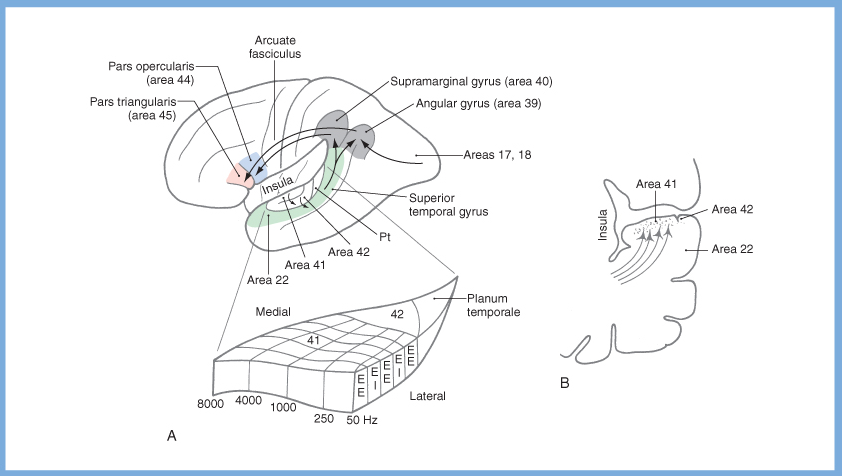
Efferent auditory system
The efferent auditory system serves several functions, including modulating the activity of the ascending auditory system (through the descending auditory pathways via type 2 cochlear hair cells), and mediating protective acoustic reflexes (the stapedius reflex and the tensor tympani reflex).
Descending auditory pathways
Descending pathways from the auditory cortex make reciprocal connections throughout the auditory pathway, forming feedback loops that modulate information arriving from the periphery.
In particular, neurons in the auditory cortex project to the inferior colliculus and synapse there. Neurons in the inferior colliculus in turn project to the lateral and medial periolivary nuclei, and synapse there. Finally, neurons in the periolivary nuclei project to the cochlear nuclei and to the type 2 (outer) cochlear hair cells.
These descending auditory pathways are depicted in the Figure below, from Haines (Haines 2006).
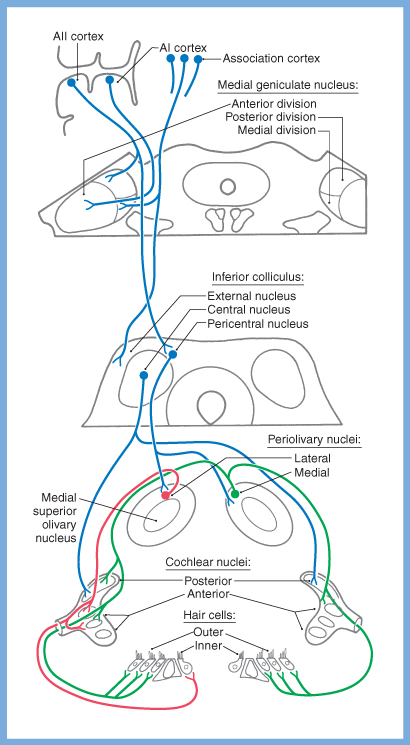
The Figures below are schematics depicting the termination of descending auditory pathways, mostly on type 2 (outer) cochlear hair cells, less on type 1 (inner) cochlear hair cells.
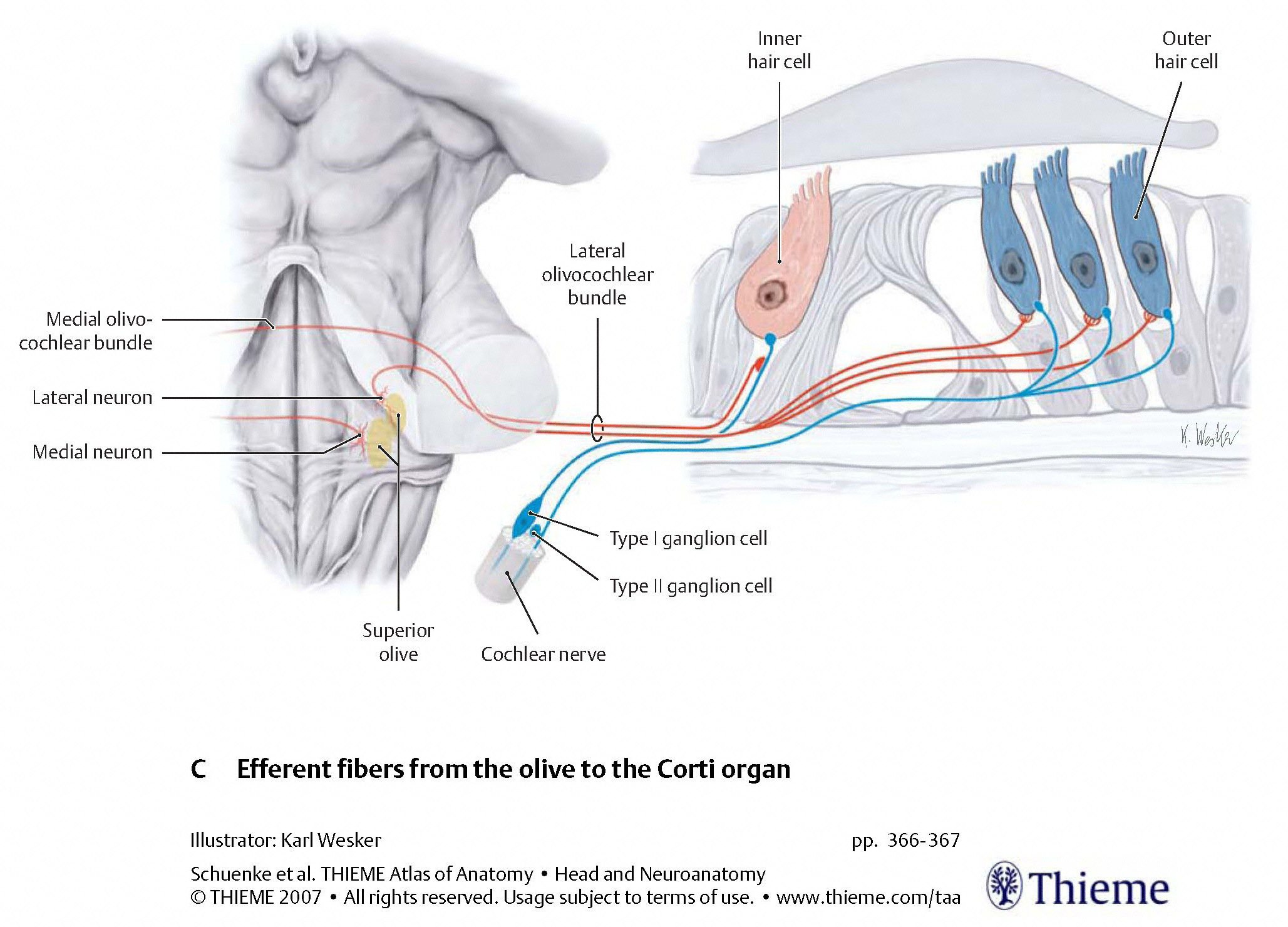 |
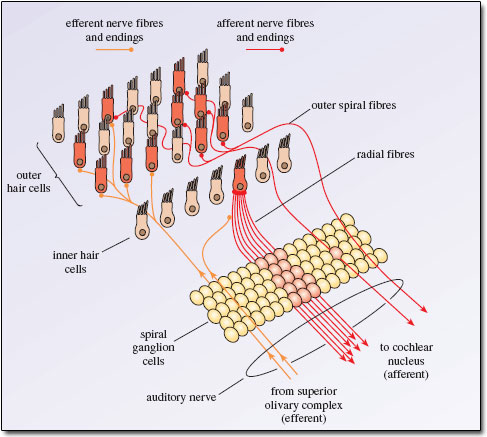 |
The type 2 (outer) cochlear hair cells serve to modulate the activity of type 1 (inner) cochlear hair cells, effectively serving the function of an auditory equalizer.
The activity of type 2 (outer) cochlear hair cells can be assessed by otoacoustic emissions.
Protective reflexes
Protective acoustic reflexes mechanically limit the motion of the ossicular chain in order to protect the cochlea from loud stimuli that might otherwise damage it. The reflexes mechanically limit the ossicular chain at its mechanical “beginning” (dampening the movement of the tympanic membrane via the tensor tympani reflex) and at its mechanical “end” (dampening the movement of the stapes footplate via the stapedial reflex).
Stapedial reflex
Afferent auditory signals from one cochlear nucleus project to the ipsilateral and contralateral superior olivary nuclei. If that afferent signal is too strong, then the stapedial reflex is triggered. The first order neuron (whose cell body resides in the superior olivary nucleus) projects to the facial nucleus. The second order neuron projects through the facial nerve, and the nerve to the stapedius, causing the stapedius muscle to contract, and thereby limit the movement of the stapes footplate on the oval window.
The pathways for the stapedial reflex are depicted in the Figures below, from Schunke and colleagues (Schünke et al. 2007) and Haines (Haines 2006).
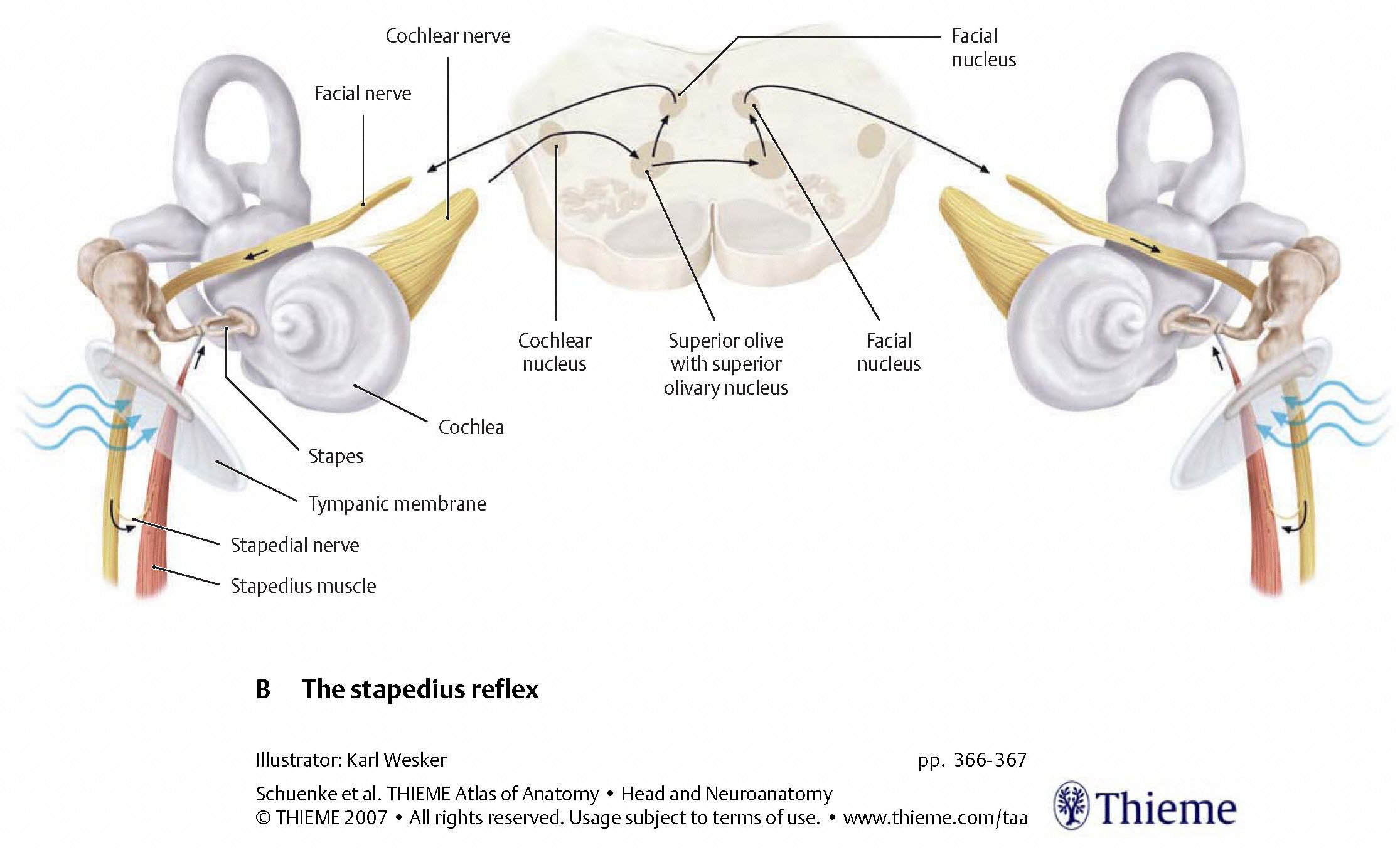 |
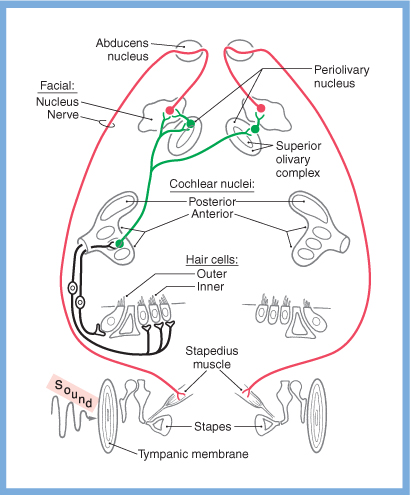 |
Tensor tympani reflex
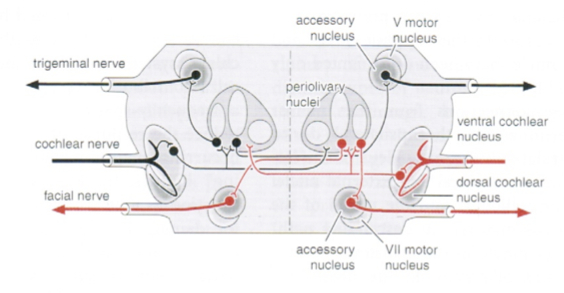
Each superior olive also sends projections to the ipsilateral trigeminal nucleus. From there, the trigeminal nucleus projects through the trigeminal nerve to the ipsilateral tensor tympani muscle, which contracts and thereby limits the movement of the tympanic membrane.
The pathways for the tensor tympani reflex are depicted in the Figure below, from Niewenhuys and colleagues (Nieuwenhuys et al. 2008).
Neurotransmitters in the auditory system
The auditory system uses a range of excitatory and inhibitory neurotransmitters (Haines 2006; Nieuwenhuys et al. 2008).
- First order neurons (in the spiral ganglion) project to the cochlear nuclei and release glutamate and aspartate (excitatory).
- There are many projections to the cochlear nuclei:
- From the hypothalamus, releasing histamine.
- From the nucleus locus coeruleus, releasing noradrenaline.
- There are also afferent projections that release dynorphin.
- There are many projections to the inferior colliculus:
- From the superior olive, releasing cholecystokinin.
- From the nucleus of the lateral lemniscus, releasing dynorphin.
- The olivocochlear bundle contains enkephalin.
- The medial superior olive projects ipsilaterally to the dorsal nucleus of the lateral lemniscus and to the inferior colliculus; this is excitatory.
- The lateral superior olive projects bilaterally to the dorsal nucleus of the lateral lemniscus and to the inferior colliculus. The ipsilateral projection is glycinergic (inhibitory); the contralateral projection is excitatory.
- The neurons of the nucleus of the trapezoid body release glycine (inhibitory).
- Commissural connections between the cochlear nuclei release glycine (inhibitory).
- The dorsal nucleus of the lateral lemniscus projects bilaterally to the inferior colliculi and releases GABA (inhibitory).
- In the descending auditory pathway, projections from the superior olive to the cochlea release acetylcholine (excitatory), cGRP (excitatory) and GABA (inhibitory).
References
Haines DE (2006) Fundamental neuroscience, 3rd edn. Churchill Livingstone, Philadelphia
Helpard L, Li H, Rohani SA, Zhu N, Rask-Andersen H, Agrawal SK, Ladak H (2021) An Approach for Individualized Cochlear Frequency Mapping Determined from 3D Synchrotron Radiation Phase-Contrast Imaging. IEEE Trans Biomed Eng PP. doi: 10.1109/TBME.2021.3080116
Li H, Helpard L, Ekeroot J, Rohani SA, Zhu N, Rask-Andersen H, Ladak HM, Agrawal S (2021) Three-dimensional tonotopic mapping of the human cochlea based on synchrotron radiation phase-contrast imaging. Sci Rep 11: 4437. doi: 10.1038/s41598-021-83225-w
Nieuwenhuys R, Voogd J, Huijzen Cv (2008) The human central nervous system, 4th edn. Springer, New York
Saenz M, Langers DR (2014) Tonotopic mapping of human auditory cortex. Hear Res 307: 42-52. doi: 10.1016/j.heares.2013.07.016
Schünke M, Ross LM, Lamperti ED, Schulte E, Schumacher U (2007) Head and neuroanatomy. Thieme, Stuttgart ; New York
![]()