By Marcello Cherchi, MD PhD
For patients
Electronystagmography (ENG) is a test that measures eye movements, and may be used when evaluating for diseases that can cause disequilibrium. During the test you will have several surface electrodes applied around your eyes. You will perform a number of tasks involving eye movements. Depending on the diagnoses being considered, there may be a section of the test in which the audiologist or technician may squirt warm and/or cool air or water in your ears, which some patients find uncomfortable because it can make you feel dizzy for a few minutes. The test overall takes about 45 minutes. If you have wax in your ear, this should be removed prior to the test; otherwise there are no special preparations required. There are no specific post-procedure instructions.
For clinicians
Overview
Electronystagmography (ENG) is an evoked potential test that can quantify horizontal and vertical eye movements. Equipment for performing ENG is relatively inexpensive, but the test is technically demanding for the audiologist or ototechnician, and is subject to errors from artifact and miscalibration. ENG can be helpful in identifying eye movements that are diagnostic of a variety of vestibular and neurologic disorders. ENG is gradually being replaced by videonystagmography because the latter test is easier to perform.
Introduction
Electronystagmography (ENG) was the earliest methodology for recording and quantifying eye movements. ENG probably remains the most widely used methodology worldwide because it is relatively inexpensive. It is gradually being replaced by videonystagmography (VNG) which, although more expensive, is far less cumbersome to use, is easier for the technician or audiologist to perform, and is less uncomfortable for the patient. Although audiology graduate programs in the United States still teach their students about ENG, very few audiologists use this methodology in their clinical practice.
ENG and VNG, while not identical (Pietkiewicz et al. 2012), overlap significantly in their ability to detect pathology (Bielinska et al. 2018; Gananca et al. 2010).
Although the term “electronystagmography” implies that this test only measures nystagmus, in fact it evaluates many aspects of oculomotor function beyond simply patterns of nystagmus; nevertheless, we will continue to use the conventional term ENG even though its utility extends beyond what the name suggests.
Overview of ENG
Electronystagmography (ENG) is regarded as an evoked potential test, but unlike more conventional evoked potential tests (that measure voltage differences resulting from biologically generated electrochemical gradients), electronystagmography measures the degree to which a voltage gradient (across surface electrodes placed on the face immediately around the eye) is affected by the physical orientation of an electrical dipole arising from the corneal-retinal potential (CRP).
History of ENG
The CRP, first recognized by Emil Heinrich du Bois-Reymond in 1848 (du Bois-Reymond 1848, 1849), exists because the metabolic activity in the retinal pigment epithelium results in a net negative charge, whereas the cornea has a net positive charge. This net electrical difference between cornea and retina generates an electrical dipole along the anterior (corneal) — posterior (retinal) axis of the globe. As the orientation of the globe (relative to the orbit) changes (and thus also the vector of its electrical dipole), it influences the voltage difference between the surface electrodes placed around the eye. Meyers (Meyers 1929) and Jacobson (Jacobson 1930) are usually credited as having been the first to recognize, apparently independently (Mowrer et al. 1936), that the CRP could be used in detection of eye movements.
Technique of ENG
Generally, one pair of diametrically opposed electrodes is placed to the right and left of the eye (for detecting horizontal eye movements) while another pair of electrodes is placed above and below the eye (for detecting vertical eye movements), as shown in the Figure below, from Duchowski (Duchowski 2003).
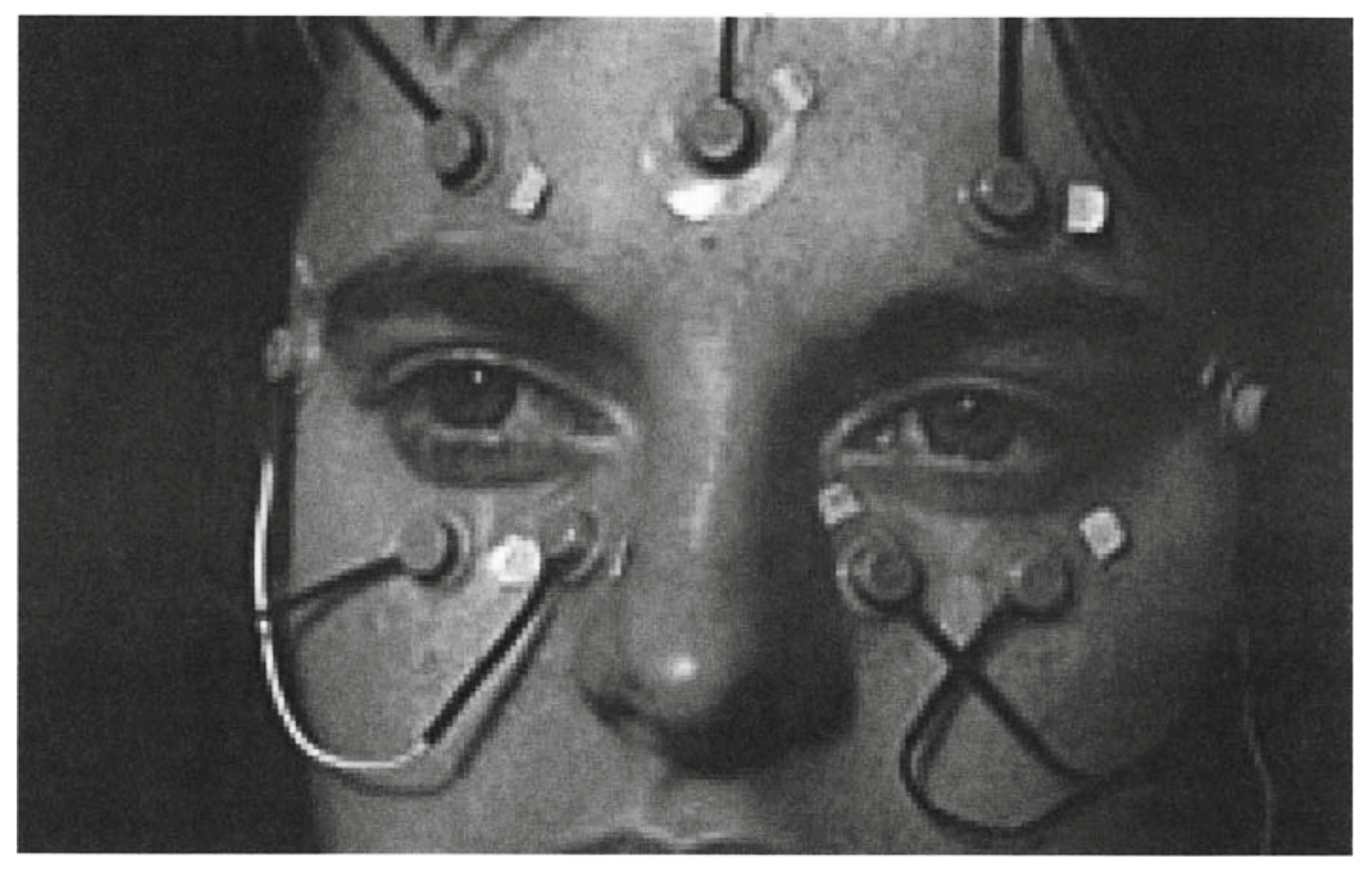
The magnitude of the electrical potential across a pair of electrodes is plotted over time, generating a tracing that is taken as a proxy for the orientation of the eye.
Advantages of ENG
Advantages of ENG include that the equipment is relatively inexpensive, and that it can to some degree detect eye movements even when the eye is closed (Mulch and Bonner 1976). Disadvantages include that it is more technically demanding than videonystagmography (VNG); calibration (discussed below) is difficult and often needs to be repeated during a study; the results are subject to a variety of electrical artifacts; and it does not detect ocular torsion.
ENG will probably continue to have some role in clinical medicine insofar as it is the only practical technique to use during a sleep study to analyze eye movements behind closed eyelids. Aserinsky and Kleitman originally inferred rapid eye movements during sleep from electroencephalography studies (Aserinsky and Kleitman 1953), and shortly thereafter began to measure these eye movements directly using electronystagmography (Aserinsky and Kleitman 1955).
Disadvantages of ENG
The magnitude of the CRP is not static. The negative electrical potential of the retina changes depending on its metabolic activity. Specifically, when less light is stimulating the retina, it is less metabolically active (and thus its electronegativity is reduced); when more light is stimulating the retina, it is more metabolically active (and thus its electronegativity is augmented). In contrast, the electrical charge of the cornea is relatively static. Thus, as the electronegativity of the retina changes, so does the electrical difference between it and the cornea (Arden and Kelsey 1962). In an individual who is dark-adapted the CRP is smaller, while in an individual who is light-adapted the CRP is larger. Consequently, when exposed to different illumination levels, the magnitude of the corneal-retinal electrical dipole changes, as does its influence on the surface electrodes placed around the eye that detect it. Moreover, the change (depending on the level of illumination) in the retinal potential is not instantaneous; it occurs gradually, often over several minutes (Lightfoot 2004; Norman and Brown 1999). These characteristics of the CRP pose a problem for calibration of ENG. If an individual is calibrated for ENG at an intermediate level of illumination but is then examined in the dark, a given eye excursion will falsely appear (on the ENG tracing) to be smaller in magnitude, whereas if the individual is examined in a brighter environment, the same eye excursion will falsely appear (on the ENG tracing) to be larger in magnitude. Unless this variability is accounted for, the measurement of the magnitude of the eye movements will be incorrect, introducing error into calculations of eye position, and thus velocity, corrupting many of the results.
From the perspective of the test’s accuracy it would be desirable to recalibrate the ENG for each subsection of the test during which the level of illumination reaching the retina differs from the previous subsection, allowing several minutes for the retinal potential to stabilize. From the perspective of the test’s efficiency, this is undesirable as it would greatly increase the test duration. Professional societies differ on recommendations of how often, and at what points during the ENG, recalibration should be performed ([No authors] 1999; Acoustical Society of America and American National Standards Institute 2009; British Society of Audiology 2015).
Other physiologic, non-pathological factors can influence ENG. When the eye blinks, the electrochemical activity of contraction in the frontalis muscle can influence the voltage difference detected by the leads (Eckert and Gizzi 1998; Gananca et al. 2010), introducing artifact; this eyelid artifact affects detection of vertical eye movements more unpredictably than horizontal eye movements (Barry and Jones 1965).
Ophthalmological disease affecting the retina can influence the CRP (Jacobson and McCaslin 2004).
There are also several sources of error arising from technology and technique. As with any evoked potential test involving surface leads, additional sources of error include inadequately cleaned skin (resulting in higher impedance), improperly placed leads (Hakkinen et al. 1993), and lead migration. If there is inadequate electrical shielding, ambient electrical noise can also result in interference (Eckert and Gizzi 1998).
Equipment needed
Electronystagmography is performed by an audiologist or ototechnician. Required equipment includes surface electrodes and multi-channel evoked potential detector. If caloric testing will be performed, then an irrigator is needed.
How to perform the test
While there is no standardized set of subtests for oculomotor testing, most clinical vestibular laboratories include spontaneous nystagmus, eccentric gaze, horizontal and vertical saccades, horizontal and vertical smooth pursuit, upright positional testing (sometimes referred to as vertebral artery testing), the Dix-Hallpike maneuver and horizontal optokinetic testing. This is a reasonable selection of subtests for detecting a fairly broad range of disorders.
What this test assesses
Electronystagmography quantifies horizontal and vertical eye movements during various states and tasks.
How to interpret the test results
Output is generally a tracing in which the X‑axis is time, and the Y‑axis is degrees of eye displacement from primary position of gaze; in some systems the horizontal eye movement tracing is depicted in one color, while the vertical eye movement tracing is displayed in another color.
Limitations
Electronystagmography does not detect torsional eye movements.
Contraindications
If the patient is known to be allergic to the electrode adhesive, then it may not be feasible to perform electronystagmography.
Pitfalls
As discussed earlier, calibration and re-calibration can be challenging. Other confounding factors include the level of illumination.
When is the test indicated
Electronystagmography (ENG) is reasonable to check in patients with the symptom of disequilibrium. If videonystagmography is available, it is usually preferable to ENG.
Diseases that may be diagnosed by this test
Results from electronystagmography may be helpful in diagnosing a number of vestibular and neurological disorders.
References
[No authors] (1999) Caloric test protocol. Br J Audiol 33: 179-84.
Acoustical Society of America, American National Standards Institute (2009) American National Standard Procedures for Testing Basic Vestibular Function, ASA-ANSI/ASA S3.45-2009. Standards Secretariat, Melville, NY
Arden GB, Kelsey JH (1962) Changes produced by light in the standing potential of the human eye. J Physiol 161: 189-204. doi: 10.1113/jphysiol.1962.sp006881
Aserinsky E, Kleitman N (1953) Regularly occurring periods of eye motility, and concomitant phenomena, during sleep. Science 118: 273-4. doi: 10.1126/science.118.3062.273
Aserinsky E, Kleitman N (1955) Two types of ocular motility occurring in sleep. J Appl Physiol 8: 1-10. doi: 10.1152/jappl.1955.8.1.1
Barry W, Jones GM (1965) Influence of Eye Lid Movement Upon Electro-Oculographic Recording of Vertical Eye Movements. Aerosp Med 36: 855-8.
Bielinska M, Polaczkiewicz A, Pietkiewicz P, Milonski J, Olszewski J (2018) Concurrent ENG and VNG recording in healthy people – preliminary report. Otolaryngol Pol 72: 45-49. doi: 10.5604/01.3001.0011.7251
British Society of Audiology (2015) Recommended Procedure: Vestibular assessment — eye movement recordings.
du Bois-Reymond EH (1848) Untersuchungen über thierische Elektricität, erster Band. Verlag von G. Reimer, Berlin
du Bois-Reymond EH (1849) Untersuchungen über thierische Elektricität, zweiter Band. Verlag von G. Reimer, Berlin
Duchowski AT (2003) Eye tracking methodology : theory and practice. Springer, New York
Eckert AM, Gizzi M (1998) Video-oculography as part of the ENG battery. Br J Audiol 32: 411-6. doi: 10.3109/03005364000000092
Gananca MM, Caovilla HH, Gananca FF (2010) Electronystagmography versus videonystagmography. Braz J Otorhinolaryngol 76: 399-403.
Hakkinen V, Hirvonen K, Hasan J, Kataja M, Varri A, Loula P, Eskola H (1993) The effect of small differences in electrode position on EOG signals: application to vigilance studies. Electroencephalogr Clin Neurophysiol 86: 294-300. doi: 10.1016/0013-4694(93)90111-8
Jacobson E (1930) Electrical measurements of neuromuscular states during mental activities. III. Visual imagination and recollection. American Journal of Physiology 95: 694-702. doi: 10.1152/ajplegacy.1930.95.3.694
Jacobson GP, McCaslin DL (2004) Detection of ophthalmic impairments indirectly with electronystagmography. J Am Acad Audiol 15: 258-63. doi: 10.3766/jaaa.15.3.8
Lightfoot GR (2004) The origin of order effects in the results of the bi-thermal caloric test. Int J Audiol 43: 276-82. doi: 10.1080/14992020400050037
Meyers IL (1929) Electronystagmography: a graphic study of the action currents in nystagmus. Archives of Neurology and Psychiatry 21: 901-918. doi: 10.1001/archneurpsyc.1929.02210220172009
Mowrer OH, Ruch TC, E MN (1936) The corneo-retinal potential difference as the basis of the galvanometric method of recording eye movements. American Journal of Physiology 114: 422. doi: 10.1152/ajplegacy.1935.114.2.423
Mulch G, Bonner C (1976) The effect of eye closure upon the pathological vestibular spontaneous nystagmus. Acta Otolaryngol 81: 376-85.
Norman M, Brown E (1999) Variations in calibration for computerized electronystagmography. Br J Audiol 33: 1-7. doi: 10.3109/03005364000000095
Pietkiewicz P, Pepas R, Sulkowski WJ, Zielinska-Blizniewska H, Olszewski J (2012) Electronystagmography versus videonystagmography in diagnosis of vertigo. Int J Occup Med Environ Health 25: 59-65. doi: 10.2478/s13382-012-0002-1
![]()