By Marcello Cherchi, MD PhD
For patients
Vogt-Koyanagi-Harada syndrome (VKHS) is a rare autoimmune disorder that can cause eye problems, hearing problems, neurological problems and skin problems. If your doctor suspects VKHS, then they may check several tests and an MRI. Depending on the particular symptoms, treatment may be with an ophthalmologist or rheumatologist.
For clinicians
Overview
Vogt-Koyanagi-Harada syndrome (VKHS) is a rare autoimmune disorder of unknown etiology in which T-cell autoimmunity is directed against melanocytes in multiple tissues, including the uvea, meninges, skin and labyrinth, correspondingly manifesting with some combination of visual deficits, neurological symptoms, audiovestibular symptoms and integumentary symptoms. VKHS patients can exhibit symmetrical or asymmetrical hearing loss. Vestibular workup variably reveals spontaneous nystagmus, unilateral or bilateral caloric weakness, abnormalities on cervical and/or ocular vestibular evoked myogenic potentials. This profile of audiologic and vestibular findings is neither sensitive nor specific for the diagnosis. MRI may reveal ophthalmologic, meningeal, brain or spine parenchymal, and labyrinthine findings. Management is multidisciplinary and often involves ophthalmology and rheumatology. Treatment typically begins with systemic corticosteroids.
Introduction
Urzua and colleagues (Urzua et al. 2022), and Herbort and Mochizuki (Herbort and Mochizuki 2007) summarize the sequence of early investigators who helped characterize this syndrome as follows:
- The Swiss ophthalmologist Alfred Vogt (1879 – 1943) described a case of whitening of the eyelashes (for which the medical term is “poliosis”) in 1906 (Vogt 1906).
- The first Japanese researcher to publish about this was the professor of ophthalmology, Dr. Jujiro Komoto, in 1911 (Komoto 1911).
- The Japanese researcher Yoshizo Koyanagi (1880 – 1954) published two cases in 1914, then a series of 16 in 1929 (Koyanagi 1929).
- The Japanese researcher Einosuke Harada (1892 – 1946) published one case in 1923, and 5 cases in 1926 (Harada 1926).
Subsequent descriptions recognized additional features which, when taken together, comprise a uveomeningoencephalitic syndrome characterized by chronic bilateral panuveitis and variable neurological symptoms, auditory symptoms (such as tinnitus and hearing loss) and cutaneous symptoms (such as vitiligo), which came to be known as Vogt-Koyanagi-Harada syndrome (VKHS). Unfortunately, Dr. Jujiro Komoto was not included in the eponymous designation of this syndrome.
|
Figure : Alfred Vogt (1879-1943). From Gloor (Gloor 2008). |
Figure : Yoshizo Koyanagi (1880-1954). From Herbort and Mochizuki (Herbort and Mochizuki 2007). |
Figure : Einosuke Harada (1892-1946). From Urzua et al. (Urzua et al. 2022). |
Epidemiology
VKHS is rare. It occurs more commonly in populations of East Asia, South Asia, the Middle East, and some Native American groups. Peak onset is the third decade, with a range of second to fifth decade (Mota and Santos 2010). It rarely begins in childhood, and when it does, it has a more aggressive clinical course (Tabbara et al. 1998).
Genetics
Numerous genetic and epigenetic factors have been associated with an increased susceptibility to VKHS (Su et al. 2023; Wu et al. 2023; Zhou et al. 2023).
Pathophysiological mechanism of disease
VKHS is a T-cell mediated autoimmune disorder of unknown etiology in which the misdirected autoimmune attack targets melanocytes in multiple tissue types, including the uvea, meninges, skin and labyrinth (Fujiwara et al. 2017; Kimura et al. 1996).
Clinical presentation
Revised diagnostic criteria for VKHS published in 2001 (Read et al. 2001), displayed below, are rather complex.
Complete Vogt-Koyanagi-Harada disease (criteria 1 to 5 must be present):
- No history of penetrating ocular trauma or surgery preceding the initial onset of uveitis.
- No clinical or laboratory evidence suggestive of other ocular disease entities.
- Bilateral ocular involvement (a or b must be met, depending on the stage of disease when the patient is examined).
- Early manifestations of disease.
- There must be evidence of a diffuse choroiditis (with or without anterior uveitis, vitreous inflammatory reaction, or optic disk hyperemia), which may manifest as one of the following:
- Focal areas of subretinal fluid, or
- Bullous serous retinal detachments.
- With equivocal fundus findings; both of the following must be present as well:
- Focal areas of delay in choroidal perfusion, multifocal areas of pinpoint leakage, large placoid areas of hyperfluorescence, pooling within subretinal fluid, and optic nerve staining (listed in order of sequential appearance) by fluorescein angiography, and
- Diffuse choroidal thickening, without evidence of posterior scleritis by ultrasonography.
- There must be evidence of a diffuse choroiditis (with or without anterior uveitis, vitreous inflammatory reaction, or optic disk hyperemia), which may manifest as one of the following:
- Late manifestations of disease.
- History suggestive of prior presence of findings from 3a, and either both (2) and (3) below, or multiple signs from (3):
- Ocular depigmentation (either of the following manifestations is sufficient):
- Sunset glow fundus, or
- Sugiura sign.
- Other ocular signs:
- Nummular chorioretinal depigmented scars, or
- Retinal pigment epithelium clumping and/or migration, or
- Recurrent or chronic anterior uveitis.
- Early manifestations of disease.
- Neurological/auditory findings (may have resolved by time of examination).
- Meningismus (malaise, fever, headache, nausea, abdominal pain, stiffness of the neck and back, or a combination of these factors; headache alone is not sufficient to meet definition of meningismus, however), or
- Tinnitus, or
- Cerebrospinal fluid pleocytosis.
- Integumentary finding (not preceding onset of central nervous system or ocular disease).
- Alopecia, or
- Poliosis, or
- Vitiligo.
Incomplete Vogt-Koyanagi-Harada disease (criteria 1 to 3 and either 4 or 5 must be present):
-
- No history of penetrating ocular trauma or surgery preceding the initial onset of uveitis, and
- No clinical or laboratory evidence suggestive of other ocular disease entities, and
- Bilateral ocular involvement.
- Neurologic/auditory findings; as defined for complete Vogt-Koyanagi-Harada disease above, or
- Integumentary findings; as defined for complete Vogt-Koyanagi-Harada disease above.
Probable Vogt-Koyanagi-Harada disease (isolated ocular disease; criteria 1 to 3 must be present):
-
- No history of penetrating ocular trauma or surgery preceding the initial onset of uveitis.
- No clinical or laboratory evidence suggestive of other ocular disease entities.
- Bilateral ocular involvement as defined for complete Vogt-Koyanagi-Harada disease above.
Fujiwara and colleagues state that, “Otological symptoms, including hearing loss, tinnitus, and vertigo, typically coincide with the onset of ocular pathology” (Fujiwara et al. 2017). The ophthalmologic involvement is bilateral, though the corresponding visual symptoms may be asymmetric (Urzua et al. 2022).
Noguchi and colleagues (Noguchi et al. 2014) studied 41 VKHS patients and found that 28/41 (68%) complained of auditory symptoms. Of those 28 patients, 68% complained of tinnitus, 43% complained of hearing loss, 21% complained of aural fullness and 11% complained of hyperacusis.
The audiologic and vestibular symptoms can be episodic, and may be metachronous (Gaudreau et al. 2012).
The profile of audiologic and vestibular symptoms is neither sensitive nor specific for the diagnosis.
Physical examination
Physical examination findings change as the disease evolves.
The Figure below, taken from the original article published by Vogt in 1906 (Vogt 1906), demonstrates poliosis (whitening of the eyelashes).
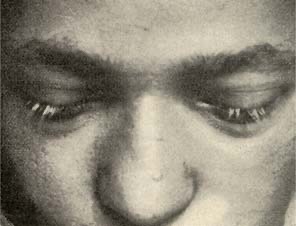
Figure : Poliosis (whitening of the eyelashes). From Vogt (Vogt 1906).
Testing: auditory
Fujiwara and colleagues (Fujiwara et al. 2017) studied 15 VKHS patients with pure tone audiometry and reported that 9/15 (60%) had age-appropriate hearing, 4/15 (27%) had some degree of unilateral hearing loss out of proportion to age, and 2/15 (13%) had some degree of bilateral hearing loss out of proportion to age.
Testing: vestibular
Fujiwara and colleagues (Fujiwara et al. 2017) studied vestibular tests (oculography, caloric testing, cervical vestibular evoked myogenic potentials and ocular vestibular evoked myogenic potentials) in 42 VKHS patients and reported the rates of abnormalities displayed in the Table below.
|
Feature |
Number of patients (out of 42) |
Percentage of patients |
|
Subjective symptoms (n=42) |
||
|
Yes |
8 |
19% |
|
No |
34 |
81% |
|
Nystagmus tests (n=28) |
||
|
Abnormal |
12 |
43% |
|
Normal |
16 |
57% |
|
Stepping tests (n=16) |
||
|
Abnormal |
6 |
38% |
|
Normal |
10 |
63% |
|
Caloric tests (n=15) |
||
|
Bilateral weakness |
3 |
20% |
|
Unilateral weakness |
4 |
27% |
|
Normal |
8 |
53% |
|
Cervical vestibular evoked myogenic potentials (cVEMP) (n=16) |
||
|
Abnormal |
7 |
44% |
|
Normal |
9 |
56% |
|
Ocular vestibular evoked myogenic potentials (oVEMP) (n=16) |
||
|
Abnormal |
8 |
50% |
|
Normal |
8 |
50% |
Table : Rates of abnormal vestibular test results in 42 Vogt-Koyanagi-Harada syndrome patients. From Fujiwara et al. (Fujiwara et al. 2017).
Noguchi and colleagues (Noguchi et al. 2014) studied oculography 24 VKHS patients, and reported that 11/24 (46%) showed spontaneous horizontal nystagmus.
Oku and Ishikawa (Oku and Ishikawa 1994) studied oculography in 15 VKHS patients, and reported that 8/15 (53%) exhibited spontaneous horizontal nystagmus. Of the 8 patients with spontaneous nystagmus, 6 (75%) exhibited low gain in smooth pursuit “on the side ipsilateral to the nystagmus,” and 7 (88%) exhibited elevated gain of the vestibulo-ocular reflex. Of the 7 patients with no spontaneous nystagmus, only 1 (14%) exhibited elevated gain of the vestibulo-ocular reflex.
Noguchi and colleagues (Noguchi et al. 2014) studied caloric testing in 6 VKHS patients, and reported that 2/6 (33%) exhibited bilateral caloric weakness, 2/6 (33%) exhibited unilateral caloric weakness, and 2/6 (33%) showed normal caloric responses.
Fujiwara and colleagues (Fujiwara et al. 2017) studied one VKHS patient with video head impulse testing (vHIT) and found low gain (0.80) on one side and normal gain (0.87) on the other side, and no compensatory saccades.
Testing: other
Treated VKHS patients in the chronic (quiescent) stage of the disease exhibit decreased retinal and choroidal vessel density on optical coherence tomography (Guo et al. 2025).
Imaging
Numerous MRI findings have been reported in VKHS, including:
- Ophthalmologic findings:
- Retinal detachment (Al Banna et al. 2019; McGehee et al. 2005)
- Choroidal thickening (Ando et al. 2018; Ibanez et al. 1994; Phagura and Bhikoo 2021)
- Meningeal findings:
- Pachymeningeal enhancement (Han et al. 2010)
- Leptomeningeal enhancement (Le et al. 2019; Lohman et al. 2011)
- Labyrinthine enhancement (Hida et al. 2017)
- Brain parenchymal findings:
- Intracerebral hemorrhage (Baheti et al. 2009)
- Intraparenchymal FLAIR hyperintensities (Keles et al. 2013)
- Intraparenchymal enhancement (Ikeda and Tsukagoshi 1992)
- Myelitis (Dahbour 2009)
Differential diagnosis
The differential diagnosis of VKHS is broad and includes choroidal melanoma, sympathetic ophthalmia, infectious uveitis, systemic lupus erythematosus, sarcoidosis, Cogan syndrome, and others.
Treatment
Management of VKHS is multidisciplinary and often involves ophthalmology and rheumatology. Initial treatment is usually with systemic of corticosteroids, and may also include immunosuppressants or immunomodulatory agents.
Prognosis
Mota and Santos (Mota and Santos 2010) state:
“With this disease, prognosis depends greatly on how early it is diagnosed and correctly treated. Disorders of the auditory system respond well to treatment and are generally completely reversed in 2 to 3 months, whereas the cutaneous lesions are permanent. Prognosis for the vision is extremely variable, although generally favorable. It has been found that the three most important prognostic factors are: good visual acuity 1 month after starting treatment, early high-dose corticosteroids and age at disease onset, with evidence that younger patients suffered a lower rate of ocular complications” (Mota and Santos 2010).
References
Al Banna M, Reeder S, Ghannam M, Robertson J, Stutz A (2019) Teaching NeuroImages: A case of Vogt-Koyanagi-Harada disease with bilateral retinal detachment. Neurology 93: e421. doi: 10.1212/wnl.0000000000007830
Ando T, Kato H, Mochizuki K, Ozawa K, Goshima S, Matsuo M (2018) MR findings of the orbit in patients with Vogt-Koyanagi-Harada disease. Neuroradiology 60: 421-426. doi: 10.1007/s00234-018-1999-z
Baheti NN, Cherian A, Kate M, Krishnan S, Thomas B (2009) Intracerebral hemorrhages in Vogt-Koyanagi-Harada disease. Neurol India 57: 815-7. doi: 10.4103/0028-3886.59491
Dahbour SS (2009) MRI documented acute myelitis in a patient with Vogt-Koyanagi-Harada syndrome: first report. Clin Neurol Neurosurg 111: 200-2. doi: 10.1016/j.clineuro.2008.09.006
Fujiwara K, Morita S, Hoshino K, Fukuda A, Nakamaru Y, Homma A (2017) Evaluation of Vestibular Functions in Patients with Vogt-Koyanagi-Harada Disease. Audiol Neurootol 22: 190-195. doi: 10.1159/000481426
Gaudreau P, Moy J, Lindsay F (2012) An unusual cause of vertigo, tinnitus, and hyperacusis: Vogt-Koyanagi-Harada syndrome. Ear Nose Throat J 91: E7-9. doi: 10.1177/014556131209101213
Gloor BP (2008) Alfred vogt (1879-1943). Surv Ophthalmol 53: 655-63. doi: 10.1016/j.survophthal.2008.08.014
Guo S, Xia L, Hu R, Wang J, Yang P (2025) VASCULAR CHANGES AND IRREVERSIBLE COMPLICATIONS IN 120 degrees FUNDUS USING WIDEFIELD SWEPT-SOURCE OPTICAL COHERENCE TOMOGRAPHY ANGIOGRAPHY IN VOGT-KOYANAGI-HARADA DISEASE. Retina 45: 79-87. doi: 10.1097/IAE.0000000000004259
Han HJ, Kim HY, Park JH, Lee EJ, Kim DG, Shin DI (2010) Magnetic resonance imaging of pachymeningeal enhancement in Vogt-Koyanagi-Harada disease. Neurol Sci 31: 785-8. doi: 10.1007/s10072-010-0236-2
Harada E (1926) Beitrag zur klinischen Kenntnis von nichteitriger Choroiditis (Choroiditis diffusa acuta) [Clinical study of nonsuppurative choroiditis]. Nippon Ganka Gakkai Zasshi [Acta Societatis Ophthalmologica Japonica] 30: 356-361.
Herbort CP, Mochizuki M (2007) Vogt–Koyanagi–Harada disease: inquiry into the genesis of a disease name in the historical context of Switzerland and Japan. International Ophthalmology 27: 67-79. doi: 10.1007/s10792-007-9083-4
Hida K, Takano K, Yoshimitsu K, Fukae J, Hokao K (2017) Inner ear enhancement on gadolinium-enhanced 3D FLAIR images in a patient with Vogt-Koyanagi-Harada disease. BJR Case Rep 3: 20160090. doi: 10.1259/bjrcr.20160090
Ibanez HE, Grand MG, Meredith TA, Wippold FJ, 2nd (1994) Magnetic resonance imaging findings in Vogt-Koyanagi-Harada syndrome. Retina 14: 164-8.
Ikeda M, Tsukagoshi H (1992) Vogt-Koyanagi-Harada disease presenting meningoencephalitis. Report of a case with magnetic resonance imaging. Eur Neurol 32: 83-5. doi: 10.1159/000116797
Keles S, Ogul H, Pinar LC, Kantarci M (2013) Teaching neuroimages: cerebral white matter involvement in a patient with Vogt-Koyanagi-Harada syndrome. Neurology 81: e85-6. doi: 10.1212/WNL.0b013e3182a43b01
Kimura H, Ohashi N, Aso S, Watanabe Y (1996) Clinical study of the role of melanocytes in the inner ear of patients with Harada’s disease. ORL J Otorhinolaryngol Relat Spec 58: 233-7. doi: 10.1159/000276843
Komoto J (1911) Über Vitiligo und Auge. Klinische Monatsblätter für Augenheilkunde [Monthly Clinical Journal of Ophthalmology] 49: 139-142.
Koyanagi Y (1929) Dysakusis, Alopecie und Poliosis bei schwerer Uveitis nicht traumatischen Ursprungs [Dysacusis, alopecia and poliosis in severe uveitis of non-traumatic origin]. Klinische Monatsblätter für Augenheilkunde [Monthly Clinical Journal of Ophthalmology] 82: 194-211.
Le TA, Simon S, Gilhotra J, Hissaria P (2019) Vogt-Koyanagi-Harada syndrome presenting with bilateral optic disc swelling and leptomeningeal enhancement. BMJ Case Rep 12. doi: 10.1136/bcr-2019-229719
Lohman BD, Gustafson CA, McKinney AM, Sarikaya B, Silbert SC (2011) MR imaging of Vogt-Koyanagi-Harada syndrome with leptomeningeal enhancement. AJNR Am J Neuroradiol 32: E169-71. doi: 10.3174/ajnr.A2279
McGehee BE, Chaljub G, Shah RK, Oto A, Camacho AC, Tang RA (2005) Bilateral retinal detachment in a patient with Vogt-Koyanagi-Harada syndrome. Emerg Radiol 11: 366-71. doi: 10.1007/s10140-005-0430-3
Mota LA, Santos AB (2010) Vogt-Koyanagi-Harada’s syndrome and its multisystem involvement. Rev Assoc Med Bras 56: 590-5.
Noguchi Y, Nishio A, Takase H, Miyanaga M, Takahashi H, Mochizuki M, Kitamura K (2014) Audiovestibular findings in patients with Vogt-Koyanagi-Harada disease. Acta Otolaryngol 134: 339-44. doi: 10.3109/00016489.2013.868604
Oku H, Ishikawa S (1994) Vestibulo-ocular reflex abnormality in Vogt-Koyanagi-Harada syndrome. Br J Ophthalmol 78: 912-6. doi: 10.1136/bjo.78.12.912
Phagura RS, Bhikoo R (2021) MRI Choroidal Thickening as Presenting Sign of Vogt-Koyanagi-Harada Disease. Asia Pac J Ophthalmol (Phila) 10: 127-128. doi: 10.1097/apo.0000000000000359
Read RW, Holland GN, Rao NA, Tabbara KF, Ohno S, Arellanes-Garcia L, Pivetti-Pezzi P, Tessler HH, Usui M (2001) Revised diagnostic criteria for Vogt-Koyanagi-Harada disease: report of an international committee on nomenclature. Am J Ophthalmol 131: 647-52. doi: 10.1016/s0002-9394(01)00925-4
Su G, Du L, Yu H, Li M, Huang R, Yang X, Wang D, Wang Q, Yang P (2023) Epigenome-wide association study identifies Vogt-Koyanagi-Harada disease-associated DNA methylation loci in Chinese. Exp Eye Res 233: 109553. doi: 10.1016/j.exer.2023.109553
Tabbara KF, Chavis PS, Freeman WR (1998) Vogt-Koyanagi-Harada syndrome in children compared to adults. Acta Ophthalmol Scand 76: 723-6. doi: 10.1034/j.1600-0420.1998.760619.x
Urzua CA, Herbort CP, Jr., Takeuchi M, Schlaen A, Concha-Del-Rio LE, Usui Y, Cuitino L, Papasavvas I (2022) Vogt-Koyanagi-Harada disease: the step-by-step approach to a better understanding of clinicopathology, immunopathology, diagnosis, and management: a brief review. J Ophthalmic Inflamm Infect 12: 17. doi: 10.1186/s12348-022-00293-3
Vogt A (1906) Frühzeitiges Ergrauen der Zilien und Bemerkungen über den sogenannten plötzlichen Eintritt dieser Veränderung [Premature whitening of eyelashes and remarks on the so-called sudden onset of this change]. Klinische Monatsblätter für Augenheilkunde [Monthly Clinical Journal of Ophthalmology] 44: 228-242.
Wu Q, Zhong Z, Zhou C, Cao Q, Su G, Yang P (2023) Association of ZC3HAV1 single nucleotide polymorphisms with the susceptibility of Vogt-Koyanagi-Harada Disease. BMC Med Genomics 16: 113. doi: 10.1186/s12920-023-01546-3
Zhou C, Cai S, Xie Y, Zeng Z, Zhang J, Su G, Wu Q, Ye X, Cao Q, Yang P, Hu J (2023) Genetic association of PRKCD and CARD9 polymorphisms with Vogt-Koyanagi-Harada disease in the Chinese Han population. Hum Genomics 17: 9. doi: 10.1186/s40246-023-00459-7
![]()


