By Marcello Cherchi, MD PhD
For patients
Granulomatosis with polyangiitis (GPA), previously known as Wegener’s granulomatosis, is an autoimmune disease that can cause symptoms involving the eyes, ears, nose, throat, lungs, kidneys and skin. Diagnosis and treatment is managed by rheumatology. Some patients with GPA get hearing loss. Less commonly they develop tinnitus or disequilibrium.
For clinicians
Overview
Granulomatosis with polyangiitis (GPA) is a member of a broader family of antineutrophil cytoplasmic autoantibody (ANCA)-associated vasculidites. It causes granulomas in numerous tissues including the eye, ear, nose throat, lungs, kidneys and skin. Some patients with GPA experience hearing loss that is usually bilateral, can be in any pattern (sensorineural, conductive or mixed), and any tempo (abrupt in onset, or progressive). Fewer patients experience tinnitus and even fewer experience vestibular symptoms. The hearing loss may respond to immunosuppressive treatment, if initiated early. For residual hearing loss, amplification or cochlear implantation can be considered. Management of GPA is typically overseen by a rheumatologist. Practitioners from other subspecialties may be required depending on the specific manifestations (e.g., otolaryngology, nephrology, dermatology).
Introduction
Granulomatosis with polyangiitis (GPA) is a member of a broader family of antineutrophil cytoplasmic autoantibody (ANCA)-associated vasculidites (which also includes eosinophilic granulomatosis with polyangiitis, microscopic polyangiitis and renal-limited vasculitis).
Historical background
In the 1930s, pathologist Dr. Friedrich Wegener (1907 – 1990) began publishing descriptions (Wegener 1936, 1939) of a disease that came to bear his name for a time, and he provided an overview of this period of his career in an article published in the year of his death (Wegener 1990). In light of subsequent research revealing that Wegener had been a member of the Nazi party (Woywodt and Matteson 2006), in 2010 the eponymous designation was retired and the diagnosis was renamed granulomatosis with polyangiitis (GPA) (Arkuszewski and Cieslak-Arkuszewska 2025).

Epidemiology
GPA is rare. Kitching and colleagues state that studies cite a prevalence ranging from 48 to 421 cases per million (Kitching et al. 2020).
Genetics
Some research suggests that there are genetic distinctions between the various antineutrophil cytoplasmic autoantibody (ANCA)-associated vasculidites (Lyons et al. 2012).
Pathophysiological mechanism of disease
GPA damages tissues through its vasculitic effects and the formation of granulomas.
Clinical presentation
GPA is a multi-system disease with variable manifestations. Presentation can include:
- Constitutional symptoms (fever, weight loss).
- Ophthalmologic symptoms (visual loss, conjunctivitis, corneal ulceration, scleritis, episcleritis, uveitis, retinal vasculitis, optic neuropathy).
- Otorhinolaryngological symptoms (hearing loss, disequilibrium, sinusitis, otalgia, rhinorrhea, epistaxis, otorrhea).
- Tracheo-pulmonary symptoms (cough, stridor, wheezing, dyspnea, hemoptysis, chest pain).
- Musculoskeletal symptoms (myalgias, arthralgias).
- Dermatologic symptoms (purpura, ulcers, urticaria, livedo reticularis, skin nodules).
- Renal symptoms (hematuria).
Patients with GPA can develop hearing loss that is usually bilateral (Sahyouni et al. 2019), and can be in any pattern (Sahyouni et al. 2019): sensorineural (Gomes et al. 2022; Guyot et al. 1990; Illum and Thorling 1982; Kempf 1989; Koenen et al. 2022; Kornblut et al. 1982; McCaffrey et al. 1980; Nakamaru et al. 2016; Ratmeyer et al. 2021; Takagi et al. 2002; Wierzbicka et al. 2011), conductive (Kiessling et al. 2020; McCaffrey et al. 1980; Rahne et al. 2017) or mixed (Bakthavachalam et al. 2004; Busch et al. 2022; Chen et al. 2024; Nakamaru et al. 2016). The conductive hearing loss appears to result from the ability of GPA to cause otitis media (Alkhaldi et al. 2024; Sahyouni et al. 2019; Takagi et al. 2002) with middle ear effusion (Uppal et al. 2014; Wierzbicka et al. 2011) and Eustachian tube dysfunction. GPA can also cause otitis externa (Illum and Thorling 1982). The hearing loss can be gradually progressive (Uppal et al. 2014; Yamazaki et al. 2012) or abrupt in onset (Kim et al. 2016; Ratmeyer et al. 2021).
Some GPA patients develop hypertrophic pachymeningitis (Martinez-Pina et al. 2024; Yildirim et al. 2008), which can cause hearing loss that is mixed (Chen et al. 2024).
Hearing loss is the most common aural symptom in GPA (Rahne et al. 2020; Safavi Naini et al. 2017). Less common aural symptoms include tinnitus (Rahne et al. 2020) and aural fullness (Vainutiene et al. 2024).
Rarely, the aural symptoms may precede other symptoms of GPA (Yamazaki et al. 2012).
A small minority of GPA patients experience vestibular symptoms (Goderis et al. 2015; Kim et al. 2016; Rahne et al. 2020).
Physical examination
Physical examination varies depending on organ system involvement.
Imaging
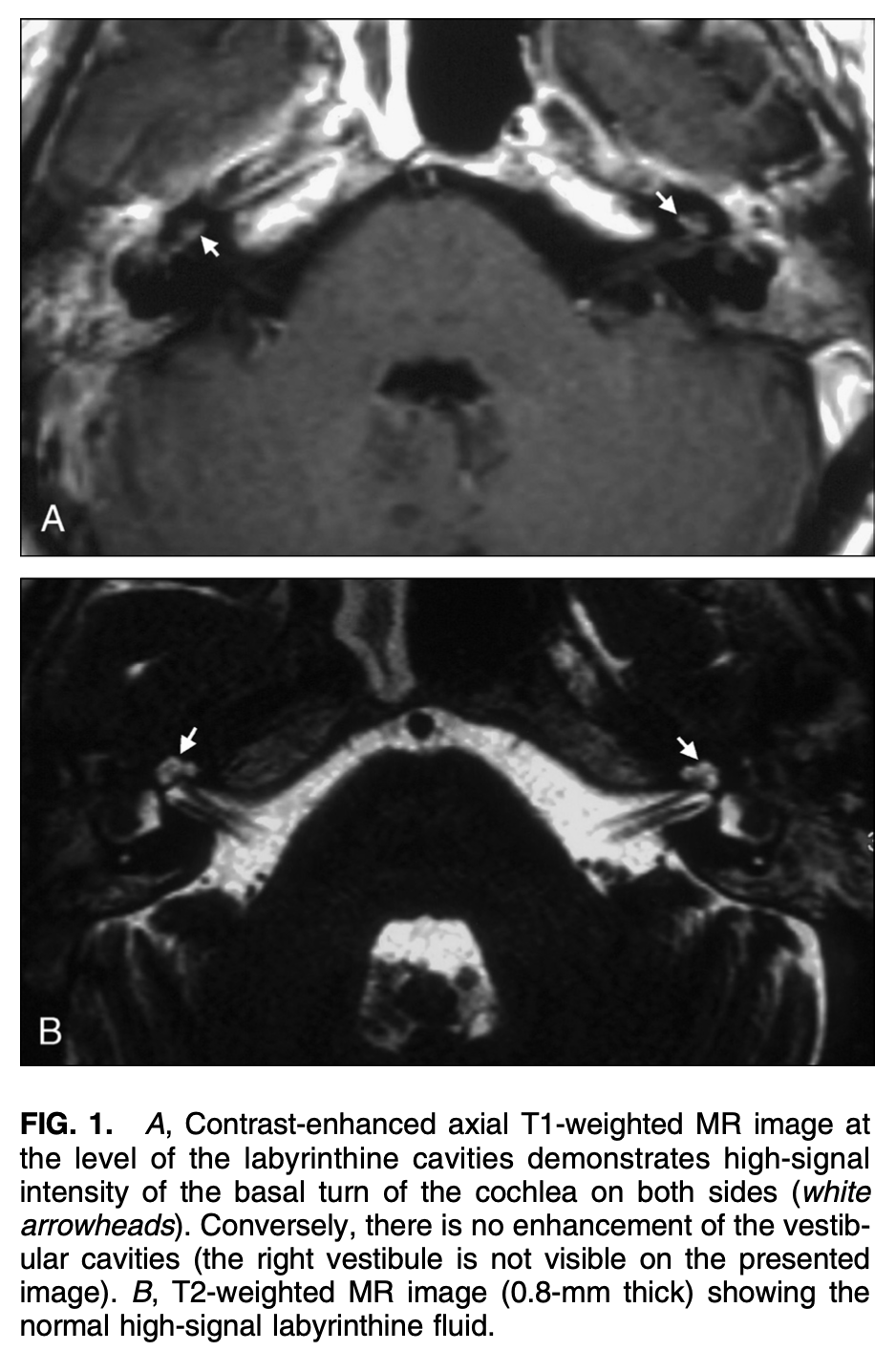
When GPA has a labyrinthitis-type of presentation, MRI of the internal auditory canals with contrast may show enhancement in the labyrinths, as displayed in the Figure below from Teszler and colleagues (Teszler et al. 2008).
Histopathology
Definitive diagnosis of GPA requires biopsy.
Temporal bone histopathology has reported granulation in the tympanic cavity (Ohtani et al. 2000). Yoon and colleagues (Yoon et al. 1989) studied temporal bone specimens from three GPA patients and reported serous middle ear effusion, and abnormalities involving the tympanic membrane, vascular thickening, tensor tympani muscle, Eustachian tube, mesotympanum, epitympanum, mastoid, round window, round window niche and labyrinth .
Differential diagnosis
The differential diagnosis includes:
- Other members of the family of antineutrophil cytoplasmic autoantibody (ANCA)-associated vasculidites (eosinophilic granulomatosis with polyangiitis, microscopic polyangiitis and renal-limited vasculitis).
- Polyarteritis nodosa.
- Anti-glomerular basement membrane antibody disease.
Management
Management of GPA is typically overseen by a rheumatologist. Practitioners from other subspecialties may be required depending on the specific manifestations (e.g., otolaryngology, nephrology, dermatology).
If hearing loss related to GPA is permanent, then consultation with audiology is appropriate to be evaluated for amplification (Dwyer and Janzen 1981). Cochlear implantation is sometimes offered (Bartov et al. 2022; Cacco et al. 2021; Elmas et al. 2017).
Prognosis
When treated with immunosuppressants (such as prednisone and cyclophosphamide), the hearing loss in GPA may improve (Guyot et al. 1990) or resolve (Clements et al. 1989), especially if treatment is initiated early (Sriskandarajah et al. 2012).
References
Alkhaldi A, Alaraifi A, Alsalamah S, Nassar J (2024) Atypical presentation of granulomatosis with polyangiitis: a case report and review of the literature. AME Case Rep 8: 82. doi: 10.21037/acr-24-47
Arkuszewski P, Cieslak-Arkuszewska A (2025) Undisclosed facts in Friedrich Wegener’s links with Nazism. Histopathology 86: 317-326. doi: 10.1111/his.15296
Bakthavachalam S, Driver MS, Cox C, Spiegel JH, Grundfast KM, Merkel PA (2004) Hearing loss in Wegener’s granulomatosis. Otol Neurotol 25: 833-7. doi: 10.1097/00129492-200409000-00030
Bartov N, Dahan T, Halperin D, Katzenell U (2022) Cochlear Implantation in a Patient with Granulomatosis with Polyangiitis. Isr Med Assoc J 25: 834-835.
Busch BH, Wilhelm D, Johnson P, Pfeifer M (2022) Granulomatosis With Polyangiitis Presenting as Complete Hearing Loss. Cureus 14: e24711. doi: 10.7759/cureus.24711
Cacco T, Castello E, Canevari FRM, Laborai A, Grillone A, Zanetti D, Peretti G (2021) Cochlear Implantation as a Treatment for Sudden Autoimmune Sensorineural Hearing Loss in a Patient Affected by Eosinophilic Granulomatosis with Polyangiitis: A Case Report and A Review of Literature. Ann Otol Rhinol Laryngol 130: 112-115. doi: 10.1177/0003489420938827
Chen Y, Gou L, Wang L, Qian M, Wu Q, Zheng W, Li M, Zeng X, Zhang F (2024) Clinical characteristics of hypertrophic cranial pachymeningitis in granulomatosis with polyangiitis: a retrospective single-center study in China. Arthritis Res Ther 26: 6. doi: 10.1186/s13075-023-03239-9
Clements MR, Mistry CD, Keith AO, Ramsden RT (1989) Recovery from sensorineural deafness in Wegener’s granulomatosis. J Laryngol Otol 103: 515-8. doi: 10.1017/s0022215100156750
Dwyer J, Janzen VD (1981) Wegener’s granulomatosis with otological and nervous system involvement. J Otolaryngol 10: 476-80.
Elmas F, Shrestha BL, Linder TE (2017) Subtotal Petrosectomy and Cochlear Implant Placement in Otologic Presentation of “Wegener’s Granulomatosis”. Kathmandu Univ Med J (KUMJ) 15: 94-98.
Goderis J, De Schepper S, Vannieuwenhuyze P, Schmelzer B (2015) Wegener granulomatosis as possible cause of vertigo: case report and review. B-ENT 11: 67-72.
Gomes I, Girao A, Gomes J, Rebelo O, Jesus-Ribeiro J (2022) Neurological impact of eosinophilic granulomatosis with polyangiitis. Acta Neurol Belg 122: 123-128. doi: 10.1007/s13760-021-01683-5
Guyot JP, Baud C, Montandon P (1990) Wegener’s granulomatosis with otological disorders as primary symptoms. ORL J Otorhinolaryngol Relat Spec 52: 327-34. doi: 10.1159/000276159
Illum P, Thorling K (1982) Otological manifestations of Wegener’s granulomatosis. Laryngoscope 92: 801-4. doi: 10.1288/00005537-198207000-00017
Kempf HG (1989) Ear involvement in Wegener’s granulomatosis. Clin Otolaryngol Allied Sci 14: 451-6. doi: 10.1111/j.1365-2273.1989.tb00403.x
Kiessling PT, Marinelli JP, Peters PA, DeLone DR, Lane JI, Koster MJ, Carlson ML (2020) Cranial Base Manifestations of Granulomatosis with Polyangiitis. Otolaryngol Head Neck Surg 162: 666-673. doi: 10.1177/0194599820912025
Kim SH, Jung AR, Kim SI, Yeo SG (2016) Refractory Granulomatosis with Polyangiitis Presenting as Facial Paralysis and Bilateral Sudden Deafness. J Audiol Otol 20: 55-8. doi: 10.7874/jao.2016.20.1.55
Kitching AR, Anders H-J, Basu N, Brouwer E, Gordon J, Jayne DR, Kullman J, Lyons PA, Merkel PA, Savage COS, Specks U, Kain R (2020) ANCA-associated vasculitis. Nature Reviews Disease Primers 6: 71. doi: 10.1038/s41572-020-0204-y
Koenen L, Elbelt U, Olze H, Zappe S, Dommerich S (2022) Granulomatosis with polyangiitis in a patient with polydipsia, facial nerve paralysis, and severe otologic complaints: a case report and review of the literature. J Med Case Rep 16: 291. doi: 10.1186/s13256-022-03492-7
Kornblut AD, Wolff SM, Fauci AS (1982) Ear disease in patients with Wegener’s granulomatosis. Laryngoscope 92: 713-7. doi: 10.1288/00005537-198207000-00001
Lyons PA, Rayner TF, Trivedi S, Holle JU, Watts RA, Jayne DR, Baslund B, Brenchley P, Bruchfeld A, Chaudhry AN, Cohen Tervaert JW, Deloukas P, Feighery C, Gross WL, Guillevin L, Gunnarsson I, Harper L, Hruskova Z, Little MA, Martorana D, Neumann T, Ohlsson S, Padmanabhan S, Pusey CD, Salama AD, Sanders JS, Savage CO, Segelmark M, Stegeman CA, Tesar V, Vaglio A, Wieczorek S, Wilde B, Zwerina J, Rees AJ, Clayton DG, Smith KG (2012) Genetically distinct subsets within ANCA-associated vasculitis. N Engl J Med 367: 214-23. doi: 10.1056/NEJMoa1108735
Martinez-Pina DA, Calderon-Garciduenas AL, Gama-Lizarraga E, Enriquez-Peregrino KG, Curiel-Zamudio JM (2024) Hypertrophic Pachymeningitis, Associated with Eosinophilic Granulomatosis with Polyangiitis, and ANCA-Negative Serology. Eur J Case Rep Intern Med 11: 004595. doi: 10.12890/2024_004595
McCaffrey TV, McDonald TJ, Facer GW, DeRemee RA (1980) Otologic manifestations of Wegener’s granulomatosis. Otolaryngol Head Neck Surg (1979) 88: 586-93. doi: 10.1177/019459988008800512
Nakamaru Y, Takagi D, Suzuki M, Homma A, Morita S, Homma A, Fukuda S (2016) Otologic and Rhinologic Manifestations of Eosinophilic Granulomatosis with Polyangiitis. Audiol Neurootol 21: 45-53. doi: 10.1159/000442040
Ohtani I, Baba Y, Suzuki C, Sakuma H, Kano M (2000) Temporal bone pathology in Wegener’s granulomatosis. Fukushima J Med Sci 46: 31-9. doi: 10.5387/fms.46.31
Rahne T, Clauss F, Plontke SK, Keysser G (2017) Prevalence of hearing impairment in patients with rheumatoid arthritis, granulomatosis with polyangiitis (GPA, Wegener’s granulomatosis), or systemic lupus erythematosus. Clin Rheumatol 36: 1501-1510. doi: 10.1007/s10067-017-3651-4
Rahne T, Plontke S, Keysser G (2020) Vasculitis and the ear: a literature review. Curr Opin Rheumatol 32: 47-52. doi: 10.1097/BOR.0000000000000665
Ratmeyer PR, Johnson BR, Roldan LP, Kraai TL (2021) Granulomatosis with Polyangiitis as a Cause of Sudden-Onset Bilateral Sensorineural Hearing Loss: Case Report and Recommendations for Initial Assessment. Case Rep Otolaryngol 2021: 6632344. doi: 10.1155/2021/6632344
Safavi Naini A, Ghorbani J, Montazer Lotfe Elahi S, Beigomi M (2017) Otologic Manifestations and Progression in Patients with Wegener’s granulomatosis: A Survey in 55 Patients. Iran J Otorhinolaryngol 29: 327-331.
Sahyouni R, Moshtaghi O, Abouzari M, Le P, Birkenbeuel J, Cheung D, Lin HW, Djalilian HR (2019) A Case Series of Granulomatosis With Polyangiitis Primarily Diagnosed by Otological Manifestations. Ann Otol Rhinol Laryngol 128: 263-266. doi: 10.1177/0003489418815517
Sriskandarajah V, Bansal RA, Yeoh R, Bansal AS (2012) Early intervention in localized Wegener’s granulomatosis with sensorineural hearing loss preserves hearing. Am J Audiol 21: 121-6. doi: 10.1044/1059-0889(2012/12-0003)
Takagi D, Nakamaru Y, Maguchi S, Furuta Y, Fukuda S (2002) Otologic manifestations of Wegener’s granulomatosis. Laryngoscope 112: 1684-90. doi: 10.1097/00005537-200209000-00029
Teszler CB, Williams MT, Belange G, Ayache D (2008) Labyrinthitis related to Wegener granulomatosis: magnetic resonance imaging findings. Otol Neurotol 29: 721-2. doi: 10.1097/mao.0b013e31815f665B
Uppal P, Taitz J, Wainstein B, Soma M, Belessis Y, Gray P (2014) Refractory otitis media: an unusual presentation of childhood granulomatosis with polyangiitis. Pediatr Pulmonol 49: E21-4. doi: 10.1002/ppul.22746
Vainutiene V, Ivaska J, Dadoniene J, Beleskiene V, Ivaskiene T, Lesinskas E (2024) Audiological Manifestations in Patients with Granulomatosis with Polyangiitis. Medicina (Kaunas) 60. doi: 10.3390/medicina60020267
Wegener F (1936) Über generalisierte, septische Gefässerkrankungen [About generalized septic vascular diseases]. Verhandlungen der deutschen pathologischen Gesellschaft [Proceedings of the German Pathological Society] 29: 202-210.
Wegener F (1939) Über eine eigenartige rhinogene Granulomatose mit besonderer Beteiligung des Arteriensystems und der Nieren [On a peculiar rhinogenic granulomatosis with particular involvement of the arterial system and the kidneys]. Beiträge zur pathologischen Anatomie und zur allgemeinen Pathologie [Contributions to pathological anatomy and general pathology] 102: 36-38. doi: 10.1016/S0005-8165(76)80082-0
Wegener F (1990) Wegener’s granulomatosis. European Archives of Oto-Rhino-Laryngology 247: 133-142. doi: 10.1007/BF00175962
Wierzbicka M, Szyfter W, Puszczewicz M, Borucki L, Bartochowska A (2011) Otologic symptoms as initial manifestation of wegener granulomatosis: diagnostic dilemma. Otol Neurotol 32: 996-1000. doi: 10.1097/MAO.0b013e31822558fd
Woywodt A, Matteson EL (2006) Wegener’s granulomatosis–probing the untold past of the man behind the eponym. Rheumatology (Oxford) 45: 1303-6. doi: 10.1093/rheumatology/kel258
Yamazaki H, Fujiwara K, Shinohara S, Kikuchi M, Kanazawa Y, Kurihara R, Kishimoto I, Naito Y (2012) Reversible cochlear disorders with normal vestibular functions in three cases with Wegener’s granulomatosis. Auris Nasus Larynx 39: 236-40. doi: 10.1016/j.anl.2011.03.010
Yildirim N, Arslanoglu A, Aygun N (2008) Otologic and leptomeningeal involvements as presenting features in seronegative Wegener granulomatosis. Am J Otolaryngol 29: 147-9. doi: 10.1016/j.amjoto.2007.04.007
Yoon TH, Paparella MM, Schachern PA (1989) Systemic vasculitis: a temporal bone histopathologic study. Laryngoscope 99: 600-9. doi: 10.1288/00005537-198906000-00007
![]()