By Marcello Cherchi, MD PhD
For patients
Ménière’s disease is a progressive degenerative disease of the labyrinth. Its cause remains unknown. The disease presents with recurrent episodes of vertigo accompanied by ear symptoms (usually just on one side) of abrupt hearing loss, ear fullness and tinnitus. These episodes can last hours and then spontaneously resolve. The time pattern is often erratic. The diagnosis is based partly on the clinical history, and partly on test results (usually an audiogram, electrocochleogram, and tests of vestibular function). Management includes prophylactic therapies (intended to prevent the episodes) and abortive therapies (treatments for individual episodes when they occur).
For clinicians
Overview
Ménière’s disease is a progressive degenerative disease of the labyrinth whose underlying cause is unknown. The disease presents with a constellation of symptoms consisting of variably frequent episodes of (usually unilateral) aural fullness, hearing loss and tinnitus accompanying or preceding (usually spinning) disequilibrium (often with) nausea and vomiting that can last for several hours, after which the symptom cluster resolves. Patients return to baseline in between episodes, but over time there is cumulative hearing loss in the affected ear. A minority of patients progress to bilateral involvement. In late stages, patients may develop drop attacks, termed “otolithic catastrophes of Tumarkin.” Acute management of individual episodes with vestibular suppressants and anti-emetics is reasonable. Early in the disease course, prophylaxis (to reduce the frequency of the episodes) is sometimes achievable with daily oral medications (such as a loop diuretic, verapamil or betahistine), but in later stages of the disease, invasive interventions (by an otolaryngologist) may be required, such as a transtympanic injection of dexamethasone or gentamicin.
Introduction
Prosper Ménière originally described the constellation of symptoms in 1861 (Méniere 1861) as follows:
- An auditory organ, otherwise perfectly healthy, may suddenly become the site of a functional disturbance consisting of variable tones, continuous or intermittent, and these noises are soon accompanied by a greater or lesser diminution of hearing.
- These functional disturbances take place in the internal ear, can cause symptoms [previously] imputed to the brain, such as vertigo, lightheadedness, uncertainty of gait, spins and drops, also accompanied by nausea, vomiting and fainting.
- These events, which are intermittent, are soon followed by progressive hearing loss, and often hearing is completely abolished.
- This all leads one to believe that the main lesion of these episodes localizes to the semicircular canals.
In other words, he listed the cardinal symptoms of (1) episodic vertigo, (2) episodic or chronic tinnitus, and (3) episodic worsening of hearing (with an overall trajectory of decline between episodes). He also explicitly articulated a relationship between the symptoms that practitioners had previously “imputed to the brain” (vertigo, impaired gait, nausea, vomiting, fainting) and the aural symptoms (tinnitus and hearing loss), which is what led him to hypothesize that the disease “resides” in the ear — an idea which was novel at the time.
Ménière’s original description has basically been carried forward to the current criteria articulated by the American Academy of Otolaryngology in 1995 (American Academy of Otolaryngology-Head and Neck Foundation 1995), and the criteria articulated by the Bárány Society (Lopez-Escamez, Carey et al. 2015), which are (1) recurrent, spontaneous episodes of vertigo, (2) hearing loss in at least one ear, and (3) aural fullness, or tinnitus, or both, on the same side as the hearing loss.
Epidemiology
There is considerable controversy regarding the prevalence and incidence of Ménière’s disease.
Alexander and Harris (Alexander and Harris 2010) reviewed a range of literature on this topic (Cawthorne and Hewlett 1954; Celestino and Ralli 1991; Harris and Alexander 2010; Havia, Kentala, Pyykko 2005; Nakae et al. 1980; Radtke et al. 2008; Shojaku et al. 2005; Stahle, Stahle, Arenberg 1978; Tokumasu et al. 1982; Watanabe et al. 1995; Wladislavosky-Waserman et al. 1984) and found that reported prevalence rates per 100,000 people ranged from 3.5 to 513 — in other words, more than two orders of magnitude.
Alexander and Harris point out several potential sources of variability, including the biases in different study methodologies (e.g., retrospective series, population-based cross-sectional studies). In addition to methodological differences, studies use different methods of data collection and use variably stringent diagnostic criteria.
To take an example of a study of both prevalence and incidence, Wladislavosky-Wasserman and colleagues (Wladislavosky-Waserman et al. 1984), reporting on patients seen at Mayo Clinic over a 30-year period, listed a prevalence of 218.2 and an annual incidence of 15.3 per 100,000. In other words, about 0.2% of the population (1 in 500 people) at any given time has Ménière’s disease (prevalence); and every year about 0.015% of the population (1 in 6,536 people) newly develops Ménière’s disease (incidence).
The age distribution of incident Ménière’s disease is a separate question. An epidemiologic study by Havia and colleagues (Havia and Kentala 2004) noted that the age of symptom onset ranged from 15 to 80 years, with a peak age of incidence in the fifth decade (age 40 – 50 years), in an approximately bell-curve-like distribution, as displayed in the Figure below.

Pathophysiology: General considerations
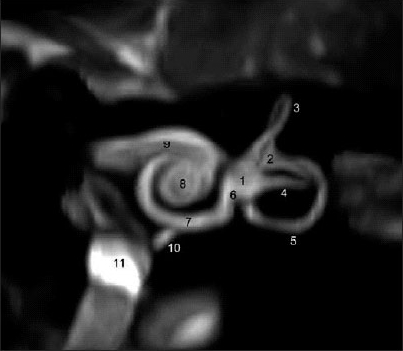
Despite a fair amount of research, the underlying cause of Ménière’s disease remains poorly understood. Early autopsy studies of patients with Ménière’s disease consistently described finding a dilated inner ear structure; this finding is called “hydrops.” This anatomical abnormality is sometimes detectable on high resolution MRI, as displayed in the Figure below, though to be clear, this type of imaging findings is not yet considered a standard diagnostic tool for this disease.
 |
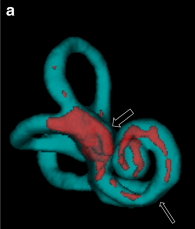 |
Rauch and colleagues (1989) reported that of 13 patients who in life had been confidently diagnosed with Ménière’s disease, at autopsy all 13 had “hydrops.” However, autopsies of the general population report that up to 6% of individuals (30 out of 500) have “hydrops,” whereas only around 0.2% of people (1 out of 500) have Ménière’s disease — a difference greater than one order of magnitude. Consequently, the state of “hydrops” is thought to be necessary but not sufficient for Ménière’s disease. In other words, something else (in addition to hydrops) must go wrong in order for Ménière’s disease to manifest clinically — but exactly what that “something else” is, remains unknown.
Numerous candidate etiologies have been suggested, but none is universally accepted. These candidates have included:
- Genetic
- Immunological disorders
- Autoimmune
- Allergic
- Infectious
- Viral
- Bacterial
- “Hydraulic”
- Over-production of endolymphatic fluid
- Under-absorption of endolymphatic fluid (e.g., obstruction of vestibular and cochlear aqueducts)
- Fluctuations of intracranial pressure
Regarding genetic causes, Eppsteiner et al. (Eppsteiner and Smith 2011) reported that of individuals affected by Ménière’s disease, 7% – 15% of report having a relative with the disease. Although such numbers are greater than what is likely by chance, it should be borne in mind that family members share more than just their genes; they also share environments and exposures. Genes encoding a range of proteins — including human leukocyte antigens, potassium channels, sodium channels, and others — have been proposed, but none has been widely accepted.
Although allergies and Ménière’s disease have frequently been noted to be comorbid (Banks, McGinness et al. 2012), so far there is little evidence that allergies cause Ménière’s disease (Weinreich and Agrawal 2014). Nevertheless, some researchers advocate allergy treatment (Derebery and Berliner 2010), at least in patients who otherwise have a clear history of allergies (such as to foods or environmental factors).
Although some patients with Ménière’s disease also have autoimmune diseases, the link between the two remains uncertain (Greco, Gallo et al. 2012), and so far there is no firm rationale for attempting to treat Ménière’s disease with immunomodulatory strategies.
Although DNA to herpes viruses has been reported in the vestibular ganglion in patients with Ménière’s disease (Gartner, Bossart et al. 2008), this finding does not appear to be higher than in the general population, so no causal link has been firmly established.
The rationale for proposing a “hydraulic” cause of Ménière’s disease is that hydrops must be due to either an excess production of fluid (in this case, endolymph), or an insufficient resorption of said fluid, or both. Histopathological studies of the labyrinth in Ménière’s disease show a variety of findings, including changes in the cells lining the endolymphatic duct (Michaels, Soucek et al. 2009), raising the question of whether alteration in the functions of these cells may contribute to the “fluid imbalance” of the labyrinth; this type of finding is often viewed by otolaryngologists as a rationale for attempting a procedure called endolymphatic sac decompression, whose putative goal would be to give any excessively produced endolymph another mechanism for escape (beyond the usual resorption).
Pathophysiology: Can Meniere’s disease result from noise exposure?
There are scattered case reports and case series in the literature of patients who appeared to develop Ménière’s disease after noise exposure, typically at a distance of some years from the acoustic insult (Paparella and Mancini 1983; Roitman et al. 1989; Ylikoski 1988). Ménière’s disease is relatively uncommon, whereas noise exposure is extraordinarily common, so this raises the question of whether the association between the two is simply one of chance. This question often comes up in medico-legal cases and disability claims.
Segal and colleagues (Segal et al. 2003) reviewed and critiqued the analytical flaws in the cases reported by Paparella and Mancini (Paparella and Mancini 1983), Roitman and colleagues (Roitman et al. 1989) and Ylikoski (Ylikoski 1988). They elected an epidemiologically-based approach to answer this question:
“We considered that the search for an association between NIHL [noise-induced hearing loss] and subsequent endolymphatic hydrops should be based on epidemiologic research. Our reasoning was based mainly on the small number of reported cases of MD [Ménière’s disease] compared with the relatively huge numbers of reported cases of NIHL. Only a significant difference between the incidence of MD among persons with NIHL, in comparison with the incidence seen in the general population, would provide definitive evidence one way or the other” (Segal et al. 2003).
They studied 17,245 military veterans who had all suffered noise exposure and among them identified 11 individuals who met clinical criteria for Ménière’s disease, amounting to a prevalence of 1.9 per 100,000 (which is actually less than the prevalence in the studies reviewed earlier). They concluded that:
“Our survey showed that the prevalence of MD among patients suffering from acute acoustic trauma or NIHL was 1.9:100000, comparable to that of the general population. We contend that the late appearance of MD in these patients is coincidental” (Segal et al. 2003).
Thus, in this large-scale epidemiologically-based study, “No support was found for the hypothesis that Ménière’s Disease may be causally related to previous acoustic trauma or noise-induced hearing loss” (Segal et al. 2003).
Natural history of Ménière’s disease
Although Ménière’s disease is not common, it is rare enough that it is difficult to establish what the “natural history” of the disease is — in other words, how the disease progresses over time if untreated. Specific questions include whether Ménière’s disease “burns out,” and how often it “spreads” from unilateral involvement to bilateral involvement.
For many decades practitioners were under the impression that Ménière’s disease eventually “burns out,” in the sense that patients eventually cease experiencing vertiginous episodes. However, more recent longitudinal research suggests that this is probably not true (Havia and Kentala 2004).
Most cases of Ménière’s disease begin unilaterally. Stahle and colleagues (Stahle, Friberg et al. 1991) followed a cohort of 167 patients, and found that only 3 (1.7%) presented with bilateral ear symptoms from the outset. Of the cases that begin unilaterally, there is controversy regarding what proportion of cases progresses to bilateral involvement. The cited figures are generally in the range of one-third to one-half of cases. The longitudinal study by Wladislavosky-Wasserman and colleagues (Wladislavosky-Waserman, Facer et al. 1984) reported 34% of patients eventually developed bilateral involvement. Havia and colleagues (Havia and Kentala 2004) reported that of their patients who were followed for at least 20 years, 43% developed bilateral ear symptoms. Stahle and colleagues (Stahle, Friberg et al. 1991) reported that of the 34 patients who were followed for at least 20 years, 47% developed bilateral ear symptoms.
Otolithic catastrophe of Tumarkin
Some patients, generally at more advanced stages of Ménière’s disease, begin experiencing abrupt “drop attacks” without any prodrome of aural symptoms. Such an attack is sometimes referred to as an “otolithic catastrophe of Tumarkin,” after the first investigator to describe them in 1936, Alexis Tumarkin (Tumarkin 1936). Patients who have reached this stage of symptoms generally can no longer control the vertiginous symptoms with medical therapy (in the sense of oral medications), and must move on to more invasive procedures for management, such as a transtympanic injection of gentamicin (Wu, Li et al. 2019).
Diagnostic testing for Ménière’s disease
When a patient experiences a first-ever episode of disequilibrium and unilateral ear symptoms (hearing loss, fullness, tinnitus), that clinical presentation in itself does not yet meet criteria for Ménière’s disease because the disequilibrium has not yet recurred — the presentation could be compatible with, for example, labyrinthitis. When a patient begins to have recurrent attacks, a diagnosis of Ménière’s disease becomes more likely. In an attempt to confirm the diagnosis, several diagnostic tests may be useful.
- Audiometry. This usually shows sensorineural hearing loss. This hearing loss more commonly begins by involving the lower frequencies. This hearing loss also tends to fluctuate, with an overall course (over years) of gradual decline.
-
Tests of unilateral vestibular function
(vestibular evoked myogenic potentials, caloric testing). These tests often, though not always, identify a vestibular weakness on the same side as the ear symptoms (Lee et al. 2024). There is emerging literature (Cheon et al. 2025; Mavrodiev et al. 2024; McGarvie et al. 2015; Rubin et al. 2018; Shugyo et al. 2020) regarding the observation that an ear affected by Ménière’s disease often exhibits a caloric weakness (which assesses the low frequency of the vestibular tuning spectrum), yet has normal gain on video head impulse testing (which assesses the high frequencies of the vestibular tuning spectrum).
- Electrocochleography. There is controversy in the literature regarding the diagnostic utility of this test (Eggermont 2017), which supposedly detects hydrops.
- MRI of the internal auditory canals without and with contrast. Although research studies (mentioned earlier) sometimes identify anatomical evidence of hydrops, this is not yet considered a standard test for Ménière’s disease. When a diagnosis of Ménière’s disease is being considered, the main role of this imaging study is to help exclude competing diagnosis, such as a vestibular schwannoma.
When the clinical history is compatible with Ménière’s disease, and audiometry shows fluctuating low frequency sensorineural hearing loss, and electrocochleography shows hydrops, and competing diagnoses have been excluded, then one can diagnose Ménière’s disease with a high degree of confidence.
Often the data do not turn out this neatly — in other words, the results do not all “line up” perfectly in support of Ménière’s disease. In this circumstance, it is sometimes worthwhile to repeat selected parts of the workup later. For example, since the hearing loss in Ménière’s disease fluctuates, if an initial audiogram shows normal hearing, this may be worth repeating later, on the possibility that you will “catch” the hearing loss.
Management of acute attacks.
The treatment of Ménière’s disease consists of both (1) management of acute episodes, and (2) prophylactic strategies.
Management of acute episodes usually consists of medications for disequilibrium and (if that fails) nausea/vomiting.
The medications for acute disequilibrium include antihistamines (meclizine, dimenhydrinate) or benzodiazepines (such as clonazepam or diazepam). For patients whose nausea/vomiting make it difficult to ingest medications, they may benefit from an orally disintegrating formulation (such as clonazepam) or suppository formulation (such as diazepam).
The medications for nausea/vomiting include ondansetron, promethazine and prochlorperazine. For patients whose nausea/vomiting make it difficult to ingest medication, they may benefit from either an orally disintegrating formulation (such as clonazepam) or suppository formulation (such as promethazine or prochlorperazine).
If a patient’s episodes are very infrequent, then they may be successful in simply treating each attack as it happens.
Longer-term (prophylactic) management, non-invasive
For many patients the frequency of the episodes is unacceptably high, and these patients may benefit from prophylactic strategies. The goal of prophylaxis is to decrease the frequency of the episodes. Note that even when a patient is using appropriate prophylactic strategies, this may not completely eliminate the episodes.
A common initial therapy is restriction of total daily dietary sodium intake to ≤2000 mg (Santos, Hall et al. 1993).
If dietary sodium restriction fails, a typical first-line medication is a diuretic, such as the combination of hydrochlorothiazide-triamterene. Results of studies on this have been mixed. For instance, van Deelen and colleagues (van Deelen and Huizing 1986) reported that hydrochlorothiazide/triamterene reduced the frequency of attacks. In contrast, a meta-analysis by Thirlwall and colleagues (Thirlwall and Kundu 2006) found insufficient evidence of efficacy for diuretics in management of Ménière’s.
Another medical approach involves calcium channel blockers — such as flunarizine (Fraysse, Bebear et al. 1991), nimodipine (Lassen, Hirsch et al. 1996) or verapamil (Kaya, Eraslan et al. 2019) — or drugs whose actions modulate both calcium channels and histamine channels — such as cinnarizine (Djelilovic-Vranic, Alajbegovic et al. 2012).
Betahistine is also commonly used oral medication in the prophylaxis for Ménière’s disease. This drug is not currently FDA approved in the US, but is still available through compounding pharmacies. Animal experiments suggest that some of betahistine’s catabolites (aminoethylpyridine and hydroxyethylpyridine) promote blood flow in the cochlea (Bertlich, Ihler et al. 2014). Even if this is also true in humans, it is unclear why this would help Ménière’s disease. A meta-analysis by James and colleagues (James and Burton 2001) concluded that there is insufficient evidence for efficacy of betahistine in treatment of Ménière’s disease.
If a patient has attempted reasonable trials of these approaches (dietary sodium restriction, diuretic, calcium channel blocker, betahistine) without success, then in the interest of avoiding invasive procedures (see below), some practitioners at this stage may provisionally change strategies and attempt treatment for migraine associated vertigo (MAV), on the rationale that there is significant clinical overlap between the disequilibrium caused by MAV and that caused by Ménière’s disease. If attempts at treating for migraine also fail, then they may return to the idea of Ménière’s disease, and advance to invasive therapies.
Longer-term (prophylactic) management, invasive
If reasonable attempts at non-invasive prophylactic treatments have failed, then patients are often referred to otolaryngology for further management.
Some otolaryngologists advocate the use of transtympanic injections of steroids such as dexamethasone (Garduno-Anaya, Couthino De Toledo et al. 2005). In favor of this approach is that dexamethasone should not have a destructive effect on the inner ear. Against this idea is the fact that it is difficult to see why this approach would be effective, and even if it provides benefit, that benefit should be transient since the steroids are catabolized and eliminated. In patients who seem to benefit from steroids, they often appear to require repeated steroid injections.
Other otolaryngologists are willing to consider a transtympanic injection of gentamicin, an aminoglycoside antibiotic that is preferentially vestibulotoxic. In favor of this approach is well-documented efficacy (Driscoll, Kasperbauer et al. 1997, Casani, Piaggi et al. 2012, Martin-Sanz, Diaz et al. 2018, Webster, Harrington-Benton et al. 2021, Wu, Zhou et al. 2021). Against this approach is its destructive nature, and otolaryngologists often voice understandable concern that there is the potential for further hearing loss, though most studies show the risk of additional hearing loss to be very low (Driscoll, Kasperbauer et al. 1997, Casani, Piaggi et al. 2012), and hearing preservation may be enhanced if gentamicin is administered concomitantly with dexamethasone (Bae, Lee et al. 2021).
Longer-term management, other
Patients often ask whether they should consider vestibular rehabilitation therapy (VRT) for Ménière’s disease. Earlier systematic reviews of this question (van Esch et al. 2017) concluded that there was insufficient evidence of any benefit. Some guidelines (Dubernard et al. 2026) suggest that insofar as a person with Ménière’s disease has any degree of vestibular weakness on the affected side, vestibular rehabilitation therapy may have some role. Clinicians should be sure to set expectations by advising the patient that vestibular rehabilitation therapy would not have any prophylactic efficacy against vertiginous episodes.
References
Alexander TH, Harris JP (2010) Current epidemiology of Meniere’s syndrome. Otolaryngol Clin North Am 43: 965-70. doi: 10.1016/j.otc.2010.05.001
American Academy of Otolaryngology-Head and Neck Foundation I (1995) Committee on Hearing and Equilibrium guidelines for the diagnosis and evaluation of therapy in Meniere’s disease. American Academy of Otolaryngology-Head and Neck Foundation, Inc. Otolaryngol Head Neck Surg 113: 181-5. doi: 10.1016/S0194-5998(95)70102-8
Bae S-H, Lee J-M, Lee H-J, Na G, Kim S-H (2021) Effect of Dexamethasone Combination with Gentamicin in Chemical Labyrinthectomy on Hearing Preservation and Vertigo Control in Patients with Unilateral Meniere’s Disease: A Randomized Controlled Clinical Trial. Journal of Clinical Medicine 10. doi: 10.3390/jcm10235581
Banks C, McGinness S, Harvey R, Sacks R (2012) Is allergy related to Meniere’s disease? Curr Allergy Asthma Rep 12: 255-60. doi: 10.1007/s11882-012-0258-3
Bertlich M, Ihler F, Sharaf K, Weiss BG, Strupp M, Canis M (2014) Betahistine metabolites, Aminoethylpyridine, and Hydroxyethylpyridine increase cochlear blood flow in guinea pigs in vivo. Int J Audiol 53: 753-9. doi: 10.3109/14992027.2014.917208
Bulakbasi N, Pabuscu Y (2007) Neuro-otologic applications of MRI. Diagn Interv Radiol 13: 109-20.
Casani AP, Piaggi P, Cerchiai N, Seccia V, Franceschini SS, Dallan I (2012) Intratympanic treatment of intractable unilateral Meniere disease: gentamicin or dexamethasone? A randomized controlled trial. Otolaryngol Head Neck Surg 146: 430-7. doi: 10.1177/0194599811429432
Cawthorne T, Hewlett AB (1954) Meniere’s disease. Proc R Soc Med 47: 663-70.
Celestino D, Ralli G (1991) Incidence of Meniere’s disease in Italy. Am J Otol 12: 135-8.
Cheon TU, Park JH, Lee JS, Bae SH (2025) Defining diagnostic thresholds for dissociation between caloric test and vHIT in Meniere’s disease. Front Neurol 16: 1651714. doi: 10.3389/fneur.2025.1651714
Derebery MJ, Berliner KI (2010) Allergy and its relation to Meniere’s disease. Otolaryngol Clin North Am 43: 1047-58. doi: 10.1016/j.otc.2010.05.004
Djelilovic-Vranic J, Alajbegovic A, Tiric-Campara M, Volic A, Sarajlic Z, Osmanagic E, Todorovic L, Beslagic O (2012) Betahistine or Cinnarizine for treatment of Meniere’s disease. Med Arch 66: 396-8. doi: 10.5455/medarh.2012.66.396-398
Driscoll CL, Kasperbauer JL, Facer GW, Harner SG, Beatty CW (1997) Low-dose intratympanic gentamicin and the treatment of Meniere’s disease: preliminary results. Laryngoscope 107: 83-9. doi: 10.1097/00005537-199701000-00017
Dubernard X, Ortega Solis J, Fraysse MJ, Tronche S, Darrouzet V, Mom T (2026) Guidelines of the French Society of ENT (short version) on the role and modalities of vestibular rehabilitation in Meniere’s disease. Eur Ann Otorhinolaryngol Head Neck Dis 143: 50-55. doi: 10.1016/j.anorl.2025.12.001
Eggermont JJ (2017) Ups and Downs in 75 Years of Electrocochleography. Front Syst Neurosci 11: 2. doi: 10.3389/fnsys.2017.00002
Eppsteiner RW, Smith RJ (2011) Genetic disorders of the vestibular system. Curr Opin Otolaryngol Head Neck Surg 19: 397-402. doi: 10.1097/MOO.0b013e32834a9852
Fraysse B, Bebear JP, Dubreuil C, Berges C, Dauman R (1991) Betahistine dihydrochloride versus flunarizine. A double-blind study on recurrent vertigo with or without cochlear syndrome typical of Meniere’s disease. Acta Otolaryngol Suppl 490: 1-10.
Garduno-Anaya MA, Couthino De Toledo H, Hinojosa-Gonzalez R, Pane-Pianese C, Rios-Castaneda LC (2005) Dexamethasone inner ear perfusion by intratympanic injection in unilateral Meniere’s disease: a two-year prospective, placebo-controlled, double-blind, randomized trial. Otolaryngol Head Neck Surg 133: 285-94. doi: 10.1016/j.otohns.2005.05.010
Gartner M, Bossart W, Linder T (2008) Herpes virus and Meniere’s disease. ORL J Otorhinolaryngol Relat Spec 70: 28-31; discussion 31. doi: 10.1159/000111045
Greco A, Gallo A, Fusconi M, Marinelli C, Macri GF, de Vincentiis M (2012) Meniere’s disease might be an autoimmune condition? Autoimmun Rev 11: 731-8. doi: 10.1016/j.autrev.2012.01.004
Gurkov R, Berman A, Dietrich O, Flatz W, Jerin C, Krause E, Keeser D, Ertl-Wagner B (2015) MR volumetric assessment of endolymphatic hydrops. Eur Radiol 25: 585-95. doi: 10.1007/s00330-014-3414-4
Harris JP, Alexander TH (2010) Current-day prevalence of Meniere’s syndrome. Audiol Neurootol 15: 318-22. doi: 10.1159/000286213
Havia M, Kentala E (2004) Progression of symptoms of dizziness in Meniere’s disease. Arch Otolaryngol Head Neck Surg 130: 431-5. doi: 10.1001/archotol.130.4.431
Havia M, Kentala E, Pyykko I (2005) Prevalence of Meniere’s disease in general population of Southern Finland. Otolaryngol Head Neck Surg 133: 762-8. doi: 10.1016/j.otohns.2005.06.015
James AL, Burton MJ (2001) Betahistine for Meniere’s disease or syndrome. Cochrane Database Syst Rev: CD001873. doi: 10.1002/14651858.CD001873
Kaya I, Eraslan S, Tarhan C, Bilgen C, Kirazli T, Gokcay F, Karapolat H, Celebisoy N (2019) Can verapamil be effective in controlling vertigo and headache attacks in vestibular migraine accompanied with Meniere’s disease? A preliminary study. J Neurol 266: 62-64. doi: 10.1007/s00415-019-09309-w
Lassen LF, Hirsch BE, Kamerer DB (1996) Use of nimodipine in the medical treatment of Meniere’s disease: clinical experience. Am J Otol 17: 577-80.
Lee SU, Park E, Kim HJ, Choi JY, Kim JS (2024) Evolution of Vestibular Findings During and Between the Attacks of Meniere Disease: Update. Neurol Clin Pract 14: e200235. doi: 10.1212/CPJ.0000000000200235
Lopez-Escamez JA, Carey J, Chung WH, Goebel JA, Magnusson M, Mandala M, Newman-Toker DE, Strupp M, Suzuki M, Trabalzini F, Bisdorff A, Classification Committee of the Barany S, Japan Society for Equilibrium R, European Academy of O, Neurotology, Equilibrium Committee of the American Academy of O-H, Neck S, Korean Balance S (2015) Diagnostic criteria for Meniere’s disease. J Vestib Res 25: 1-7. doi: 10.3233/VES-150549
Martin-Sanz E, Diaz JY, Esteban-Sanchez J, Sanz-Fernandez R, Perez-Fernandez N (2018) Delayed Effect and Gain Restoration After Intratympanic Gentamicin for Meniere’s Disease. Otol Neurotol. doi: 10.1097/MAO.0000000000001973
Mavrodiev V, Strupp M, Vinck A-S, van de Berg R, Lehner L (2024) The dissociation between pathological caloric testing and a normal video head impulse test helps differentiate between Menière’s disease, vestibular migraine, and other vestibular disorders: a confirmatory study in a large cohort of 2,101 patients. Frontiers in Neurology 15.
McGarvie LA, Curthoys IS, MacDougall HG, Halmagyi GM (2015) What does the dissociation between the results of video head impulse versus caloric testing reveal about the vestibular dysfunction in Meniere’s disease? Acta Otolaryngol 135: 859-65. doi: 10.3109/00016489.2015.1015606
Méniere P (1861) Sur une forme de surdité grave dépendant d’une lésion de l’oreille interne. Bulletin de l’Adadémie Impériale de médecine 26: 241.
Michaels L, Soucek S, Linthicum F (2009) The intravestibular source of the vestibular aqueduct: Its structure and pathology in Meniere’s disease. Acta Otolaryngol 129: 592-601. doi: 10.1080/00016480802342416
Nakae K, Nitta H, Hattori Y, Maeda K, Watanabe I, Mizukoshi K, Komatsuzaki A (1980) [The Prevalence of Ménière’s Disease in Japan]. Practica Oto-Rhino-Laryngologica 73: 1023-1029. doi: 10.5631/jibirin.73.6special_1023
Paparella MM, Mancini F (1983) Trauma and Meniere’s syndrome. Laryngoscope 93: 1004-12. doi: 10.1288/00005537-198308000-00006
Radtke A, von Brevern M, Feldmann M, Lezius F, Ziese T, Lempert T, Neuhauser H (2008) Screening for Meniere’s disease in the general population – the needle in the haystack. Acta Otolaryngol 128: 272-6. doi: 10.1080/00016480701509933
Roitman R, Talmi YP, Finkelstein Y, Silver S, Sadov R, Zohar Y (1989) Acoustic trauma-induced Meniere’s syndrome. ORL J Otorhinolaryngol Relat Spec 51: 246-50. doi: 10.1159/000276066
Rubin F, Simon F, Verillaud B, Herman P, Kania R, Hautefort C (2018) Comparison of Video Head Impulse Test and Caloric Reflex Test in advanced unilateral definite Meniere’s disease. Eur Ann Otorhinolaryngol Head Neck Dis 135: 167-169. doi: 10.1016/j.anorl.2017.08.008
Santos PM, Hall RA, Snyder JM, Hughes LF, Dobie RA (1993) Diuretic and diet effect on Meniere’s disease evaluated by the 1985 Committee on Hearing and Equilibrium guidelines. Otolaryngol Head Neck Surg 109: 680-9. doi: 10.1177/019459989310900408
Segal S, Eviatar E, Berenholz L, Kessler A, Shlamkovitch N (2003) Is there a relation between acoustic trauma or noise-induced hearing loss and a subsequent appearance of Meniere’s Disease? An epidemiologic study of 17245 cases and a review of the literature. Otol Neurotol 24: 387-91. doi: 10.1097/00129492-200305000-00007
Shojaku H, Watanabe Y, Fujisaka M, Tsubota M, Kobayashi K, Yasumura S, Mizukoshi K (2005) Epidemiologic characteristics of definite Meniere’s disease in Japan. A long-term survey of Toyama and Niigata prefectures. ORL J Otorhinolaryngol Relat Spec 67: 305-9. doi: 10.1159/000089413
Shugyo M, Ito T, Shiozaki T, Nishikawa D, Ohyama H, Fujita H, Yamanaka T, Kitahara T (2020) Comparison of the video head impulse test results with caloric test in patients with Meniere’s disease and other vestibular disorders. Acta Otolaryngol 140: 728-735. doi: 10.1080/00016489.2020.1766700
Stahle J, Friberg U, Svedberg A (1991) Long-term progression of Meniere’s disease. Acta Otolaryngol Suppl 485: 78-83.
Stahle J, Stahle C, Arenberg IK (1978) Incidence of Meniere’s disease. Arch Otolaryngol 104: 99-102.
Thirlwall AS, Kundu S (2006) Diuretics for Meniere’s disease or syndrome. Cochrane Database Syst Rev: CD003599. doi: 10.1002/14651858.CD003599.pub2
Tokumasu K, Tashiro N, Goto K, Saito A, Kawano R, Hirose Y, Yoneda S (1982) [Incidence and prevalence of Meniere’s disease in Sagamihara city, kanagawa-ken]. Prac Otol (Kyoto) 75: 1165-73.
Tumarkin A (1936) The Otolithic Catastrophe: A New Syndrome. Br Med J 2: 175-7.
van Deelen GW, Huizing EH (1986) Use of a diuretic (Dyazide) in the treatment of Meniere’s disease. A double-blind cross-over placebo-controlled study. ORL J Otorhinolaryngol Relat Spec 48: 287-92.
van Esch BF, van der Scheer-Horst ES, van der Zaag-Loonen HJ, Bruintjes TD, van Benthem PP (2017) The Effect of Vestibular Rehabilitation in Patients with Meniere’s Disease. Otolaryngol Head Neck Surg 156: 426-434. doi: 10.1177/0194599816678386
Watanabe Y, Mizukoshi K, Shojaku H, Watanabe I, Hinoki M, Kitahara M (1995) Epidemiological and clinical characteristics of Meniere’s disease in Japan. Acta Otolaryngol Suppl 519: 206-10. doi: 10.3109/00016489509121906
Webster KE, Harrington-Benton NA, Judd O, Kaski D, Maarsingh OR, MacKeith S, Ray J, Van Vugt VA, Burton MJ (2021) Intratympanic aminoglycosides for Ménière’s disease. Cochrane Database of Systematic Reviews. doi: 10.1002/14651858.CD015246
Weinreich HM, Agrawal Y (2014) The link between allergy and Meniere’s disease. Curr Opin Otolaryngol Head Neck Surg 22: 227-30. doi: 10.1097/MOO.0000000000000041
Wladislavosky-Waserman P, Facer GW, Mokri B, Kurland LT (1984) Meniere’s disease: a 30-year epidemiologic and clinical study in Rochester, Mn, 1951-1980. Laryngoscope 94: 1098-102.
Wu Q, Li X, Sha Y, Dai C (2019) Clinical features and management of Meniere’s disease patients with drop attacks. Eur Arch Otorhinolaryngol 276: 665-672. doi: 10.1007/s00405-018-5260-5
Wu Y, Zhou Y, Yu J, Song Z, Dai C, Wang J (2021) Intratympanic gentamicin injection for refractory ménière’s disease (MD) has potential effect in preventing contralateral MD occurrence. Acta Oto-Laryngologica: 1-6. doi: 10.1080/00016489.2021.1915499
Ylikoski J (1988) Delayed endolymphatic hydrops syndrome after heavy exposure to impulse noise. Am J Otol 9: 282-5.
![]()