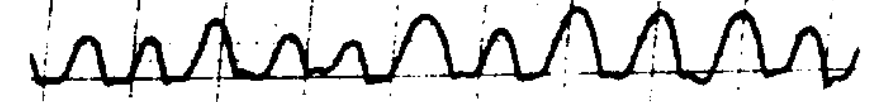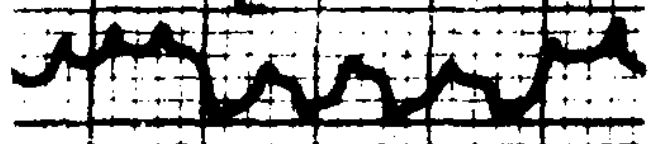By Marcello Cherchi, MD PhD
For patients
Congenital nystagmus (CN) is an eye movement abnormality that usually begins in infancy. It consists of an involuntary, somewhat rhythmic, oscillation of the eyes that tends to worsen when a person tries to focus (visually) on a target. The most common symptom of CN is poor vision. Since there are numerous causes of poor vision, your doctor may consider checking for other diseases, and may refer you to an ophthalmologist or neuro-ophthalmologist. The condition is lifelong. Some patients with CN seem to benefit from oral medications (gabapentin or memantine), though these have only been studied in small numbers of patients. Sometimes patients consider treatment with surgery of the muscles around the eyes.
For clinicians
Overview
In otoneurology the phrase, “congenital nystagmus” (CN), refers to an eye movement abnormality that becomes evident around 3 months of age, consists of involuntary cyclical jerk nystagmus or pendular nystagmus that is usually conjugate and usually predominantly horizontal, which worsens on attempted fixation, and may improve when the eyes are in a certain orientation within the orbits (called a “null point”). CN is rare. CN can occur in isolation, or in conjunction with other diseases (such as albinism), including ophthalmologic disease (such as achromatopsia). The most common presenting symptom is poor vision, which results from the fact that the involuntary eye movements interfere with foveation of a target. The underlying pathophysiologic mechanism of CN is unknown. A number of genetic mutations have been reported in association with CN. CN is usually evident on face-to-face examination, though accurate characterization requires instrumented oculographic testing. Some imaging studies report increased gray matter volume in certain temporo-occipital regions and parts of the cerebellum. Patients with CN can also have vestibular disease, and it is reasonable to screen for these. CN patients can also have unrelated ophthalmic disease, so evaluation by ophthalmology or neuro-ophthalmology is appropriate. Small case series report improvement of nystagmus with gabapentin or memantine. Surgery is sometimes attempted. CN is a lifelong condition.
Introduction
The unqualified phrase “congenital nystagmus” (CN) refers broadly to any abnormal pattern of eye movements identified at or shortly after birth. However, in otoneurology, the term is often used to refer more narrowly to a set of oscillatory eye movement disorders (recognized at birth or during infancy) of variable rhythmicity that result from a deficit of the visual fixation and smooth pursuit systems. Clinically what distinguishes these abnormal eye movements is that they worsen (become more pronounced) when the patient is attempting to fixate (Abadi and Dickinson 1986; Dell’Osso and Daroff 1975). This contrasts with most forms of nystagmus arising from peripheral vestibular disease, in which fixation tends to reduce or suppress the nystagmus.
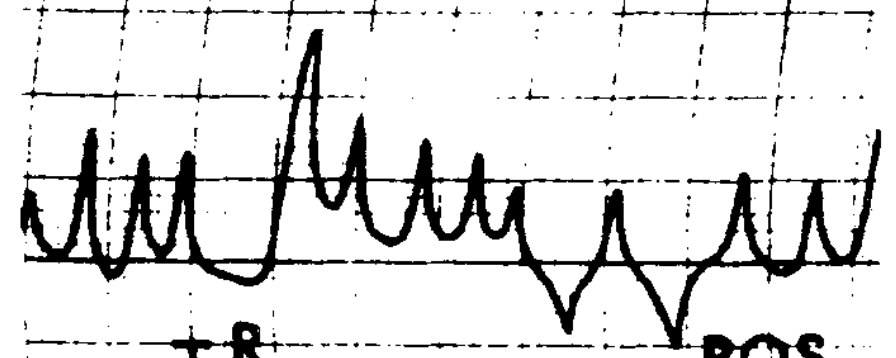
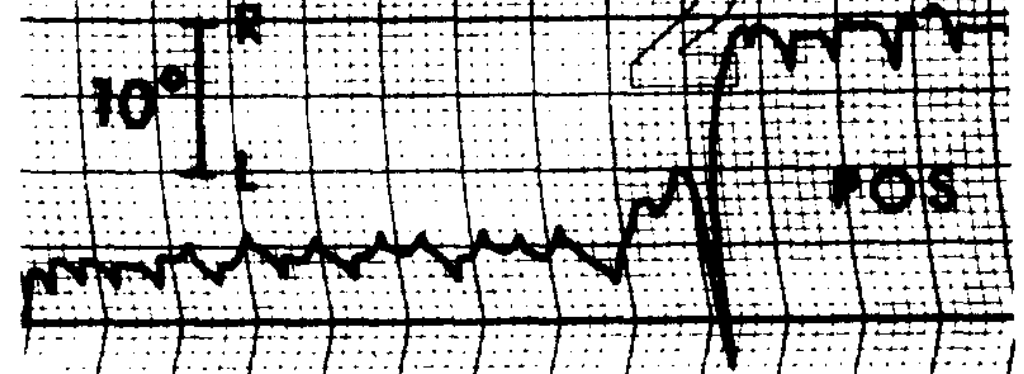
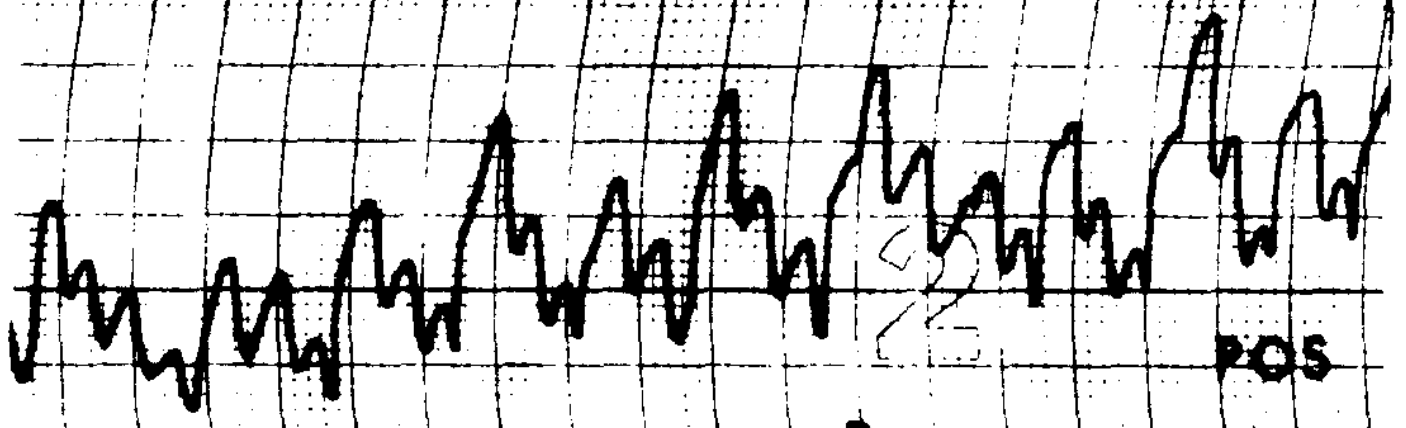
Congenital nystagmus can appear in a variety of forms, and is often difficult to characterize. Dell’Osso and Daroff (Dell’Osso and Daroff 1975) offer the following classification of congenital nystagmus. The tracings show horizontal eye movements.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
The classification described by Dell’Osso and Daroff (Dell’Osso and Daroff 1975) has been adopted by other investigators (Abadi and Dickinson 1986).
The nystagmus is nearly always conjugate, though monocular CN has been described (Kamel 1974). The eye movements are predominantly horizontal, though modest torsional (Abadi and Dickinson 1986; Averbuch-Heller et al. 2002) or vertical (Gresty et al. 1984) components may also be present.
Some forms of CN can be modulated by vergence (Abadi and Dickinson 1986) — typically the spontaneous nystagmus improves with convergence.
Epidemiology
Huang and colleagues (Huang et al. 2018) identified 50 patients with congenital nystagmus out of a clinic population of 32,050 patients who had presented for, “vertigo/dizziness, tinnitus, hearing loss or pathological eye movements,” amounting to 0.16% of that population, and they described this as comparable to the prevalence rate of 1 in 1000 – 1500, citing Self and Lotery (Self and Lotery 2006). In their 50 patients (27 male, 23 female), the mean age at presentation was 36 years, with a range of 3 to 76 years.
Genetics
A range of genetic defects has been reported in association with CN (Arshad et al. 2023; Du et al. 2011; Gupta et al. 2015; Gutmann et al. 1991; He et al. 2008; Hoffmann et al. 2004; Hu et al. 2011; Hu et al. 2012; Kerrison et al. 1996; Kerrison et al. 1998; Kim et al. 2016; Kohmoto et al. 2015; Li et al. 2020; Liu et al. 2007; Patton et al. 1993; Pearce 1978; Peng et al. 2009; Schorderet et al. 2007; Self and Lotery 2006, 2007; Wang et al. 2021; Xiu et al. 2018). The mechanism by which these mutations cause or contribute to CN is unknown.
Pathophysiological mechanism of disease
The underlying pathophysiological mechanism of CN is unknown, though Optican and Zee (Optican and Zee 1984) have proposed a model based on control systems analysis.
Clinical presentation
“The commonest symptom of congenital nystagmus is reduced visual acuity which can range from mild to severe” (Gresty et al. 1984), but this symptom in isolation is not a common reason for referral to otoneurology.
Common reasons for CN patients to be referred to otoneurology include observation (by a family member or another clinician) of abnormal eye movements. It is uncommon for CN patients to be referred for the symptom of disequilibrium (“dizziness,” “vertigo”), but insofar as poor vision can interfere with equilibrium, this is sometimes the basis for the referral.
CN may be apparent at birth, but typically becomes evident around the age of 3 months when infants begin visually to fixate (Huang et al. 2018), and it is a lifelong condition.
It has been well documented that CN reduces visual acuity (Simmers et al. 1999), and in fact, “For all cases of idiopathic congenital nystagmus visual acuity is reduced” (Abadi and Sandikcioglu 1975). This is because, “Normal visual acuity requires a stationary retinal image on the fovea. If fixation instabilities cause movement of the retinal image across the fovea for a few degrees, visual acuity is diminished” (Dorn et al. 2005).
Since untreated congenital nystagmus is constant and lifelong, some investigators have suggested that the chronic interference with foveation may additionally introduce a component of amblyopia, further interfering with vision (Bedell 2006).
The intrusive involuntary eye movements make it difficult to execute visual tasks requiring precise control of eye movements, such as reading (Kudo et al. 1995).
Despite the spontaneous oscillatory nature of eye movements in CN, oscillopsia is not a common complaint. Gresty and colleagues comment that, “When viewing a stationary target under normal illumination oscillopsia is generally not a symptom of congenital nystagmus regardless of eye position” (Gresty et al. 1984). However, patients whose CN manifests as periodic alternating nystagmus (PAN) may intermittently complain of oscillopsia (Abadi and Dickinson 1986).
Physical examination
In a patient with CN who is otherwise healthy, aside from the ocular motor abnormalities and possible head tilt (see below), the remainder of the physical examination should be normal.
However, note that CN is often comorbid with other pathologies (such as albinism) and other ophthalmic diseases (such as achromatopsia).
Ocular motor examination
CN can often be identified on face-to-face examination.
The Video below, from https://www.youtube.com/watch?v=MJgiptV_l3c (accessed 4/2/23), shows a face-to-face examination of a patient with CN.
CN often has a “null point,” meaning a position of the eyes within the orbits at which the nystagmus waveform is smallest. “If the null point is eccentric this leads to a head turn in the opposite direction” (Gresty et al. 1984), which is why many CN patients assume some type of head tilt or rotation when looking “straight ahead.”
Testing: oculographic
Detailed oculographic recordings are often helpful in characterizing CN and distinguishing it from alternative diagnoses.
CN patients often have impaired optokinetic responses, with decreased gain of optokinetic nystagmus and reduced or absent optokinetic afternystagmus (Yee et al. 1980). These findings are sensitive but not specific for CN.
Some studies of patients with CN report the vestibulo-ocular reflex (VOR) to be normal (Yee et al. 1981), while others report it to be consistently abnormal (Gresty et al. 1985). There have been very few studies of the high frequency end of the vestibular spectrum (Denia-Lafuente and Lombardero 2021).
Gresty and colleagues (Gresty et al. 1984) report that, “Abnormalities of responses to rotational testing occur in about 50% of asymptomatic patients with congenital nystagmus” (Gresty et al. 1984).
Curiously, CN patients have reduced motion aftereffects (Shallo-Hoffmann et al. 1998). Presumably this is because consistent foveation (which CN patients lack) is required to build the habituation that generates motion aftereffect illusions when the visual stimulus stops moving.
Imaging
Hufner and colleagues (Hufner et al. 2011) compared MRIs of 14 CN patients (7 males, mean age 28.3 ± 9.38 years) and 18 healthy controls (11 males, mean age 28.56 ± 8.58 years) using voxel-based morphometry. Compared to controls, the CN patients exhibited “significant volume increases in [the gray matter of] the human motion sensitive complex V5/MT+, the fusiform gyrus, and the middle occipital gyrus bilaterally,” and speculate that, “These volume increases may be associated with excess visual motion stimulation due to involuntary retinal slip of the visual scene.”

They further observed a “positive correlation (linear model) of nystagmus sway path with cerebellar GM [gray matter] volume… in the following areas: vermal parts VIII-X as well as hemisphere lobule II, hemisphere VI, crus I, crus II, and lobule VII-IX bilaterally,” and speculated that this, “might be related to the subjects’ attempt to maintain fixation, rather than be due to the generation of nystagmus.”

Differential diagnosis
CN is rare. It can occur in isolation, though is sometimes associated with other pathologies (such as albinism) and other ophthalmic diseases (such as achromatopsia).
It should be kept in mind that CN patients are just as vulnerable to common otovestibular diseases as the general population. Of the 50 CN patients studied by Huang and Hsu (Huang et al. 2018), they report that:
“Audiometry revealed normal mean hearing level (MHL) in 66 ears and abnormal MHL in 34 ears (34%). The caloric test showed normal responses in 75 ears, canal paresis in 6 ears and caloric areflexia in 19 ears, representing 25% (25/100) abnormality. The oVEMP test was performed in 8 patients (16 ears) and showed normal oVEMPs in 11 ears, reduced oVEMPs in 1 ear and absent oVEMP in 4 ears, accounting for 31% (5/16) abnormality. The cVEMP test was performed in 16 patients (32 ears) and identified normal, delayed and absent responses in 22, 2 and 8 ears, respectively, representing 31% (10/32) abnormality. In sum, approximately one-third of the congenital nystagmus patients (n = 17) had comorbid inner ear disorders including labyrinthine insufficiency 4, endolymphatic hydrops 4, sequela of otitis media 2, congenital deafness 2, acoustic trauma 2 and others 3. The remaining 33 patients, about two-thirds of the congenital nystagmus patient, lacked inner ear disorders” (Huang et al. 2018).
This emphasizes the point that screening for other otovestibular disorders may be worthwhile, as some may be treatable; for example, vestibular weakness may be managed with vestibular rehabilitation therapy.
Since poor vision is a common reason for referral, it should be kept in mind that the differential diagnosis of poor vision is very broad, and includes (non-otoneurological) primary ophthalmologic diseases as well. Holmstrom and colleagues (Holmstrom et al. 2014) studied 62 patients referred for “nystagmus,” and found that only 2 (3%) were ultimately diagnosed with CN; the remaining diagnoses included foveal hypoplasia, albinism, achromatopsia, rod-cone dystrophy and degenerative high myopia — all primary ophthalmological diseases. This emphasizes the point that even in a patient with CN, evaluation by ophthalmology or neuro-ophthalmology is important.
Treatment
In a retrospective review Sherry and colleagues (Shery et al. 2006) observed improvement in visual acuity and nystagmus in two patients with CN treated with gabapentin and memantine. Consequently, McLean and colleagues (McLean et al. 2007) performed a prospective randomized, double-blinded study of 48 CN patients treated with memantine (n=16), gabapentin (n=16) and placebo (n=15). They reported that at 56 days, outcomes in the treatment groups (memantine and gabapentin) were similar to each other, and were statistically significantly superior to placebo in terms of improvement in visual acuity, nystagmus intensity and foveation.
Periodic alternating nystagmus (PAN) can be acquired, in some cases appearing after cerebellar lesions, and in some such cases the nystagmus may improve with baclofen (Halmagyi et al. 1980). Case reports (Solomon et al. 2002) and case series (Comer et al. 2006) have also observed improvement in patients with congenital periodic alternating nystagmus (cPAN) treated with baclofen.
A variety of non-pharmacologic approaches have been attempted in CN, including contact lenses (Allen and Davies 1983), acupuncture (Blekher et al. 1998), biofeedback (Mezawa et al. 1990), auditory feedback (Sharma et al. 2000), extraocular muscle injection with onabotulinum toxin (Carruthers 1995) and cannabis (Pradeep et al. 2008).
A variety of surgical approaches to CN has been proposed (Bagheri et al. 2008; Dell’Osso and Flynn 1979; Erbagci et al. 2004; Flynn and Dell’Osso 1979; Nelson et al. 1985; Spielmann 2000; Taylor and Jesse 1987; Wagdy et al. 2017; Whittle 1999).
Prognosis
CN is a lifelong condition.
References
Abadi RV, Dickinson CM (1986) Waveform characteristics in congenital nystagmus. Doc Ophthalmol 64: 153-67. doi: 10.1007/BF00159990
Abadi RV, Sandikcioglu M (1975) Visual resolution in congenital pendular nystagmus. Am J Optom Physiol Opt 52: 573-81. doi: 10.1097/00006324-197509000-00001
Allen ED, Davies PD (1983) Role of contact lenses in the management of congenital nystagmus. Br J Ophthalmol 67: 834-6. doi: 10.1136/bjo.67.12.834
Arshad MW, Shabbir MI, Asif S, Shahzad M, Leydier L, Rai SK (2023) FRMD7 Gene Alterations in a Pakistani Family Associated with Congenital Idiopathic Nystagmus. Genes (Basel) 14. doi: 10.3390/genes14020346
Averbuch-Heller L, Dell’Osso LF, Leigh RJ, Jacobs JB, Stahl JS (2002) The torsional component of “horizontal” congenital nystagmus. J Neuroophthalmol 22: 22-32. doi: 10.1097/00041327-200203000-00007
Bagheri A, Ale-Taha M, Abrishami M, Salour H (2008) Effect of horizontal rectus surgery on clinical and paraclinical indices in congenital nystagmus. J Ophthalmic Vis Res 3: 6-15.
Bedell HE (2006) Visual and perceptual consequences of congenital nystagmus. Semin Ophthalmol 21: 91-5. doi: 10.1080/08820530600614181
Blekher T, Yamada T, Yee RD, Abel LA (1998) Effects of acupuncture on foveation characteristics in congenital nystagmus. Br J Ophthalmol 82: 115-20. doi: 10.1136/bjo.82.2.115
Carruthers J (1995) The treatment of congenital nystagmus with Botox. J Pediatr Ophthalmol Strabismus 32: 306-8. doi: 10.3928/0191-3913-19950901-09
Comer RM, Dawson EL, Lee JP (2006) Baclofen for patients with congenital periodic alternating nystagmus. Strabismus 14: 205-9. doi: 10.1080/09273970601026045
Dell’Osso LF, Daroff RB (1975) Congenital nystagmus waveforms and foveation strategy. Doc Ophthalmol 39: 155-82. doi: 10.1007/BF00578761
Dell’Osso LF, Flynn JT (1979) Congenital nystagmus surgery. A quantitative evaluation of the effects. Arch Ophthalmol 97: 462-9. doi: 10.1001/archopht.1979.01020010212004
Denia-Lafuente A, Lombardero B (2021) Vestibular Function Measured Using the Video Head Impulse Test in Congenital Nystagmus and Vertigo: A Case Report. Front Neurol 12: 690402. doi: 10.3389/fneur.2021.690402
Dorn L, Curkovic T, Dorn V (2005) Foveation period and waveforms of congenital ocular nystagmus. Coll Antropol 29 Suppl 1: 99-101.
Du W, Bu J, Dong J, Jia Y, Li J, Liang C, Si S, Wang L (2011) A novel frame-shift mutation in FRMD7 causes X-linked idiopathic congenital nystagmus in a Chinese family. Mol Vis 17: 2765-8.
Erbagci I, Gungor K, Bekir NA (2004) Effectiveness of retroequatorial recession surgery in congenital nystagmus. Strabismus 12: 35-40. doi: 10.1076/stra.12.1.35.29007
Flynn JT, Dell’Osso LF (1979) The effects of congenital nystagmus surgery. Ophthalmology 86: 1414-27. doi: 10.1016/s0161-6420(79)35379-9
Gresty M, Page N, Barratt H (1984) The differential diagnosis of congenital nystagmus. J Neurol Neurosurg Psychiatry 47: 936-42. doi: 10.1136/jnnp.47.9.936
Gresty MA, Barratt HJ, Page NG, Ell JJ (1985) Assessment of vestibulo-ocular reflexes in congenital nystagmus. Ann Neurol 17: 129-36. doi: 10.1002/ana.410170205
Gupta S, Pathak E, Chaudhry VN, Chaudhry P, Mishra R, Chandra A, Mukherjee A, Mutsuddi M (2015) A novel mutation in FRMD7 causes X-linked idiopathic congenital nystagmus in a North Indian family. Neurosci Lett 597: 170-5. doi: 10.1016/j.neulet.2015.04.037
Gutmann DH, Brooks ML, Emanuel BS, McDonald-McGinn DM, Zackai EH (1991) Congenital nystagmus in a (46,XX/45,X) mosaic woman from a family with X-linked congenital nystagmus. Am J Med Genet 39: 167-9. doi: 10.1002/ajmg.1320390210
Halmagyi GM, Rudge P, Gresty MA, Leigh RJ, Zee DS (1980) Treatment of periodic alternating nystagmus. Ann Neurol 8: 609-11. doi: 10.1002/ana.410080611
He X, Gu F, Wang Z, Wang C, Tong Y, Wang Y, Yang J, Liu W, Zhang M, Ma X (2008) A novel frameshift mutation in FRMD7 causing X-linked idiopathic congenital nystagmus. Genet Test 12: 607-13. doi: 10.1089/gte.2008.0070
Hoffmann S, Becker A, Hoerle S, Metz A, Oertel WH, Sommer N, Hemmer B (2004) Autosomal dominant congenital nystagmus is not linked to 6p12, 7p11, and 15q11 in a German family. Am J Ophthalmol 138: 439-43. doi: 10.1016/j.ajo.2004.04.065
Holmstrom G, Bondeson ML, Eriksson U, Akerblom H, Larsson E (2014) ‘Congenital’ nystagmus may hide various ophthalmic diagnoses. Acta Ophthalmol 92: 412-6. doi: 10.1111/aos.12250
Hu J, Liang D, Xue J, Liu J, Wu L (2011) A novel GPR143 splicing mutation in a Chinese family with X-linked congenital nystagmus. Mol Vis 17: 715-22.
Hu Y, Shen J, Zhang S, Yang T, Huang S, Yuan H (2012) A novel splicing mutation of the FRMD7 gene in a Chinese family with X-linked congenital nystagmus. Mol Vis 18: 87-91.
Huang YC, Hsu SY, Young YH (2018) Comorbid inner ear disorders in 50 patients with congenital nystagmus. Clin Otolaryngol 43: 1171-1177. doi: 10.1111/coa.13127
Hufner K, Stephan T, Flanagin VL, Deutschlander A, Dera T, Karch C, Linn J, Glasauer S, Dieterich M, Strupp M, Brandt T (2011) Cerebellar and visual gray matter brain volume increases in congenital nystagmus. Front Neurol 2: 60. doi: 10.3389/fneur.2011.00060
Kamel KS (1974) Congenital nystagmus. Report of a rare case of uniocular congenital nystagmus. Bull Ophthalmol Soc Egypt 67: 349-52.
Kerrison JB, Arnould VJ, Barmada MM, Koenekoop RK, Schmeckpeper BJ, Maumenee IH (1996) A gene for autosomal dominant congenital nystagmus localizes to 6p12. Genomics 33: 523-6. doi: 10.1006/geno.1996.0229
Kerrison JB, Koenekoop RK, Arnould VJ, Zee DS, Maumenee IH (1998) Clinical features of autosomal dominant congenital nystagmus linked to chromosome 6p12. Am J Ophthalmol 125: 64-70. doi: PMID: 9437315
Kim US, Cho E, Kim HJ (2016) A novel nonsense mutation of GPR143 gene in a Korean kindred with X-linked congenital nystagmus. Int J Ophthalmol 9: 1367-70. doi: 10.18240/ijo.2016.09.25
Kohmoto T, Okamoto N, Satomura S, Naruto T, Komori T, Hashimoto T, Imoto I (2015) A FRMD7 variant in a Japanese family causes congenital nystagmus. Hum Genome Var 2: 15002. doi: 10.1038/hgv.2015.2
Kudo Y, Kaga K, Ito K, Nakamura M (1995) Ocular movement during reading in patients with congenital nystagmus. Acta Otolaryngol Suppl 520 Pt 2: 282-4. doi: 10.3109/00016489509125249
Li Y, Jiang L, Wang L, Wang C, Liu C, Guo A, Liu M, Zhang L, Ma C, Zhang X, Gao S, Liu JY (2020) p.His16Arg of STXBP1 (MUNC18-1) Associated With Syntaxin 3B Causes Autosomal Dominant Congenital Nystagmus. Front Cell Dev Biol 8: 591781. doi: 10.3389/fcell.2020.591781
Liu JY, Ren X, Yang X, Guo T, Yao Q, Li L, Dai X, Zhang M, Wang L, Liu M, Wang QK (2007) Identification of a novel GPR143 mutation in a large Chinese family with congenital nystagmus as the most prominent and consistent manifestation. J Hum Genet 52: 565-570. doi: 10.1007/s10038-007-0152-3
McLean R, Proudlock F, Thomas S, Degg C, Gottlob I (2007) Congenital nystagmus: randomized, controlled, double-masked trial of memantine/gabapentin. Ann Neurol 61: 130-8. doi: 10.1002/ana.21065
Mezawa M, Ishikawa S, Ukai K (1990) Changes in waveform of congenital nystagmus associated with biofeedback treatment. Br J Ophthalmol 74: 472-6. doi: 10.1136/bjo.74.8.472
Nelson LB, Wagner RS, Harley RD (1985) Congenital nystagmus surgery. Int Ophthalmol Clin 25: 133-8. doi: 10.1097/00004397-198502540-00012
Optican LM, Zee DS (1984) A hypothetical explanation of congenital nystagmus. Biol Cybern 50: 119-34. doi: 10.1007/BF00337159
Patton MA, Jeffery S, Lee N, Hogg C (1993) Congenital nystagmus cosegregating with a balanced 7;15 translocation. J Med Genet 30: 526-8. doi: 10.1136/jmg.30.6.526
Pearce WG (1978) Congenital nystagmus–genetic and environmental causes. Can J Ophthalmol 13: 1-9.
Peng Y, Meng Y, Wang Z, Qin M, Li X, Dian Y, Huang S (2009) A novel GPR143 duplication mutation in a Chinese family with X-linked congenital nystagmus. Mol Vis 15: 810-4.
Pradeep A, Thomas S, Roberts EO, Proudlock FA, Gottlob I (2008) Reduction of congenital nystagmus in a patient after smoking cannabis. Strabismus 16: 29-32. doi: 10.1080/09273970701821063
Schorderet DF, Tiab L, Gaillard MC, Lorenz B, Klainguti G, Kerrison JB, Traboulsi EI, Munier FL (2007) Novel mutations in FRMD7 in X-linked congenital nystagmus. Mutation in brief #963. Online. Hum Mutat 28: 525. doi: 10.1002/humu.9492
Self J, Lotery A (2006) The molecular genetics of congenital idiopathic nystagmus. Semin Ophthalmol 21: 87-90. doi: 10.1080/08820530600614017
Self J, Lotery A (2007) A review of the molecular genetics of congenital Idiopathic Nystagmus (CIN). Ophthalmic Genet 28: 187-91. doi: 10.1080/13816810701651233
Shallo-Hoffmann J, Wolsley CJ, Acheson JF, Bronstein AM (1998) Reduced duration of a visual motion aftereffect in congenital nystagmus. Doc Ophthalmol 95: 301-14. doi: 10.1023/a:1001860524648
Sharma P, Tandon R, Kumar S, Anand S (2000) Reduction of congenital nystagmus amplitude with auditory biofeedback. J AAPOS 4: 287-90. doi: 10.1067/mpa.2000.107900
Shery T, Proudlock FA, Sarvananthan N, McLean RJ, Gottlob I (2006) The effects of gabapentin and memantine in acquired and congenital nystagmus: a retrospective study. Br J Ophthalmol 90: 839-43. doi: 10.1136/bjo.2005.086322
Simmers AJ, Gray LS, Winn B (1999) The effect of abnormal fixational eye movements upon visual acuity in congenital nystagmus. Curr Eye Res 18: 194-202. doi: 10.1076/ceyr.18.3.194.5374
Solomon D, Shepard N, Mishra A (2002) Congenital periodic alternating nystagmus: response to baclofen. Ann N Y Acad Sci 956: 611-5.
Spielmann A (2000) Clinical rationale for manifest congenital nystagmus surgery. J AAPOS 4: 67-74. doi: 10.1067/mpa.2000.103433
Taylor JN, Jesse K (1987) Surgical management of congenital nystagmus. Aust N Z J Ophthalmol 15: 25-34. doi: 10.1111/j.1442-9071.1987.tb00302.x
Wagdy FM, Ismael ME, Sarhan AE (2017) Evaluation of the Role of Displacement Surgery in the Management of Congenital Nystagmus. Electron Physician 9: 3672-3677. doi: 10.19082/3672
Wang XF, Chen H, Huang PJ, Feng ZK, Hua ZQ, Feng X, Han F, Xu XT, Shen RJ, Li Y, Jin ZB, Yu HY (2021) Genotype-Phenotype Analysis and Mutation Spectrum in a Cohort of Chinese Patients With Congenital Nystagmus. Front Cell Dev Biol 9: 627295. doi: 10.3389/fcell.2021.627295
Whittle J (1999) The effectiveness of surgery for congenital nystagmus. Eye (Lond) 13 ( Pt 1): 1-2. doi: 10.1038/eye.1999.1
Xiu Y, Yao Y, Yang T, Pan M, Yang H, Fang W, Gu F, Zhao J, Zhu Y (2018) Identification of a novel idiopathic congenital nystagmus‑causing missense mutation, p.G296C, in the FRMD7 gene. Mol Med Rep 18: 2816-2822. doi: 10.3892/mmr.2018.9260
Yee RD, Baloh RW, Honrubia V (1980) Study of congenital nystagmus: optokinetic nystagmus. Br J Ophthalmol 64: 926-32. doi: 10.1136/bjo.64.12.926
Yee RD, Baloh RW, Honrubia V, Kim YS (1981) A study of congenital nystagmus: vestibular nystagmus. J Otolaryngol 10: 89-98.
![]()