By Marcello Cherchi, MD PhD
For patients
In Fahr disease (FD) calcium accumulates in parts of the brain. Patients with FD can have problems with movement and thinking. If your doctor suspects FD, they may check brain imaging and sometimes tests of balance function. There is no cure.
For clinicians
Overview
Fahr disease (FD) is a rare neurodegenerative disorder in which symmetrical calcification occurs in various brain regions, particularly the basal ganglia and cerebellum. Some cases are idiopathic, others occur in a familial pattern (autosomal dominant or recessive). Clinical presentation includes movement disorders, cognitive and psychiatric disorders and ataxia. Some patients exhibit imbalance but this has not been well studied from the otovestibular perspective. The intracranial calcifications can be identified on head CT. Treatment is supportive.
Introduction
In 1930 the German pathologist, Karl Theodor Fahr (Pirmasens, Germany 1877 – Hamburg, Germany 1945) published a case report of a patient suffering from what was eventually designated Fahr disease (FD) or syndrome (Fahr 1930-1931), though Manyam and colleagues (Manyam et al. 2001) credit Delacour (Delacour 1850) with having published the first description of calcification of the basal ganglia.
FD is a neurodegenerative disorder in which calcium deposits accumulate, usually symmetrically, in various areas of the brain, including the basal ganglia, striatum, internal capsule, cerebral cortex and subcortical regions, thalamus, hippocampus and cerebellum (usually the dentate nucleus).
Some cases of FD occur sporadically, and these are sometimes termed idiopathic basal ganglia calcification (IBGC).
Other cases of FD occur in familial patterns, and these are sometimes called primary familial brain calcification (PFBC).
Epidemiology
FD is rare. Age at symptom onset ranges from the third to the seventh decades (Ellie et al. 1989; Manyam et al. 2001).
Genetics
Several genes have been implicated in the familial form of FD, and exhibit autosomal dominant and autosomal recessive patterns.
Autosomal dominant variants:
- IBGC1 (OMIM 213600) is caused by a mutation in the solute carrier family 20 member 2 (SLC20A2) on chromosome 8p11.2 (OMIM 158378) (Wang et al. 2012).
- IBGC4 (OMIM 615007) is caused by a mutation in the platelet-derived growth factor receptor beta (PDGFRB) gene on chromosome 5q32 (OMIM 173410) (Nicolas et al. 2013).
- IBGC6 (OMIM 616413) is caused by a mutation in the xenotropic and polytropic retrovirus receptor 1 (XPR1) gene on chromosome 1q25.3 (OMIM 605237) (Legati et al. 2015).
- IBGC5 (OMIM 615483) is caused by a mutation in the platelet-derived growth factor subunit B (PDGFB) gene on chromosome 22q13.1 (OMIM 190040) (Keller et al. 2013; Keogh et al. 2015).
Autosomal recessive variants:
- IBGC7 (OMIM 618317) is caused by a mutation in the myogenesis regulating glycosidase (MYORG) gene on chromosome 9p13.3 (OMIM 618255) and is transmitted in an autosomal recessive pattern (Yao et al. 2018).
- IBGC8 (OMIM 618824) is caused by a mutation in the junctional adhesion molecule 2 (JAM2) gene on chromosome 21q21.3 (OMIM 606870) and is transmitted in an autosomal recessive pattern (Cen et al. 2020; Schottlaender et al. 2020).
Clinical presentation
In view of the diverse neuroanatomical areas that can be involved by FD it is unsurprising that the clinical presentation is variable.
Manyam and colleagues (Manyam et al. 2001) conducted a literature and registry review of patients with FD. Of 67 symptomatic patients they documented:
- 37 (55%) exhibited movement disorders
- 26 (39%) exhibited cognitive deficits
- 24 (36%) exhibited speech deficits
- 24 (36%) exhibited cerebellar dysfunction
- 21 (31%) exhibited psychiatric disorders
- 15 (22%) exhibited pyramidal disorders
- 12 (18%) exhibited gait disorders
- 11 (16%) complained of pain and other sensory disorders
- 9 (13%) exhibited genitourinary dysfunction
- 8 (12%) exhibited gastrointestinal dysfunction
- 6 (9%) exhibited seizures.
Of the 37 patients with movement disorders they documented:
- 21 (57%) had parkinsonism
- 7 (19%) had chorea
- 3 (8%) had tremor
- 3 (8%) had dystonia
- 2 (5%) had athetosis
- 1 (3%) had orofacial dyskinesias
There are several case reports of FD patients suffering from imbalance (Castello and Pallestrini ; Parasram et al. 2020; Salli et al. 2016; Wang et al. 2015).
Testing: vestibular
As of this writing there were no published reports of instrumented vestibular testing in FD patients.
Testing: other
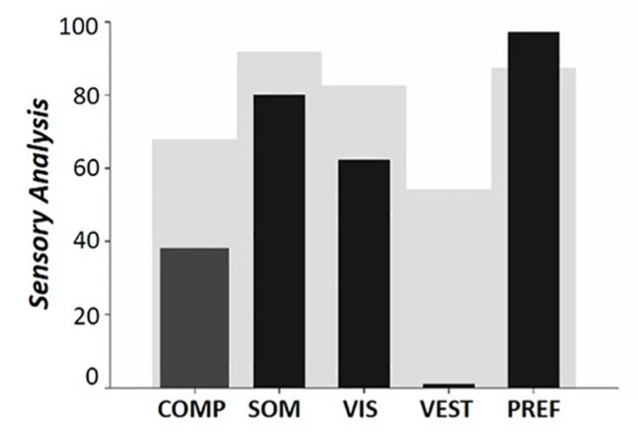
Scarano and colleagues studied computerized dynamic posturography in a 69 year old male with FD (Scarano et al. 2022). Sensory organization testing showed primarily a vestibular pattern, as shown in the Figure below. This pattern is also found in other disorders with basal ganglionic involvement (even though they are not primary otovestibular diseases).
Imaging
The calcifications in Fahr disease are revealed on CT scans.
The Figure below, from Scarano and colleagues (Scarano et al. 2022), shows approximately symmetrical calcification in the basal ganglia and cerebellum.
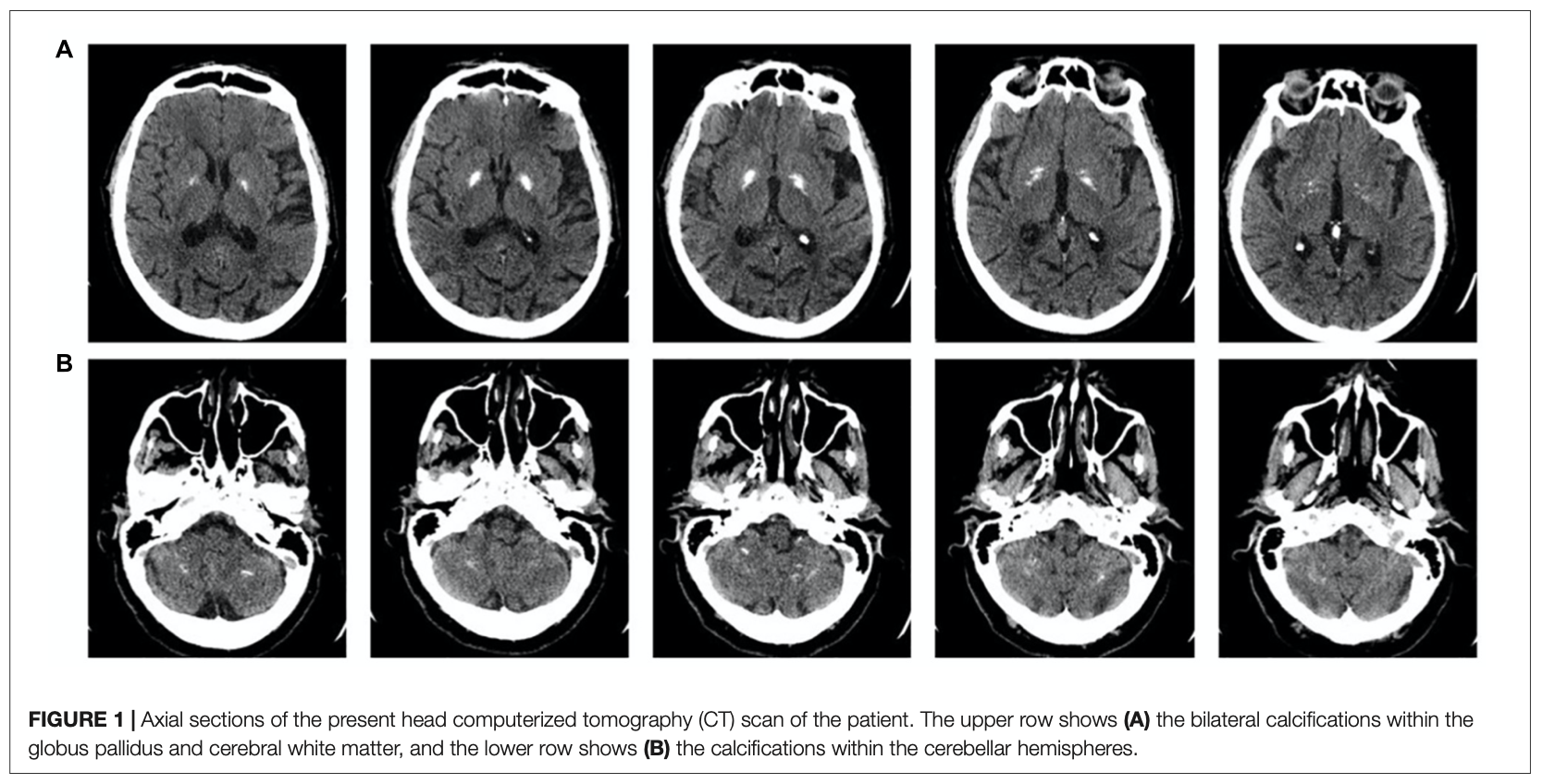
Note that abnormal calcium deposits, including in patterns compatible with FD, are found incidentally on anywhere from 0.3% (Koller et al. 1979) to 1.2% (Forstl et al. 1992) of head CTs of patients exhibiting no clinical features of FD.
Differential diagnosis
The differential diagnosis of FD includes basal ganglionic and cerebellar disorders.
Treatment and prognosis
There is no direct therapy for FD. Treatment is supportive.
References
Castello E, Pallestrini EA [Otoneurological manifestations in Fhar’s disease. A case report].
Cen Z, Chen Y, Chen S, Wang H, Yang D, Zhang H, Wu H, Wang L, Tang S, Ye J, Shen J, Wang H, Fu F, Chen X, Xie F, Liu P, Xu X, Cao J, Cai P, Pan Q, Li J, Yang W, Shan PF, Li Y, Liu JY, Zhang B, Luo W (2020) Biallelic loss-of-function mutations in JAM2 cause primary familial brain calcification. Brain 143: 491-502. doi: 10.1093/brain/awz392
Delacour A (1850) Ossification des capillaires du cerveau. Ann Med Psychol (Paris): 458-461.
Ellie E, Julien J, Ferrer X (1989) Familial idiopathic striopallidodentate calcifications. Neurology 39: 381-5. doi: 10.1212/wnl.39.3.381
Fahr KT (1930-1931) Idiopathische Verkalkung der Hirngefässe. Zentralblatt für allgemeine Pathologie und pathologische Anatomie 50: 129-133.
Forstl H, Krumm B, Eden S, Kohlmeyer K (1992) Neurological disorders in 166 patients with basal ganglia calcification: a statistical evaluation. J Neurol 239: 36-8. doi: 10.1007/BF00839209
Keller A, Westenberger A, Sobrido MJ, Garcia-Murias M, Domingo A, Sears RL, Lemos RR, Ordonez-Ugalde A, Nicolas G, da Cunha JE, Rushing EJ, Hugelshofer M, Wurnig MC, Kaech A, Reimann R, Lohmann K, Dobricic V, Carracedo A, Petrovic I, Miyasaki JM, Abakumova I, Mae MA, Raschperger E, Zatz M, Zschiedrich K, Klepper J, Spiteri E, Prieto JM, Navas I, Preuss M, Dering C, Jankovic M, Paucar M, Svenningsson P, Saliminejad K, Khorshid HR, Novakovic I, Aguzzi A, Boss A, Le Ber I, Defer G, Hannequin D, Kostic VS, Campion D, Geschwind DH, Coppola G, Betsholtz C, Klein C, Oliveira JR (2013) Mutations in the gene encoding PDGF-B cause brain calcifications in humans and mice. Nat Genet 45: 1077-82. doi: 10.1038/ng.2723
Keogh MJ, Pyle A, Daud D, Griffin H, Douroudis K, Eglon G, Miller J, Horvath R, Chinnery PF (2015) Clinical heterogeneity of primary familial brain calcification due to a novel mutation in PDGFB. Neurology 84: 1818-20. doi: 10.1212/WNL.0000000000001517
Koller WC, Cochran JW, Klawans HL (1979) Calcification of the basal ganglia: computerized tomography and clinical correlation. Neurology 29: 328-33. doi: 10.1212/wnl.29.3.328
Legati A, Giovannini D, Nicolas G, Lopez-Sanchez U, Quintans B, Oliveira JR, Sears RL, Ramos EM, Spiteri E, Sobrido MJ, Carracedo A, Castro-Fernandez C, Cubizolle S, Fogel BL, Goizet C, Jen JC, Kirdlarp S, Lang AE, Miedzybrodzka Z, Mitarnun W, Paucar M, Paulson H, Pariente J, Richard AC, Salins NS, Simpson SA, Striano P, Svenningsson P, Tison F, Unni VK, Vanakker O, Wessels MW, Wetchaphanphesat S, Yang M, Boller F, Campion D, Hannequin D, Sitbon M, Geschwind DH, Battini JL, Coppola G (2015) Mutations in XPR1 cause primary familial brain calcification associated with altered phosphate export. Nat Genet 47: 579-81. doi: 10.1038/ng.3289
Manyam BV, Walters AS, Narla KR (2001) Bilateral striopallidodentate calcinosis: clinical characteristics of patients seen in a registry. Mov Disord 16: 258-64. doi: 10.1002/mds.1049
Nicolas G, Pottier C, Maltete D, Coutant S, Rovelet-Lecrux A, Legallic S, Rousseau S, Vaschalde Y, Guyant-Marechal L, Augustin J, Martinaud O, Defebvre L, Krystkowiak P, Pariente J, Clanet M, Labauge P, Ayrignac X, Lefaucheur R, Le Ber I, Frebourg T, Hannequin D, Campion D (2013) Mutation of the PDGFRB gene as a cause of idiopathic basal ganglia calcification. Neurology 80: 181-7. doi: 10.1212/WNL.0b013e31827ccf34
Parasram M, Levi Chazen J, Sarva H (2020) Primary familial brain calcification. J Am Osteopath Assoc. doi: 10.7556/jaoa.2020.116
Salli M, D’Arienzo A, Bonanno M, Morello S, Sanfilippo A, Mauro GL, D’Arienzo M (2016) Femoral neck’s fracture in Fahr’s Syndrome: case report. Clin Cases Miner Bone Metab 13: 265-267. doi: 10.11138/ccmbm/2016.13.3.265
Scarano S, Rota V, Tesio L, Perucca L, Robecchi Majnardi A, Caronni A (2022) Balance Impairment in Fahr’s Disease: Mixed Signs of Parkinsonism and Cerebellar Disorder. A Case Study. Front Hum Neurosci 16: 832170. doi: 10.3389/fnhum.2022.832170
Schottlaender LV, Abeti R, Jaunmuktane Z, Macmillan C, Chelban V, O’Callaghan B, McKinley J, Maroofian R, Efthymiou S, Athanasiou-Fragkouli A, Forbes R, Soutar MPM, Livingston JH, Kalmar B, Swayne O, Hotton G, Group SS, Pittman A, Mendes de Oliveira JR, de Grandis M, Richard-Loendt A, Launchbury F, Althonayan J, McDonnell G, Carr A, Khan S, Beetz C, Bisgin A, Tug Bozdogan S, Begtrup A, Torti E, Greensmith L, Giunti P, Morrison PJ, Brandner S, Aurrand-Lions M, Houlden H (2020) Bi-allelic JAM2 Variants Lead to Early-Onset Recessive Primary Familial Brain Calcification. Am J Hum Genet 106: 412-421. doi: 10.1016/j.ajhg.2020.02.007
Wang C, Li Y, Shi L, Ren J, Patti M, Wang T, de Oliveira JR, Sobrido MJ, Quintans B, Baquero M, Cui X, Zhang XY, Wang L, Xu H, Wang J, Yao J, Dai X, Liu J, Zhang L, Ma H, Gao Y, Ma X, Feng S, Liu M, Wang QK, Forster IC, Zhang X, Liu JY (2012) Mutations in SLC20A2 link familial idiopathic basal ganglia calcification with phosphate homeostasis. Nat Genet 44: 254-6. doi: 10.1038/ng.1077
Wang H, Shao B, Wang L, Ye Q (2015) Fahr’s disease in two siblings in a family: A case report. Exp Ther Med 9: 1931-1933. doi: 10.3892/etm.2015.2356
Yao XP, Cheng X, Wang C, Zhao M, Guo XX, Su HZ, Lai LL, Zou XH, Chen XJ, Zhao Y, Dong EL, Lu YQ, Wu S, Li X, Fan G, Yu H, Xu J, Wang N, Xiong ZQ, Chen WJ (2018) Biallelic Mutations in MYORG Cause Autosomal Recessive Primary Familial Brain Calcification. Neuron 98: 1116-1123 e5. doi: 10.1016/j.neuron.2018.05.037
![]()