By Marcello Cherchi, MD PhD
For patients
The human immunodeficiency virus (HIV) can affect many aspects of a person’s health, including hearing and balance. When a patient infected with HIV develops symptoms such as hearing loss, tinnitus, or disequilibrium, the doctor may consider checking several tests of hearing and balance to make sure that no other diseases (besides HIV) are present. If no other causes are found, then the doctor may refer the patient to an audiologist for management of the ear symptoms (hearing loss, tinnitus) or a physical therapist (for disequilibrium).
For clinicians
Overview
The human immunodeficiency virus (HIV), which is responsible for acquired immunodeficiency syndrome (AIDS), compromises a patient’s immune system and increases the risk of opportunistic infections, as well as non-infectious problems. The presence of such HIV-associated medical problems often makes it difficult to study which symptoms and deficits are due to HIV itself. Nevertheless, it appears that HIV may be responsible increased rates of auditory symptoms (hearing loss, tinnitus) and vestibular symptoms (disequilibrium) in all age groups. In otoneurology the usual clinical scenario is that a patient already diagnosed with HIV is referred for evaluation of the auditory and vestibular symptoms. Workup may reveal audiologic abnormalities (hearing loss on audiometry and otoacoustic emissions; abnormalities on brainstem auditory evoked responses) and vestibular abnormalities (vestibular weakness on caloric testing). Screening for other (non-HIV-related) causes of these symptoms is reasonable. If other causes are excluded with a reasonable degree of confidence, then one may conclude that HIV itself is responsible for the auditory and vestibular symptoms. The auditory symptoms are usually managed by an audiologist. If vestibular symptoms are present, referral to vestibular rehabilitation therapy is reasonable.
Epidemiology
Auditory and vestibular symptoms appear to be more common in HIV+ patients than in healthy individuals. Fokouo and colleagues (Fokouo et al. 2015) studied 90 HIV+ patients and 90 healthy controls. They reported that, “The HIV-positive patients had more otologic symptoms (hearing loss, dizziness, tinnitus, and otalgia) than HIV-negative patients (41 vs. 13, p=0.04).”
Pathophysiological mechanism of disease
As we discuss below, most, though not all, studies of otoacoustic emissions in HIV+ patients find a greater rate of abnormalities compared to healthy controls (Maro et al. 2014; Niemczak et al. 2022; Soucek and Michaels 1996), suggesting cochlear (end-organ) dysfunction, as opposed to dysfunction of the auditory nerve or higher auditory processing. Furthermore, temporal bone studies (also discussed below) report evidence of HIV infection in the cochlea itself (Pappas et al. 1994). Taken together, these data suggest a direct effect of HIV infection on the peripheral auditory system.
There is also evidence (discussed below) of vestibular dysfunction on clinical testing, but no corresponding histopathological evidence from temporal bone studies.
Patients with HIV+ are at increased risk for other infectious and non-infectious diseases, some of which may also affect the ear. For example, Michaels and colleagues (Michaels et al. 1994) studied 49 temporal bones from 25 AIDS patients and found evidence of labyrinthine infection by cryptococcosis and cytomegalovirus, and Kaposi’s sarcoma in the vestibulocochlear nerve. This often makes it difficult to isolate the effects attributable to HIV alone.
Clinical presentation: audiologic
Hearing loss occurring in the context of HIV generally begins insidiously and progresses gradually, though abrupt-onset cases have been reported (Grimaldi et al. 1993).
Millar and colleagues (Millar et al. 2020) compared the case histories and pure tone audiometry in 60 HIV+ patients and 32 HIV‑ subjects. They found no statistically significant difference in auditory thresholds, but did find a significantly higher rate of tinnitus in the HIV+ patients compared to controls.
Soucek and Michaels (Soucek and Michaels 1996) studied 62 AIDS patients (mean age 39 years, length of HIV positivity ranging from 3 to 10 years), and reported that, “Ten [16%] patients, all of whom had sensorineural hearing loss, complained of tinnitus. This was bilateral in six patients and unilateral in four.”
Clinical presentation: vestibular
Soucek and Michaels (Soucek and Michaels 1996) studied 62 AIDS patients (mean age 39 years, length of HIV positivity ranging from 3 to 10 years), and reported that, “Sixteen [26%] patients had vertigo.”
Palacios and colleagues (Palacios et al. 2008) studied 23 HIV+ children (median age 4.5 years, range 5 months to 16 years) and reported that, “Four children (17.4%), all over 5 years old, referred to disequilibrium disorders when questioned.”
Testing: general comments
There are case reports (Palacios et al. 2008) in the literature of HIV+ patients presenting with auditory and vestibular symptoms, whose workups reveal various deficits in auditory tests and vestibular tests. While case reports are a good start, since HIV+ patients are vulnerable to a variety of other illnesses, such deficits are better explored through case series or cohort studies.
Testing: audiologic
Many studies report no statistically significant differences in air-conducted pure tone audiometry in HIV+ patients (Luque et al. 2014; Maro et al. 2014; Niemczak et al. 2022).
In contrast, Fokouo and colleagues (Fokouo et al. 2015) studied pure tone audiometry in 90 HIV+ patients (divided into three sub-groups of HAART-naïve patients, patients receiving first-line HAART, and patients receiving second-line HAART) versus 90 healthy control subjects, all age 15 – 49 years. They reported a statistically significantly higher odds ratio of hearing loss among all subgroups of HIV+ patients compared to controls.
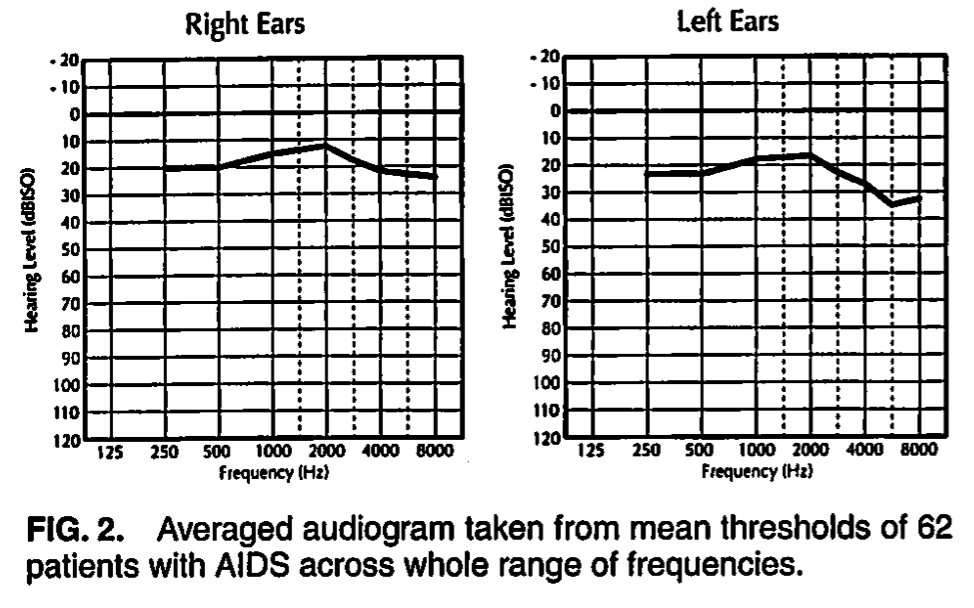
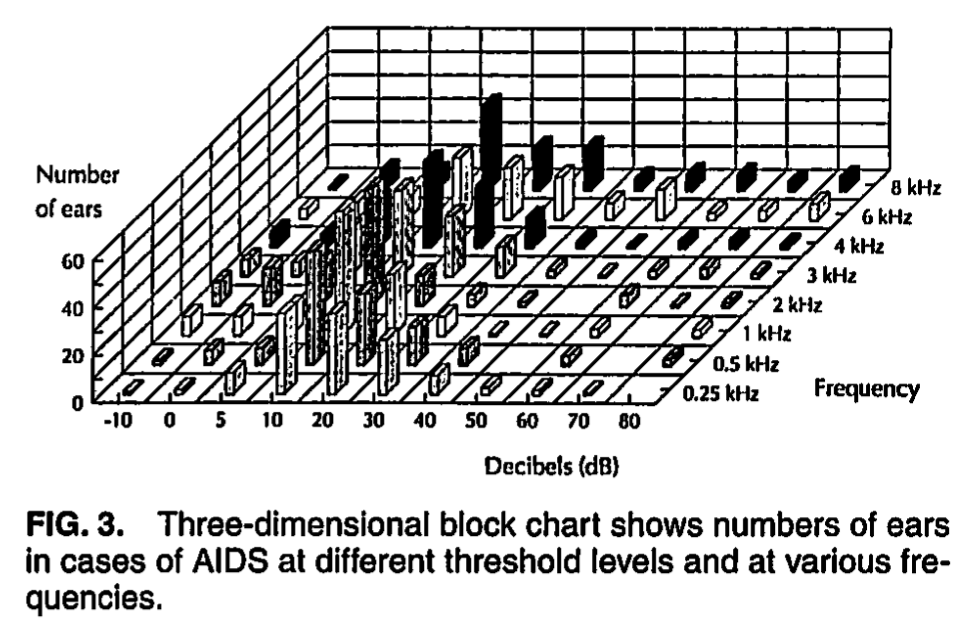
Similarly, Soucek and Michaels (Soucek and Michaels 1996) studied 62 adult AIDS patients (mean age 39 years, length of HIV positivity ranging from 3 to 10 years). The Figures below show a composite audiogram, and a 3-dimensional block chart of air-conducted hearing thresholds.
 |
 |
Soucek and Michaels interpreted the findings as follows:
“Figure 2 shows a composite audiogram taken from the average hearing thresholds of left and right ears of 62 patients with AIDS at 250, 500, 1000, 2000, 3000, 4000, 6000 and 8000 Hz. Referring to life tables, it can be seen that at the average age of 39 years for this group, only thresholds >20 dB can be considered abnormal. In the averaged levels of patients with AIDS this threshold is exceeded slightly in the lower and higher frequencies on the left side; on the right side there is a slight elevation at 6 and 8 kHz.
In Figure 3 the thresholds of all ears are presented as a three-dimensional block chart, giving the numbers of ears showing thresholds at the different frequencies and intensities of hearing loss. The chart illustrates that the largest single intensity group across the whole range of frequencies is in the region of 10 – 20 dB. This is normal at the mean age of 39 years of the patients studied clinically. There is a hearing impairment present (losses >20 dB) in many. This is mainly mild, but there are significant numbers with more severe losses. All frequencies are represented in the hearing losses, but, as the mean threshold audiograms also indicate (Figure 2), the higher and lower frequencies have greater numbers of ears with losses than the middle range. A survey of the individual audiograms showed that 39% of the patients tested had this type of audiogram, with notable losses at higher or lower frequencies but little or no loss at middle frequency range. Accepting a threshold of 20 dB or worse at any frequency as an indication of hearing loss in this group, the total number of subjects affected was 43 (69%)” (Palacios et al. 2008).
Soucek and Michaels (Soucek and Michaels 1996) additionally studied brainstem auditory evoked responses and found that:
“In 16 patients investigated by auditory brainstem evoked responses (ABR), five [31%] were abnormal. All patients tested had some degree of sensorineural hearing loss. In three of the five patients there was an abnormal I‑III interpeak interval suggesting a lower brainstem lesion. In one case, wave III on both sides was delayed. This patient had a cryptococcal meningitis. In the fifth patient, who had migraine and had had head injury in the past, there was an abnormal I‑V central conduction time” (Soucek and Michaels 1996).
Palacios and colleagues (Palacios et al. 2008) studied 23 HIV+ children (median age 4.5 years, range 5 months to 16 years) and reported that, “Pure tone audiometry was carried out in 12 children over 4 years of age: 4 (33%) showed hearing loss, 2 were conductive.”
Studies of otoacoustic emissions provide (OAEs) mixed results. Most studies report a greater rate of abnormalities in HIV+ patients compared to controls (Maro et al. 2014; Niemczak et al. 2022; Soucek and Michaels 1996). Some report no difference in OAEs between HIV+ and HIV‑ patients (Buckey et al. 2019).
Some studies find evidence of central auditory problems rather than peripheral auditory dysfunction. For instance, Buckey and colleagues (Buckey et al. 2019) studied 107 HIV+ individuals (age 37±11.5 years) and 72 HIV‑ individuals (age 27±17 years) using two tests of peripheral auditory function (pure tone audiometry and distortion product otoacoustic emissions) and one test of central auditory function (gaps-in-noise detection). The authors reported that compared to the HIV‑ subjects, in the HIV+ patients the peripheral tests were normal while the central test was abnormal. They concluded that, “The gap detection results from the study show effects on central processing in HIV+ individuals, supporting the origin of HIV-related hearing complaints in the central auditory system” rather than the peripheral auditory system.
The studies cited above mostly pertain to adult HIV+ patients. Data from pediatric HIV+ patients may suggest different, or additional mechanisms of auditory dysfunction. Maro and colleagues (Maro et al. 2016) studied 131 HIV+ patients and 113 HIV‑ subjects, all under age 18 years, and found that, “Children with HIV+ were more likely to have a history of ear drainage, and to have abnormal audiograms… [and] the HIV+ group… did have significantly lower DPOAE [distortion product otoacoustic emissions] magnitudes.” They concluded, “These data suggest that (1) HIV+ children often have middle ear damage which complicates understanding the direct effects of HIV on the hearing system, and (2) even when corrected for confounders DPOAEs were lower in the HIV+ group.” Palacios and colleagues (Palacios et al. 2008) similarly reported evidence of “conductive hearing loss associated with previous otitis media events” in a population of 23 HIV+ children (median age 4.5 years, range 5 months to 16 years).
Testing: vestibular
Palacios and colleagues (Palacios et al. 2008) studied 23 HIV+ children (median age 4.5 years, range 5 months to 16 years) and reported that, “ENG [electronystagmography] was carried out in 6 of 23 patients; alterations in smooth pursuit were detected in 1 [17%] patient of the 6 tested. Caloric tests were abnormal in the four [100%] patients tested, showing unilateral vestibular hyporeflexia. Rotatory tests were abnormal in the two [100%] patients for whom tests could be performed, with asymmetries in both vestibular and optovestibular tests.”
Karaca and colleagues (Karaca et al. 2022) studied video head impulse testing in 48 HIV+ patients and 24 healthy controls, and found no differences between the two groups. They commented that, “Because the video head impulse test is considered a high-frequency test of vestibulo-ocular reflex, it is possible that vestibular deficits of the human immunodeficiency virus can be confined to low frequencies.”
Millar and colleagues (Millar et al. 2021) studied video head impulse testing in 60 HIV+ patients and 32 healthy controls, and reported that, “The prevalence of peripheral vestibular impairment in the HIV-positive group was not significantly different from that of the HIV-negative group.”
Histopathology
There is some evidence that HIV can directly involve the cochlea. Pappas and colleagues (Pappas et al. 1994) studied 8 temporal bones and reported that:
“Extracellular viral-like particles with morphologic characteristics of HIV-1 were identified on the tectorial membrane in three cases. Numerous viral-like particles with cores appeared trapped within lacunae along the tectorial membrane in one specimen. Intracellular viral-like particles, appearing essentially similar to identified HIV-1 particles of infected lymphocyte cultures, were found within the cytoplasm of connective tissue cells. These same cells had gradients of enlarged viral envelopes forming cisterns that contained cores and varying amounts of cistern particulate. Some sectioned profiles of enlarged cytoplasmic cisterns revealed the attachment and inward protrusion of the core from the cistern wall. Other ultrastructural findings included cytoplasmic globular-dense particulate bodies, aggregates of smaller viral-like particles, and mitochondrial changes” (Pappas et al. 1994).
Differential diagnosis
As far as HIV is concerned, the usual clinical scenario in otoneurology is that a patient already diagnosed with HIV is referred for auditory or vestibular complaints. In this circumstance, as discussed below, it is appropriate to undertake an otovestibular workup to screen for other treatable diseases. If the workup is unrevealing, then by exclusion, it is reasonable to conclude that HIV itself is the mechanism of the symptoms in question.
The reverse scenario is extraordinarily rare — that is, a patient with auditory or vestibular complaints is found to have (previously undiagnosed) HIV. Auditory and vestibular symptoms in themselves do not constitute grounds to screen for HIV; rather, the decision to monitor for HIV would be driven by the presence of other risk factors, and the patient’s HIV status would usually be checked by their internist.
Treatment
Most of the studies that have compared audiologic findings in HIV+ patients who have received anti-retroviral therapies to those in untreated HIV+ patients found no statistically significant differences (Maro et al. 2016), suggesting that HIV infection itself (rather than the anti-retroviral therapies) is responsible. However, not all studies reach the same conclusion; for example, Maro and colleagues (Maro et al. 2014) report that HIV+ patients receiving ART (anti-retroviral therapy) had “significantly higher mean gap-detection threshold [a central auditory processing test] compared with the ART‑ group.” Despite our interest in preserving and improving auditory and vestibular function, we believe that treatment of HIV should take priority, even if there is a minority of literature suggesting potential audiovestibular risks to anti-retroviral therapy.
Patients who are HIV+ and have subjective hearing loss should be evaluated and managed by an audiologist. Such management may include amplification for hearing loss, and masking for tinnitus.
In patients who are HIV+ and have subjective disequilibrium, it is medically reasonable to consider undertaking a screening vestibular workup (cervical and ocular vestibular evoked myogenic potentials, video head impulse testing, videonystagmography, rotatory chair testing). If evidence of vestibular weakness is found, then a trial of vestibular rehabilitation therapy is appropriate.
Prognosis
Absent sufficient longitudinal studies, the prognosis of auditory and vestibular symptoms in HIV+ patients is unknown.
References
Buckey JC, Fellows AM, Magohe A, Maro I, Gui J, Clavier O, Massawe E, Moshi N (2019) Hearing complaints in HIV infection originate in the brain not the ear. AIDS 33: 1449-1454. doi: 10.1097/QAD.0000000000002229
Fokouo JV, Vokwely JE, Noubiap JJ, Nouthe BE, Zafack J, Minka Ngom ES, Dalil AB, Ngo Nyeki AR, Bengono G, Njock R (2015) Effect of HIV Infection and Highly Active Antiretroviral Therapy on Hearing Function: A Prospective Case-Control Study From Cameroon. JAMA Otolaryngol Head Neck Surg 141: 436-41. doi: 10.1001/jamaoto.2015.125
Grimaldi LM, Luzi L, Martino GV, Furlan R, Nemni R, Antonelli A, Canal N, Pozza G (1993) Bilateral eighth cranial nerve neuropathy in human immunodeficiency virus infection. J Neurol 240: 363-6.
Karaca S, Kalcioglu MT, Sargin F, Topcu MT, Uzun L, Tekin M, Barin K (2022) Evaluation of Effects of the Human Immunodeficiency Virus on the Vestibular System by Using the Video Head Impulse Test. Eurasian J Med 54: 138-144. doi: 10.5152/eurasianjmed.2022.20289
Luque AE, Orlando MS, Leong UC, Allen PD, Guido JJ, Yang H, Wu H (2014) Hearing function in patients living with HIV/AIDS. Ear Hear 35: e282-90. doi: 10.1097/AUD.0000000000000064
Maro, II, Fellows AM, Clavier OH, Gui J, Rieke CC, Wilbur JC, Chambers RD, Jastrzembski BG, Mascari JE, Bakari M, Matee M, Musiek FE, Waddell RD, von Reyn CF, Palumbo PE, Moshi N, Buckey JC (2016) Auditory Impairments in HIV-Infected Children. Ear Hear 37: 443-51. doi: 10.1097/AUD.0000000000000276
Maro, II, Moshi N, Clavier OH, MacKenzie TA, Kline-Schoder RJ, Wilbur JC, Chambers RD, Fellows AM, Jastrzembski BG, Mascari JE, Bakari M, Matee M, Musiek FE, Waddell RD, von Reyn CF, Buckey JC (2014) Auditory impairments in HIV-infected individuals in Tanzania. Ear Hear 35: 306-17. doi: 10.1097/01.aud.0000439101.07257.ed
Michaels L, Soucek S, Liang J (1994) The ear in the acquired immunodeficiency syndrome: I. Temporal bone histopathologic study. Am J Otol 15: 515-22.
Millar A, Joubert K, Naude A (2020) Prevalence of hearing loss and tinnitus in a group of adults with Human Immunodeficiency Virus. S Afr J Commun Disord 67: e1-e7. doi: 10.4102/sajcd.v67i1.631
Millar A, Joubert K, Naude A (2021) Prevalence of Peripheral Vestibular Impairment in Adults with Human Immunodeficiency Virus. J Audiol Otol 25: 36-42. doi: 10.7874/jao.2020.00164
Niemczak CE, White-Schwoch T, Fellows A, Magohe A, Gui J, Rieke C, Nicol T, Massawe ER, Moshi N, Kraus N, Buckey JC (2022) Peripheral Auditory Function in Young HIV-Positive Adults With Clinically Normal Hearing. Otolaryngol Head Neck Surg 167: 155-162. doi: 10.1177/01945998211047147
Palacios GC, Montalvo MS, Fraire MI, Leon E, Alvarez MT, Solorzano F (2008) Audiologic and vestibular findings in a sample of human immunodeficiency virus type-1-infected Mexican children under highly active antiretroviral therapy. Int J Pediatr Otorhinolaryngol 72: 1671-81. doi: 10.1016/j.ijporl.2008.08.002
Pappas DG, Jr., Chandra Sekhar HK, Lim J, Hillman DE (1994) Ultrastructural findings in the cochlea of AIDS cases. Am J Otol 15: 456-65.
Soucek S, Michaels L (1996) The ear in the acquired immunodeficiency syndrome: II. Clinical and audiologic investigation. Am J Otol 17: 35-9.
![]()