By Marcello Cherchi, MD PhD
For patients
Latent nystagmus (LN) is a pattern of abnormal eye movements that can occur in people who, during infancy, had strabismus (misalignment of the eyes). Usually the abnormal eye movement only becomes apparent when a patient is trying to view out of one eye. Since most people use both eyes most of the time, patients with LN usually function normally, and treatment is generally not needed.
For clinicians
Overview
Latent nystagmus (LN) is disorder of ocular motility thought to result from developmental failure of binocular vision early in life, the most common cause of which is infantile strabismus or amblyopia. The characteristic findings include that (1) when one eye is occluded there will be nystagmus whose fast phase beats towards the viewing eye; (2) optokinetic testing exhibits low gain in the temporalward direction of the viewing eye. The mechanism of LN is not known, but several lines of evidence implicate binocular disparity (due to failure to develop stereopsis early in life). Since most individuals use binocular vision for most activities, most patients with LN can function normally, and may come to attention if the abnormal eye movement is observed by an examiner. Since most patients with LN can function normally, treatment is usually unnecessary. In cases where LN is believed to be the cause of symptoms, a variety of cycloplegic procedures have been attempted.
Introduction
Latent nystagmus (LN), sometimes also called “infantile nystagmus,” is an abnormal eye movement that appears (manifests) under monocular viewing conditions and disappears (returns to its latent state) when binocular vision is available. The nystagmus is conjugate, horizontal, and beats towards the viewing eye, and thus, the direction changes depending upon which eye is viewing. In other words, when the patient is viewing out of the right eye, there will be conjugate right beat nystagmus; when the patient is viewing out of the left eye, there will be conjugate left beat nystagmus. In some, though not all patients, the nystagmus obeys Alexander’s law (Dell’Osso et al. 1979).
Latent nystagmus (LN) was initially described by Fromaget and Fromaget (Fromaget and Fromaget 1912). They wrote:
“[When] the patient, [with] both eyes open, looks at the eye chart, both eyes are motionless. We place a screen in front of the right eye which is still open. Instantly an oscillatory, horizontal, very rapid nystagmus appears, of 18 to 20 beats every quarter of a minute. The vision [visual acuity] also drops to 0.4. If you uncover the right eye, immediately the nystagmus disappears. If we repeat the experiment by closing the left eye then a horizontal, slow and irregular nystagmus appears of 5 to 6 beats per quarter of a minute and the visual acuity does not reach 0.1.”
This description is incomplete, in the sense that Fromaget and Fromaget merely note that the nystagmus appears when one eye is occluded, but they do not specify the direction of the nystagmus that they observed. Fromaget and Fromaget reported on several other cases of latent nystagmus (Fromaget and Fromaget 1916; Fromaget 1913). The cases reported by Fromaget and Fromaget are reviewed by Sorsby (Sorsby 1931).
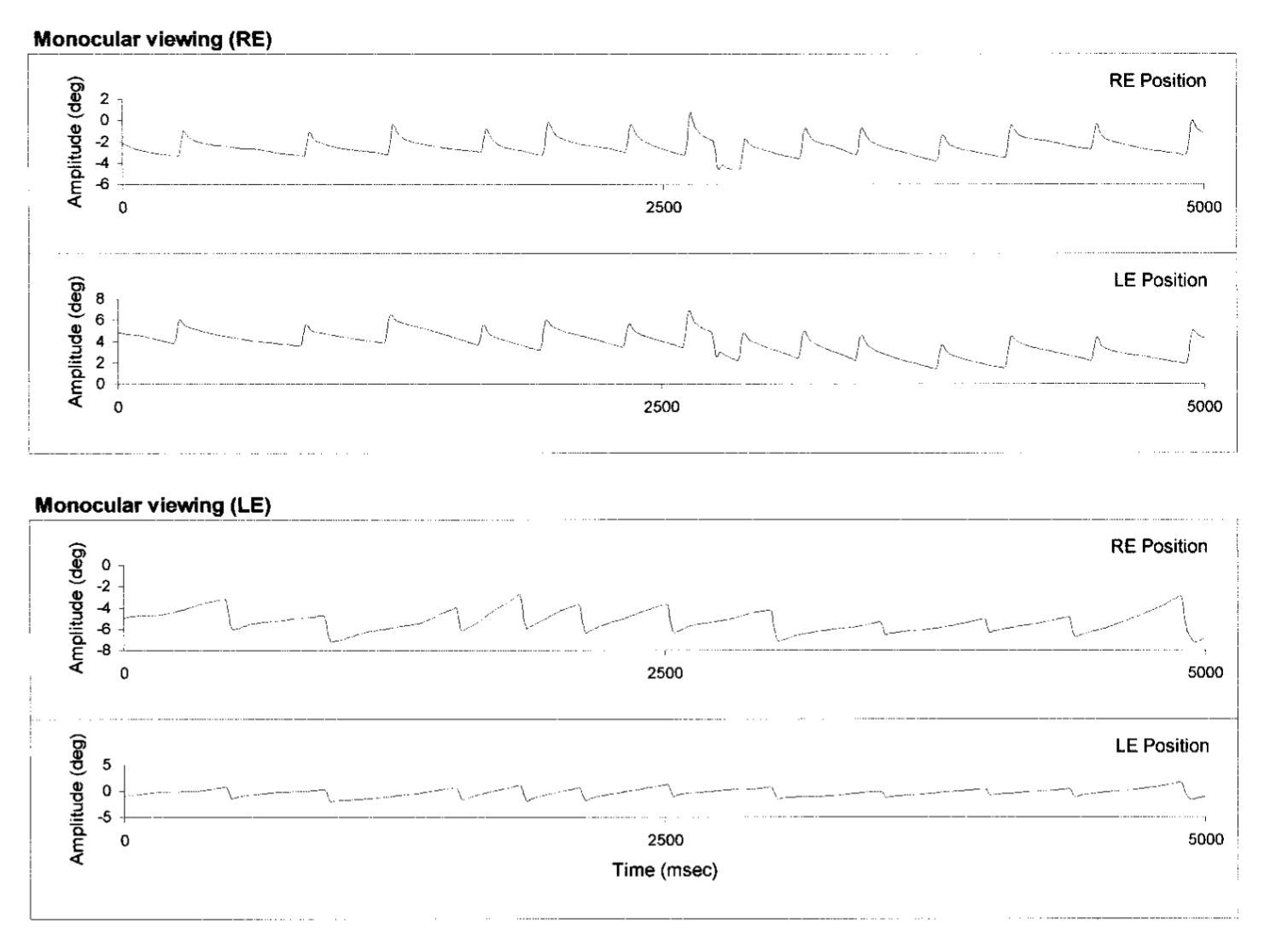
The Figure below, from Abadi and Scallan (Abadi and Scallan 2000), demonstrates LN. By convention in horizontal eye tracings, an upward deflection represents rightward eye movement, and a downward deflection represents leftward eye movement. The top pair of tracings shows conjugate right beat nystagmus when the patient is viewing only out of the right eye. The bottom pair of tracings shows conjugate left beat nystagmus when the patient is viewing only out of the left eye.
The following Video, from https://www.youtube.com/watch?v=Vp0E19yjZf8 (accessed 4/3/23), demonstrates latent nystagmus.
As described above, LN becomes evidence when one eye is occluded. However, in some circumstances it can become apparent even when vision is available to both eyes, and in this situation it is called manifest latent nystagmus (MLN) (Tychsen et al. 2010), first described by Faucon (Faucon 1872).
Epidemiology
Latent nystagmus (LN) is “strongly linked to binocular maldevelopment in infancy, from either strabismus or deprivation of monocular spatial vision (amblyopia)” (Tychsen et al. 2010).
Latent nystagmus can occur in patients with uncorrected childhood strabismus (Dell’Osso et al. 2009). Gresty and colleagues (Gresty et al. 1992) studied 30 patients with LN and found that 29 reported a history of strabismus. Many, though not all, of these patients have amblyopia (Gresty et al. 1992).
Genetics
Latent nystagmus (LN) has been described in familial patterns, such as in identical twins (Abramson 1973), but it is unknown whether there is a genetic susceptibility to developing LN.
Pathophysiological mechanism of disease
The mechanism of latent nystagmus is unknown (Kommerell and Zee 1993). At some level, both the vestibular system and the optokinetic system are probably involved. As Brodsky ant Tusa note:
“Visual stabilization mechanisms act in concert with labyrinthine reflexes. In normal life, optokinetic responses are elicited mainly by head movements, which also stimulate the vestibular system. Because vestibular neurons receive such prominent visual and vestibular inputs, disrupting either input reduces the tonic activation of these neurons, with the effect of disturbing the responses to the other sensory modality… According to Miles, each sensory modality ‘has played such a major role in the evolution of the other that it is impossible to understand the operation of either one in isolation.’” (Brodsky and Tusa 2004).
Brodsky and Tusa further state:
“Various theories have been advanced to explain latent nystagmus. These include a primitive tonus imbalance, an egocentric disorder, a disorder of the subcortical optokinetic system, a subcortical maldevelopment of retinal slip control, abnormal cortical motion processing, a disorder of proprioception, and an evolutionary preponderance of the nasal half of the retina” (Brodsky and Tusa 2004).
Several proposals rely on the idea that in strabismic patients there has been a developmental failure of visual fusion, such that no synaptic connections provide one neuron with binocular information.
This synaptic architecture is illustrated in the Figures below, from Tychsen and colleagues (Tychsen et al. 2010), which show the normal synaptic configuration in the visual cortex for binocular vision, and abnormal synaptic configuration in failure of binocular vision.
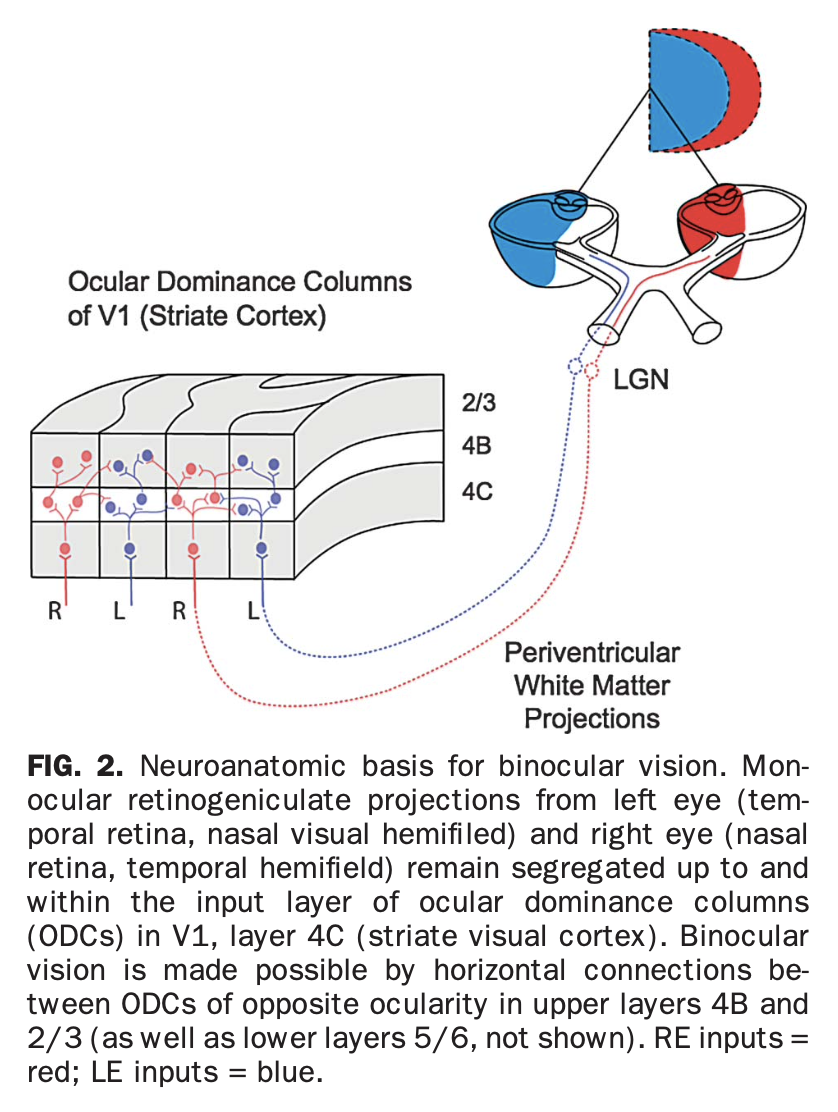 |
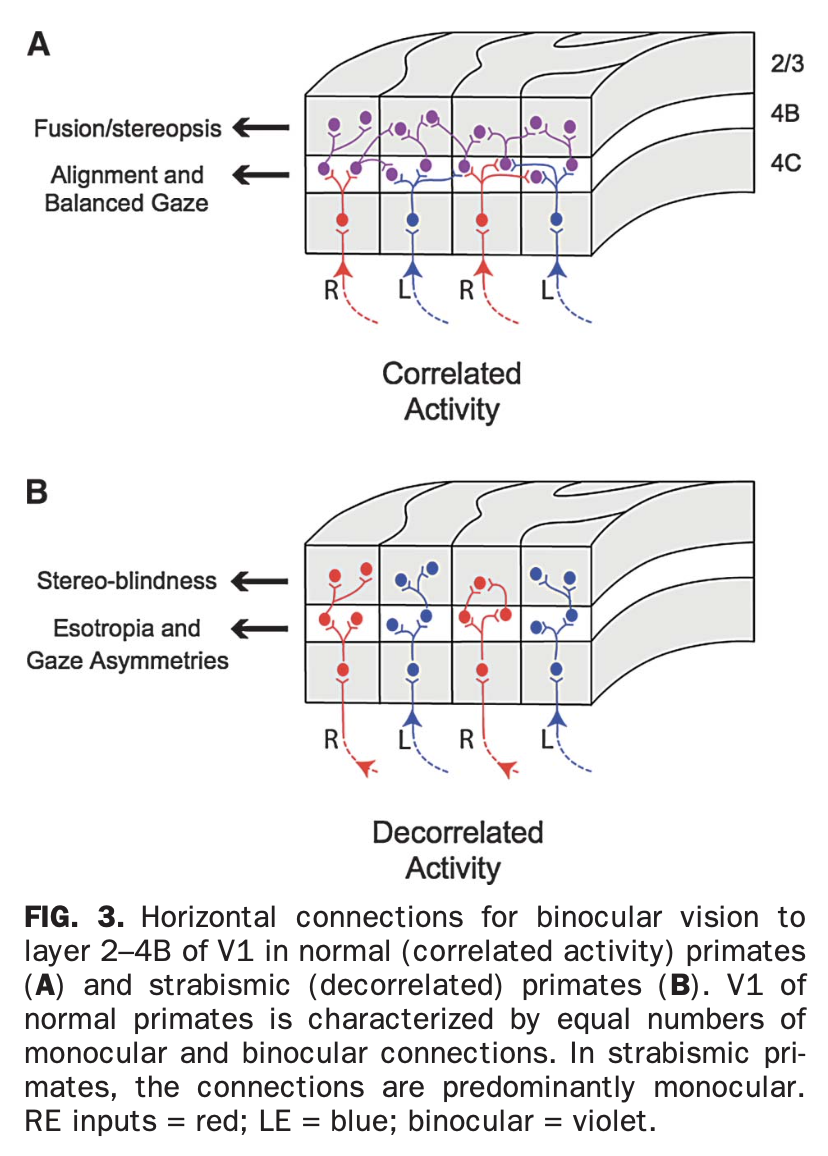 |
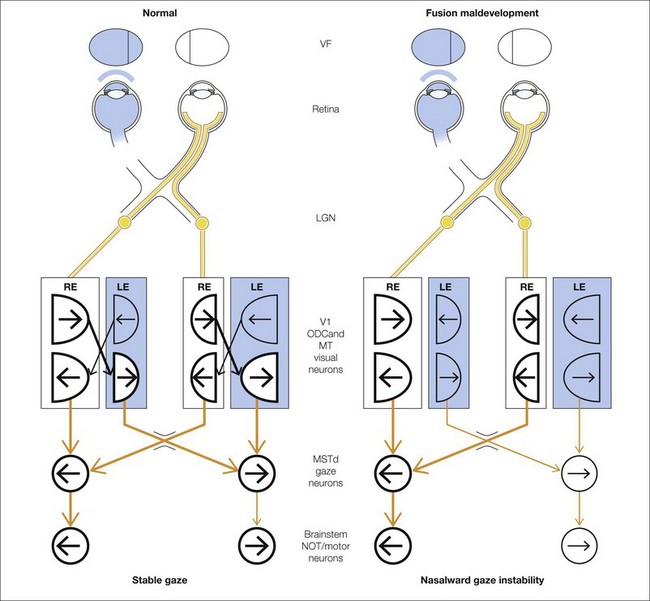
Tychsen then provides the Figure below, with the following accompanying explanation:
“Neural network diagrams showing visual signal flow for pursuit and gaze holding in strabismic and normal primates. A paucity of mature binocular connections explains behavioral asymmetries evident as asymmetric pursuit/optokinetic nystagmus and latent fixation nystagmus. In all primates, pursuit area neurons in each hemisphere encode ipsilaterally directed pursuit. Signal flow is initiated by a moving stimulus in the monocular visual field (VF), which evokes a response in visual area neurons V1 and MT. Each eye at birth has access, through innate monocular connections, to the pursuit area neurons in MSTd of the contralateral hemisphere. Access to pursuit neurons of the ipsilateral hemisphere requires mature binocular connections. In fusion maldevelopment (right column), retinal ganglion cell fibers from the nasal and temporal hemiretina (eye) decussate at the optic chiasm (chi), synapse at the lateral geniculate nucleus (LGN), and project to alternating rows of ODCs in V1 (visual area rectangles). In each V1, ODCs representing the nasal hemiretina (temporal visual hemifield) occupy slightly more cortical territory than those representing the temporal hemiretina (nasal hemifield), but each ODC contains neurons sensitive to nasally directed and temporally directed motion (half circles shaped like the matching hemifield; arrows indicate directional preference). Visual area neurons, including those beyond V1 in area MT, encoding nasally directed motion are wired innately—through monocular connections—to the pursuit area. In normal primates (left column), binocular connections are present, linking neurons with similar orientation/directional preferences within ODCs of opposite ocularity (diagonal lines be- tween columns). When the subject views with the right eye, visual neurons preferring nasally directed motion project to the left hemisphere pursuit area and visual neurons preferring temporally directed motion project to the right hemisphere pursuit area. Temporally directed visual area neurons gain access to pursuit area neurons only through binocular connections. Call = corpus callosum. Bold lines = active neurons and neuronal projections” (Tychsen et al. 2010).
Schor (Schor 1983) alludes to this idea when stating:
“Latent nystagmus and fixation drifts of amblyopic patients could be described in terms of functional suppression of cortical projections to the pretectal OKN [optokinetic nystagmus] system. Bilateral reductions of cortical projections to the pretectum would, during attempted steady monocular fixation, result in subcortical stimulation of the contralateral NOT [nucleus of the optic tract] without a counterbalancing stimulation from the cortical projection to the ipsilateral NOT. Thus, stimulation of either eye alone would cause a nasal drift bias to the side of the closed eye” (Schor 1983).
Similarly, Kommerell and Zee (Kommerell and Zee 1993) reported on two patients who could voluntarily elicit LN. They hypothesized that, “This phenomenon could be the result of voluntary control of the visual input contributed by the amblyopic eye and/or a direct influence of will on the slow eye movement and fixation systems.” In other words, these two patients could “choose” to pay attention to the visual input of only one eye and ignore that of the fellow eye, bringing about the same effect as “occluding” the ignored eye. Abadi (Abadi 1980) comments that van Vliet (van Vliet 1972, 1973) also reported that some LN patients could induce nystagmus “by the ‘intention’ of looking with one eye.”
Brodsky and Tusa (Brodsky and Tusa 2004) describe this as follows:
“The vestibular nucleus presides over motion input from the eyes and labyrinths. Latent nystagmus corresponds to the optokinetic component of ocular rotation that is driven monocularly by nasal optic flow during a turning movement of the body in lateral-eyed animals. Congenital esotropia alters visual pathway development from the visual cortex to subcortical centers that project to the vestibular nucleus, allowing this primitive subcortical motion detection system to generate latent nystagmus under conditions of monocular fixation” (Brodsky and Tusa 2004).
A different view is expressed by Gresty and colleagues (Gresty et al. 1992), who studied the combinations of LN, strabismus, stereopsis, amblyopia and optokinetic abnormalities in 29 patients and conclude that:
“Dissociations found between the presence and severity of strabismus, stereopsis, amblyopia and optokinetic abnormalities point to these features being relatively independent although associated in typical clusterings. This is evidence against the theory that strabismus and LN are directly caused by nasotemporal optokinetic imbalance which persists because of failure to develop binocular vision. The variability of findings favours the view that LN and CN [congenital nystagmus] arise from a genetic or acquired embryological disorder with various degrees and directions of expression” (Gresty et al. 1992).
Brodsky and Tusa comment that, “The magnitude of the resulting manifest latent nystagmus is proportional to the degree of the interocular visual disparity” (Brodsky and Tusa 2004).
Clinical presentation
Most, though not all, patients with latent nystagmus lack binocular vision. Gresty and colleagues (Gresty et al. 1992) studied 30 LN patients and reported that 24 of them lacked binocular vision, while the remaining 6 had “varying degrees of stereopsis.”
LN patients do not usually complain of disequilibrium, but the lack of stereopsis can interfere with depth perception, which may cause difficulties that the patient reports as unsteadiness, difficulty navigating stairs, etc.
Physical examination
In a patient with latent nystagmus who is otherwise healthy, general physical examination should be normal.
Ocular motor examination
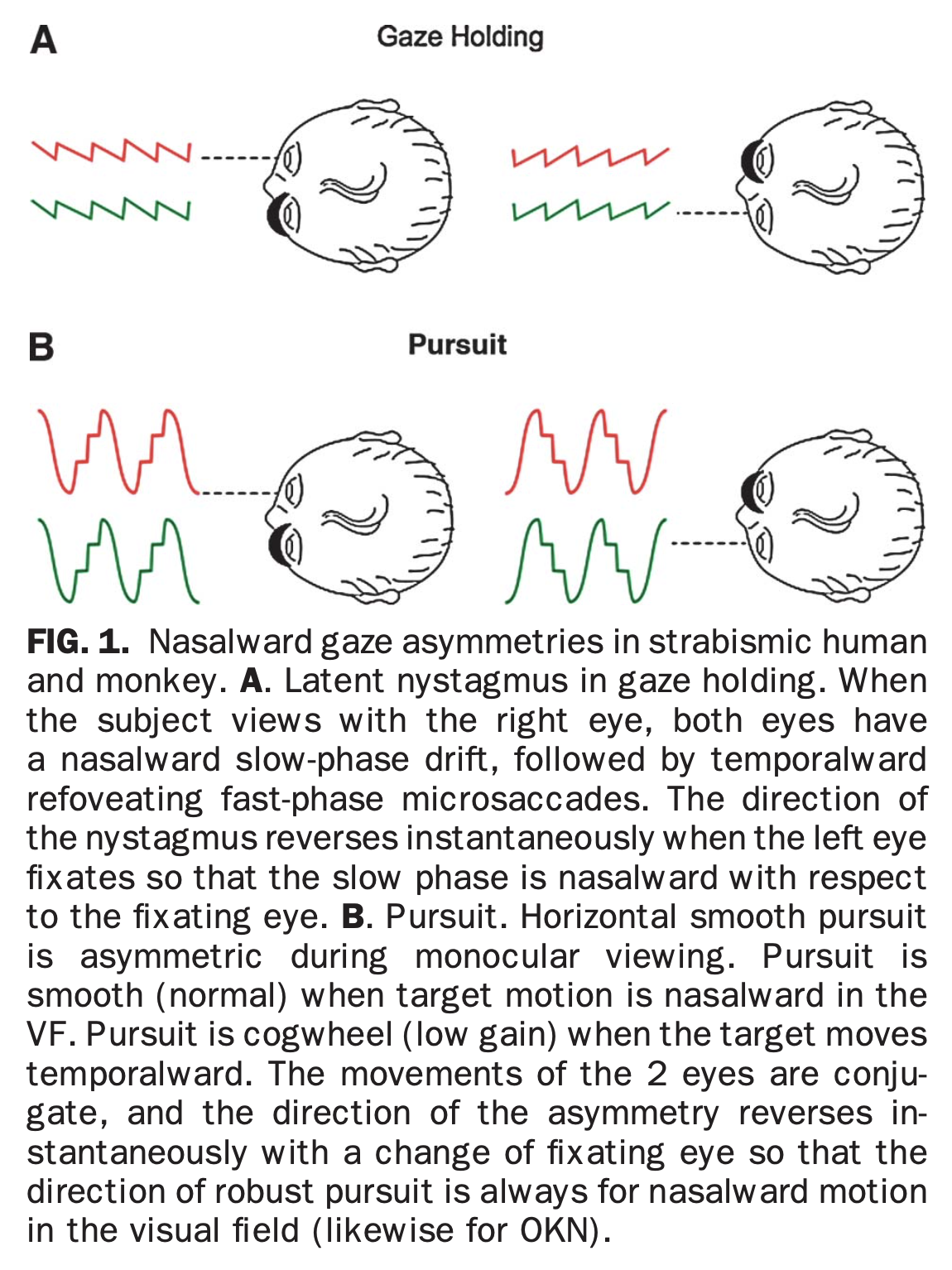
In a patient with latent nystagmus (LN), at baseline, when one eye is occluded, on attempted visual fixation on primary position of gaze, relative to the viewing eye there should be conjugate nasalward slow drift and temporalward corrective saccades (in other words, nystagmus beating toward the side of the viewing eye).
Aside from the baseline pattern of LN evident with monocular viewing, LN patients also have difficulty with smooth pursuit during monocular viewing that Brodsky and Tusa describe as, “nasotemporal asymmetry of the horizontal optokinetic response during monocular viewing” (Brodsky and Tusa 2004), which they say was first reported by Roelofs (Roelofs 1928). During modular viewing the baseline eye movement consists of slow nasalward drift (relative to the viewing eye) with a corrective temporalward saccade (relative to the viewing eye); thus nasalward smooth pursuit appears normal, whereas temporalward smooth pursuit exhibits low gain and thus corrective temporalward saccades are triggered.
These two patterns (attempted fixed gaze during monocular viewing, and attempted smooth pursuit during monocular viewing) are demonstrated in the Figure below, from Tychsen and colleagues (Tychsen et al. 2010).
There are several studies of LN involving vertical and torsional components (Guyton et al. 1998; Hertle and Dell’Osso 1999; Irving et al. 1998).
Testing: vestibular
Vestibular testing is usually unrevealing in latent nystagmus (LN). However, note that ocular motor studies can be misleading if they are performed with the camera always over the same eye, because this will give the mistaken impression of a unidirectional nystagmus at baseline — in other words, it may miss the alternating nature of LN.
Differential diagnosis
Latent nystagmus (LN) is usually quite distinctive and most easily detected on face-to-face examination.
LN can co-occur with other disorders, including ophthalmic and other ocular motor disorders, so it is reasonable to consult ophthalmology or neuro-ophthalmology.
Treatment
In most patients, latent nystagmus (LN) is present only with monocular viewing. Since most people use binocular vision for most daily activities, LN usually remains latent and patients have no symptoms, or minimal symptoms.
For patients in whom LN provokes symptoms, various treatments have been attempted, including injection of extraocular muscles with onabotulinum toxin (Liu et al. 1993) and cyclopentolate eyedrops (Windsor 1968; Windsor and Burian 1968). Occlusion therapy (blocking vision in one eye) has also been reported as sometimes effective (von Noorden et al. 1987), which is unexpected given that monocular viewing generally worsens the nystagmus of LN.
Prognosis
Latent nystagmus (LN) is usually a lifelong condition. In the majority of patients LN in itself does not interfere with daily functioning.
References
Abadi RV (1980) Pattern contrast thresholds in latent nystagmus. Acta Ophthalmol (Copenh) 58: 210-20. doi: 10.1111/j.1755-3768.1980.tb05711.x
Abadi RV, Scallan CJ (2000) Waveform characteristics of manifest latent nystagmus. Invest Ophthalmol Vis Sci 41: 3805-17.
Abramson DH (1973) Latent nystagmus in identical twins. Arch Ophthalmol 89: 256-7. doi: 10.1001/archopht.1973.01000040258017
Brodsky MC, Tusa RJ (2004) Latent nystagmus: vestibular nystagmus with a twist. Arch Ophthalmol 122: 202-9. doi: 10.1001/archopht.122.2.202
Dell’Osso L, Traccis S, Abel L (2009) Strabismus – a necessary condition for latent and manifest latent nystagmus. Neuro-Ophthalmology 3: 247-257. doi: 10.3109/01658108308997312
Dell’Osso LF, Schmidt D, Daroff RB (1979) Latent, manifest latent, and congenital nystagmus. Arch Ophthalmol 97: 1877-85. doi: 10.1001/archopht.1979.01020020325008
Faucon A (1872) Nystagmus par insuffisance des droits externes. J Ophthal (Paris) 1: 223-229.
Fromaget C, Fromaget H (1912) Nystagmus latent (nystagmus et strabisme). Annales d’Oculistique 147: 344-352.
Fromaget C, Fromaget H (1916) Nystagmus latent. Annales d’Oculistique 153: 465-472.
Fromaget H (1913) Nystagmus latent. Annales d’Oculistique 149: 241-250.
Gresty MA, Metcalfe T, Timms C, Elston J, Lee J, Liu C (1992) Neurology of latent nystagmus. Brain 115 ( Pt 5): 1303-21. doi: 10.1093/brain/115.5.1303
Guyton DL, Cheeseman EW, Jr., Ellis FJ, Straumann D, Zee DS (1998) Dissociated vertical deviation: an exaggerated normal eye movement used to damp cyclovertical latent nystagmus. Trans Am Ophthalmol Soc 96: 389-424; discussion 424-9.
Hertle RW, Dell’Osso LF (1999) Vertical latent nystagmus component and vertical saccadic asymmetries in subjects with dissociated vertical deviation. J AAPOS 3: 257-8. doi: 10.1016/s1091-8531(99)70014-5
Irving EL, Goltz HC, Steinbach MJ, Kraft SP (1998) Vertical latent nystagmus component and vertical saccadic asymmetries in subjects with dissociated vertical deviation. J AAPOS 2: 344-50. doi: 10.1016/s1091-8531(98)90032-5
Kommerell G, Zee DS (1993) Latent nystagmus. Release and suppression at will. Invest Ophthalmol Vis Sci 34: 1785-92.
Liu C, Gresty M, Lee J (1993) Management of symptomatic latent nystagmus. Eye (Lond) 7 ( Pt 4): 550-3. doi: 10.1038/eye.1993.120
Roelofs CO (1928) Nystagmus latens. Archiv für Augenheilkunde 98: 401-447.
Schor CM (1983) Subcortical binocular suppression affects the development of latent and optokinetic nystagmus. Am J Optom Physiol Opt 60: 481-502. doi: 10.1097/00006324-198306000-00009
Sorsby A (1931) Latent Nystagmus. Br J Ophthalmol 15: 1-18. doi: 10.1136/bjo.15.1.1
Tychsen L, Richards M, Wong A, Foeller P, Bradley D, Burkhalter A (2010) The neural mechanism for Latent (fusion maldevelopment) nystagmus. J Neuroophthalmol 30: 276-83. doi: 10.1097/WNO.0b013e3181dfa9ca
van Vliet AG (1972) On the central mechanism of latent nystagmus. Monogr Hum Genet 6: 204. doi: 10.1159/000392707
van Vliet AG (1973) On the central mechanism of latent nystagmus. Acta Ophthalmol (Copenh) 51: 772-81. doi: 10.1111/j.1755-3768.1973.tb06046.x
von Noorden GK, Avilla C, Sidikaro Y, LaRoche R (1987) Latent nystagmus and strabismic amblyopia. Am J Ophthalmol 103: 87-9. doi: 10.1016/s0002-9394(14)74174-1
Windsor CE (1968) Modification of latent nystagmus. 2. Arch Ophthalmol 80: 352-3. doi: 10.1001/archopht.1968.00980050354011
Windsor CE, Burian HM (1968) Modification of latent nystagmus. I. Arch Ophthalmol 80: 657-63. doi: 10.1001/archopht.1968.00980050659014
![]()