By Marcello Cherchi, MD PhD
For patients
The term opsoclonus refers to a pattern of chaotic, rapid eye movement abnormalities. This sometimes occurs with unintentional spasms of muscles in the limbs or trunk. In children opsoclonus is usually associated with a cancer called neuroblastoma. In adults it can be associated with other cancers and with infections. Symptoms usually develop over hours to days. Workup should look for cancer and infection and, if those are found, they should be treated. Whether or not cancer or infection is found, some doctors will attempt treatment with therapies aimed at the immune system. Sometimes the muscle spasms are treated with anti-seizure medications.
For clinicians
Overview
Opsoclonus is an eye movement abnormality consisting of bursts of involuntary, seemingly random, high-frequency, conjugate saccades that are multidirectional (with horizontal, vertical and torsional components), occurring in sequence without any inter-saccadic interval delay. It often occurs in conjunction with myoclonus involving any combination of limbs, trunk and cranio-cervical region. In the pediatric population this is almost always a paraneoplastic syndrome related to neuroblastoma. In adults the differential diagnosis includes paraneoplastic (with various cancers), para-infectious (with various infections) and other rare causes. The underlying pathophysiology is unclear, but likely involves dysfunction of omnipause neurons or their upstream regulators. The clinical presentation is distinct; onset can be over hours to days, and may consist of the ocular motor findings, myoclonus, and encephalopathy or other behavioral disturbances. Workup should include a neoplastic workup and (if that is normal) a search for paraneoplastic onconeuronal antibodies and infection. If neoplasm or infection is discovered, then those should be treated. Whether or not neoplasm and infection are found, many investigators also advocate intervention with immunosuppressive or immunomodulatory therapies (steroids, plasma exchange, intravenous immunoglobulin). Treatment of myoclonus is sometimes attempted with clonazepam and/or anti-seizure medications (gabapentin, levetiracetam, valproate).
Introduction
Opsoclonus, sometimes referred to as saccadomania (Espinosa and Berger 2005), is an unusual eye movement abnormality consisting of bursts of involuntary, seemingly random, high-frequency, conjugate saccades that are multidirectional (with horizontal, vertical and torsional components), occurring in sequence without any inter-saccadic interval delay (Oh, Kim et al. 2019).
The Polish neurologist, Dr. Kazimierz Orzechowski (1878 – 1942) (Magowska, Owecki et al. 2018) was the first to suggest the term “opsoclonus” in a 1927 paper (Orzechowski 1927) as a description for this pattern of eye movements.
The term opsoclonus is probably more broadly recognized by clinicians as one part of the broader opsoclonus-myoclonus syndrome (OMS) later described by the Austrian pediatric neurologist, Dr. Marcel Kinsbourne (Kinsbourne 1962).
In children, OMS usually occurs as a paraneoplastic manifestation of neuroblastoma (Rossor, Yeh et al. 2022). Most neurologists have seen the historical Video of an infant with OMS being held by a nurse.
Though even rarer, opsoclonus (with or without myoclonus) can also occur in adults (Digre 1986, Klaas, Ahlskog et al. 2012). In many cases the underlying etiology is unknown. Among cases with an identified etiology, most appear due to a paraneoplastic syndrome, less to a para-infectious process.
The Video below, from Hsu and Young (Hsu and Young 2017), shows opsoclonus in a 39-year-old woman that was attributed to a para-infectious phenomenon related to herpes simplex virus infection.
Epidemiology
Adult-onset OMS is rare. It occurs either as a paraneoplastic syndrome (more common) or para-infectious syndrome (less common). Armangue and colleagues (Pike 2013) reviewed records of 114 adult patients with OMS and determined that 69 (61%) were “idiopathic,” meaning that no underlying cause could be discerned.
Klaas and colleagues (Hsu and Young 2017) reviewed 21 adult cases of OMS from their own institution (Mayo Clinic) and 116 previously reported adult cases in the literature. Of their own 21 cases, the associated malignancies included primary breast adenocarcinoma and small cell lung cancer. Of the 116 previous cases they reviewed, malignancies were identified in 55 (47.4%), and these included lung carcinoma (small cell and non-small cell), breast adenocarcinoma, melanoma, ovarian carcinoma, ovarian teratoma, renal cell carcinoma, thyroid medullary carcinoma, esthesioneuroblastoma, B‑cell lymphoma, cervical carcinoma, bladder carcinoma and gastric adenocarcinoma.
Armangue and colleagues (Armangue, Sabater et al. 2016) reviewed 114 adult-onset cases of OMS and reported that 45 (39%) were paraneoplastic. The most common neoplasms were lung cancer in 19 (42%), breast cancer in 10 (22%), other cancers in 5 (11%) and ovarian teratoma in 8 (18%).
Of the 116 cases of adult-onset OMS reviewed by Klaas and colleagues (Hsu and Young 2017), infectious causes were identified in 15 (12.9%), and these included HIV in 7 (47%), Borrelia burgdorferi in 2 (13%), Streptococcus in 1 (7%), Salmonella in 1 (7%), cytomegalovirus in 1 (7%), Epstein-Barr virus in 1 (7%), Coxsackie virus in 1 (7%) and West Nile Virus in 1 (7%). Reports are also emerging of OMS occurring in the context of COVID‑19 infection (Sanguinetti and Ramdhani 2021).
Pathophysiological mechanism of disease
Whether paraneoplastic or para-infectious, opsoclonus (with or without myoclonus) is thought to result from a mis-directed immune response that mistakenly targets healthy “self” cells, rather than only “non-self” cancerous cells or infectious pathogens, presumably due to epitope similarity (molecular mimicry).
Klaas and colleagues (Klaas, Ahlskog et al. 2012) note very rare reports of adult-onset OMS occurring apparently due to non-paraneoplastic, non-para-infectious causes, such as in association with anti‑GAD65 antibody (Markakis, Alexiou et al. 2008), and during pregnancy (Koide, Sakamoto et al. 2004) or the post-partum period (Adhikari, Thuringer et al. 2021). There is also a reported case of OMS occurring after the use of intranasal cocaine (Scharf 1989).
Oh and colleagues (Oh, Kim et al. 2019) reviewed three theories regarding the pathophysiologic mechanism of opsoclonus.
- The “omnipause neuron” hypothesis is that opsoclonus results from damage of omnipause neurons in the nucleus raphe interpositus of the pons. In the healthy state these neurons suppress unwanted saccades by inhibiting burst neurons in the paramedian pontine reticular formation and the rostral interstitial nucleus of Cajal.
- The “brainstem theory” suggests that saccadic oscillations result from either (1) alterations in the membrane properties of saccadic burst neurons that make them prone to excessive post-inhibitory rebound excitation after sustained inhibition from the omnipause neurons, or (2) reduction in the efficacy of omnipause neuron inhibition. The second possibility sounds similar to the “omnipause neuron” hypothesis.
- The ”cerebellar theory” hypothesizes that dysfunctional cerebellar Purkinje cells fail to inhibit the cerebellar fastigial nuclei, which in turn reinforces inhibition of omnipause neurons, thereby releasing (disinhibiting) burst neurons.
While these hypotheses have merit, they remain speculative.
Clinical presentation
Symptoms usually develop over hours to days.
Klaas and colleagues (Klaas, Ahlskog et al. 2012) reviewed 21 cases of adult-onset OMS from their own institution (Mayo Clinic) and stated that “a flulike prodrome before neurologic symptom onset was reported by 6 patients” (28%). They reported that the initial neurologic symptoms were dizziness in 14 (67%), imbalance from myoclonus in 14 (67%), nausea/vomiting in 10 (48%), visual abnormalities from opsoclonus in 6 (28%), tremor in 4 (19%) and altered speech due to myoclonus in 2 (10%).
Klaas and colleagues (Klaas, Ahlskog et al. 2012) also reviewed 116 previously reported cases of adult-onset OMS. Of 93 cases for which presenting symptoms were documented, these included dizziness in 44 (47%), imbalance in 35/93 (38%), nausea in 32 (34%), tremor in 20 (22%), visual disturbance in 15 (16%), seizure in 3 (3%), fever in 2/93 (2%), headache in 2 (2%), hearing loss in 1 (1%) and anxiety in 1 (1%); some patients had multiple presenting symptoms.
Armangue and colleagues (Armangue, Sabater et al. 2016) retrospectively reviewed the records of 45 patients with adult-onset OMS and noted that 6 (13%) had an infectious prodrome, 22(49%) presented with “acute vertigo,” 11 (24%) presented with “subacute ataxia or myoclonus.” Other features at some point in the course of the disease included encephalopathy in 13 (29%) and “severe behavioral changes” in 6 (13%).
Physical examination
Opsoclonus is usually evident on face-to-face examination and is not subtle.
In patients who also have myoclonus, there is considerable variability in the anatomical area of involvement (Klaas, Ahlskog et al. 2012); this can include any combination of upper extremities, lower extremities, cranio-cervical muscles, and trunk.
Testing: serological
Klaas and colleagues (Klaas, Ahlskog et al. 2012) also reviewed 116 previously reported cases of adult-onset OMS. Of the 60 cases though to be paraneoplastic in nature, the associated antibodies included ANNA‑2 (anti‑Ri), NMDA‑R, calcium channel P/Q‑type or N‑type, ANNA‑1 (anti‑Hu) and amphiphysin IgG. Among these 116 cases they reviewed, and in the 21 cases from their own institution (Mayo Clinic), they stated that, “lung and breast carcinomas were the most common cancers identified, and ANNA‑2 [anti‑Ri] was the most commonly reported paraneoplastic antibody.”
Armangue and colleagues (Armangue, Sabater et al. 2016) similarly reported that in paraneoplastic cases of OMS, 7/10 (70%) of breast cancer patients had anti-ANNA2 (anti‑Ri) antibodies.
Differential diagnosis
Opsoclonus (with or without myoclonus) is usually such a distinctive presentation that there is a relatively limited number of categories of differential diagnoses. In the pediatric population the main diagnostic consideration is neuroblastoma. In adults the differential is somewhat broader (multiple neoplasms, multiple infections).
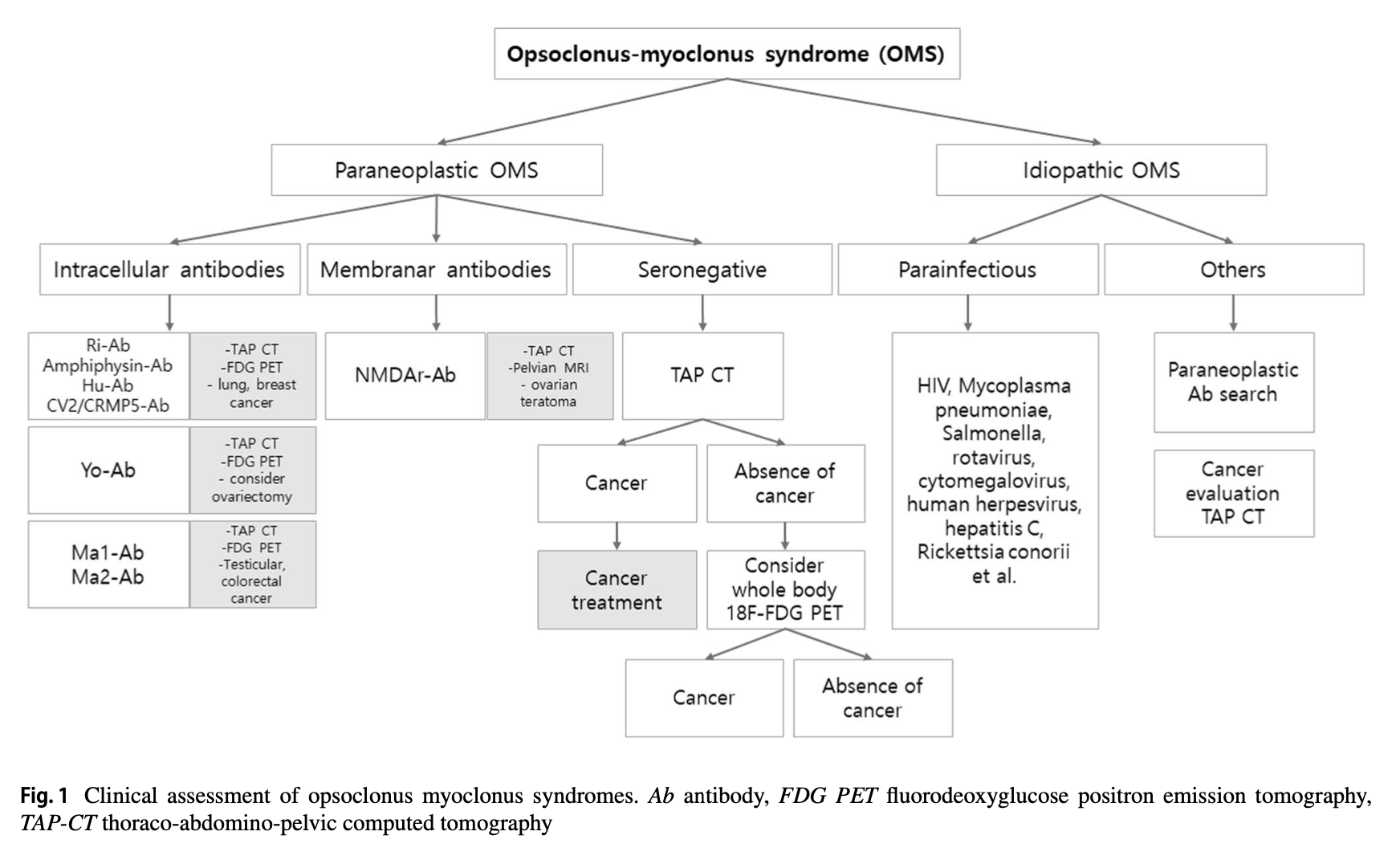
The Figure below shows the diagnostic algorithm suggested by Oh and colleagues (Armangue, Sabater et al. 2016).
Treatment
For adult-onset OMS cases in which malignancy is identified, treatment should include removal of the cancer. For cases in which infection is identified, treatment should include anti-microbial agents (where available).
There is otherwise no standardized protocol for management. Given the presumed immunologically-mediated mechanism of disease, a variety of immunosuppressive interventions have been attempted, including plasma exchange, corticosteroids (prednisone, methylprednisolone) and intravenous immunoglobulin. In the 116 cases of adult-onset OMS reviewed by Klaas and colleagues (Klaas, Ahlskog et al. 2012) they calculated that 79% of patients who received some kind of immunotherapy achieved remission or improvement.
Acute (symptomatic) management of myoclonus is often attempted with clonazepam (Bartos 2006), gabapentin (Moretti, Torre et al. 2000), levetiracetam (Lou, Hensley et al. 2010) or valproate (Digre 1986, Scharf 1989, Lou, Hensley et al. 2010).
Prognosis
Of the 21 cases of adult-onset OMS studied by Klaas and colleagues (Klaas, Ahlskog et al. 2012), 12 (57%) achieved “neurologic remission,”, 3 (14%) died, 1 (5%) had “mild symptoms,” 1 (5%) was “improved,” and in 4 (19%) the outcome was unknown.
References
Adhikari S, Thuringer A, Maali L, Jassam Y (2021) Opsoclonus myoclonus syndrome in a postpartum period. Mult Scler Relat Disord 50: 102862. doi: 10.1016/j.msard.2021.102862
Armangue T, Sabater L, Torres-Vega E, Martinez-Hernandez E, Arino H, Petit-Pedrol M, Planaguma J, Bataller L, Dalmau J, Graus F (2016) Clinical and Immunological Features of Opsoclonus-Myoclonus Syndrome in the Era of Neuronal Cell Surface Antibodies. JAMA Neurol 73: 417-24. doi: 10.1001/jamaneurol.2015.4607
Bartos A (2006) Effective high-dose clonazepam treatment in two patients with opsoclonus and myoclonus: GABAergic hypothesis. Eur Neurol 56: 240-2. doi: 10.1159/000096494
Digre KB (1986) Opsoclonus in adults. Report of three cases and review of the literature. Arch Neurol 43: 1165-75. doi: 10.1001/archneur.1986.00520110055016
Espinosa PS, Berger JR (2005) Volitional opsoclonus. Neurology 65: E11. doi: 10.1212/01.wnl.0000180351.66206.a6
Hsu SY, Young YH (2017) Opsoclonus Recorded by a Smartphone. JAMA Otolaryngol Head Neck Surg 143: 847-848. doi: 10.1001/jamaoto.2017.0510
Kinsbourne M (1962) Myoclonic encephalopathy of infants. J Neurol Neurosurg Psychiatry 25: 271-6. doi: 10.1136/jnnp.25.3.271
Klaas JP, Ahlskog JE, Pittock SJ, Matsumoto JY, Aksamit AJ, Bartleson JD, Kumar R, McEvoy KF, McKeon A (2012) Adult-onset opsoclonus-myoclonus syndrome. Arch Neurol 69: 1598-607. doi: 10.1001/archneurol.2012.1173
Koide R, Sakamoto M, Tanaka K, Hayashi H (2004) Opsoclonus-myoclonus syndrome during pregnancy. J Neuroophthalmol 24: 273. doi: 10.1097/00041327-200409000-00024
Lou E, Hensley ML, Lassman AB, Aghajanian C (2010) Paraneoplastic opsoclonus-myoclonus syndrome secondary to immature ovarian teratoma. Gynecol Oncol 117: 382-4. doi: 10.1016/j.ygyno.2010.01.017
Magowska AM, Owecki MK, Bogusz H (2018) Kazimierz Orzechowski (1878-1942). J Neurol 265: 1953-1955. doi: 10.1007/s00415-017-8724-4
Markakis I, Alexiou E, Xifaras M, Gekas G, Rombos A (2008) Opsoclonus-myoclonus-ataxia syndrome with autoantibodies to glutamic acid decarboxylase. Clin Neurol Neurosurg 110: 619-21. doi: 10.1016/j.clineuro.2008.03.005
Moretti R, Torre P, Antonello RM, Nasuelli D, Cazzato G (2000) Opsoclonus-myoclonus syndrome: gabapentin as a new therapeutic proposal. Eur J Neurol 7: 455-6. doi: 10.1046/j.1468-1331.2000.00091.x
Oh SY, Kim JS, Dieterich M (2019) Update on opsoclonus-myoclonus syndrome in adults. J Neurol 266: 1541-1548. doi: 10.1007/s00415-018-9138-7
Orzechowski K (1927) De l’ataxie des yeux dite myoclonique (opsoclonic, opsochorie). J. Psychol. Neurol.(Lpz.) 35: 1.
Pike M (2013) Opsoclonus-myoclonus syndrome. Handb Clin Neurol 112: 1209-11. doi: 10.1016/B978-0-444-52910-7.00042-8
Rossor T, Yeh EA, Khakoo Y, Angelini P, Hemingway C, Irani SR, Schleiermacher G, Santosh P, Lotze T, Dale RC, Deiva K, Hero B, Klein A, de Alarcon P, Gorman MP, Mitchell WG, Lim M, Group OMSS (2022) Diagnosis and Management of Opsoclonus-Myoclonus-Ataxia Syndrome in Children: An International Perspective. Neurol Neuroimmunol Neuroinflamm 9. doi: 10.1212/NXI.0000000000001153
Sanguinetti SY, Ramdhani RA (2021) Opsoclonus-Myoclonus-Ataxia Syndrome Related to the Novel Coronavirus (COVID-19). J Neuroophthalmol 41: e288-e289. doi: 10.1097/WNO.0000000000001129
Scharf D (1989) Opsoclonus-myoclonus following the intranasal usage of cocaine. J Neurol Neurosurg Psychiatry 52: 1447-8. doi: 10.1136/jnnp.52.12.1447-a
![]()