By Marcello Cherchi, MD PhD
For patients
Persistent postural perceptual dizziness (abbreviated PPPD) usually occurs in the wake of some other “dizzy disease.” Most patients describe the symptom as a non-spinning disequilibrium, which fluctuates in intensity, and tends to be worse when upright, when in active or passive motion, and when exposed to complex or moving visual stimuli. Since there is not yet any confirmatory diagnostic test for PPPD, and since the symptoms can resemble those of other disorders, it is medically reasonable to undertake a screening workup (to confirm or exclude competing diagnoses). If workup reveals no more compelling diagnosis, then treatment is generally attempted with vestibular rehabilitation therapy and/or cognitive behavioral therapy. If those fail, then some practitioners advocate a trial of medications such as sertraline.
For clinicians
Overview
Persistent postural perceptual dizziness (PPPD) generally follows on the heels of another “dizzy disease” and is thought to reflect failure of the vestibular system to re-calibrate to the premorbid state, though the underlying pathophysiology is unknown. Current diagnostic criteria require that symptoms (non-spinning disequilibrium) be present >15 days per month for ≥3 months, are exacerbated by several factors (upright posture, active/passive motion, complex/moving visual stimuli), that the symptoms cause distress or functional impairment, and that the symptoms are not better explained by another disorder. There is no confirmatory diagnostic test for PPPD, though research studies have observed non-specific abnormalities on volumetric MRI and posturography. There is a fairly extensive differential diagnosis which includes a number of objectively verifiable diseases, so we generally undertake a screening otovestibular workup with cervical and ocular vestibular evoked myogenic potentials, video head impulse testing, videonystagmography, rotatory chair testing and computerized dynamic posturography, and (if ear symptoms are present) audiometry. Treatments studies are generally hampered by the lack of placebo arms, so we generally proceed in order of escalating risk, starting with vestibular rehabilitation therapy, then cognitive behavioral therapy. If those fail, then a trial of sertraline is reasonable.
Introduction
Persistent postural perceptual dizziness (PPPD) is currently believed to be a disorder of equilibrium precipitated by another discrete vestibular illness. PPPD may emerge while the other vestibular illness is present, or resolving. PPPD is thought to reflect a maladaptive failure to recalibrate the vestibular system to the pre-morbid state. The symptoms consist of non-spinning disequilibrium, are present more than half the time, tend to be most obvious when a patient is upright, can be exacerbated by visual stimuli and spatial cues, and are frequently comorbid with psychiatric disorders (particularly anxiety). There is currently no confirmatory diagnostic test, so at present PPPD is a diagnosis of exclusion.
With the caveat that “as a rule, only poorly understood disorders require classification by a committee” (Fenichel 2005), the diagnostic criteria for PPPD were based on a committee consensus document of the Barany Society (Staab, Eckhardt-Henn et al. 2017), and drew on the work of several previously described entities, including phobic postural vertigo (PPV), space motion discomfort (SMD), chronic subjective dizziness (CSD) and visual vertigo (VV). PPPD has some features of each of these other entities, but is not synonymous with any of them.
Precipitating disorders
In principle, PPPD can follow nearly any other disorder of equilibrium. In descending order of frequency, the precipitating disorders are (Staab, Eckhardt-Henn et al. 2017):
- 25 – 30%: Peripheral or central vestibular disorders
- 15 – 20%: Vestibular migraine
- 15%: Panic attacks manifesting with dizziness
- 15%: Anxiety manifesting with dizziness
- 10 – 15%: Brain concussion and whiplash injury
- 7%: Autonomic disorders
- 3%: Cardiac dysrhythmias, adverse drug reactions
This distribution is fairly close to the prevalence of each category, so it is unclear whether this distribution reflects a general predisposition/vulnerability to simply any precipitant (and thus is just more commonly found following more common diseases), or whether specific diseases are actually more likely than others to trigger PPPD.
Epidemiology
The Barany consensus document states that:
“No epidemiologic studies are available for PPPD, but its prevalence and incidence may be estimated from research done on patients with PPV, VV, CST and chronic dizziness following acute vestibular syndromes. Clinical epidemiologic data from tertiary care centers with special interest in PPV and CSD showed their prevalence to be 15 – 20% among all patients presenting for evaluation of vestibular symptoms, making them the most common diagnoses among young adults and the second most common among all adults” (Staab, Eckhardt-Henn et al. 2017).
The Barany consensus document further states that symptoms, “may last for months to years,” and that “the average duration of illness at the time of tertiary consultation was 4.5 years.”
Diagnostic criteria
As articulated in the Barany Society consensus document (Staab, Eckhardt-Henn et al. 2017), the diagnostic criteria for PPPD are:
- [Symptoms and chronology] One or more symptoms of dizziness, unsteadiness, or non-spinning vertigo are present on most days for 3 months or more.
- Symptoms last for prolonged (hours-long) periods of time, but may wax and wane in severity.
- Symptoms need not be present continuously throughout the entire day.
- [Exacerbators] Persistent symptoms occur without specific provocation, but are exacerbated by three factors:
- Upright posture,
- Active or passive motion without regard to direction or position, and
- Exposure to moving visual stimuli or complex visual patterns.
- [Precipitants] The disorder is precipitated by conditions that cause vertigo, unsteadiness, dizziness, or problems with balance including acute, episodic, or chronic vestibular syndromes, other neurologic or medical illnesses, or psychological distress.
- When the precipitant is an acute or episodic condition, symptoms settle into the pattern of criterion A as the precipitant resolves, but they may occur intermittently at first, and then consolidate into a persistent course.
- When the precipitant is a chronic syndrome, symptoms may develop slowly at first and worsen gradually.
- [Emotional/functional sequelae] Symptoms cause significant distress or functional impairment.
- [Diagnosis of exclusion] Symptoms are not better accounted for by another disease or disorder.
Differential diagnosis
The Barany Society consensus document (Staab, Eckhardt-Henn et al. 2017) offers the following structured differential diagnosis:
- “Chronic sequelae of acute precipitants.” This refers to symptoms attributable to whatever antecedent “dizzy disease” triggered PPPD.
- “Recurrent attacks of episodic precipitants,” such as vestibular migraine, Ménière’s disease, benign paroxysmal positional vertigo and others.
- “Ongoing manifestations of chronic precipitants,” such as:
- “Chronic anxiety and depressive disorders.”
- “Post-concussive syndrome.”
- “Autonomic disorders.”
- “Other chronic vestibular syndromes,” such as:
- Bilateral vestibular weakness.
- Chronic neurologic disorders.
- Mal de debarquement syndrome.
- “Adverse effects of medications.”
- “Other functional forms of vestibular disorders.”
- “Gait disorders, falls, and near falls.”
Diagnostic testing
One study (Sohsten, Bittar et al. 2016) compared results of computerized dynamic posturography in patients diagnosed with PPPD versus patients with “acute vestibular syndromes” and normal controls. The study noted that, “Patients with PPPD had the greatest likelihood of abnormal sensory analyses,” and “Patients with PPPD had significantly lower mean scores than normal individuals on conditions 2-6 and the composite.” These patterns are very non-specific.
Several studies have commented on changes in brain MRIs in patients with PPPD (Sun and Xiang 2020). For example, a volumetric MRI study of PPPD patients (Wurthmann, Naegel et al. 2017) found that, “PPPD patients showed gray matter volume decrease in the temporal cortex, cingulate cortex, precentral gyrus, hippocampus, dorsolateral prefrontal cortex, caudate nucleus and the cerebellum. A negative correlation of disease duration and gray matter volume was observed in the visual cortex, supplementary motor area and somatosensory processing structures,” which the authors summarized as, “In patients with PPPD areas involved in multisensory vestibular processing show gray matter volume decrease,” noting that, “Longer duration of disease leads to a more pronounced gray matter alteration,” and commented that, “These brain regions resemble those previously described for other vestibular disorders.” In other words, these volumetric brain MRI changes do not distinguish PPPD from other vestibular disorders.
One study explored posturographic changes induced by galvanic vestibular stimulation (GVS) in patients with PPPD (Woll, Sprenger et al. 2019). The study found that, “The sway increase in the baseline of PPPD may be related to an earlier transition from open- to closed-loop mode of postural control. In contrast, the smaller sway of PPPD in the foam condition under visual deprivation is in line with the known balance improvement under more demanding postural challenges in PPPD,” concluding that, “GVS discloses differences between simple and complex balance tasks in PPPD.”
None of these studies identified a specific pattern (of MRI volumetric changes, or posturographic changes) that was diagnostic of PPPD. Given the differential diagnosis reviewed earlier, and the fact that PPPD is a diagnosis of exclusion, when a diagnosis of PPPD is being entertained, it is medically reasonable to undertake a screening otovestibular workup with cervical and ocular vestibular evoked myogenic potentials, video head impulse testing, rotatory chair testing and computerized dynamic posturography. If a patient complains of lateralizing hearing symptoms, then audiometry is also reasonable.
Pathophysiology
The underlying pathophysiology of PPPD is not understood.
Staab and colleagues (Staab, Eckhardt-Henn et al. 2017) proposed the theoretical conceptual framework depicted in the Figure below.
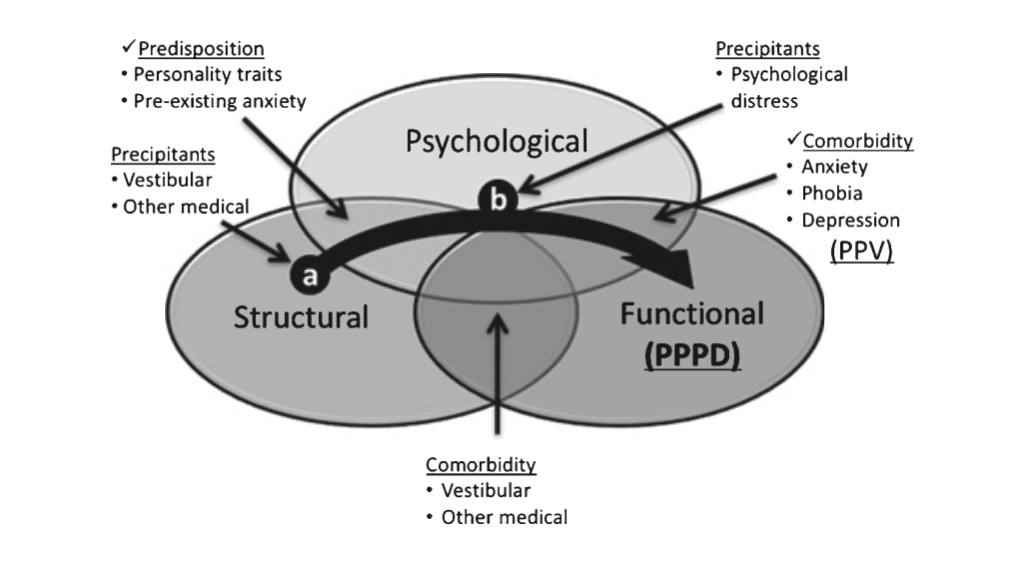 |
 |
Treatments
Despite the poor understanding of the underlying pathophysiology of PPPD, a number of treatment modalities have been explored in multiple studies, including physical therapy, cognitive behavioral therapy and pharmacotherapy (Trinidade and Goebel 2018). Single studies of other modalities, such as non-invasive vagus nerve stimulation, have also been undertaken.
Treatment: Physical therapy
Several studies have explored whether physical therapy is beneficial in PPPD.
One study (Nada, Ibraheem et al. 2019) divided PPPD into two treatment groups, both of which underwent vestibular rehabilitation therapy, and one of which received additional “placebo” treatment. The study reported that, “There was a significant decrease in functional, physical, and total scores on the DHI in both groups after VRT. Adding the placebo did not have supplementary outcomes.” The structure of this study strikes us as odd, as it is not truly placebo-controlled (there was no comparison of “treatment alone” versus “placebo alone”).
A pilot study (Teh, Abdullah et al. 2022) offered vestibular rehabilitation therapy (VRT) to 30 patients and assessed outcomes using several validated scoring systems including the Dizziness Handicap Inventory (DHI), Depression, Anxiety and Stress Scale (DASS‑21) and the EuroQuol quality of life scale (EQ5D). The investigators concluded, “VRT is an effective modality in significantly improving quality of life, dizziness handicap, depression, and anxiety levels within 3 months in PPPD.”
These studies are encouraging but hindered by the absence of true placebo arms. However, since vestibular rehabilitation therapy incurs no significant medical risk, it is still reasonable.
Treatment: Cognitive behavioral therapy
Cognitive behavioral therapy (CBT) has been attempted in the treatment of PPPD. These studies are somewhat difficult to interpret because they generally combine CBT with some other treatment modality, such as vestibular rehabilitation therapy (Popkirov, Stone et al. 2018) or pharmacotherapy (Yu, Xue et al. 2018). Nevertheless, since CBT incurs no medical risk, we feel it is reasonable to attempt.
Treatment: Pharmacotherapy
Several studies have explored the utility of pharmacotherapy in treating PPPD; the agents investigated are generally SSRIs (typically sertraline) or SNRIs. The majority of these publications are case-control studies, case series and retrospective reviews (Trinidade and Goebel 2018). There has been a single double-blind randomized controlled trial of sertraline versus verapamil (Staab, Eggers et al. 2015). As of this writing there have been no randomized, prospective, double-blinded, placebo-controlled trials.
Treatment: Vagus nerve stimulation
One study (Eren, Filippopulos et al. 2018) took the fact that many PPPD patients complain of symptoms that are potentially autonomically mediated (nausea, tachycardia, diaphoresis) as a rationale for attempting non-invasive vagus nerve stimulation (nVNS) in a protocol similar to what had been explored for cluster headaches. The investigators reported that, “Results showed that non-invasive vagus nerve stimulation significantly improved quality of life, as measured by the EQ-5D-3L (p = 0.04), and depression, as measured by the HADS-D (p = 0.002), in the nVNS group,” further observing that, “in the pooled analysis (additional 4 weeks of stimulation…), less severe vertigo attacks/exacerbations (p = 0.04), a decrease in total postural sway path as measured by posturography (p = 0.02), as well as tendentious less anxiety (p = 0.08), occurred after stimulation,” and concluded that, “These data imply that short term nVNS is a safe and promising treatment option in patients with otherwise refractory PPPD.”
References
Eren OE, Filippopulos F, Sonmez K, Mohwald K, Straube A, Schoberl F (2018) Non-invasive vagus nerve stimulation significantly improves quality of life in patients with persistent postural-perceptual dizziness. J Neurol 265: 63-69. doi: 10.1007/s00415-018-8894-8
Fenichel G (2005) Clinical Pediatric Neurology: A Signs and Symptoms Approach, 5 edn. W.B. Saunders, Philadelphia
Nada EH, Ibraheem OA, Hassaan MR (2019) Vestibular Rehabilitation Therapy Outcomes in Patients With Persistent Postural-Perceptual Dizziness. Ann Otol Rhinol Laryngol 128: 323-329. doi: 10.1177/0003489418823017
Popkirov S, Stone J, Holle-Lee D (2018) Treatment of Persistent Postural-Perceptual Dizziness (PPPD) and Related Disorders. Curr Treat Options Neurol 20: 50. doi: 10.1007/s11940-018-0535-0
Sohsten E, Bittar RS, Staab JP (2016) Posturographic profile of patients with persistent postural-perceptual dizziness on the sensory organization test. J Vestib Res 26: 319-26. doi: 10.3233/VES-160583
Staab J, Eggers S, Neff B, Shepard N Vestibular migraine and persistent postural-perceptual dizziness: results of a double blind, parallel group, pharmacologic dissection trial using verapamil and sertraline International Headache Congress of the International Headache Society 2015, pp 65-65
Staab JP, Eckhardt-Henn A, Horii A, Jacob R, Strupp M, Brandt T, Bronstein A (2017) Diagnostic criteria for persistent postural-perceptual dizziness (PPPD): Consensus document of the committee for the Classification of Vestibular Disorders of the Barany Society. J Vestib Res 27: 191-208. doi: 10.3233/VES-170622
Sun L, Xiang K (2020) A review on the alterations in the brain of persistent postural-perceptual dizziness patients and non-pharmacological interventions for its management. Rev Neurosci 31: 675-680. doi: 10.1515/revneuro-2019-0109
Teh CS, Abdullah NA, Kamaruddin NR, Mohd Judi KB, Fadzilah I, Zainun Z, Prepageran N (2022) Home-based Vestibular Rehabilitation: A Feasible and Effective Therapy for Persistent Postural Perceptual Dizziness (A Pilot Study). Ann Otol Rhinol Laryngol: 34894221111408. doi: 10.1177/00034894221111408
Trinidade A, Goebel JA (2018) Persistent Postural-Perceptual Dizziness-A Systematic Review of the Literature for the Balance Specialist. Otol Neurotol 39: 1291-1303. doi: 10.1097/MAO.0000000000002010
Woll J, Sprenger A, Helmchen C (2019) Postural control during galvanic vestibular stimulation in patients with persistent perceptual-postural dizziness. J Neurol 266: 1236-1249. doi: 10.1007/s00415-019-09255-7
Wurthmann S, Naegel S, Schulte Steinberg B, Theysohn N, Diener HC, Kleinschnitz C, Obermann M, Holle D (2017) Cerebral gray matter changes in persistent postural perceptual dizziness. J Psychosom Res 103: 95-101. doi: 10.1016/j.jpsychores.2017.10.007
Yu YC, Xue H, Zhang YX, Zhou J (2018) Cognitive Behavior Therapy as Augmentation for Sertraline in Treating Patients with Persistent Postural-Perceptual Dizziness. Biomed Res Int 2018: 8518631. doi: 10.1155/2018/8518631
![]()