By Marcello Cherchi, MD PhD
For patients
Syncope (“fainting”) is usually caused by cardiovascular disease. Rarely, syncope can result from vestibular disease. Before arriving at this conclusion, your doctors may want to check several tests of your heart and your inner ear balance function.
For clinicians
Practical summary
The phrase “vestibular syncope” refers to the circumstance in which a vestibular event causes a subsequent episode of syncope. The mechanism of this is unknown, but proposals have included maladaptive cardiovascular/hemodynamic changes in response to the vestibular hallucination (induced by vestibular disease). Primary cardiovascular problems are more common causes of syncope than vestibular problems, so a cardiovascular workup (such as with tilt table testing and cardiac event monitoring) is reasonable. If no cardiovascular cause is found, then it is reasonable to undertake a workup for vestibular disease; if such is found, then treatment (if it exists) is appropriate.
Introduction
The term syncope refers to an unexpected loss of consciousness. The phrase “vestibular syncope” implies that vestibular dysfunction causes the syncopal event.
Epidemiology
There is only a modest literature regarding vestibular syncope, none of which details the epidemiology of this clinical entity.
Pathophysiological mechanism of disease
Most instances of syncope result from transient hypoperfusion of the brain, so the proximal mechanism is inadequate blood flow.
One study of recurrent “cardiogenic vertigo” found that the “vertigo” was unassociated with syncope 52% of the time, preceded syncope 37% of the time, and followed syncope 11% of the time (Kim et al. 2021).
For cases in which “vertigo” is followed by syncope, the sequence of events raises the possibility of a causal relationship. The mechanism underlying such a relationship (“vertigo” causing syncope) remains unclear, but several proposals have emerged.
Raphan, summarizing work done with Bernard Cohen, suggests, “the idea that there was a velocity storage mechanism or integrator that not only is a focus for visual-vestibular interaction but also codes spatial orientation relative to gravity as referenced by the otoliths” (Raphan 2020).
Developing this idea further, Kwon and colleagues suggest that, “By integrating the velocity-storage (VS) circuit in the brainstem and cerebellum, we propose that the vestibular syncope develops as a result of dyssynergia of the vestibulosympathetic and baroreflexes in which centrally estimated downward inertial acceleration during the vertigo attacks acts as a trigger” (Kwon et al. 2022a). In particular, “The dual reflex pathways incorporating the function of the velocity-storage circuit in the brainstem and cerebellum suggest that vestibular syncope is a neurally mediated reflex syncope associated with a sudden hemodynamic change during vertigo. This change can be arterial hypertension triggered by a false downward inertial cue, as suggested previously, or hypotension driven by a false upward inertial cue” (Kwon et al. 2022b).
The key scenarios for this are:
- In normal circumstances, when a healthy individual is subjected to true upward acceleration, that acceleration will cause pooling of intravascular volume in the lower extremities, which results in correspondingly less intravascular volume available in the thorax for the heart to pump upward and perfuse the brain adequately. In order to offset this, several cardiovascular mechanisms are activated (increased heart rate, increased ejection fraction, peripheral vasoconstriction). In contrast, if the individual is subjected to a hallucination (false perception) of upward acceleration (when in fact there is no such acceleration), then the same cardiovascular mechanisms will (mistakenly) be activated, resulting in hyperperfusion of the brain.
- In normal circumstances, when a healthy individual is subjected to true downward acceleration, that acceleration will cause pooling of intravascular volume in the upper body and brain. In order to offset this, several cardiovascular mechanisms are activated (decreased heart rate, decreased ejection fraction, peripheral vasodilation). In contrast, if the individual is subjected to a hallucination (false perception) of downward acceleration (when in fact there is no such acceleration), then the same cardiovascular mechanisms will (mistakenly) be activated, resulting in hypoperfusion of the brain.
A reasonable “test scenario” for the latter circumstance would be a drop attack — a hallucination of abrupt downward acceleration. In otoneurology and neurotology a paradigm example of such drop attacks can be found in the otolithic catastrophe of Tumarkin, which can occur in Meniere’s disease, and this has been documented in several studies (Pyykkö et al. 2017, 2018, 2019).
A drop attack is an abrupt and very pronounced hallucination of downward acceleration, but it is likely that the severity of such hallucinations can occur over a continuum, and thus it is logically plausible that less abrupt and less severe vestibular hallucinations may still provoke some degree of maladaptive (inappropriate compensatory) hemodynamic changes.
While the above logic may seem speculative, it is grounded in theoretical considerations (Balaban 1999; Balaban and Porter 1998; Previc 1993; Yates 1998) and broader evidence from studies regarding the relationship between the vestibular system and the autonomic nervous system, which has been reviewed elsewhere in some detail (Bogle et al. 2022; Carter and Ray 2008; Furman et al. 1998; Kerman et al. 2000; Yates 1992; Yates et al. 2014; Yates and Bronstein 2005; Yates and Miller 1998). Briefly, there is some experimental and clinical evidence in humans that vestibular lesions can interfere with autonomic function (Carter and Ray 2008; Cooke et al. 2004; Jáuregui-Renaud et al. 2003; Kim et al. 2022; Radtke et al. 2003), though not all studies reach the same conclusion (Aghababaei Ziarati et al. 2020; Biaggioni et al. 1998; Heidenreich et al. 2009; Kalderon et al. 2023; Watenpaugh et al. 2002). It appears that autonomic dysfunction following vestibular lesions can recover quickly in some cases (Yates and Bronstein 2005). There is evidence that with ageing, the vestibular contribution to autonomic control declines (Monahan and Ray 2002; Ray and Monahan 2002; Sauder et al. 2008). A variety of neuroanatomical structures and pathways have been implicated in the relationship between vestibular function and autonomic function, including the medial vestibular nucleus, the rostral ventrolateral medulla and caudal medullary raphe nuclei to the preganglionic neurons in the thoracic spinal cord (Yates 1992), the insular cortex (Nagai et al. 2021) and regions in the medial aspect of the cerebellar cortex (Balaban and Porter 1998).
Some of these, and other neuroanatomical pathways are depicted in the Figure below from Yates and colleagues (Yates et al. 2014).
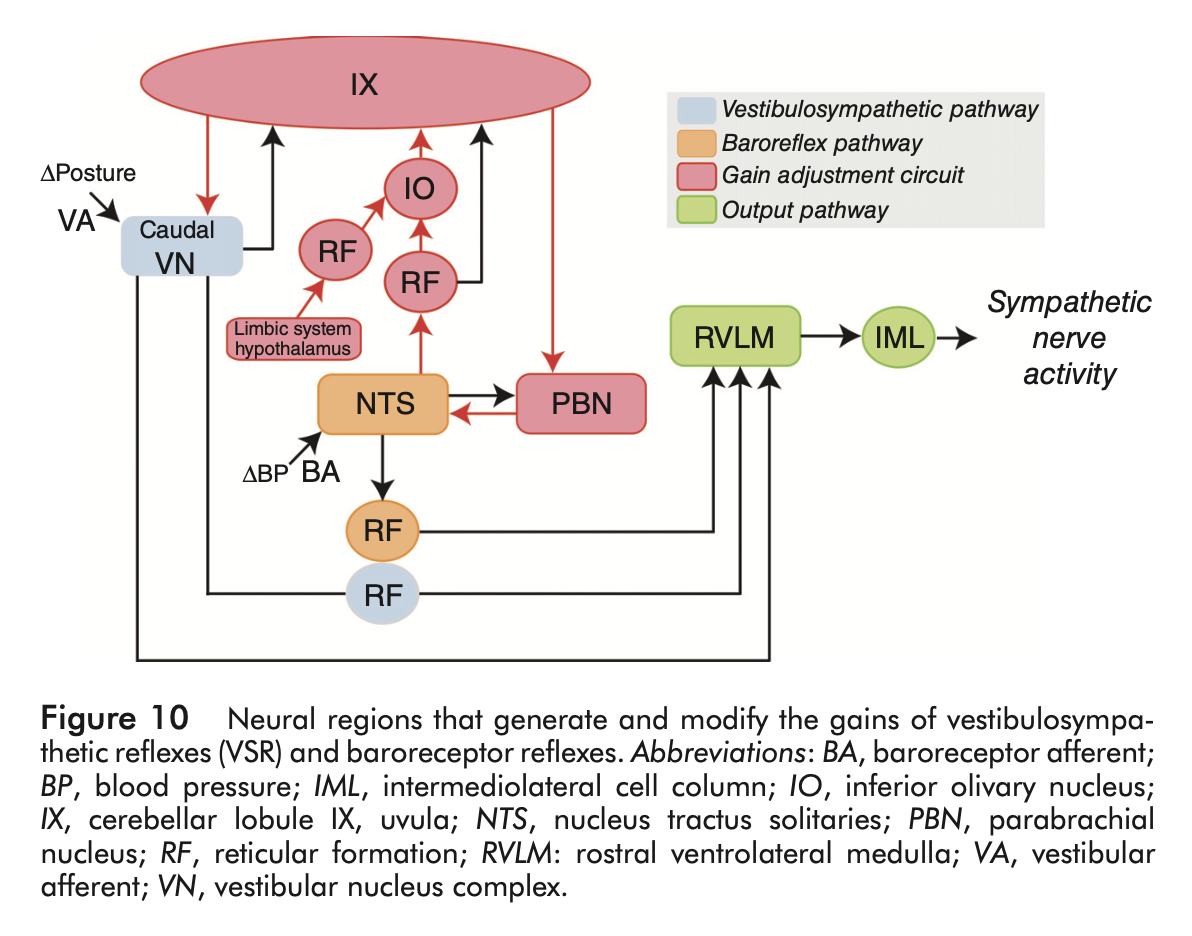
Clinical presentation
In order for vestibular-mediated syncope to occur, there must be a vestibular event of some kind. Absent such a vestibular event, it is difficult to make a convincing case that the syncope is mediated by the vestibular system.
Testing: vestibular
For patients in whom vestibular-mediated syncope is suspected, and whose cardiovascular workup has been unremarkable, it is medically reasonable to undertake a screening otovestibular workup with cervical vestibular evoked myogenic potentials, ocular vestibular evoked myogenic potentials, video head impulse testing and videonystagmography.
Testing: other
If a patient is referred for a clinical query of vestibular-mediated syncope but has not had a cardiac workup, then this should be considered, since primary cardiovascular causes of syncope are more common than vestibular ones. This aspect of the workup is usually undertaken by cardiology, and may include tilt table testing and cardiac event monitoring.
Differential diagnosis
As mentioned earlier, in order for vestibular-mediated syncope to occur, there must be a vestibular event of some kind. If there is no such vestibular event preceding the syncopal episode, then the differential diagnosis must include primary cardiac factors (arrhythmia, orthostatic intolerance, etc.).
Treatment and prognosis
If an adequate cardiovascular workup fails to reveal a cause for syncope, and if the history is compatible with vestibular syncope, and if an adequate otovestibular workup reveals evidence for vestibular disease, then it is medically reasonable to entertain a diagnosis of vestibular syncope.
There are no guidelines for treatment in this circumstance, but it logical to treat the identified vestibular disease, if such treatment exists.
The prognosis depends on the vestibular disease itself.
Final comments
Earlier we alluded to the logically plausible view that vestibular influences on the autonomic nervous system probably operate along a continuum. Thus, although we have been discussing vestibularly mediated syncope, it is plausible that vestibular dysfunction could provoke more modest autonomic changes that cause presyncope, rather than culminating in frank loss of consciousness. We speculate that this may be part of the mechanism by which diseases that usually cause sensations of rotation (such as benign paroxysmal positional vertigo or vestibular neuritis) may additionally provoke some degree of presyncope.
To this point our discussion has pertained to vestibular influences on the autonomic nervous system function. There is additionally some literature regarding autonomic etiologies of vestibular sensations (Nakagawa et al. 1993; Pappas 2003). Thus, there is the potential for a maladaptive positive feedback loop.
References
Aghababaei Ziarati M, Taziki MH, Hosseini SM (2020) Autonomic laterality in caloric vestibular stimulation. World J Cardiol 12: 144-154. doi: 10.4330/wjc.v12.i4.144
Balaban CD (1999) Vestibular autonomic regulation (including motion sickness and the mechanism of vomiting). Curr Opin Neurol 12: 29-33. doi: 10.1097/00019052-199902000-00005
Balaban CD, Porter JD (1998) Neuroanatomic substrates for vestibulo-autonomic interactions. J Vestib Res 8: 7-16.
Biaggioni I, Costa F, Kaufmann H (1998) Vestibular influences on autonomic cardiovascular control in humans. J Vestib Res 8: 35-41.
Bogle JM, Benarroch E, Sandroni P (2022) Vestibular-autonomic interactions: beyond orthostatic dizziness. Curr Opin Neurol 35: 126-134. doi: 10.1097/WCO.0000000000001013
Carter JR, Ray CA (2008) Sympathetic responses to vestibular activation in humans. Am J Physiol Regul Integr Comp Physiol 294: R681-8. doi: 10.1152/ajpregu.00896.2007
Cooke WH, Carter JR, Kuusela TA (2004) Human cerebrovascular and autonomic rhythms during vestibular activation. Am J Physiol Regul Integr Comp Physiol 286: R838-43. doi: 10.1152/ajpregu.00562.2003
Furman JM, Jacob RG, Redfern MS (1998) Clinical evidence that the vestibular system participates in autonomic control. J Vestib Res 8: 27-34.
Heidenreich KD, Weisend S, Fouad-Tarazi FM, White JA (2009) The incidence of coexistent autonomic and vestibular dysfunction in patients with postural dizziness. Am J Otolaryngol 30: 225-9. doi: 10.1016/j.amjoto.2008.04.011
Jáuregui-Renaud K, Hermosillo AG, Gómez A, Márquez MF, Cárdenas M, Bronstein AM (2003) Vestibular function interferes in cardiovascular reflexes [corrected]. Arch Med Res 34: 200-4. doi: 10.1016/s0188-4409(03)00023-7
Kalderon L, Chaimoff M, Katz-Leurer M (2023) The cardiac autonomic control system response to vestibular stimulation in subjects with BPPV compared with healthy controls. NeuroRehabilitation 52: 273-278. doi: 10.3233/nre-220021
Kerman IA, McAllen RM, Yates BJ (2000) Patterning of sympathetic nerve activity in response to vestibular stimulation. Brain Res Bull 53: 11-6. doi: 10.1016/s0361-9230(00)00303-8
Kim HA, Ahn J, Park HS, Lee SM, Choi SY, Oh EH, Choi JH, Kim JS, Choi KD (2021) Cardiogenic vertigo: characteristics and proposed diagnostic criteria. J Neurol 268: 1070-1075. doi: 10.1007/s00415-020-10252-4
Kim JG, Lee JH, Lee SU, Choi JY, Kim BJ, Kim JS (2022) Utricular dysfunction in patients with orthostatic hypotension. Clin Auton Res 32: 431-444. doi: 10.1007/s10286-022-00890-1
Kwon E, Lee JY, Kim HJ, Choi JY, Kim JS (2022a) Can Dyssynergia of Vestibulosympathetic and Baroreflexes Cause Vestibular Syncope? The Hypothesis Based on the Velocity-Storage Function. Cerebellum 21: 244-252. doi: 10.1007/s12311-021-01296-x
Kwon H, Kwon E, Kim HJ, Choi JY, Kim JS (2022b) Vestibular syncope: clinical characteristics and mechanism. Ann Clin Transl Neurol 9: 1616-1625. doi: 10.1002/acn3.51661
Monahan KD, Ray CA (2002) Vestibulosympathetic reflex during orthostatic challenge in aging humans. Am J Physiol Regul Integr Comp Physiol 283: R1027-32. doi: 10.1152/ajpregu.00298.2002
Nagai M, Scheper V, Lenarz T, Förster CY (2021) The insular cortex as a vestibular area in relation to autonomic function. Clin Auton Res 31: 179-185. doi: 10.1007/s10286-020-00744-8
Nakagawa H, Ohashi N, Kanda K, Watanabe Y (1993) Autonomic nervous system disturbance as etiological background of vertigo and dizziness. Acta Otolaryngol Suppl 504: 130-3. doi: 10.3109/00016489309128138
Pappas DG, Jr. (2003) Autonomic related vertigo. Laryngoscope 113: 1658-71. doi: 10.1097/00005537-200310000-00005
Previc FH (1993) Do the organs of the labyrinth differentially influence the sympathetic and parasympathetic systems? Neurosci Biobehav Rev 17: 397-404. doi: 10.1016/s0149-7634(05)80116-2
Pyykkö I, Manchaiah V, Zou J, Levo H, Kentala E (2017) Do patients with Ménière’s disease have attacks of syncope? J Neurol 264: 48-54. doi: 10.1007/s00415-017-8452-9
Pyykkö I, Manchaiah V, Zou J, Levo H, Kentala E (2018) Vestibular syncope: A disorder associated with drop attack in Ménière’s disease. Auris Nasus Larynx 45: 234-241. doi: 10.1016/j.anl.2017.03.023
Pyykkö I, Manchaiah V, Zou J, Levo H, Kentala E (2019) Association between Syncope and Tumarkin Attacks in Ménière’s Disease. J Int Adv Otol 15: 135-140. doi: 10.5152/iao.2019.6094
Radtke A, Popov K, Bronstein AM, Gresty MA (2003) Vestibulo-autonomic control in man: Short- and long-latency vestibular effects on cardiovascular function. J Vestib Res 13: 25-37.
Raphan T (2020) Vestibular, locomotor, and vestibulo-autonomic research: 50 years of collaboration with Bernard Cohen. J Neurophysiol 123: 329-345. doi: 10.1152/jn.00485.2019
Ray CA, Monahan KD (2002) Aging attenuates the vestibulosympathetic reflex in humans. Circulation 105: 956-61. doi: 10.1161/hc0802.104289
Sauder CL, Conboy EE, Chin-Sang SA, Ray CA (2008) Otolithic activation on visceral circulation in humans: effect of aging. Am J Physiol Renal Physiol 295: F1166-9. doi: 10.1152/ajprenal.90408.2008
Watenpaugh DE, Cothron AV, Wasmund SL, Wasmund WL, Carter R, 3rd, Muenter NK, Smith ML (2002) Do vestibular otolith organs participate in human orthostatic blood pressure control? Auton Neurosci 100: 77-83. doi: 10.1016/s1566-0702(02)00142-x
Yates BJ (1992) Vestibular influences on the sympathetic nervous system. Brain Res Brain Res Rev 17: 51-9. doi: 10.1016/0165-0173(92)90006-8
Yates BJ (1998) Autonomic reaction to vestibular damage. Otolaryngol Head Neck Surg 119: 106-12. doi: 10.1016/s0194-5998(98)70179-2
Yates BJ, Bolton PS, Macefield VG (2014) Vestibulo-sympathetic responses. Compr Physiol 4: 851-87. doi: 10.1002/cphy.c130041
Yates BJ, Bronstein AM (2005) The effects of vestibular system lesions on autonomic regulation: observations, mechanisms, and clinical implications. J Vestib Res 15: 119-29.
Yates BJ, Miller AD (1998) Physiological evidence that the vestibular system participates in autonomic and respiratory control. J Vestib Res 8: 17-25.
![]()