By Marcello Cherchi, MD PhD
For patients
Normal pressure hydrocephalus (NPH) is an uncommon condition in which certain fluid-filled spaces within the brain enlarge even though pressure within the skull remains normal. Most patients develop this in their 70s, and symptoms include some combination of cognitive impairment, difficulty walking, and urinary incontinence. The diagnosis is based on a combination of clinical criteria and findings on brain imaging. Some patients benefit from a neurosurgical procedure that involves placing a tube in the brain that allows the extra fluid to pass into the abdomen. Not all patients are good candidates for this procedure.
For clinicians
Overview
Idiopathic normal pressure hydrocephalus (NPH) is, as the name suggests, hydrocephalus with normal intracranial pressure. The mean age of onset is in the mid-70s. Symptoms begin insidiously and consist of some combination of (1) cognitive impairment, (2) gait impairment and (3) urinary incontinence. Most cases occur without any obvious trigger, while some appear to follow a specific neurological insult (such as head trauma or infection). The mechanism of the disease is unclear. It is also unclear whether any genetic factors are contributory. The tempo of symptoms is variable; in some patients it is progressive, while in others it is relatively static. Several radiographic diagnostic criteria have been proposed, but none widely accepted. Treatment is placement of a shunt (usually ventriculoperitoneal, less commonly lumboperitoneal), but response rates are variable, and response is often unsustained, thus prognosis is guarded. Nevertheless, NPH is important to recognize because it remains one of the few potentially treatable causes of gait impairment and cognitive decline.
Introduction
Neurosurgeon Dr. Salomon Hakim (1922 – 2011) (Pedro, Silva et al. 2019), neurologist Dr. Raymond Delacy Adams (1911 – 2008) (Fisher 2010) and colleagues published several papers in 1965 (Adams, Fisher et al. 1965, Hakim and Adams 1965) regarding a constellation of findings associated with radiographic hydrocephalus in patients whose intracranial pressure appeared normal.
 |
 |
In the first paper (Hakim and Adams 1965) they reported on three patients, each of whom exhibited three characteristics (sometimes referred to as “Hakim’s triad”), which they described as follows:
- “Impairment of consciousness” which “varied from mild inattentiveness with failure of daily events to be registered in mind and later retained, to a state of virtual coma. In this state the patient often was manifestly unreceptive of stimuli as well as unresponsive to them. The latter was at times more prominent than the former. The psychomotor retardation also varied in degree; when mild, a lack of impulsivity, expressed as apathy, disinterest, and lack of spontaneity, was associated with reduced levels of verbal and motorial function. When severe, it amounted to total immobility and absence of consecutive psychic activity, a state akin to akinetic mutism.”
- “Incontinence of sphincters” which “was essentially of cerebral type, the patient being unaware of the contents of bladder and bowel and incapable of making any arrangement for the somewhat precipitate action of these organs.”
- “Alteration of gait,” which “varied from a mild disorder in which locomotion appeared careless, the steps slow and somewhat shortened and the balance slightly uncertain, to one in which there was total incapacity to stand and walk. With the latter the plantar reflexes were usually extensor and the tendon reflexes brisk, particularly in the legs. An element of ataxia seemed to have been present but was always difficult to evaluate, and clearcut intention tremor and dysmetria were not demonstrable.”
They also specifically noted that, “the CSF pressure was indeed at all times within normal limits.”
Epidemiology
“The precise incidence and prevalence of INPH [idiopathic normal pressure hydrocephalus] are not known” (Relkin, Marmarou et al. 2005).
Mendes and colleagues (Mendes, de Oliveira et al. 2017) prospectively studied 30 patients who underwent placement of a Strata programmable valve ventriculoperitoneal shunt at a mean age of 77.1 years (77 years and 1 month). They reported that, “Symptoms of dementia had been present for 35 months before surgery on average, whereas the average onset of gait-related symptoms and urinary incontinence was 25 months and 22 months before surgery, respectively.” Thus, in this cohort the average age of symptom onset of dementia was 74 years and 2 months, of gait problems was 75 years, and urinary incontinence was 75 years and 3 months.
Genetics
Familial occurrence of NPH has been reported (Portenoy, Berger et al. 1984, Cusimano, Rewilak et al. 2011, Takahashi, Kawanami et al. 2011, McGirr and Cusimano 2012, Huovinen, Kastinen et al. 2016), but the extent to which genetic factors play a role in NPH remains unclear.
- Yang and colleagues reported that 15% of the unrelated NPH patients they analyzed had mutations in CWH43, which “modifies the lipid anchor of glycosylphosphatidylinositol-anchored proteins” (Yang, Lee et al. 2021).
- Morimoto and colleagues reported a loss-of-function variant in CFAP43 in a Japanese family with several members who had NPH (Morimoto, Yoshida et al. 2019).
- Korhonen and colleagues reported an expansion in C9ORF72 in 8/487 (1.6%) of patients with “possible idiopathic normal pressure hydrocephalus” (Korhonen, Remes et al. 2019). Mutations in the C9ORF72 gene have also been implicated in some cases of the frontotemporal dementia – amyotrophic lateral sclerosis complex.
- Sato and colleagues reported that of 50 patients with shunt-responsive idiopathic NPH, 26% had a “copy number loss in intron 2 of the SFMBT1 gene” (Sato, Takahashi et al. 2016).
Pathophysiological mechanism of disease
Much of the literature draws a distinction between idiopathic NPH (arising without any clear precipitant) and secondary NPH (occurring possibly as a consequence of some preceding insult such as trauma or infection).
Regarding the disorder of gait, in their original paper, Hakim and Adams (Hakim and Adams 1965) stated that, “One can only vaguely speculate in general terms that the dilatation of the ventricles subjects the major long tracts in the cerebral white matter and corpus callosum to compression and stretching, with a resulting physiological block of function which is to a considerable extent reversible when the pressure is reduced. This hypothesis would be in harmony with the observation that it is the white mater of the cerebrum which appears to yield maximally to great expansion of the ventricular system.”
Subsequent research suggested that:
“Electromyographic evidence reveals contraction of antagonistic muscle groups and abnormally increased activity in the antigravity muscles acting on hip and knee joints (2, 9). This suggests that the gait disorder of INPH is a disturbance in the phased activation of muscle groups, as would be seen in a disorder of subcortical motor control rather than a primary pyramidal tract disturbance. With progression of INPH and/or the evolution of extensive subcortical white matter changes, pyramidal tract involvement may become more likely” (Relkin, Marmarou et al. 2005)
Clinical presentation
The tempo of symptom evolution is variable:
“There is considerable variation in the nature, severity, and course of progression of the symptoms of INPH. The natural history of untreated INPH has not been well characterized, and it is not clear whether all cases ultimately progress, nor is the tempo of progression established for the majority of cases. Progression of symptoms is generally expected but may not be uniform over the course of the disease. Symptoms of INPH in the early stage and late stages of the disease may differ dramatically, as may symptoms in previously treated versus untreated patients” (Relkin, Marmarou et al. 2005).
The combination of symptoms, and the intensity of any given symptom, is also variable:
“All three components of the so-called classic triad of INPH need not be present for the diagnosis of INPH to be made. Gait disturbance tends to be the most readily recognized feature of INPH. Cognitive disturbances do not occur in all patients, and the severity of cognitive and motor symptoms does not necessarily correlate at baseline or progress in parallel. Incontinence in INPH is difficult to distinguish from urinary symptoms associated with other common disorders. The signs and symptoms of INPH are typically bilateral but may appear lateralized when superimposed on coexisting conditions, such as stroke, radiculopathy, and peripheral neuropathy” (Relkin, Marmarou et al. 2005).
Regarding the quality of gait impairment:
“The pattern of gait seen in INPH patients has been variably described as ‘apractic,’ ‘bradykinetic,’ ‘glue-footed,’ ‘magnetic,’ ‘parkinsonian,’ ‘short-stepped,’ and ‘shuffling.’ Gait problems may emerge as difficulty in ascending or descending stairs and curbs and in walking at the expected pace. Patients may complain of difficulty rising from a chair, ‘give-way’ weakness of the lower extremities, and fatigue brought on by walking. As the disease progresses, turning in place becomes tenuous and typically requires multiple steps (en bloc)” (Relkin, Marmarou et al. 2005).
Physical examination
Regarding further specific characteristics of gait:
“Computerized analysis of the gait of INPH patients has shown 1) reduced cadence, 2) decreased step height, and 3) 8reduced counterrotation of the shoulders relative to the pelvis during walking. The upward angular movement of the foot during stepping is inadequate in INPH, and stride length is diminished. The step width and foot rotation angles are reportedly increased, and there is less step-to-step variability in these parameters than in normal controls” (Relkin, Marmarou et al. 2005).
Regarding further characteristics of stance:
“The stance in INPH may be more forward leaning than in healthy normal individuals, and INPH patients tend to show a wider sway and imbalance that may be accentuated by eye closure” (Relkin, Marmarou et al. 2005).
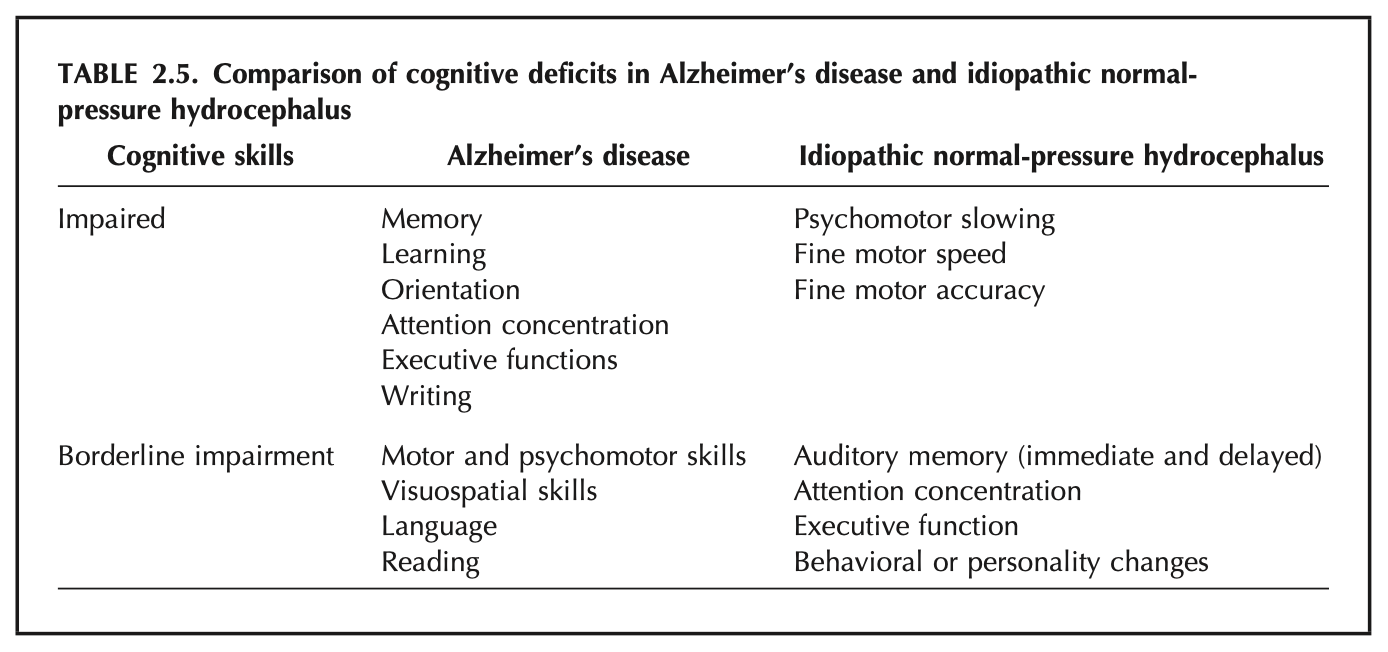
Regarding cognition, Relkin and colleagues (Relkin, Marmarou et al. 2005) state that, “The principal cognitive symptoms seen in INPH are suggestive of a subcortical dementing process, including slowing of thought, inattentiveness, and apathy, as well as encoding and recall problems,” and they offer the following Table comparing the cognitive deficits of NPH and Alzheimer’s.
Testing: vestibular
Vestibular testing does not play a significant role in confirming a diagnosis of NPH. Interestingly, Inui and colleagues (Inui, Haginomori et al. 2022) report that 25% of NPH patients exhibit a shift in the tuning curve of cervical vestibular evoked myogenic potentials, which they interpret reflecting that in hydrocephalus the “excessive CSF accumulation propagated to the endolymph and perilymph.”
The main value of vestibular testing lies in excluding competing causes of chronic disequilibrium (such as vestibular weakness).
Imaging
Several radiologic criteria have been studied for the diagnosis of NPH (Ryska, Slezak et al. 2020, Thavarajasingam, El-Khatib et al. 2023), but none has gained wide acceptance.
Differential diagnosis
Various diagnostic criteria have been proposed for NPH (Vanneste, Augustijn et al. 1993, Boon, Tans et al. 2000, Krauss and Halve 2004, Halperin, Kurlan et al. 2015, Mendes, de Oliveira et al. 2017), but none has been widely accepted.
Relkin and colleagues (Relkin, Marmarou et al. 2005) state that idiopathic NPH “should be considered in the differential diagnosis of any unexplained disturbance of gait, continence, and cognition that begins insidiously in an adult. This is most commonly a gait disturbance, followed in frequency by cognitive impairments and least often, urinary incontinence.”
Relkin and colleagues (Relkin, Marmarou et al. 2005) provide the extensive differential diagnosis of NPH in the Table below.

Table : Differential diagnosis of normal pressure hydrocephalus. From Reskin et al. (2005).
Treatment
The usual treatment of NPH is placement of a ventriculoperitoneal shunt — less commonly a lumboperitoneal shunt (Liu and Su 2020) — but these procedures come with risk, and response rates are often unsatisfactory (see below). Consequently, the question becomes one of trying to predict which patients are likely to respond to shunting, and guidance on patient selection is weak (Halperin, Kurlan et al. 2015).
Boon and colleagues (Boon, Tans et al. 2000) concluded that CSF outflow resistance was the best predictor of shunt responsiveness.
Thavarajasingam and colleagues (Thavarajasingam, El-Khatib et al. 2023) conducted a systematic literature review and meta-analysis and concluded that no radiologic criterion can serve as a sole predictor of responsiveness to shunting.
Vanneste and colleagues (Vanneste, Augustijn et al. 1993) recommended using a combination of radiologic and clinical criteria for predicting responsiveness to shunting.
Prognosis
The lack of widely accepted diagnostic criteria has been an impediment to interpreting the results of treatment trials. Response rates are variable, but difficult to compare across studies given the variability in diagnostic criteria they employ.
Relkin and colleagues (Relkin, Marmarou et al. 2005) comment that, “On the basis of clinical presentation alone, evidence shows that favorable response to shunting will vary from 46 to 63%.”
Kahlon and colleagues (Kahlon, Sjunnesson et al. 2007) studied 54 NPH patients who underwent shunting and reported that, “At the 6-month follow-up examination, 83% of the operated patients improved in gait, 65% improved in reaction time, 46% improved in memory, and 31% improved in identical forms tests; 96% found themselves subjectively improved.”
Trungu and colleagues (Trungu, Scollato et al. 2022) studied 181 NPH patients who underwent ventriculoperitoneal shunting and reported an impressive 91.2% of patients exhibited improvement after 12 months.
Even in those patients who do appear to exhibit an initial response to shunting, the improvement is usually not sustained. Kahlon and colleagues (Kahlon, Sjunnesson et al. 2007) comment that, “only 20% of the operated patients demonstrated continued functional improvements after 5 years.”
References
Adams RD, Fisher CM, Hakim S, Ojemann RG, Sweet WH (1965) Symptomatic Occult Hydrocephalus with “Normal” Cerebrospinal-Fluid Pressure. A Treatable Syndrome. N Engl J Med 273: 117-26. doi: 10.1056/NEJM196507152730301
Boon AJ, Tans JT, Delwel EJ, Egeler-Peerdeman SM, Hanlo PW, Wurzer HA, Hermans J (2000) The Dutch normal-pressure hydrocephalus study. How to select patients for shunting? An analysis of four diagnostic criteria. Surg Neurol 53: 201-7. doi: 10.1016/s0090-3019(00)00182-8
Cusimano MD, Rewilak D, Stuss DT, Barrera-Martinez JC, Salehi F, Freedman M (2011) Normal-pressure hydrocephalus: is there a genetic predisposition? Can J Neurol Sci 38: 274-81. doi: 10.1017/s031716710001146x
Fisher CM (2010) Dr. Raymond Delacy Adams (1911-2008): an appreciation. Neurologist 16: 141-2. doi: 10.1097/NRL.0b013e3181c9280a
Hakim S, Adams RD (1965) The special clinical problem of symptomatic hydrocephalus with normal cerebrospinal fluid pressure. Observations on cerebrospinal fluid hydrodynamics. J Neurol Sci 2: 307-27. doi: 10.1016/0022-510x(65)90016-x
Halperin JJ, Kurlan R, Schwalb JM, Cusimano MD, Gronseth G, Gloss D (2015) Practice guideline: Idiopathic normal pressure hydrocephalus: Response to shunting and predictors of response: Report of the Guideline Development, Dissemination, and Implementation Subcommittee of the American Academy of Neurology. Neurology 85: 2063-71. doi: 10.1212/WNL.0000000000002193
Huovinen J, Kastinen S, Komulainen S, Oinas M, Avellan C, Frantzen J, Rinne J, Ronkainen A, Kauppinen M, Lonnrot K, Perola M, Pyykko OT, Koivisto AM, Remes AM, Soininen H, Hiltunen M, Helisalmi S, Kurki M, Jaaskelainen JE, Leinonen V (2016) Familial idiopathic normal pressure hydrocephalus. J Neurol Sci 368: 11-8. doi: 10.1016/j.jns.2016.06.052
Inui T, Haginomori SI, Kajimoto Y, Kuriyama T, Shirai T, Kinoshita I, Araki M, Kawata R (2022) Asymmetry and tuning shift of the cervical vestibular evoked myogenic potential indicate saccular dysfunction in idiopathic normal pressure hydrocephalus. Clin Neurophysiol 134: 43-49. doi: 10.1016/j.clinph.2021.12.003
Kahlon B, Sjunnesson J, Rehncrona S (2007) Long-term outcome in patients with suspected normal pressure hydrocephalus. Neurosurgery 60: 327-32; discussion 332. doi: 10.1227/01.NEU.0000249273.41569.6E
Korhonen VE, Remes AM, Helisalmi S, Rauramaa T, Sutela A, Vanninen R, Suhonen NM, Haapasalo A, Hiltunen M, Jaaskelainen JE, Soininen H, Koivisto AM, Leinonen V (2019) Prevalence of C9ORF72 Expansion in a Large Series of Patients with Idiopathic Normal-Pressure Hydrocephalus. Dement Geriatr Cogn Disord 47: 91-103. doi: 10.1159/000497306
Krauss JK, Halve B (2004) Normal pressure hydrocephalus: survey on contemporary diagnostic algorithms and therapeutic decision-making in clinical practice. Acta Neurochir (Wien) 146: 379-88; discussion 388. doi: 10.1007/s00701-004-0234-3
Liu JT, Su PH (2020) The efficacy and limitation of lumboperitoneal shunt in normal pressure hydrocephalus. Clin Neurol Neurosurg 193: 105748. doi: 10.1016/j.clineuro.2020.105748
McGirr A, Cusimano MD (2012) Familial aggregation of idiopathic normal pressure hydrocephalus: novel familial case and a family study of the NPH triad in an iNPH patient cohort. J Neurol Sci 321: 82-8. doi: 10.1016/j.jns.2012.07.062
Mendes GAS, de Oliveira MF, Pinto FCG (2017) The Timed Up and Go Test as a Diagnostic Criterion in Normal Pressure Hydrocephalus. World Neurosurg 105: 456-461. doi: 10.1016/j.wneu.2017.05.137
Morimoto Y, Yoshida S, Kinoshita A, Satoh C, Mishima H, Yamaguchi N, Matsuda K, Sakaguchi M, Tanaka T, Komohara Y, Imamura A, Ozawa H, Nakashima M, Kurotaki N, Kishino T, Yoshiura KI, Ono S (2019) Nonsense mutation in CFAP43 causes normal-pressure hydrocephalus with ciliary abnormalities. Neurology 92: e2364-e2374. doi: 10.1212/WNL.0000000000007505
Pedro MKF, Silva J, Rocha S, Germiniani FMB, Ramina R, Teive HAG, Souza RKM (2019) Salomon Hakim: the man behind normal pressure hydrocephalus. Arq Neuropsiquiatr 77: 746-748. doi: 10.1590/0004-282X20190096
Portenoy RK, Berger A, Gross E (1984) Familial occurrence of idiopathic normal-pressure hydrocephalus. Arch Neurol 41: 335-7. doi: 10.1001/archneur.1984.04050150117029
Relkin N, Marmarou A, Klinge P, Bergsneider M, Black PM (2005) Diagnosing idiopathic normal-pressure hydrocephalus. Neurosurgery 57: S4-16; discussion ii-v. doi: 10.1227/01.neu.0000168185.29659.c5
Ryska P, Slezak O, Eklund A, Malm J, Salzer J, Zizka J (2020) Radiological markers of idiopathic normal pressure hydrocephalus: Relative comparison of their diagnostic performance. J Neurol Sci 408: 116581. doi: 10.1016/j.jns.2019.116581
Sato H, Takahashi Y, Kimihira L, Iseki C, Kato H, Suzuki Y, Igari R, Sato H, Koyama S, Arawaka S, Kawanami T, Miyajima M, Samejima N, Sato S, Kameda M, Yamada S, Kita D, Kaijima M, Date I, Sonoda Y, Kayama T, Kuwana N, Arai H, Kato T (2016) A Segmental Copy Number Loss of the SFMBT1 Gene Is a Genetic Risk for Shunt-Responsive, Idiopathic Normal Pressure Hydrocephalus (iNPH): A Case-Control Study. PLoS One 11: e0166615. doi: 10.1371/journal.pone.0166615
Takahashi Y, Kawanami T, Nagasawa H, Iseki C, Hanyu H, Kato T (2011) Familial normal pressure hydrocephalus (NPH) with an autosomal-dominant inheritance: a novel subgroup of NPH. J Neurol Sci 308: 149-51. doi: 10.1016/j.jns.2011.06.018
Thavarajasingam SG, El-Khatib M, Vemulapalli K, Iradukunda HAS, K SV, Borchert R, Russo S, Eide PK (2023) Radiological predictors of shunt response in the diagnosis and treatment of idiopathic normal pressure hydrocephalus: a systematic review and meta-analysis. Acta Neurochir (Wien) 165: 369-419. doi: 10.1007/s00701-022-05402-8
Trungu S, Scollato A, Ricciardi L, Forcato S, Polli FM, Miscusi M, Raco A (2022) Clinical Outcomes of Shunting in Normal Pressure Hydrocephalus: A Multicenter Prospective Observational Study. J Clin Med 11. doi: 10.3390/jcm11051286
Vanneste J, Augustijn P, Tan WF, Dirven C (1993) Shunting normal pressure hydrocephalus: the predictive value of combined clinical and CT data. J Neurol Neurosurg Psychiatry 56: 251-6. doi: 10.1136/jnnp.56.3.251
Yang HW, Lee S, Yang D, Dai H, Zhang Y, Han L, Zhao S, Zhang S, Ma Y, Johnson MF, Rattray AK, Johnson TA, Wang G, Zheng S, Carroll RS, Park PJ, Johnson MD (2021) Deletions in CWH43 cause idiopathic normal pressure hydrocephalus. EMBO Mol Med 13: e13249. doi: 10.15252/emmm.202013249
![]()