By Marcello Cherchi, MD PhD
For patients
Vestibular neuritis (VN), also called vestibular neuronitis, is thought to be due to damage of the vestibular nerve (the “balance-related nerve” between the ear and the brain) from a bout of inflammation, though the cause of that inflammation remains unknown despite considerable research. Suspicion for vestibular neuritis is based on the clinical history, and can be supported by findings on physical examination and various otovestibular tests. The standard of care for vestibular neuritis is appropriately targeted vestibular rehabilitation therapy, though even with such therapy, not all patients improve. Medications play little to no role in treatment. Recurrence of vestibular neuritis is somewhat uncommon, though an ear affected by vestibular neuritis is more susceptible to other problems, the most common of which is benign paroxysmal positional vertigo.
For clinicians
Overview
Vestibular neuritis (VN), also called vestibular neuronitis, refers to damage of the vestibular nerve from a bout of inflammation whose cause is unknown. VN usually presents abruptly, but occasionally can have a more gradual or stuttering onset. Symptoms consist of disequilibrium that often (though not always) involves a spinning sensation, nausea and vomiting, less commonly aural pain or pressure. VN can happen at any age but peaks in the 3rd – 6thdecades. Otovestibular testing (vestibular evoked myogenic potentials, video head impulse testing, caloric testing) shows unilateral vestibular hypofunction. Acute management (in the first 1 – 3 days) is with vestibular suppressants and anti-emetics, but the standard of care for subsequent management is appropriately targeted vestibular rehabilitation therapy. Prognosis is extremely variable in terms of the time required for recovery. The rate of true recurrence is low (about 2% over 10 years).
§1: Introduction
§1.1: What is vestibular neuritis?
Most literature credits Margaret Ruth Dix (1902 – 1991) and Charles Skinner Hallpike (1900 – 1979) in 1952 (Dix and Hallpike 1952) as having provided the first complete description of vestibular neuritis (VN), sometimes also referred to as vestibular neuronitis, as a discrete clinicopathological entity. Some commentators (Bae et al. 2022; Strupp et al. 2022) note that other investigators provided similar descriptions a few decades earlier, including Erich Ruttin (1880 – 1940) (Ruttin 1909) and Carl Olof Siggesson Nylen (1882 – 1978) (Nylén 1924).
Vestibular neuritis is currently suspected to be an inflammatory-mediated insult (damage) to the balance-related nerve (vestibular nerve) between the ear and the brain that manifests with abrupt-onset, severe dizziness that lasts days to weeks, and occasionally recurs.
Although vestibular neuritis is usually regarded as a process affecting the vestibular nerve itself, damage restricted to the vestibule (balance components of the inner ear) would manifest clinically in a similar way, and might be termed “vestibulitis,” although that term is seldom applied (Izraeli, Rachmel et al. 1989). Thus, distinguishing between “vestibular neuritis” (inflammation of the vestibular nerve) and “vestibulitis” (inflammation of the balance-related components of the inner ear) would be difficult.
§1.2: What is the cause of vestibular neuritis?
The short answer is that the underlying cause of vestibular neuritis is unknown. Viral and vascular causes have been proposed, but reviews of this literature reveal the supporting evidence to be circumstantial at best (Nadol 1995, Strupp and Brandt 2009). Placebo-controlled trials attempting to treat for a presumed viral cause (herpes simplex 1 infection) showed no difference in outcomes (Strupp, Zingler et al. 2004).
There is a modest literature regarding a possible relationship between ganglioside autoantibodies and vestibular neuritis. Gangliosides are glycosphingolipid structures bearing one or more sialic acid residues; these are most abundantly expressed in the central nervous system (Schnaar 2019), and are “believed to play a role in neuroplasticity through influences on neurogenesis, synaptic plasticity, neurotransmission and axonal growth. Furthermore, they also mediate neuronal differentiation, maintenance and repair” (Wanleenuwat et al. 2020). A group of researchers has reported a case (Lee et al. 2019b), a first series of 90 patients (Lee et al. 2019a), and a second series of 105 patients (Kim et al. 2023) of vestibular neuritis (AUPV, “acute unilateral peripheral vestibulopathy”), in a subset of whom anti-ganglioside antibodies (anti-GQ1b IgG or IgM; anti-GM1 IgM; anti-GD1a IgG) were identified. They reported a positivity rate of 10% (Lee et al. 2019a) to 11% (Kim et al. 2023). They conclude that, “The causal relationship between antiganglioside antibodies and AUPV may be inferred by the negative seroconversion of antibodies after symptom resolution” (Kim et al. 2023), yet in their discussion of previously proposed mechanisms (reactivation of type 1 herpes simplex 1 virus, vascular causes) they comment that, “An immune-mediated dysregulation may play a significant role following either cascade of vestibular failure” which (contrary to their postulated causal relationship) suggests that the presence of ganglioside autoantibodies occurs in response to some primary vestibular lesion. Another interpretation of these findings is that in 10% – 11% of vestibular neuritis patients, axonal or neuronal damage (from some as yet undiscovered etiology) exposes ganglioside antigens against which a maladaptive autoimmune response is mounted. We would be curious to know whether the presence of ganglioside autoantibodies increases the risk of subsequently developing recurrent vestibular neuritis (whether ipsilateral or contralateral), or increases the risk of sympathetic ear disease.
§2: How does vestibular present?
Most cases of vestibular neuritis present as a spinning sensation that develops quite rapidly, usually over minutes (less commonly over hours), and is usually associated with nausea and vomiting. Less commonly the disease may present with more of a “sputtering” onset (with brief bursts of dizziness over several days before the continuous symptoms begin), or the sensation may include some sensation other than spinning (such as wobbling, drifting, tilting, etc.).
In some cases of vestibular neuritis patients experience pressure, pain or other discomfort in the affected ear. The mechanism for this sensation is unclear. One possibility is that there are somatosensory fibers (mediating sensations of touch, pain, etc.) in the vestibular nerve, though this has not been described. Another possibility is that the nervus intermedius of Wrisberg, which provides somatosensory innervation to portions of the ear and is neuroanatomically in very close proximity to the vestibular nerve (Tubbs, Steck et al. 2013), is simultaneously affected by whatever inflammatory process causes vestibular neuritis. Another example of multiple adjacent neural structures being affected is Ramsay-Hunt syndrome (Hunt 1907, Kuhweide, Van de Steene et al. 2002), which affects both the vestibulocochlear nerve and the facial nerve.
Epidemiologic studies (Sekitani, Imate et al. 1993) suggest that the annual incidence of vestibular neuritis is 3.5 per 100,000 people (Strupp and Brandt 2009). Vestibular neuritis is among the most frequently diagnosed conditions in clinics specializing in dizziness. Vestibular neuritis can occur at any age, but peak incidence is in the 3rd – 6thdecades, as shown in the histogram below from Sekitani et al (Sekitani, Imate et al. 1993).

Labyrinthitis is similar to vestibular neuritis in terms of the dizziness/imbalance it causes, but it is different in that labyrinthitis additionally involves auditory symptoms (hearing loss and sometimes tinnitus or ear fullness) on one side; as such, it can be difficult to distinguish labyrinthitis from an initial bout of Ménière’s disease.
§3: Diagnosis
§3.1: How vestibular neuritis diagnosed?
Vestibular neuritis can be suspected based on a compatible clinical history.
Suspicion for vestibular neuritis increases when otovestibular testing (see below) provides evidence of a vestibular deficit on one side, and there is no evidence for a better explanation of the symptoms.
§3.2: What tests help diagnose vestibular neuritis?
In order to detect the vestibular (balance-related) deficits of vestibular neuritis, several tests may be needed. The reason is that there are multiple components of the inner ear that detect movement, each of which sends signals through a different bundle of fibers in the vestibular nerve; there does not currently exist a test to assess these components “all at once,” rather, there are individual tests for each component.
Briefly, each labyrinth includes five “organelles” that detect motion – specifically, three semicircular canals (the anterior/superior canal, the lateral/horizontal canal, and the posterior/inferior canal) that detect angular acceleration, and two otolith organs (the saccule and utricle) that detect linear acceleration.
Signals from each of these organelles travel along discrete fibers within the vestibular nerve to reach several vestibular nuclei in the brainstem. The microstructural anatomy of the vestibular nerve is difficult to analyze due to a variety of factors – including neuronal anastomoses with adjacent structures (Ozdogmus, Sezen et al. 2004), the gradual rotation in the disposition of the fibers along the course of the nerve (Silverstein, Norrell et al. 1986), the variable orientation between individuals (Rasmussen 1940), variability between genders (Moriyama, Itoh et al. 2007) and the attrition of nerve fibers with age (Bergström 1973, Nagai, Goto et al. 1999) – but a number of painstaking studies on human specimens have been conducted, such as Lee and Suarez’ study of 3 temporal bones (Lee, Suarez et al. 1990), and Lopez et al’s study of 13 temporal bones (Lopez, Ishiyama et al. 2005), and Bergstrom’s study of 40 human temporal bones (Bergström 1973).
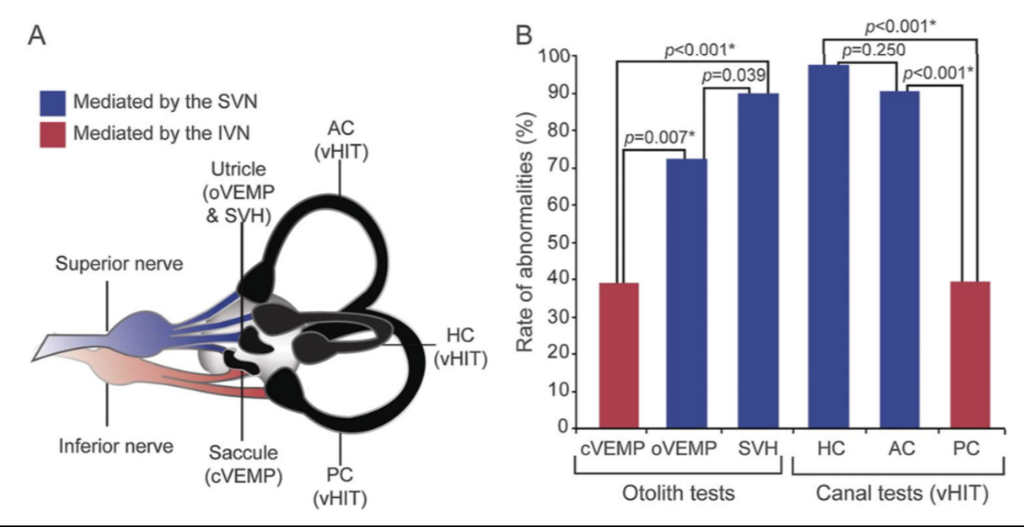
In vestibular neuritis, various subsets of the afferent fibers within the vestibular nerve suffer damage. Some studies (Jeong, Kim et al. 2013, Buki, Hanschek et al. 2017) oversimplify this (perhaps for didactic purposes) by describing vestibular neuritis that involves either the “superior division” of the vestibular nerve, or the “inferior division” of the vestibular nerve, or both, and conclude with observations that most cases involve either the superior division, or both the superior and inferior divisions (Taylor, McGarvie et al. 2016), while it is rare (2.3% (Taylor, McGarvie et al. 2016) to ≈5% (Kim and Kim 2012, Magliulo, Gagliardi et al. 2014, Magliulo, Iannella et al. 2015)) for only the inferior division to be involved. However, careful studies show that this pathology can affect nearly any combination of the subsets of fibers (Taylor, McGarvie et al. 2016, Yacovino, Zanotti et al. 2021). Taylor and McGarvie studied 43 patients with vestibular neuritis who had a fairly complete set of vestibular tests and found the data in the Figurebelow.
A slightly larger stud (59 patients) by Navari and Casani (Navari and Casani 2020) described fairly similar results, as shown in the Figure below.
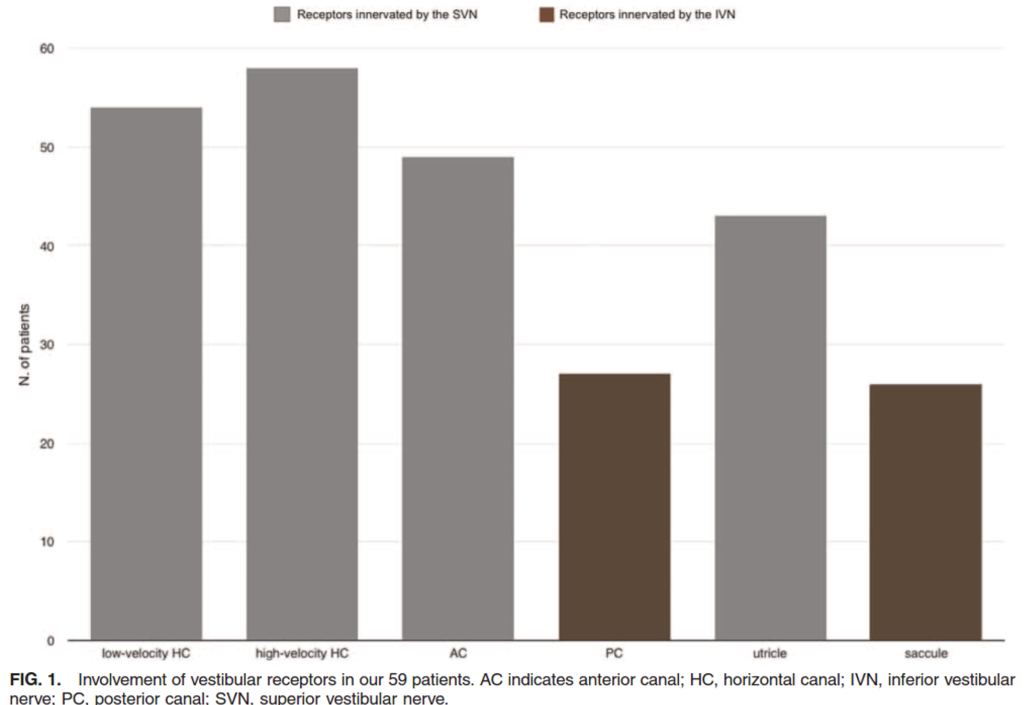
These data suggest that in cases of vestibular neuritis, the horizontal canal (or its afferent pathways) is most commonly affected (90% – 97.7%), the saccule (or its afferent pathways) is least commonly affected (39.0% – 44%), whereas the other structures (anterior canal, utricle, posterior canal) exhibit an intermediate level of affectation. The relative ordering of affectation is fairly consistent, but not perfectly so (Cherchi and Yacovino 2021).
Given this variability, and given that all tests are fallible (meaning that any test can have false positives and false negatives), we generally try not to base the diagnosis on a single test result in isolation; confidence in the diagnosis increases when multiple independent data points (from separate tests) support the same diagnosis. Tests commonly used in diagnosing vestibular neuritis include cervical vestibular evoked myogenic potentials (cVEMPs), ocular vestibular evoked myogenic potentials (oVEMPs), videonystagmography (VNG) and video head impulse testing (vHIT); this battery is a reasonable selection that assesses many of the inner ear’s “balance functions,” but it still does not provide a complete picture thereof. Research is exploring other methods of assessing vestibular neuritis (Cherchi 2019).
Occasionally, MRI of the brain and internal auditory canals without and with contrast shows enhancement of the vestibular nerve (Karlberg, Annertz et al. 2004, Fundakowski, Anderson et al. 2012, Venkatasamy, Huynh et al. 2019), though larger studies did not find this (Hasuike, Sekitani et al. 1995, Strupp, Jager et al. 1998), suggesting that this finding is uncommon. There is debate regarding which MRI protocol is optimal for detection (Hu et al. 2025). When enhancement of the vestibular nerve is observed (without any increased “bulk” of the nerve), it can be viewed as compatible with vestibular neuritis, though is not required for the diagnosis.
§4: Treatment, prognosis and recurrence
§4.1: How do you treat the dizziness/imbalance symptoms resulting from vestibular neuritis?
During the first few days of vestibular neuritis, management is supportive, sometimes including anti-emetics (such as ondansetron, metoclopramide or promethazine) and vestibular suppressants (such as meclizine or clonazepam), but these medications play no role beyond the first few days.
Meta-analyses of treatment trials (McDonnell and Hillier 2015) conclude that there is moderate to strong evidence that appropriately targeted vestibular rehabilitation therapy is safe and effective for vestibular neuritis, thus this treatment modality is generally regarded as the standard of care.
Physical therapy is thought to accelerate recovery by promoting sensory substitution and central compensation for the vestibular deficit (Strupp and Brandt 2009). Physical therapy usually is usually initiated with gaze stabilization exercises (Meldrum and Jahn 2019), but evolves depending on an individual’s progress.
There is some evidence that the earlier the vestibular rehabilitation therapy is started, the greater the likelihood that it will improve specific physical examination findings in vestibular neuritis (Michel, Laurent et al. 2019).
Note that even with appropriately-targeted vestibular rehabilitation therapy, recovery may be experienced as “uneven,” in the sense that from day to day there may be fluctuation in the intensity of baseline symptoms, though when observing symptoms over a longer time frame (typically weeks) the overall trajectory is one of improvement.
§4.2: Are medications used in the treatment of vestibular neuritis?
Aside from providing supportive care during the first few days of symptoms (see above), there is not yet any consistent evidence that medications play any role in the long-term management of vestibular neuritis.
There have been several trials of the use of steroids in the treatment of vestibular neuritis. For example, Strupp and colleagues (Strupp et al. 2004) concluded that starting steroids (specifically, methylprednisolone) within 3 days of symptom onset improved outcomes at 12 months — though the outcome measure was the degree of improvement in the caloric response (not in any measure of symptoms or level of patient function). Similarly, a trial by Kim and colleagues (Kim et al. 2022) reported improvement in caloric response, but no difference in symptomatic recovery as measured by the dizziness handicap inventory. In other words, these trials report that steroids improve a test result, but do not appear to make any difference in a patient’s level of symptoms, or any outcome of function.
Some trials report that treatment with steroids may have some short-term benefit, but no difference in prognosis or long-term outcomes (Goudakos et al. 2014; Ismail et al. 2019). Studies of vestibular rehabilitation therapy PLUS steroids found that the adjunctive steroids conferred no additional long-term benefit (Goudakos et al. 2014; Ismail et al. 2019).
Other trials were unable to detect even an improvement in caloric responses (Yoo et al. 2017). Consequently, most meta-analyses conclude that on the whole, the evidence does not support treatment of vestibular neuritis with steroids (Fishman et al. 2011; Leong et al. 2021; Yoo et al. 2017).
The above studies suggest that evidence supporting the use of steroids in the treatment of vestibular neuritis, while not completely absent, is at best weak and conflicting. In view of those conclusions, and in view of the non-trivial risks incurred by the use of steroids, we do not see a compelling rationale for treating vestibular neuritis with steroids.
§4.3: What is the prognosis of vestibular neuritis?
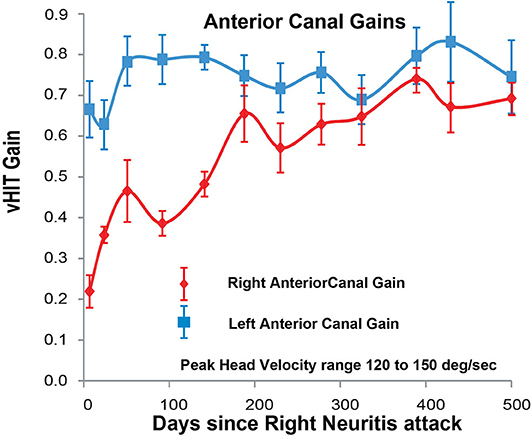
From a physiologic perspective, it appears that vestibular responses can improve, though may not fully recover. This can be detected on serial measurements of various vestibular tests. For example, on video head impulse testing, the gain may improve over time.
The Figure below, from McGarvie and colleagues (McGarvie et al. 2020), shows the gain over time (measured on 12 occasions over 500 days) of the right anterior canal (affected by right-sided vestibular neuritis) and the left anterior canal (healthy). The tracings show clear improvement (approaching normal values) in gain on the side affected by vestibular neuritis.
Measurable physiologic improvement is interesting, but most patients diagnosed with vestibular neuritis understandably want to know, “When will I get better?” Unfortunately, this is a difficult question to answer because the symptom of dizziness is hard to measure and therefore hard to study. In fact, most research of the “prognosis” of vestibular neuritis study an objective (numeric) measure such as a test result, rather than a patient’s complaint of a greater or lesser degree of severity of symptoms. For instance, the carefully conducted study by Strupp et al (Strupp, Zingler et al. 2004) measured the results of caloric testing (a subset of videonystagmography) rather than patient symptoms.
Many studies of vestibular neuritis comment that symptoms “usually” resolve within days to weeks (Kim, Kim et al. 2011). However, there are also studies documenting a less favorable prognosis. One study suggested that up to 30 – 40% of patients experience persistent dizziness after vestibular neuritis (Neuhauser 2007). Another study concluded that up to 43.3% of patients remain at least somewhat symptomatic at 5 – 10 years (Okinaka, Sekitani et al. 1993). The underlying reason for this variability in outcomes is unknown. Few studies have identified prognostic factors; Armato and colleagues (Armato et al. 2024) identified vascular risk factors (specifically hypercholesterolemia) as a poor prognostic factor.
Vestibular neuritis does not usually recur (see below). However, an ear affected by vestibular neuritis is at higher risk for developing other vestibular disorders, particularly benign paroxysmal positional vertigo (BPPV). Studies conclude that after a bout of vestibular neuritis, 9.8% (Mandala, Santoro et al. 2010) to 15.3% (Kim, Kim et al. 2011)developed benign paroxysmal positional vertigo within about 6 years.
§4.4: Does vestibular neuritis recur?
Vestibular neuritis can recur, but is thought to be uncommon. Most studies report recurrence rates of 1.9% – 2% over 10 years (Huppert et al. 2006; Mandala et al. 2010). Other studies cite rates of recurrence as high as 10.7% (Kim et al. 2011). In some cases of vestibular neuritis the recurrence affects the same ear (Emiliano De Schutter and Perez Fernandez 2017) In other cases of recurrent vestibular neuritis, the recurrence affects “the other ear” (i.e., the originally unaffected ear); this is called “bilateral sequential vestibular neuritis” (Lee et al. 2026; Ogata et al. 1993; Schuknecht and Witt 1985; Yacovino et al. 2018).
References
Armato E, Dumas G, Perottino F, Casteran M, Perrin P (2024) Determination of Recovery by Total Restitution or Compensation Using Multifrequency Vestibular Tests and Subjective Functional Scales in a Human Model of Vestibular Neuritis. Audiol Res 14: 958-982. doi: 10.3390/audiolres14060080
Bae CH, Na HG, Choi YS (2022) Current diagnosis and treatment of vestibular neuritis: a narrative review. J Yeungnam Med Sci 39: 81-88. doi: 10.12701/yujm.2021.01228
Bergström B (1973) Morphological Studies of the Vestibular Nerve, University of Uppsala
Buki B, Hanschek M, Junger H (2017) Vestibular neuritis: Involvement and long-term recovery of individual semicircular canals. Auris Nasus Larynx 44: 288-293. doi: 10.1016/j.anl.2016.07.020
Cherchi M (2019) Utricular function in vestibular neuritis: a pilot study of concordance/discordance between ocular vestibular evoked myogenic potentials and ocular cycloposition. Exp Brain Res. doi: 10.1007/s00221-019-05529-8
Cherchi M, Yacovino DA (2021) Histology and neuroanatomy suggest a unified mechanism to explain the distribution of lesion patterns in acute vestibular neuropathy. Experimental Brain Research 239: 1395–1399. doi: 10.1007/s00221-021-06094-9
Dix MR, Hallpike CS (1952) The pathology symptomatology and diagnosis of certain common disorders of the vestibular system. Proc R Soc Med 45: 341-54.
Emiliano De Schutter D, Perez Fernandez N (2017) Relapsing Ipsilateral Vestibular Neuritis. Case Rep Otolaryngol 2017: 3628402. doi: 10.1155/2017/3628402
Fishman JM, Burgess C, Waddell A (2011) Corticosteroids for the treatment of idiopathic acute vestibular dysfunction (vestibular neuritis). Cochrane Database Syst Rev: CD008607. doi: 10.1002/14651858.CD008607.pub2
Fundakowski CE, Anderson J, Angeli S (2012) Cross-sectional vestibular nerve analysis in vestibular neuritis. Ann Otol Rhinol Laryngol 121: 466-70. doi: 10.1177/000348941212100707
Goudakos JK, Markou KD, Psillas G, Vital V, Tsaligopoulos M (2014) Corticosteroids and vestibular exercises in vestibular neuritis. Single-blind randomized clinical trial. JAMA Otolaryngol Head Neck Surg 140: 434-40. doi: 10.1001/jamaoto.2014.48
Hasuike K, Sekitani T, Imate Y (1995) Enhanced MRI in patients with vestibular neuronitis. Acta Otolaryngol Suppl 519: 272-4.
Hu N, Li J, Zhao H, Yue G, Wang L, Pylypenko D, Xu T, Su S, Li C (2025) Optimal MRI protocol for acute unilateral vestibulopathy: 3D-fsFLAIR vs 3D-fsT1WI at multiple post-contrast time points. European Radiology. doi: 10.1007/s00330-025-12220-w
Hunt JR (1907) On herpetic inflammations of the geniculate gangion. A new syndrome and its complicaitons. The Journal of Nervous and Mental Disease 34.
Huppert D, Strupp M, Theil D, Glaser M, Brandt T (2006) Low recurrence rate of vestibular neuritis: a long-term follow-up. Neurology 67: 1870-1. doi: 10.1212/01.wnl.0000244473.84246.76
Ismail EI, Morgan AE, Abdel Rahman AM (2019) Corticosteroids versus vestibular rehabilitation in long-term outcomes in vestibular neuritis. J Vestib Res. doi: 10.3233/VES-180645
Izraeli S, Rachmel A, Nitzan M (1989) [Vestibular dysfunction and deafness complicating mumps parotitis]. Harefuah 117: 243-4.
Jeong SH, Kim HJ, Kim JS (2013) Vestibular neuritis. Semin Neurol 33: 185-94. doi: 10.1055/s-0033-1354598
Karlberg M, Annertz M, Magnusson M (2004) Acute vestibular neuritis visualized by 3-T magnetic resonance imaging with high-dose gadolinium. Arch Otolaryngol Head Neck Surg 130: 229-32. doi: 10.1001/archotol.130.2.229
Kim G, Seo JH, Lee SJ, Lee DH (2022) Therapeutic effect of steroids on vestibular neuritis: Systematic review and meta-analysis. Clin Otolaryngol 47: 34-43. doi: 10.1111/coa.13880
Kim JS, Kim HJ (2012) Inferior vestibular neuritis. J Neurol 259: 1553-60. doi: 10.1007/s00415-011-6375-4
Kim KT, Park E, Lee SU, Kim B, Kim BJ, Kim JS (2023) Clinical Features and Neurotologic Findings in Patients With Acute Unilateral Peripheral Vestibulopathy Associated With Antiganglioside Antibody. Neurology 101: e1913-e1921. doi: 10.1212/WNL.0000000000207814
Kim YH, Kim KS, Kim KJ, Choi H, Choi JS, Hwang IK (2011) Recurrence of vertigo in patients with vestibular neuritis. Acta Otolaryngol 131: 1172-7. doi: 10.3109/00016489.2011.593551
Kuhweide R, Van de Steene V, Vlaminck S, Casselman JW (2002) Ramsay Hunt syndrome: pathophysiology of cochleovestibular symptoms. J Laryngol Otol 116: 844-8. doi: 10.1258/00222150260293691
Lee G, Park JM, Hwang T, Lee JM (2026) Bilateral Sequential Vestibular Neuritis: Report of Three Cases. Otol Neurotol. doi: 10.1097/MAO.0000000000004829
Lee SU, Kim HJ, Choi JY, Kim JK, Kim JS (2019a) Acute vestibular syndrome associated with anti-GQ1b antibody. Neurology 93: e1085-e1092. doi: 10.1212/wnl.0000000000008107
Lee SU, Kim HJ, Choi JY, Kim JS (2019b) Anti-ganglioside antibody-associated acute unilateral peripheral vestibulopathy. J Neurol 266: 250-252. doi: 10.1007/s00415-018-9109-z
Lee WS, Suarez C, Honrubia V, Gomez J (1990) Morphological aspects of the human vestibular nerve. Laryngoscope 100: 756-64. doi: 10.1288/00005537-199007000-00012
Leong KJ, Lau T, Stewart V, Canetti EFD (2021) Systematic Review and Meta-analysis: Effectiveness of Corticosteroids in Treating Adults With Acute Vestibular Neuritis. Otolaryngol Head Neck Surg: 194599820982910. doi: 10.1177/0194599820982910
Lopez I, Ishiyama G, Tang Y, Frank M, Baloh RW, Ishiyama A (2005) Estimation of the number of nerve fibers in the human vestibular endorgans using unbiased stereology and immunohistochemistry. J Neurosci Methods 145: 37-46. doi: 10.1016/j.jneumeth.2004.11.024
Magliulo G, Gagliardi S, Ciniglio Appiani M, Iannella G, Re M (2014) Vestibular neurolabyrinthitis: a follow-up study with cervical and ocular vestibular evoked myogenic potentials and the video head impulse test. Ann Otol Rhinol Laryngol 123: 162-73. doi: 10.1177/0003489414522974
Magliulo G, Iannella G, Gagliardi S, Re M (2015) A 1-year follow-up study with C-VEMPs, O-VEMPs and video head impulse testing in vestibular neuritis. Eur Arch Otorhinolaryngol 272: 3277-81. doi: 10.1007/s00405-014-3404-9
Mandala M, Santoro GP, Awrey J, Nuti D (2010) Vestibular neuritis: recurrence and incidence of secondary benign paroxysmal positional vertigo. Acta Otolaryngol 130: 565-7. doi: 10.3109/00016480903311278
McDonnell MN, Hillier SL (2015) Vestibular rehabilitation for unilateral peripheral vestibular dysfunction. Cochrane Database Syst Rev 1: CD005397. doi: 10.1002/14651858.CD005397.pub4
McGarvie LA, MacDougall HG, Curthoys IS, Halmagyi GM (2020) Spontaneous Recovery of the Vestibulo-Ocular Reflex After Vestibular Neuritis; Long-Term Monitoring With the Video Head Impulse Test in a Single Patient. Front Neurol 11: 732. doi: 10.3389/fneur.2020.00732
Meldrum D, Jahn K (2019) Gaze stabilisation exercises in vestibular rehabilitation: review of the evidence and recent clinical advances. J Neurol 266: 11-18. doi: 10.1007/s00415-019-09459-x
Michel L, Laurent T, Alain T (2019) Rehabilitation of dynamic visual acuity in patients with unilateral vestibular hypofunction: earlier is better. Eur Arch Otorhinolaryngol. doi: 10.1007/s00405-019-05690-4
Moriyama H, Itoh M, Shimada K, Otsuka N (2007) Morphometric analysis of fibers of the human vestibular nerve: sex differences. Eur Arch Otorhinolaryngol 264: 471-5. doi: 10.1007/s00405-006-0197-5
Nadol JB, Jr. (1995) Vestibular neuritis. Otolaryngol Head Neck Surg 112: 162-72. doi: 10.1016/S0194-59989570316-0
Nagai Y, Goto N, Goto J, Kaneko Y, Suzaki H (1999) Morphometric nerve fiber analysis and aging process of the human vestibular nerve. Okajimas Folia Anat Jpn 76: 95-100.
Navari E, Casani AP (2020) Lesion Patterns and Possible Implications for Recovery in Acute Unilateral Vestibulopathy. Otol Neurotol 41: e250-e255. doi: 10.1097/MAO.0000000000002476
Neuhauser HK (2007) Epidemiology of vertigo. Curr Opin Neurol 20: 40-6. doi: 10.1097/WCO.0b013e328013f432
Nylén CO (1924) Some Cases of Ocular Nystagmus Due to Certain Positions of the Head. Acta Oto-Laryngologica 6: 106-137. doi: 10.3109/00016482409123851
Ogata Y, Sekitani T, Shimogori H, Ikeda T (1993) Bilateral vestibular neuronitis. Acta Otolaryngol Suppl 503: 57-60.
Okinaka Y, Sekitani T, Okazaki H, Miura M, Tahara T (1993) Progress of caloric response of vestibular neuronitis. Acta Otolaryngol Suppl 503: 18-22.
Ozdogmus O, Sezen O, Kubilay U, Saka E, Duman U, San T, Cavdar S (2004) Connections between the facial, vestibular and cochlear nerve bundles within the internal auditory canal. J Anat 205: 65-75. doi: 10.1111/j.0021-8782.2004.00313.x
Rasmussen AT (1940) Studies of the VIIIth cranial nerve of man. Laryngoscope 50: 67-83.
Ruttin E (1909) Zur differential diagnose der Laybrinth- und Hörnerverkrankungen [On the differential diagnosis of labyrinthine and hearing diseases]. Zeitschrift für Ohrenheilkunde [Journal of Otology] 57: 327-331.
Schnaar RL (2019) The Biology of Gangliosides. Adv Carbohydr Chem Biochem 76: 113-148. doi: 10.1016/bs.accb.2018.09.002
Schuknecht HF, Witt RL (1985) Acute bilateral sequential vestibular neuritis. Am J Otolaryngol 6: 255-7.
Sekitani T, Imate Y, Noguchi T, Inokuma T (1993) Vestibular neuronitis: epidemiological survey by questionnaire in Japan. Acta Otolaryngol Suppl 503: 9-12.
Silverstein H, Norrell H, Haberkamp T, McDaniel AB (1986) The unrecognized rotation of the vestibular and cochlear nerves from the labyrinth to the brain stem: its implications to surgery of the eighth cranial nerve. Otolaryngol Head Neck Surg 95: 543-9. doi: 10.1177/019459988609500504
Strupp M, Bisdorff A, Furman J, Hornibrook J, Jahn K, Maire R, Newman-Toker D, Magnusson M (2022) Acute unilateral vestibulopathy/vestibular neuritis: Diagnostic criteria. J Vestib Res 32: 389-406. doi: 10.3233/VES-220201
Strupp M, Brandt T (2009) Vestibular neuritis. Semin Neurol 29: 509-19. doi: 10.1055/s-0029-1241040
Strupp M, Jager L, Muller-Lisse U, Arbusow V, Reiser M, Brandt T (1998) High resolution Gd-DTPA MR imaging of the inner ear in 60 patients with idiopathic vestibular neuritis: no evidence for contrast enhancement of the labyrinth or vestibular nerve. J Vestib Res 8: 427-33.
Strupp M, Zingler VC, Arbusow V, Niklas D, Maag KP, Dieterich M, Bense S, Theil D, Jahn K, Brandt T (2004) Methylprednisolone, valacyclovir, or the combination for vestibular neuritis. N Engl J Med 351: 354-61. doi: 10.1056/NEJMoa033280
Taylor RL, McGarvie LA, Reid N, Young AS, Halmagyi GM, Welgampola MS (2016) Vestibular neuritis affects both superior and inferior vestibular nerves. Neurology 87: 1704-1712. doi: 10.1212/WNL.0000000000003223
Tubbs RS, Steck DT, Mortazavi MM, Cohen-Gadol AA (2013) The nervus intermedius: a review of its anatomy, function, pathology, and role in neurosurgery. World Neurosurg 79: 763-7. doi: 10.1016/j.wneu.2012.03.023
Venkatasamy A, Huynh TT, Wohlhuter N, Vuong H, Rohmer D, Charpiot A, Meyer N, Veillon F (2019) Superior vestibular neuritis: improved detection using FLAIR sequence with delayed enhancement (1 h). Eur Arch Otorhinolaryngol 276: 3309-3316. doi: 10.1007/s00405-019-05639-7
Wanleenuwat P, Iwanowski P, Kozubski W (2020) Antiganglioside antibodies in neurological diseases. J Neurol Sci 408: 116576. doi: 10.1016/j.jns.2019.116576
Yacovino DA, Finlay JB, Jaimes VNU, Verdecchia DH, Schubert MC (2018) Acute Bilateral Superior Branch Vestibular Neuropathy. Frontiers in Neurology 9: 6. doi: 10.3389/fneur.2018.00353
Yacovino DA, Zanotti E, Cherchi M (2021) The spectrum of acute vestibular neuropathy through modern vestibular testing: a descriptive analysis. Clinical Neurophysiology Practice. doi: https://doi.org/10.1016/j.cnp.2021.02.008
Yoo MH, Yang CJ, Kim SA, Park MJ, Ahn JH, Chung JW, Park HJ (2017) Efficacy of steroid therapy based on symptomatic and functional improvement in patients with vestibular neuritis: a prospective randomized controlled trial. Eur Arch Otorhinolaryngol 274: 2443-2451. doi: 10.1007/s00405-017-4556-1
![]()