By Marcello Cherchi, MD PhD
For patients
CANVAS results from a genetic mutation, and causes gradually progressive unsteadiness usually beginning around age 60 (though the age range is broad, ranging from 30s to 70s). Symptoms include unsteadiness, clumsiness, and reduced touch sensation. In order to distinguish this from other (similar-looking) diseases, your doctor may consider checking tests of inner ear balance function, eye movement function, brain imaging, and nerves in your arms and legs. If these results are suggestive of CANVAS, then genetic testing may help confirm the diagnosis and exclude similar-looking diagnoses. There is no effective treatment, though many patients will try vestibular rehabilitation therapy.
For clinicians
Overview
CANVAS (cerebellar ataxia, neuropathy, vestibular areflexia syndrome) is a rare progressive neurodegenerative disorder that causes multi-factorial disequilibrium through vestibular weakness (due to a vestibular ganglionopathy), sensory neuropathy (due to sensory ganglionopathy), and cerebellar ataxia. Most cases appear to be due to a mutation in the RFC1 gene; familial cases exhibit an autosomal recessive pattern more than an autosomal dominant pattern (though this latter may be pseudodominance due to compound heretozygosity). It affects men and women approximately equally. The mean age of onset is in the 6th decade (range 20s – 70s). Symptoms start insidiously and progress slowly; chronic cough may precede the other symptoms by years to decades. Physical examination findings include cerebellar ataxia, diminished small and large fiber sensation in all limbs. Ocular motor examination should show deficiencies in the visual-vestibulo-ocular reflex (VVOR), and may also show deficits in smooth pursuit, gaze-evoked nystagmus or spontaneous down beat nystagmus. Vestibular testing (rotatory chair testing, video head impulse testing) shows clear evidence of vestibular weakness. Electromyography with nerve conduction velocities shows non-length dependent small- and large-fiber sensory neuropathy which can have either axonal or demyelinating features or both, usually in all limbs. There may be evidence of dysautonomia. MRI shows cerebellar atrophy, particularly in the dorsal vermis. Genetic testing can confirm the diagnosis by identifying mutations in RFC1, and exclude competing diagnoses such as spinocerebellar ataxias and Friedreich’s ataxia. There is no effective treatment, though a trial of vestibular rehabilitation therapy is reasonable.
Introduction
CANVAS is an acronym for cerebellar ataxia, neuropathy and vestibular areflexia syndrome.
The association between bilateral vestibular weakness and cerebellar ataxia was described by Rinne and colleagues in 1995 (Rinne et al. 1995). In 2004 Migliaccio and colleagues (Migliaccio et al. 2004) reported findings in four patients with a syndrome of bilateral vestibular weakness and cerebellar ataxia. Three of the patients also had electrophysiological evidence of a peripheral sensory neuropathy and in one of those patients biopsy revealed a severe axonal neuropathy. Subsequent cases suggested that the associated sensory neuropathy was a fairly consistent feature (Szmulewicz et al. 2011b).
Additional cases were subsequently reported in the literature, and eventually the constellation of findings (cerebellar ataxia, bilateral vestibular weakness and peripheral neuropathy) came to be described as a syndrome, CANVAS.
These patients come to the attention of otoneurology by way of referral for the symptom of disequilibrium. In CANVAS the disequilibrium is multifactorial, resulting from a combination two afferent defects (sensory neuropathy, vestibular weakness) and one coordination defect (cerebellar ataxia).
Epidemiology
CANVAS was originally thought to be rare, but subsequent work has increasingly suggested that it is a common cause of late-onset ataxia (Cortese et al. 2019).
In the 27 cases reported by Szmulewicz and colleagues (Szmulewicz, Waterston et al. 2011) they found an approximately equal distribution by sex (13 males, 14 females), and reported the mean age of onset of reported symptoms was 60 years (range 33 – 71 years).
In the 105 cases reported by Cortese and colleagues (Cortese, Tozza et al. 2020) the mean age of symptom onset (excluding cough) was 52 years (range 19 – 76 years).
Genetics
CANVAS (OMIM 614575) results from a mutation resulting in a repeat expansion in the gene encoding for RFC1 (replication factor C subunit 1) (OMIM 102579) on chromosome 4p14, a pentameric DNA polymerase accessory protein involved in DNA replication and repair (Cortese et al. 2019; Davies et al. 2022).
Mutations in RFC1 are identified in 100% of familial cases of CANVAS and in 92% of sporadic cases (Cortese, Simone et al. 2019).
In a series of 44 patients with genetically confirmed CANVAS reported by Huin and colleagues (Huin, Coarelli et al. 2022), 19 (43%) exhibited a pattern of autosomal recessive transmission, 15 (34%) were sporadic, and 10 (23%) appeared to exhibit an autosomal dominant pattern, though this latter may be a pseudodominant manifestation (Falcone et al. 2024) of compound heterozygosity (Ronco et al. 2023).
Emerging literature suggests that CANVAS may be one manifestation of a broader spectrum of diseases resulting from RFC1 mutations. For example, Beijer and colleagues (Beijer, Dohrn et al. 2022) depict RFC1 diseases as a group of partly overlapping neuropathies, as depicted in the Figure below.
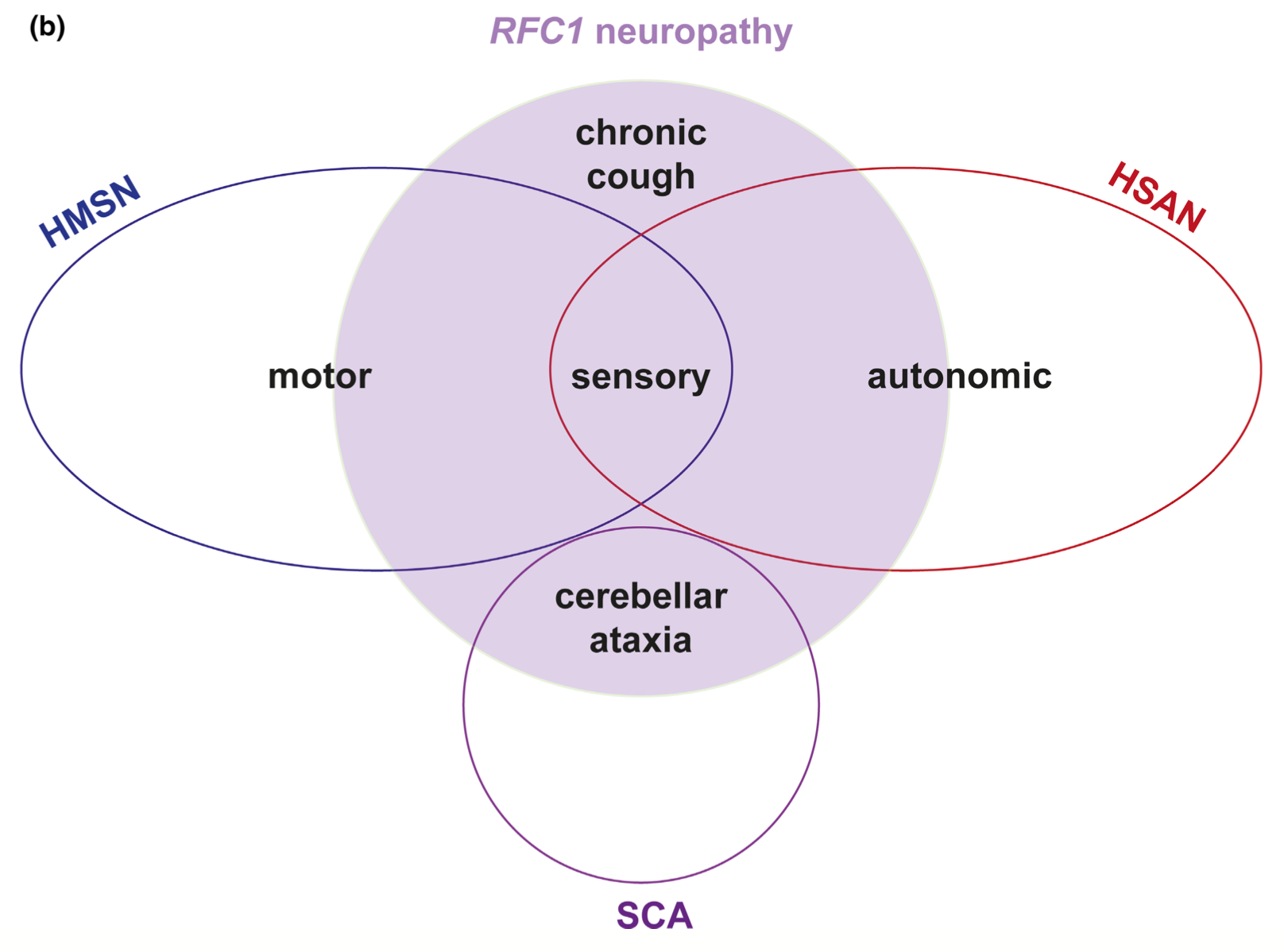
Emerging evidence suggests that RFC1 mutations may also account for isolated sensory neuropathy (Curro, Salvalaggio et al. 2021).
Pathophysiological mechanism of disease
In order to understand the key ocular motor finding in CANVAS it is helpful to review the reflex systems that drive eye movements during visual and vestibular stimulation, as shown in the Table below.
|
Stimulus |
Reflex system(s) |
|
Fixation on moving small target (vision available) |
Smooth pursuit system (SP) using visual fixation |
|
Movement of entire visual scene (vision available) |
Optokinetic system (OPK) |
|
Movement of head (vision deprived) |
Labyrinthine-to-ocular motor system: vestibulo-ocular reflex (VOR) |
|
Movement of head while vision is available |
Both visual systems (smooth pursuit, optokinetic) plus vestibulo-ocular system |
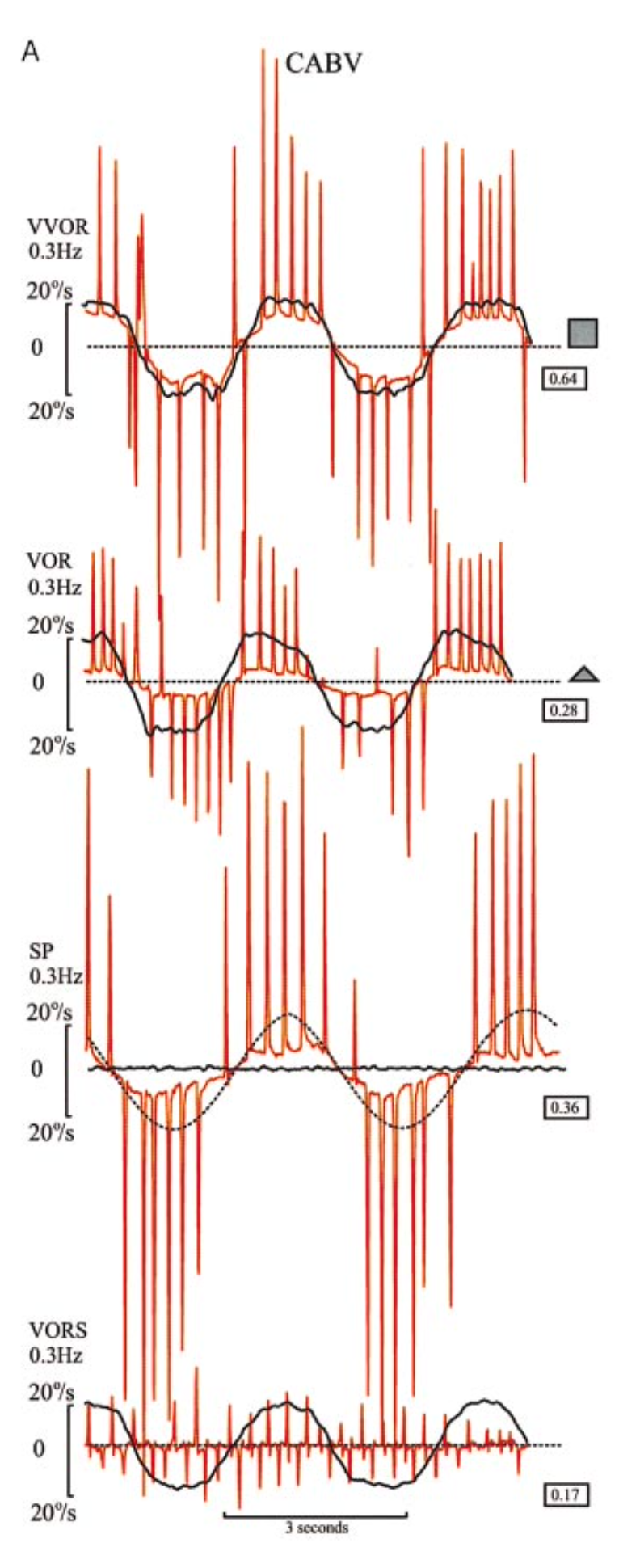
In their patients (whose diagnosis would later be designated CANVAS), Migliaccio and colleagues (Migliaccio et al. 2004) observed, “bursts of compensatory saccades when the patient’s head was slowly and smoothly oscillated from side to side while fixating an earth-fixed target straight ahead.” They interpreted this as reflecting, “low VVOR gain,” and hypothesized that, “This abnormality is due to combined impairment of VOR [vestibulo-ocular reflex] and SP [smooth pursuit].” They concluded that this was a, “characteristic sign on clinical examination: impairment of the VVOR [visual vestibulo-ocular reflex].”
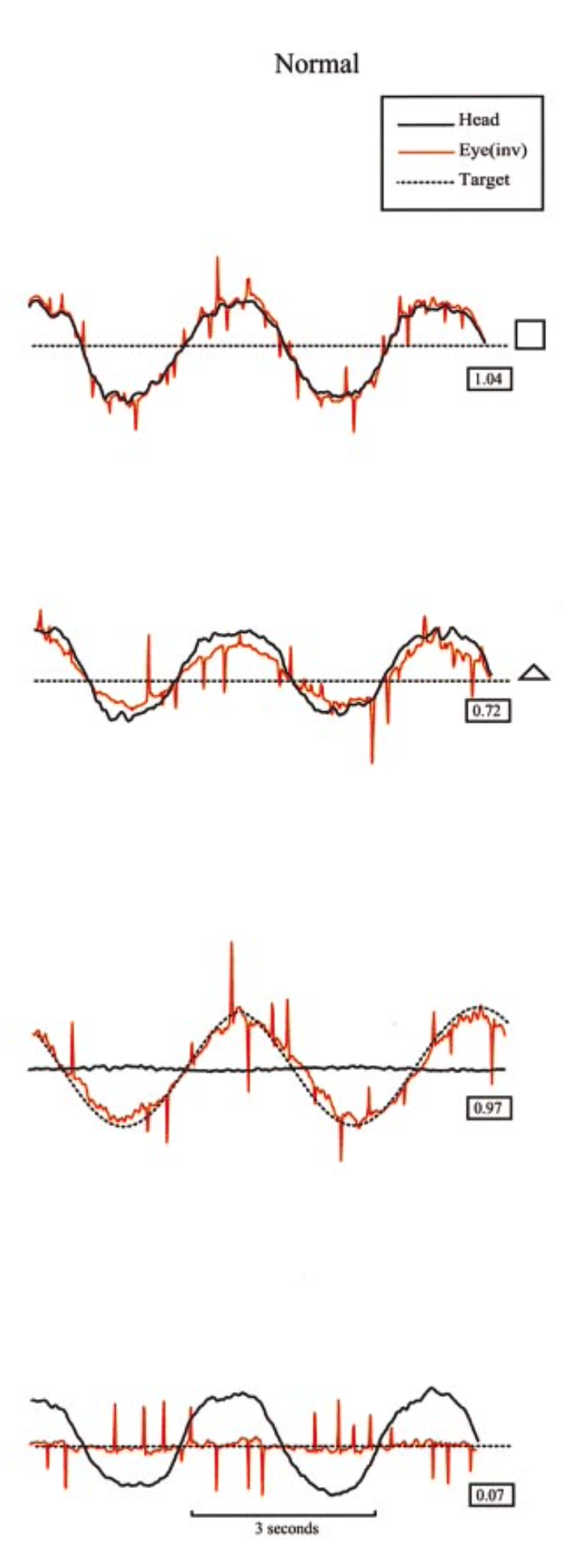
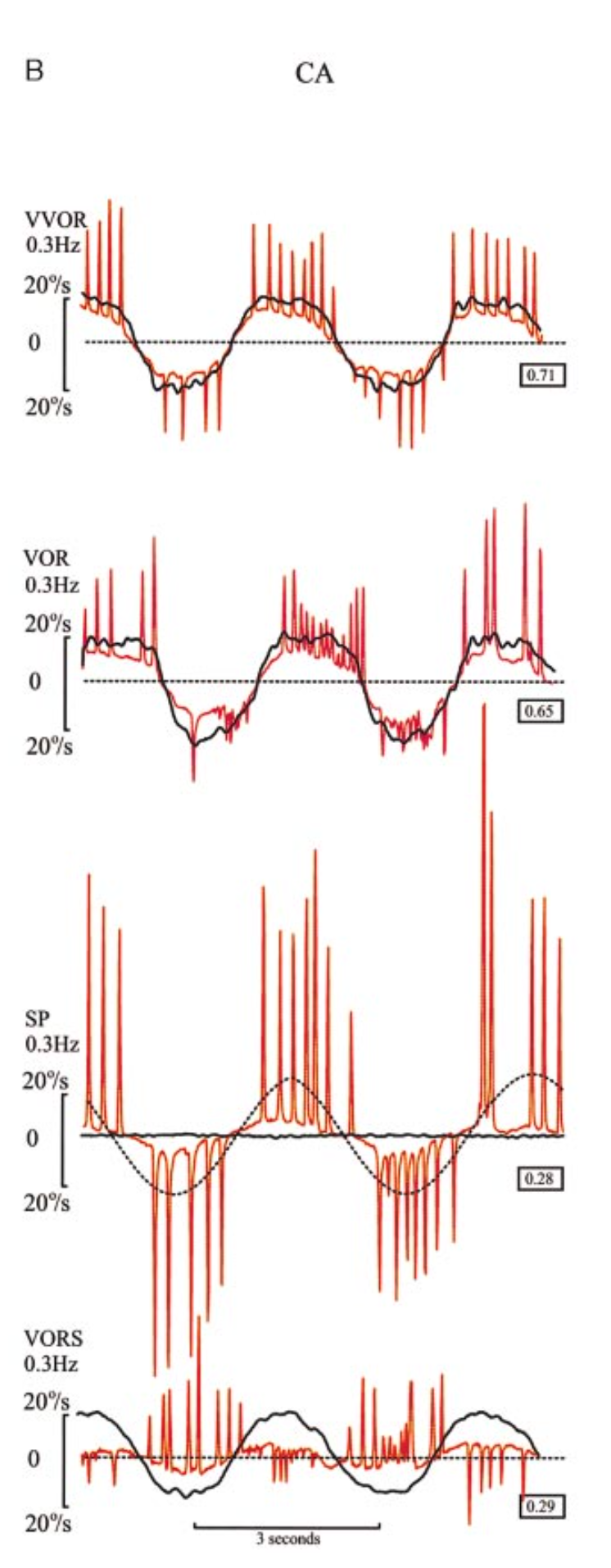
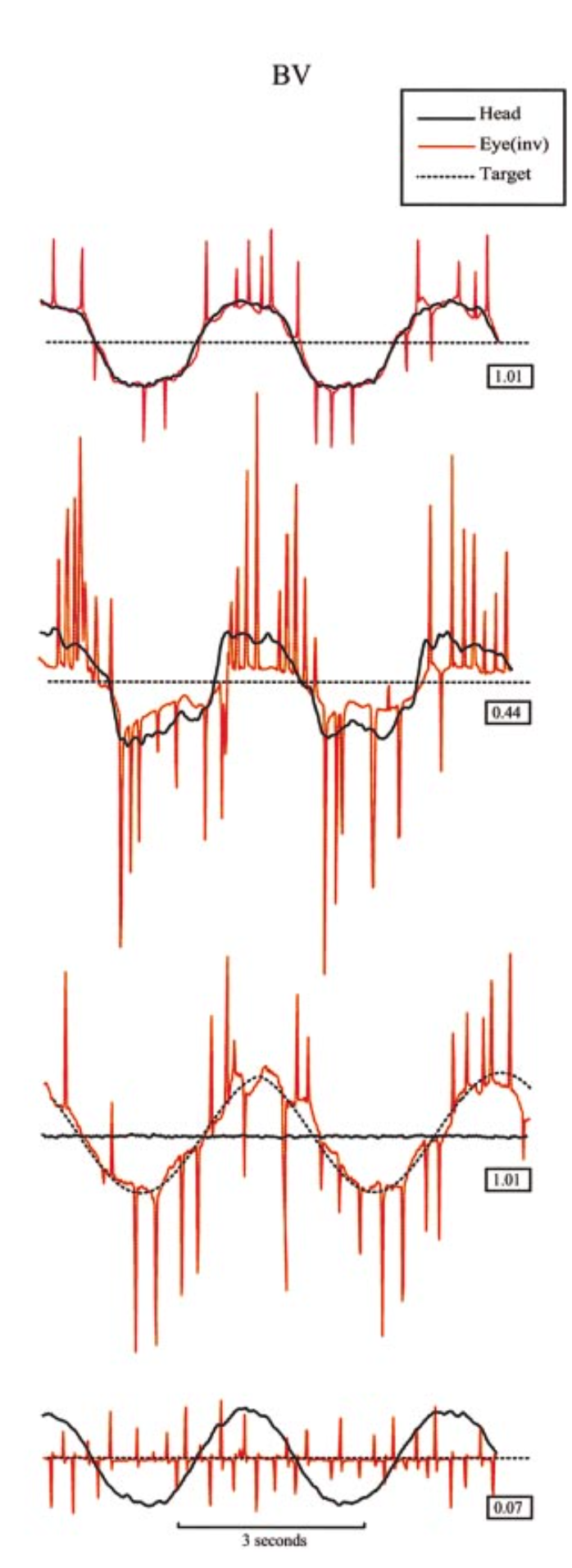
The Table below draws on data presented by Migliaccio and colleagues (Migliaccio, Halmagyi et al. 2004).
 |
 |
 |
 |
|
VVOR: Gain reduced (0.64), though similar to CA (0.71). During VVOR testing, the CABV patient makes more and larger compensatory saccades than either the CA or the BV patient. These saccades constitute the characteristic clinical sign of CABV: bursts of corrective saccades as the patient views an earth-fixed target while turning the head from side to side. |
VVOR: The normal patient makes very few compensatory saccades. |
VVOR: Gain is somewhat reduced (0.71). An intermediate number and size of compensatory saccades. |
VVOR: A modest number and size of compensatory saccades. |
|
VOR: Low gain (0.28), somewhat similar to (though worse than) BV. |
VOR: Normal gain (0.72). |
VOR: Gain (0.65) slightly lower than normal (0.72). |
VOR: Low gain (0.44) because of bilateral vestibular loss. |
|
SP: Low gain (0.36), somewhat similar to CA. |
SP: Normal gain (0.97). |
SP: Gain low (0.28). |
SP: Gain normal (1.01), as does not need to rely on (deficient) vestibular input. |
|
VORS: Despite the cerebellar ataxia, the VORS looks almost normal (relatively few corrective saccades) because there is little or no VOR to suppress, so the cerebellar dysfunction (which would usually interfere with visual fixation suppression of the vestibulo-ocular reflex) is not apparent. |
VORS: Normal gain (0.07). |
VORS: Abnormal. Gain is higher (0.29) than the others. In many cerebellar disorders (with an intact vestibular system), the cerebellar deficit interferes with visual fixation suppression of the vestibulo-ocular reflex. |
VORS: The VORS looks almost normal (relatively few corrective saccades) because there is little or no VOR to suppress. |
Clinical presentation
In the 27 cases of CANVAS described by Szmulewicz and colleagues (Szmulewicz et al. 2011b) they state that the presenting features were: gait imbalance in 20 (74%), dysesthesia in 8 (30%), oscillopsia in 5 (19%), “dizziness” in 2 (7%) and falls in 1 (4%); some patients had more than one presenting feature.
Additional studies of “RFC1-spectrum” disorders (Traschutz et al. 2021) have revealed several manifestations beyond the originally described triad, including:
- Chronic cough, which can precede the other symptoms by years or even decades (Cortese et al. 2019; Dupre et al. 2021).
- Evidence of dysautonomia, such as orthostatic hypotension (Moreno-Ajona et al. 2019; Szmulewicz et al. 2014).
- Nigrostriatal dysfunction (Azevedo et al. 2023).
- Bulbar dysfunction, such as dysphagia (Casanueva et al. 2021).
- Cognitive decline (Dujardin et al. 2024).
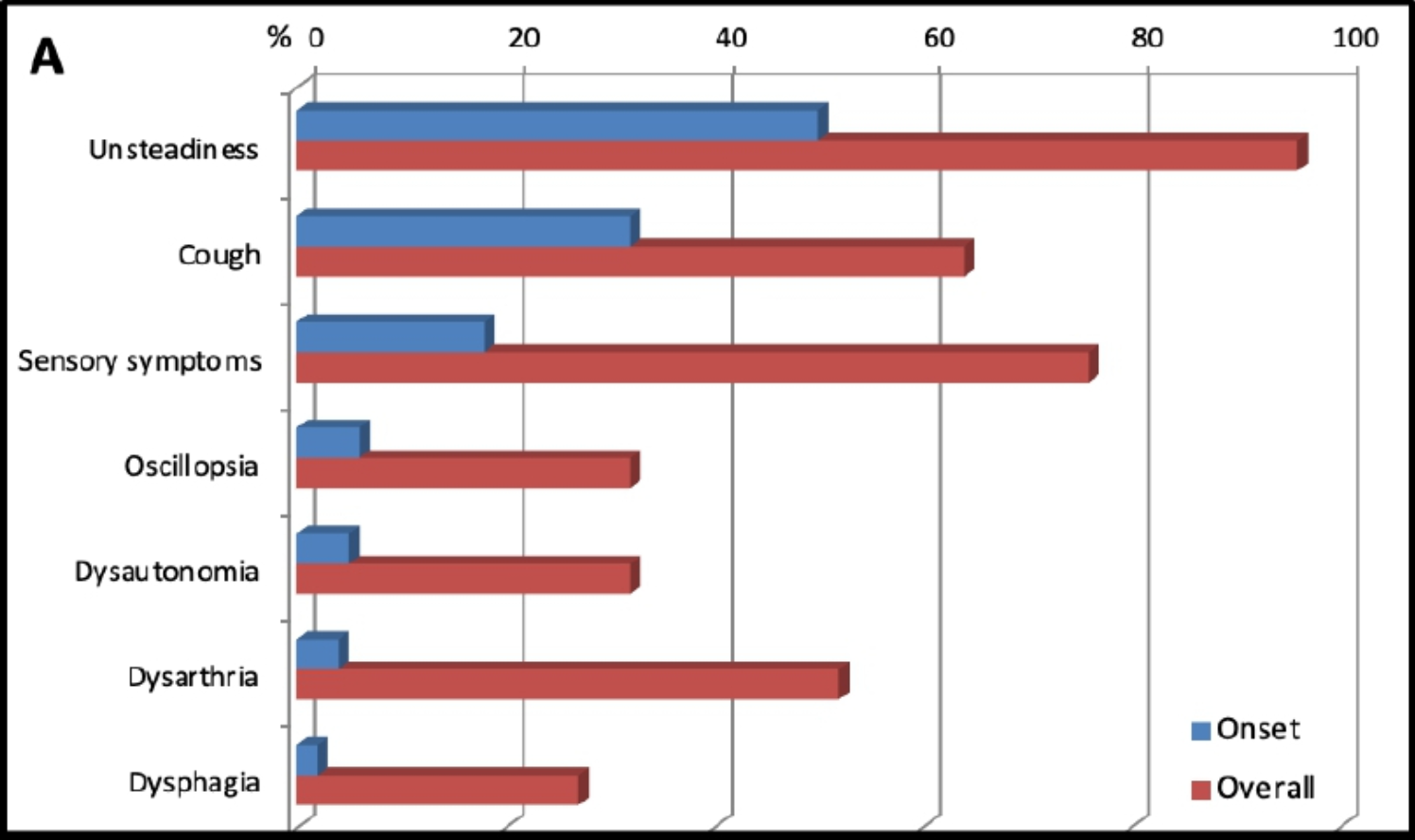
The Figure below, from Cortese and colleagues (Cortese, Curro et al. 2022), illustrates the percentage of patients that have each symptom at onset, and overall.
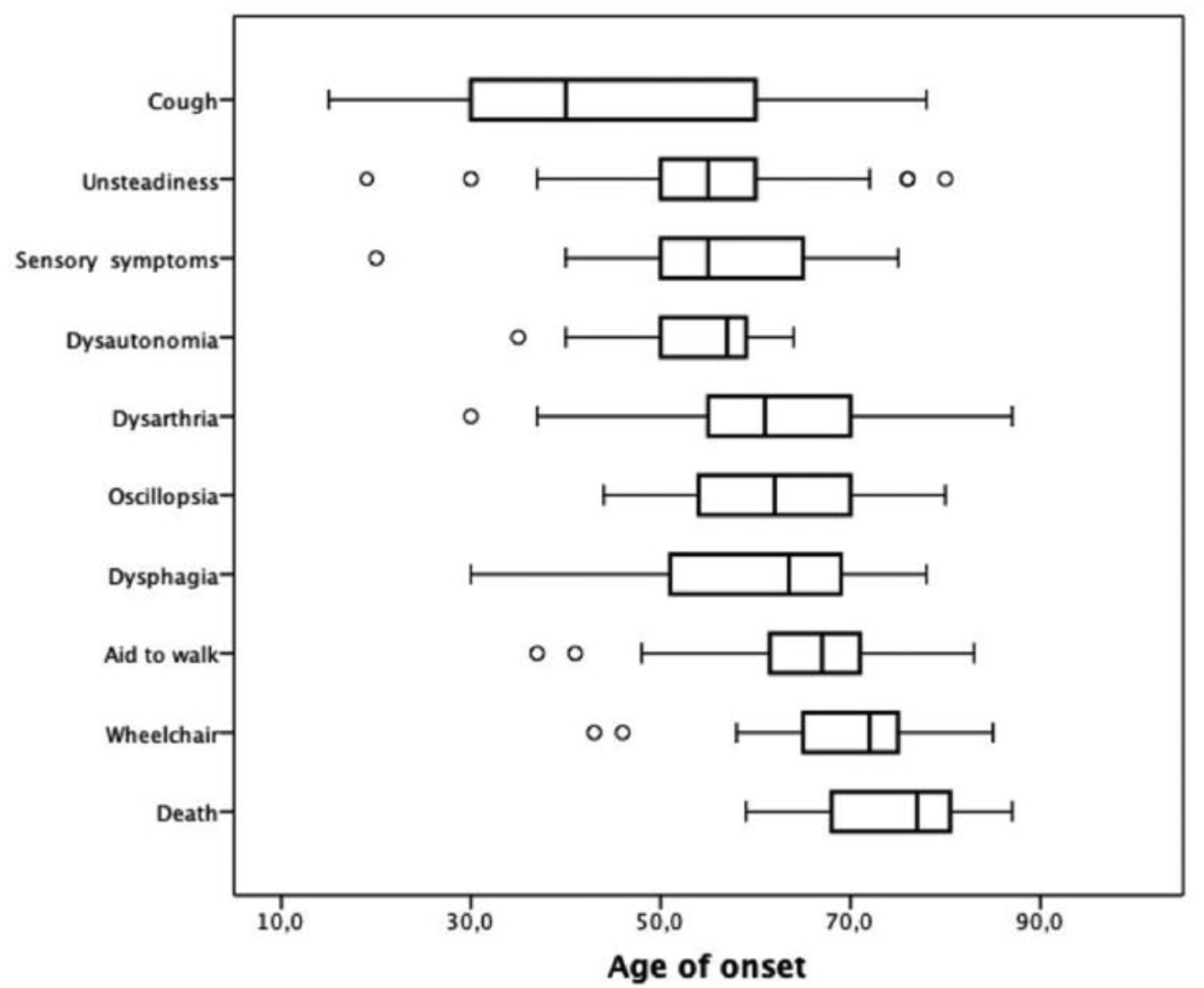
The Figure below, from Cortese and colleagues (Cortese, Tozza et al. 2020), shows the age of onset for each symptom.
Physical examination
Patients usually have signs of cerebellar appendicular ataxia.
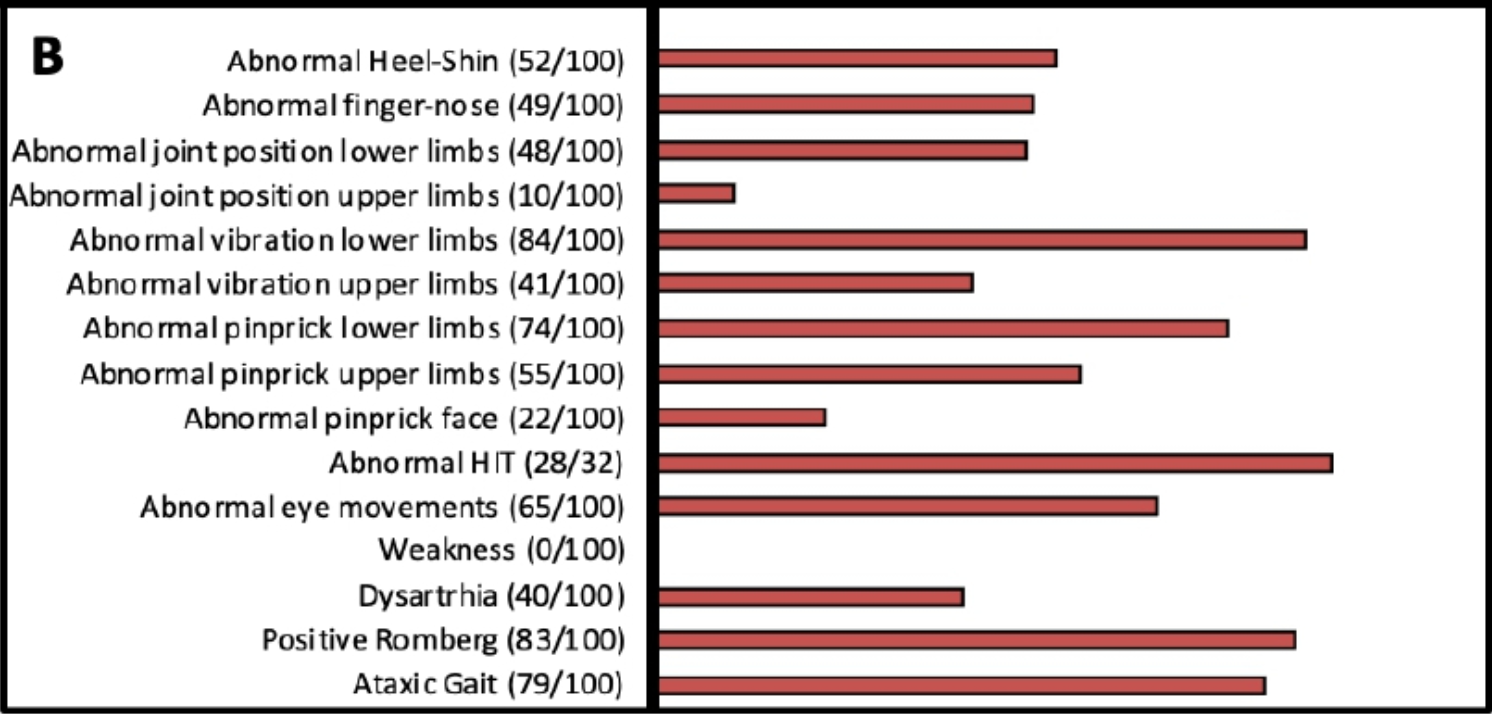
Patients usually have signs of sensory neuropathy and sensory ataxia. Regarding neuropathic signs, in the 27 cases of CANVAS described by Szmulewicz and colleagues (Szmulewicz, Waterston et al. 2011) they reported that “15/27 [56%] had absent ankle jerks, 21/27 [78%] had impaired vibration sensation (with abnormal joint position sense in a further one), and 21/27 [78%] had a deficit of pin-prick sensation,” while “only two [7%] lacked any clinical evidence of a non-length-dependent multimodality sensory deficit.”
Patients may also have evidence of dysautonomia, such as orthostatic hypotension (Szmulewicz, McLean et al. 2014, Moreno-Ajona, Alvarez-Gomez et al. 2019).
The Figure below, from Cortese and colleagues (Cortese, Curro et al. 2022), shows the percentage of patients exhibiting each physical examination finding.
Rarely, patients with RFC1 mutations exhibit fasciculations (Scriba, Beecroft et al. 2020, Huin, Coarelli et al. 2022).
Ocular motor examination
Evidence of bilateral vestibular weakness should be apparent on ophthalmoscopy and dynamic visual acuity testing.
Szmulewicz and colleagues (Szmulewicz, Waterston et al. 2011) state that, “An impaired VVOR can be demonstrated clinically by turning a patient’s head slowly (at about 0.5 Hz) from side-to-side while the patient starts at an earth-fixed target and observing that the compensatory eye movements are saccadic rather than smooth.” They comment that other clinical features may include, “saccadic smooth pursuit, gaze-evoked nystagmus, gait ataxia, dysarthria and appendicular ataxia.” They additionally commented that, “Gaze-evoked horizontal or down beat nystagmus was found in all but two patients” of the 27 they analyzed.
The Video below, from Terryn and colleagues (Terryn et al. 2020) shows an example of abnormal visual vestibulo-ocular reflex.
Huin and colleagues (Huin, Coarelli et al. 2022) reported the following ocular motor findings for a series of 34 patients with RFC1 mutations:
- Saccadic pursuit in 22 (65%)
- Saccadic dysmetria in 15 (44%)
- Down beat nystagmus in 13 (38%)
- Gaze evoked nystagmus in 11 (32%)
- Fixation instability in 9 (26%)
- Slow saccades in 8 (24%)
- Down beat nystagmus plus gaze evoked nystagmus in 3 (9%)
Testing: vestibular
Tests of the vestibulo-ocular reflex should show vestibular weakness.
The Figure below, from Szmulewicz and colleagues (Szmulewicz, Waterston et al. 2011), shows the results of video head impulse testing in a CANVAS patient.
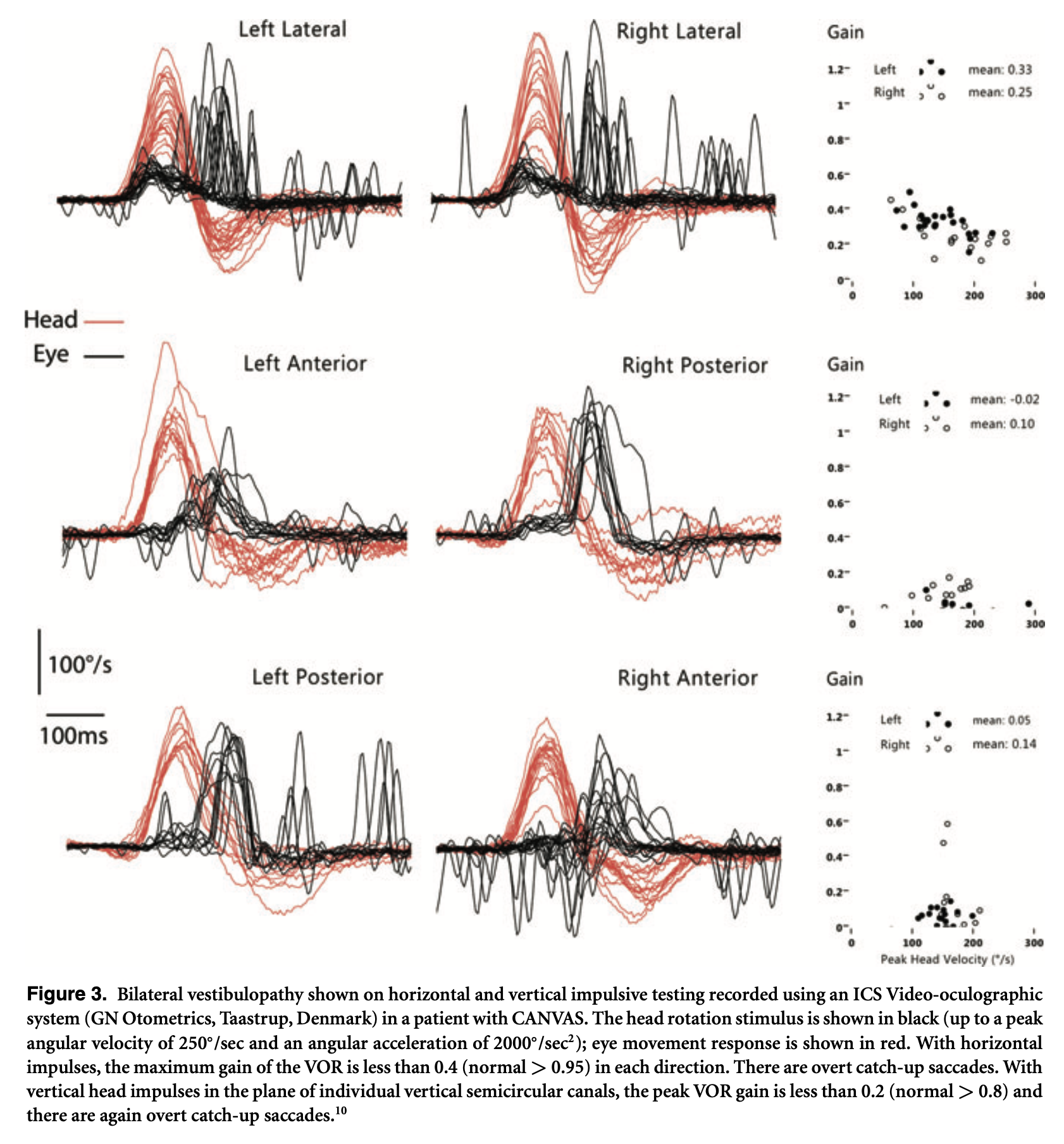
Halmagyi and Szmulewicz (Halmagyi and Szmulewicz 2021) report similar video head impulse testing findings.
In another paper of the same year, Szmulewicz and colleagues (Szmulewicz, Waterston et al. 2011) reported on 18 patients who had had some combination of rotatory chair testing, caloric testing and vestibular evoked myogenic potentials performed. In every case where one of the tests was performed there was evidence of vestibular weakness, either in the form of reduced VOR gain on rotatory chair testing, reduced caloric responses, or absent vestibular evoked myogenic potentials. The Table below summarizes their results.
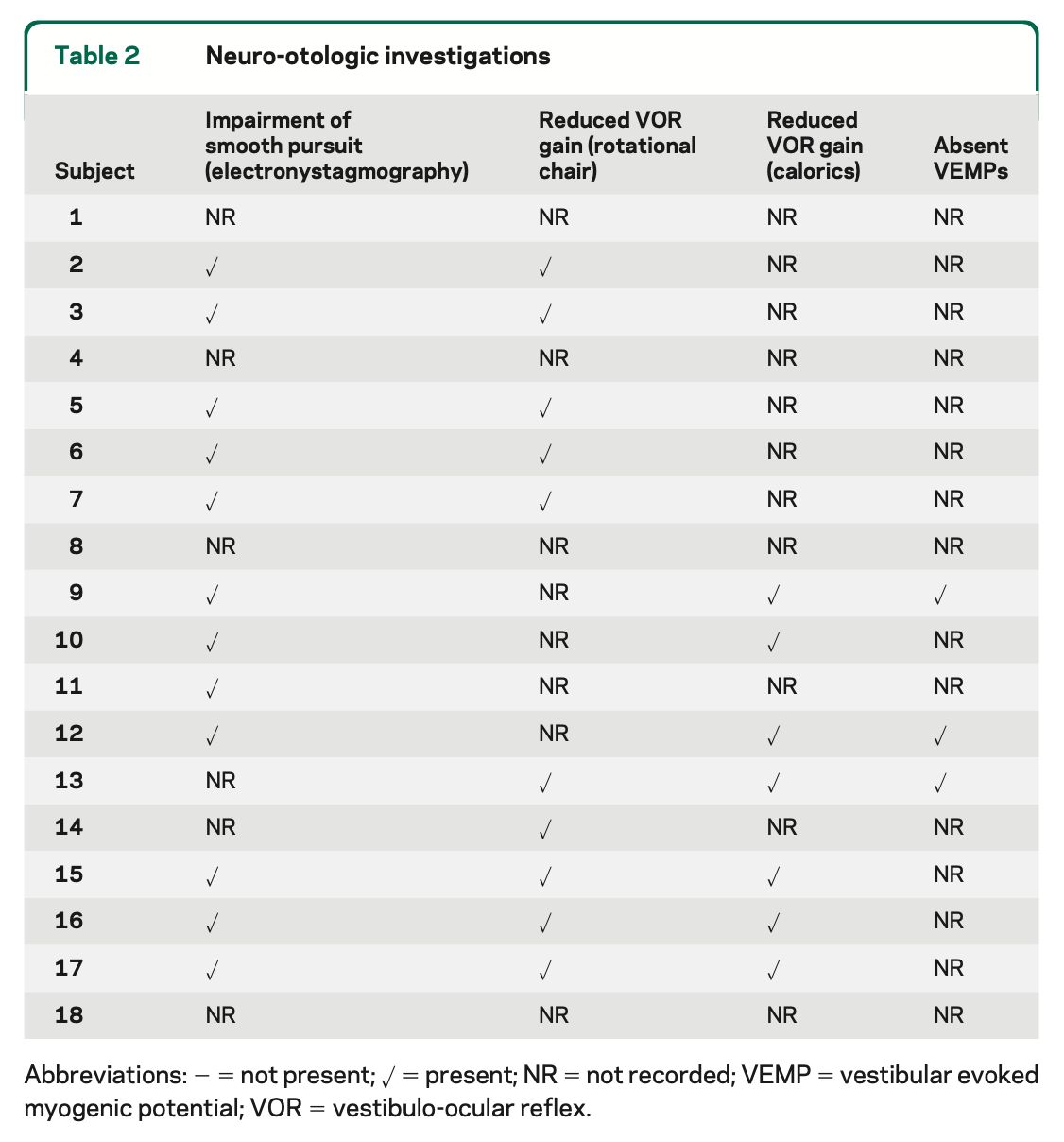
Other investigators reported only inconsistent abnormalities on vestibular evoked myogenic potentials (Moreno-Ajona, Alvarez-Gomez et al. 2019).
Testing: electromyography and nerve conduction velocities
Szmulewicz and colleagues (Szmulewicz, Waterston et al. 2011) reported that, “All 20 patients in whom a four-limb nerve conduction study was performed had a loss of upper and lower limb SNAPs” (sensory nerve action potentials). They also stated that the sensory neuropathy can be either axonal or demyelinating, noting that, “One sural nerve was biopsied in six of the patents and revealed severe axonal loss in all, with evidence of coexistent demyelination in one.”
The neuropathy affects both small and large diameter fiber types (Szmulewicz, Waterston et al. 2011).
Szmulewicz and colleagues (Szmulewicz, Waterston et al. 2011) reported further note that the neuropathy is “non-length-dependent” and affects both upper and lower limbs, and concluded that, “the total body involvement in some suggested a neuronopathy” (as opposed to merely a neuropathy).
Such observations, coupled with the histopathological findings reported in the vestibular nerves (see below), have led to speculation that the vestibular deficit may be due to a vestibular ganglionopathy (as opposed to merely vestibular neuropathy) (Yacovino, Zanotti et al. 2019), which was subsequently confirmed on temporal bone studies (Ishai, Seyyedi et al. 2021).
Testing: genetic
Mutations in RFC1 are identified in 100% of familial cases of CANVAS and in 92% of sporadic cases (Cortese, Simone et al. 2019).
Genetic testing, or at least referral to a genetic counselor, is reasonable. This may help confirm a diagnosis of CANVAS by identifying a mutation in RFC1 (Traschutz, Cortese et al. 2021, Davies, Szmulewicz et al. 2022), as well as exclude differential diagnoses such as spinocerebellar ataxia (particularly SCA3) and Friedreich’s ataxia.
Imaging
Szmulewicz and colleagues (Szmulewicz, Waterston et al. 2011) reported that, “MRI was done in all patients, and cerebellar atrophy was evident in 22/27 [81%] patients,” adding that, “While the severity varied, a consistent pattern of anterior and dorsal vermis atrophy (vermal lobules VI, VIIa, and VIIb), and laterally, atrophy predominantly affecting crus 1 (corresponding to vermal lobule VII), was observed.”
The Figure below, from Szmulewicz and colleagues (Szmulewicz, Waterston et al. 2011) shows examples of sagittal sections from brain MRIs demonstrating cerebellar atrophy.
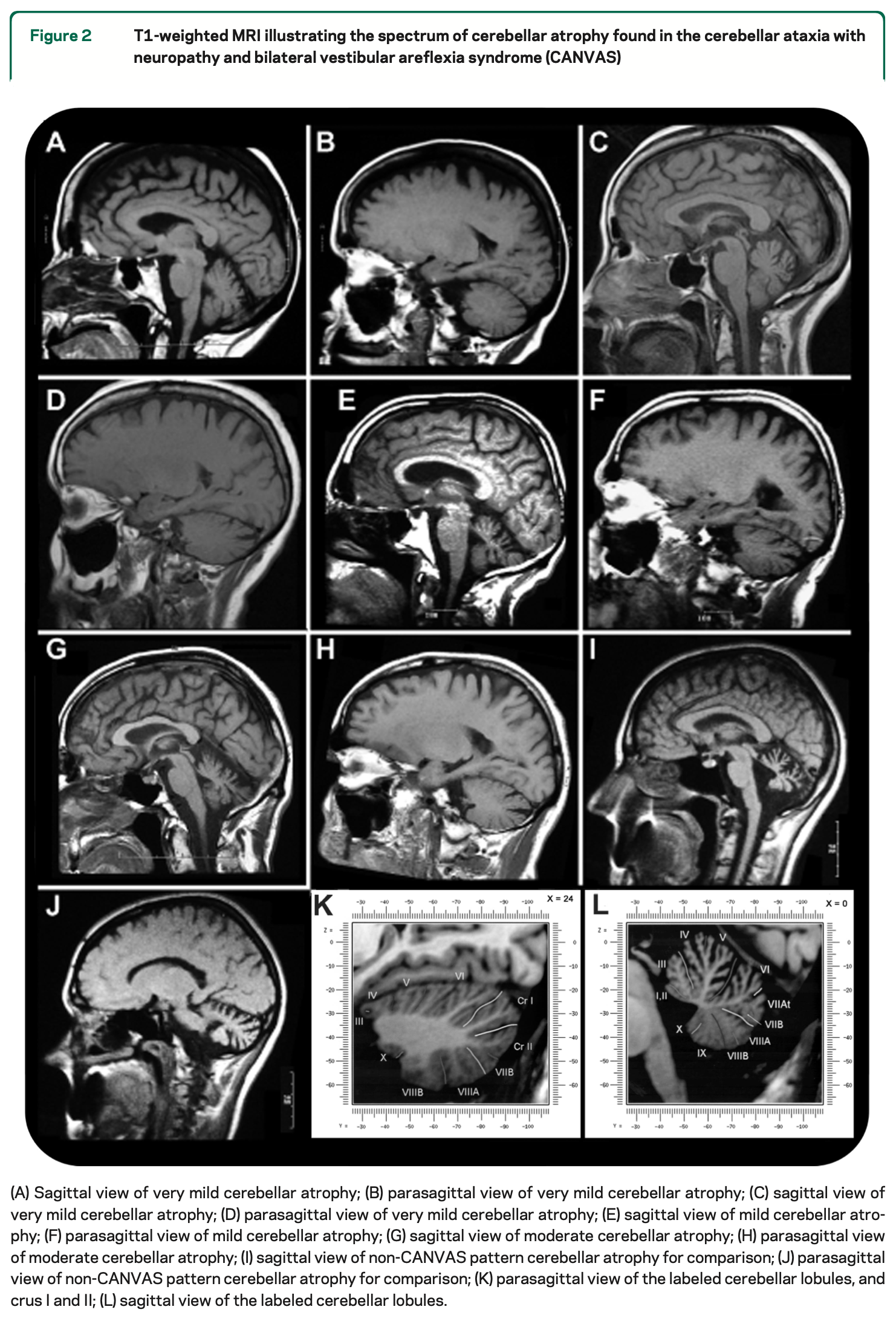
Histopathology
In a case studied by Szmulewicz and colleagues (Szmulewicz, Merchant et al. 2011), they observed “severe atrophy of both vestibular nerves,” “severe loss of Scarpa’s ganglion cells,” and “macroscopic cerebellar cortical atrophy of the anterior vermis and the superomedial aspect of the cerebellar hemispheres.”
A temporal bone series of 5 patients with CANVAS showed that the vestibular deficit is due to a vestibular ganglionopathy (Ishai, Seyyedi et al. 2021).
There is emerging evidence that some CANVAS patients (with genetically confirmed RFC1 mutations) have a component of motor neuronopathy (Huin, Coarelli et al. 2022).
Differential diagnosis
It is important to bear in mind that, although the majority of CANVAS patients eventually develop symptoms/signs/findings of all three components to which its name alludes (cerebellar, neuropathic, vestibular), not all patients with RFC1 mutations exhibit the full clinical picture.
The Figure below, from Cortese and colleagues (Cortese, Tozza et al. 2020), shows Venn diagrams of the incomplete overlap among the three components.
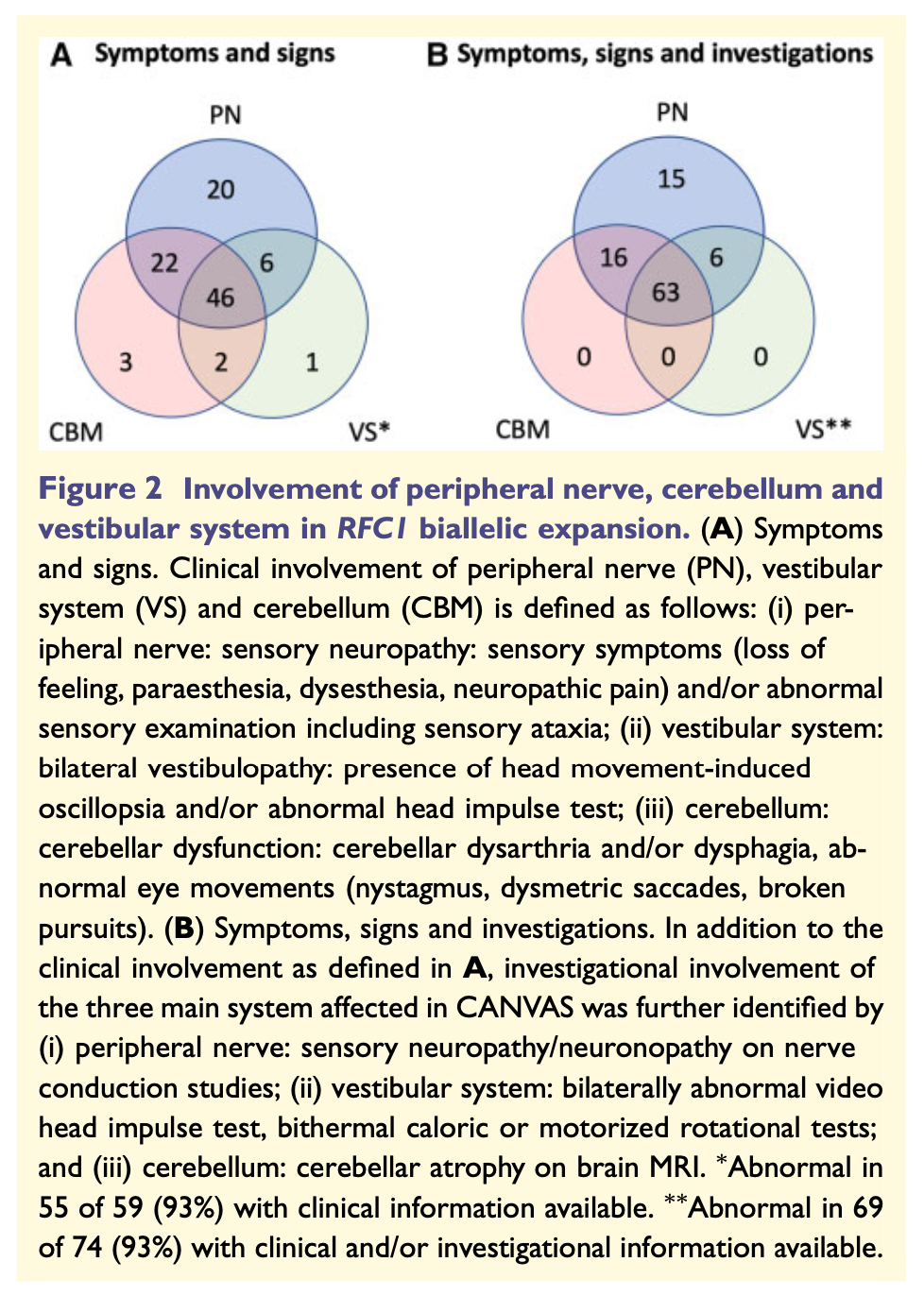
From this figure, the frequency of findings, in descending order, are:
- All 3 components are present (cerebellar, neuropathic, vestibular). This is the most common manifestation.
- Cerebellar plus neuropathic is the second most common.
- Neuropathic in isolation is the third most common.
- Neuropathic plus vestibular.
- The rarest combinations are isolated cerebellar, combined cerebellar and vestibular, and isolated vestibular.
Because of this variety, the differential diagnosis is somewhat broad.
Szmulewicz and colleagues (Szmulewicz, Waterston et al. 2011) suggest that, “The major differential diagnoses of CANVAS are SCA3 (Machado-Joseph disease), Friedreich’s ataxia (FRDA), multiple system atrophy of cerebellar type (MSA‑C), and Wernicke’s encephalopathy.” Szmulewicz and colleagues (Szmulewicz, McLean et al. 2014) state that they usually also screen for SCA1, 2, 6 and 7 and well.
Cortese and colleagues (Cortese, Curro et al. 2022) offer the more extensive differential diagnosis in the Table below.
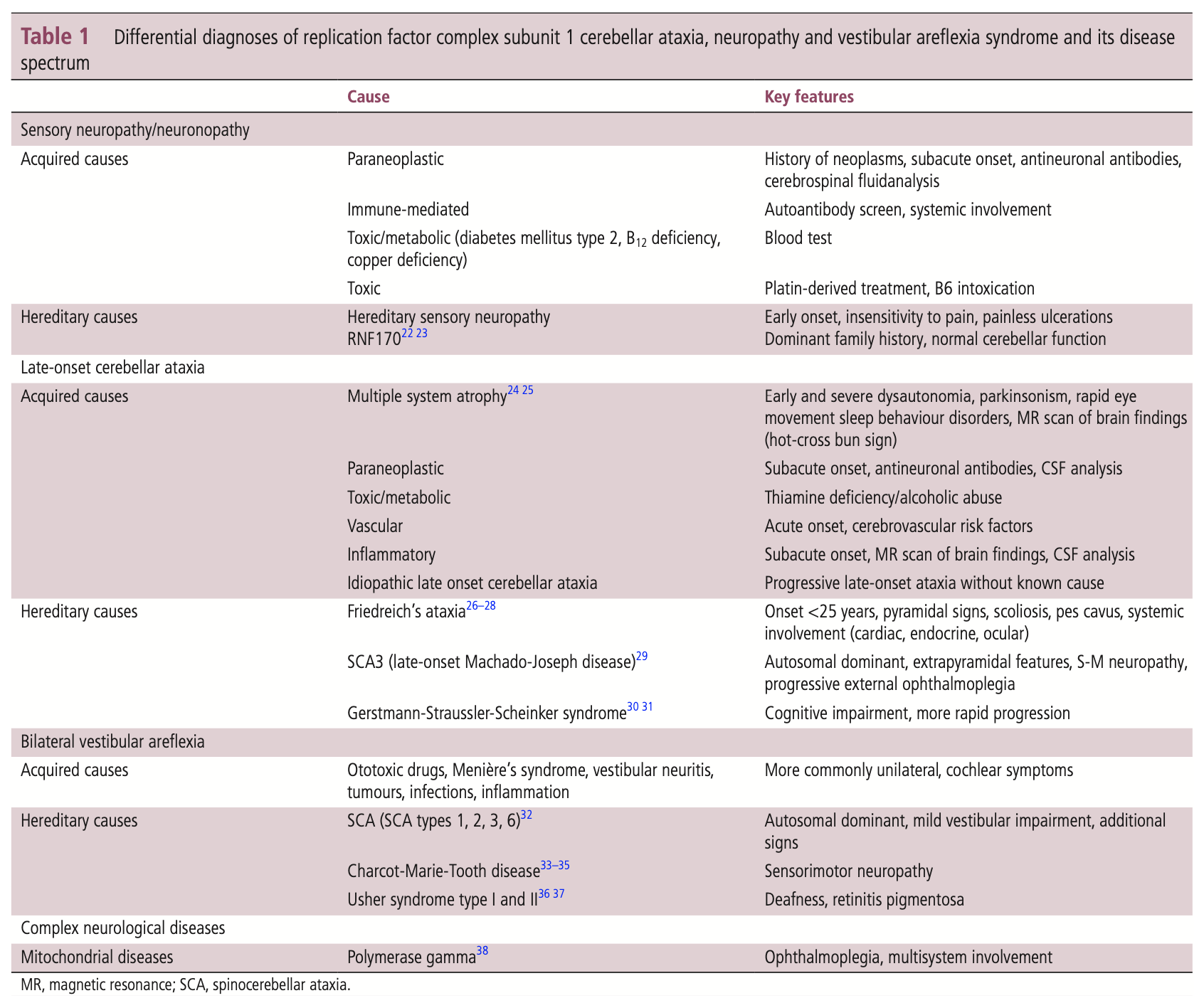
Treatment
There is no established treatment for CANVAS. Management of disequilibrium is difficult since there is multi-modal impairment.
Szmulewicz and colleagues (Szmulewicz, McLean et al. 2014) state that, “Anecdotally, we find significant benefit in referring our patients for individualised combination neurological and vestibular rehabilitation, which are performed regularly by the patient and reviewed periodically by a specialist physiotherapist.”
Practically, referral for vestibular rehabilitation therapy is reasonable, on the logic that strategies aimed at bilateral vestibular weakness can be applied — recognizing that sensory substitution strategies will be limited due to the neuropathy, and motor compensatory strategies will be limited by the incoordination from the cerebellar ataxia.
Prognosis
There is no curative or arrestive therapy. Cortese and colleagues comment that, “The disease shows a slowly progressive course. Half of the patients needed a stick when walking after 10 years, and a quarter were wheelchair-dependent 5 years latera. However, life expectancy was not affected” (Cortese, Curro et al. 2022).
References
Azevedo MPC, Lobo CC, Schmitt GS, Matos P, Barsottini OGP, Pedroso JL, Marques W, Jr., Souza JP, Amorim BJ, Franca MC, Jr. (2023) Nigrostriatal dysfunction in RFC1-related disorder/CANVAS. Parkinsonism Relat Disord 115: 105854. doi: 10.1016/j.parkreldis.2023.105854
Beijer D, Dohrn MF, De Winter J, Fazal S, Cortese A, Stojkovic T, Fernandez-Eulate G, Remiche G, Gentile M, Van Coster R, Dufke C, Synofzik M, De Jonghe P, Zuchner S, Baets J (2022) RFC1 repeat expansions: A recurrent cause of sensory and autonomic neuropathy with cough and ataxia. Eur J Neurol 29: 2156-2161. doi: 10.1111/ene.15310
Casanueva R, Lopez F, Costales M, Ordas A, Villanueva E, Llorente JL, Alvarez Marcos C (2021) The presence of dysphagia in patients with cerebellar ataxia, neuropathy and vestibular areflexia syndrome (CANVAS): a subjective and objective study. Eur Arch Otorhinolaryngol 278: 2585-2592. doi: 10.1007/s00405-020-06534-2
Cortese A, Curro R, Vegezzi E, Yau WY, Houlden H, Reilly MM (2022) Cerebellar ataxia, neuropathy and vestibular areflexia syndrome (CANVAS): genetic and clinical aspects. Pract Neurol 22: 14-18. doi: 10.1136/practneurol-2020-002822
Cortese A, Simone R, Sullivan R, Vandrovcova J, Tariq H, Yau WY, Humphrey J, Jaunmuktane Z, Sivakumar P, Polke J, Ilyas M, Tribollet E, Tomaselli PJ, Devigili G, Callegari I, Versino M, Salpietro V, Efthymiou S, Kaski D, Wood NW, Andrade NS, Buglo E, Rebelo A, Rossor AM, Bronstein A, Fratta P, Marques WJ, Zuchner S, Reilly MM, Houlden H (2019) Biallelic expansion of an intronic repeat in RFC1 is a common cause of late-onset ataxia. Nat Genet 51: 649-658. doi: 10.1038/s41588-019-0372-4
Cortese A, Tozza S, Yau WY, Rossi S, Beecroft SJ, Jaunmuktane Z, Dyer Z, Ravenscroft G, Lamont PJ, Mossman S, Chancellor A, Maisonobe T, Pereon Y, Cauquil C, Colnaghi S, Mallucci G, Curro R, Tomaselli PJ, Thomas-Black G, Sullivan R, Efthymiou S, Rossor AM, Laura M, Pipis M, Horga A, Polke J, Kaski D, Horvath R, Chinnery PF, Marques W, Tassorelli C, Devigili G, Leonardis L, Wood NW, Bronstein A, Giunti P, Zuchner S, Stojkovic T, Laing N, Roxburgh RH, Houlden H, Reilly MM (2020) Cerebellar ataxia, neuropathy, vestibular areflexia syndrome due to RFC1 repeat expansion. Brain 143: 480-490. doi: 10.1093/brain/awz418
Curro R, Salvalaggio A, Tozza S, Gemelli C, Dominik N, Galassi Deforie V, Magrinelli F, Castellani F, Vegezzi E, Businaro P, Callegari I, Pichiecchio A, Cosentino G, Alfonsi E, Marchioni E, Colnaghi S, Gana S, Valente EM, Tassorelli C, Efthymiou S, Facchini S, Carr A, Laura M, Rossor AM, Manji H, Lunn MP, Pegoraro E, Santoro L, Grandis M, Bellone E, Beauchamp NJ, Hadjivassiliou M, Kaski D, Bronstein AM, Houlden H, Reilly MM, Mandich P, Schenone A, Manganelli F, Briani C, Cortese A (2021) RFC1 expansions are a common cause of idiopathic sensory neuropathy. Brain 144: 1542-1550. doi: 10.1093/brain/awab072
Davies K, Szmulewicz DJ, Corben LA, Delatycki M, Lockhart PJ (2022) RFC1-Related Disease: Molecular and Clinical Insights. Neurol Genet 8: e200016. doi: 10.1212/NXG.0000000000200016
Dujardin K, Tard C, Digle E, Herlin V, Mutez E, Davion JB, Wissocq A, Delforge V, Kuchcinski G, Huin V (2024) Cognitive Impairment Is Part of the Phenotype of Cerebellar Ataxia, Neuropathy, Vestibular Areflexia Syndrome (CANVAS). Mov Disord 39: 892-897. doi: 10.1002/mds.29750
Dupre M, Hermann R, Froment Tilikete C (2021) Update on Cerebellar Ataxia with Neuropathy and Bilateral Vestibular Areflexia Syndrome (CANVAS). Cerebellum 20: 687-700. doi: 10.1007/s12311-020-01192-w
Falcone GMI, Tessa A, Arena IG, Barghigiani M, Migliorato A, Incensi A, Rodolico C, Donadio V, Santorelli FM, Musumeci O (2024) Pseudodominance in RFC1-Spectrum Disorder. Cerebellum 23: 2622-2628. doi: 10.1007/s12311-024-01735-5
Halmagyi GM, Szmulewicz DJ (2021) Vestibular function testing in patients with RFC1 mutations. J Neurol 268: 4894-4896. doi: 10.1007/s00415-021-10698-0
Huin V, Coarelli G, Guemy C, Boluda S, Debs R, Mochel F, Stojkovic T, Grabli D, Maisonobe T, Gaymard B, Lenglet T, Tard C, Davion JB, Sablonniere B, Monin ML, Ewenczyk C, Viala K, Charles P, Le Ber I, Reilly MM, Houlden H, Cortese A, Seilhean D, Brice A, Durr A (2022) Motor neuron pathology in CANVAS due to RFC1 expansions. Brain 145: 2121-2132. doi: 10.1093/brain/awab449
Ishai R, Seyyedi M, Chancellor AM, McLean CA, Rodriguez ML, Halmagyi GM, Nadol JB, Jr., Szmulewicz DJ, Quesnel AM (2021) The Pathology of the Vestibular System in CANVAS. Otol Neurotol 42: e332-e340. doi: 10.1097/MAO.0000000000002985
Migliaccio AA, Halmagyi GM, McGarvie LA, Cremer PD (2004) Cerebellar ataxia with bilateral vestibulopathy: description of a syndrome and its characteristic clinical sign. Brain 127: 280-93. doi: 10.1093/brain/awh030
Moreno-Ajona D, Alvarez-Gomez L, Manrique-Huarte R, Rivas E, Martinez-Vila E, Perez-Fernandez N (2019) VEMPs and Dysautonomia Assessment in Definite Cerebellar Ataxia, Neuropathy, Vestibular Areflexia Syndrome (CANVAS): a Case Series Study. Cerebellum. doi: 10.1007/s12311-019-01061-1
Rinne T, Bronstein AM, Rudge P, Gresty MA, Luxon LM (1995) Bilateral loss of vestibular function. Acta Otolaryngol Suppl 520 Pt 2: 247-50. doi: 10.3109/00016489509125239
Ronco R, Perini C, Curro R, Dominik N, Facchini S, Gennari A, Simone R, Stuart S, Nagy S, Vegezzi E, Quartesan I, El-Saddig A, Lavin T, Tucci A, Szymura A, Novis De Farias LE, Gary A, Delfeld M, Kandikatla P, Niu N, Tawde S, Shaw J, Polke J, Reilly MM, Wood NW, Crespan E, Gomez C, Chen JYH, Schmahmann JD, Gosal D, Houlden H, Das S, Cortese A (2023) Truncating Variants in RFC1 in Cerebellar Ataxia, Neuropathy, and Vestibular Areflexia Syndrome. Neurology 100: e543-e554. doi: 10.1212/WNL.0000000000201486
Scriba CK, Beecroft SJ, Clayton JS, Cortese A, Sullivan R, Yau WY, Dominik N, Rodrigues M, Walker E, Dyer Z, Wu TY, Davis MR, Chandler DC, Weisburd B, Houlden H, Reilly MM, Laing NG, Lamont PJ, Roxburgh RH, Ravenscroft G (2020) A novel RFC1 repeat motif (ACAGG) in two Asia-Pacific CANVAS families. Brain 143: 2904-2910. doi: 10.1093/brain/awaa263
Szmulewicz DJ, McLean CA, MacDougall HG, Roberts L, Storey E, Halmagyi GM (2014) CANVAS an update: clinical presentation, investigation and management. J Vestib Res 24: 465-74. doi: 10.3233/VES-140536
Szmulewicz DJ, Merchant SN, Halmagyi GM (2011a) Cerebellar ataxia with neuropathy and bilateral vestibular areflexia syndrome: a histopathologic case report. Otol Neurotol 32: e63-5. doi: 10.1097/MAO.0b013e318210b719
Szmulewicz DJ, Waterston JA, Halmagyi GM, Mossman S, Chancellor AM, McLean CA, Storey E (2011b) Sensory neuropathy as part of the cerebellar ataxia neuropathy vestibular areflexia syndrome. Neurology 76: 1903-10. doi: 10.1212/WNL.0b013e31821d746e
Terryn J, Van Eesbeeck A, Vermeer S, Vandenberghe W (2020) The Characteristic Eye Movement Disorder of RFC1-Linked CANVAS. Mov Disord Clin Pract 7: 230-231. doi: 10.1002/mdc3.12896
Traschutz A, Cortese A, Reich S, Dominik N, Faber J, Jacobi H, Hartmann AM, Rujescu D, Montaut S, Echaniz-Laguna A, Erer S, Schutz VC, Tarnutzer AA, Sturm M, Haack TB, Vaucamps-Diedhiou N, Puccio H, Schols L, Klockgether T, van de Warrenburg BP, Paucar M, Timmann D, Hilgers RD, Gazulla J, Strupp M, Moris G, Filla A, Houlden H, Anheim M, Infante J, Basak AN, Synofzik M, Group RFCS (2021) Natural History, Phenotypic Spectrum, and Discriminative Features of Multisystemic RFC1 Disease. Neurology 96: e1369-e1382. doi: 10.1212/WNL.0000000000011528
Yacovino DA, Zanotti E, Hain TC (2019) Is Cerebellar Ataxia, Neuropathy, and Vestibular Areflexia Syndrome (CANVAS) a Vestibular Ganglionopathy? J Int Adv Otol 15: 304-308. doi: 10.5152/iao.2019.7068
![]()