By Marcello Cherchi, MD PhD
For patients
The phrase “central positional nystagmus” (CPN) refers to various eye movements triggered in certain positions. CPN is not in itself a disease, but can occur in several different neurological disorders, of which the most common is migraine associated vertigo (MAV). Management depends on the underlying cause.
For clinicians
Overview
Central positional nystagmus (CPN) refers to positionally-triggered nystagmus resulting from central (neurological) disorders rather than peripheral (otologic) disorders. Of all patients with positional nystagmus, it is estimated that about 12% have CPN. Positions that elicit CPN vary, but can include Dix-Hallpike, head hanging and side-lying. Various patterns of nystagmus occur in CPN, including paroxysmal, persistent, uniplanar, or multiplanar. CPN can occur in a range of neurological disorders, of which migraine associated vertigo (MAV) is probably the most common; less common etiologies include lesions in the cerebellum or brainstem from various diagnoses, autoimmune and paraneoplastic disorders. The mechanism of CPN is unknown, but given the variety of patterns and variety of etiologies, there is probably more than one mechanism. CPN can mimic other disorders, such as atypical benign paroxysmal positional vertigo (BPPV). In the workup of CPN, if a reasonable search for peripheral causes is unrevealing, then imaging is sensible, preferably brain MRI. The management and prognosis depend on the underlying cause, if identifiable.
Introduction
The phrase “positional nystagmus” is usually taken to refer to nystagmus that is generated by a change in head position with respect to gravity. It usually (though not always) assumes that the nystagmus in question is absent on primary position of gaze when the patient is seated upright in a neutral position.
The Bárány Society consensus document regarding classification of vestibular signs defines “central positional nystagmus” (CPN) as “positional nystagmus attributed to disease affecting the central nervous system” (Eggers et al. 2019), thereby differentiating it from nystagmus due to “peripheral” pathology (i.e., disease involving the labyrinth or vestibular nerve). This nomenclature in itself neither describes a specific set of ocular motor findings, nor does it map any ocular motor findings on to a specific pathology; it merely designates that “positional nystagmus” has been identified, and that it is due to “central” (brain) disease, rather than arising from some otologic source.
Of all patients with “positional nystagmus,” up to 12% are reported to have central positional nystagmus in some series (Bertholon et al. 2006).
Epidemiology
Bertholon and colleagues conducted a prospective study of 100 adult (>18 years old) patients with positional nystagmus reported at 12 (12%) were ultimately diagnosed with central positional nystagmus (Bertholon et al. 2006). These 12 patients had a mean age of 60 years (range 49 – 81 years).
Genetics
As we are attempting to outline it, central positional nystagmus (CPN) refers to a number of ocular motor findings occurring in a variety of diseases, and as such, there is no unifying underlying genetic predisposition.
That being said, a few genetically mediated disorders have been reported to cause CPN, such as spinocerebellar ataxias type 6 (Yu-Wai-Man et al. 2009) and type 31 (Yabe et al. 2015); familial hemiplegic migraine type 1 (Suzuki et al. 2016; Yabe et al. 2008); and episodic ataxia type (Jorge et al. 2022) and type 2 (Ling et al. 2020; Muth et al. 2021).
Pathophysiological mechanism of disease
The mechanism of central positional nystagmus (CPN) is unclear, but is probably variable given the different pathologies with which it has been reported (see below on differential diagnosis).
Lemos and Strupp (Lemos and Strupp 2022) review a variety of potential mechanisms of CPN. Citing other investigators (Choi et al. 2018a; Choi et al. 2015; Glasauer et al. 2001), Lemos and Strupp broadly describe these mechanisms as “abnormal[ities in] central processing/integration of SCC [semicircular canal] and/or otolith-related signals in addition to a prolonged velocity storage due to vestibulo-cerebellar dysfunction.” The specific candidate mechanisms Lemos and Strupp discuss include:
- In cases of CPN that is paroxysmal and down beat, it has “been hypothesized that primary nodulus and uvula dysfunction leads to (1) disinhibition of the SCC-driven signals reaching the brainstem, ultimately causing a prominent post-acceleratory rebound phenomenon… (2) a deficient rotational feedback mechanism which cannot compensate in a real-time fashion for a large difference between an erroneous estimation of gravity direction… and its actual direction,” citing Choi and colleagues (Choi et al. 2015).
- In cases of CPN that is persistent and apogeotropic, this has “been ascribed to nodulus and uvula dysfunction, in this case reflecting a constant erroneous (i.e., negative bias) estimation of gravitation direction in the presence of a normal compensatory rotational feedback mechanism,” citing Choi and colleagues (Choi et al. 2018a) and Lee and colleagues (Lee et al. 2017).
- In cases of CPN that is persistent and geotropic, “tonsillar dysfunction has been hypothesized,” citing Choi and colleagues (Choi et al. 2018b).
- “CPN associated with lesion locations other than the cerebellar nodulus, uvula or tonsil (e.g., inferior cerebellar peduncle) may reflect damage of the fibers running from the aforementioned structures to the vestibular nuclei,” citing Lee and colleagues (Lee et al. 2021).
- A possible mechanism “In cases in which CPN direction is not aligned with that of head motion during the positioning, is the presence of a mismatch between eye position estimated by the burst generator and the neural integrator, hypothetically due to altered otolith input to one of these systems,” citing Glasauer and colleagues (Glasauer et al. 2001).
Lemos and Strupp (Lemos and Strupp 2022) review the various structures implicated in CPN, summarized in the Figure below.
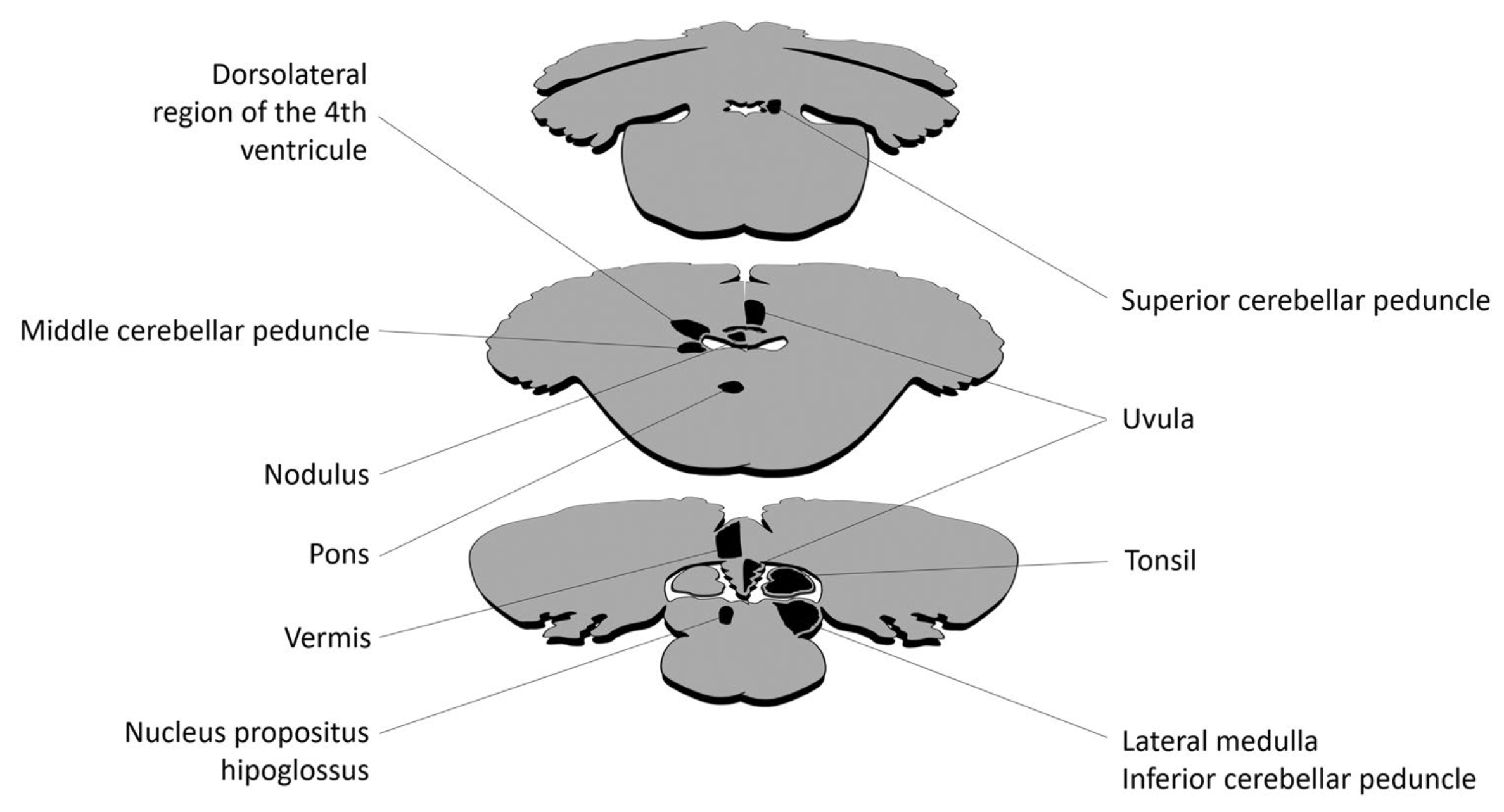
Figure : Neuroanatomical structures implicated in central positional nystagmus (CPN). For clarity, each structure is only depicted on one side. From Lemos and Strupp (Lemos and Strupp 2022).
Taking into account these possible mechanisms and neuroanatomical structures, Lemos and Strupp (Lemos and Strupp 2022) propose the model in the Figure below.
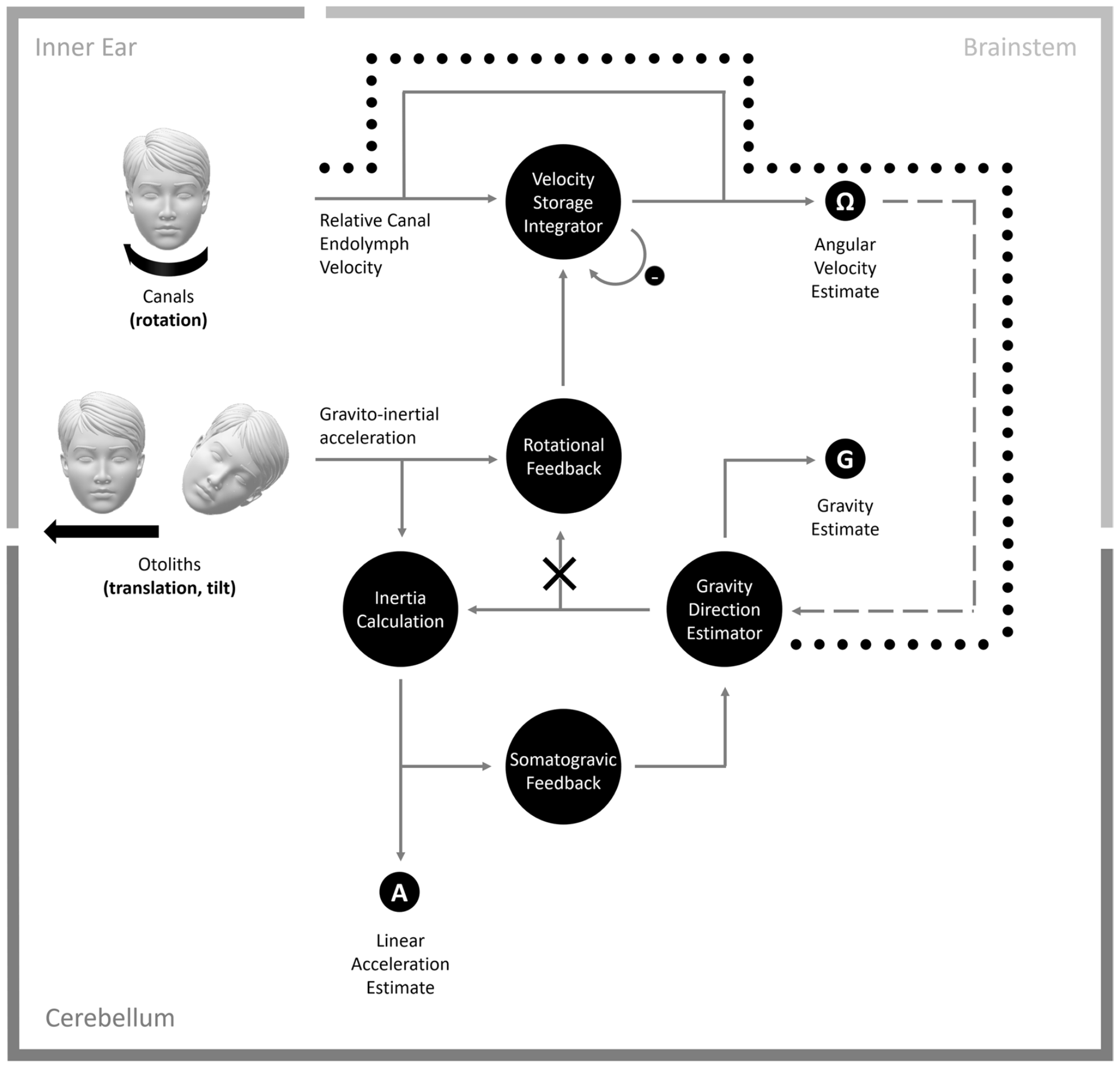
Figure : CPN model. Head rotations are sensed by the SCC in the inner ear. Here, the relative endolymph motion is detected by the cupula, which when deflected, creates a neural signal indicating the head rotation. The angular velocity estimate of the head is then centrally reconstructed by a velocity storage integrator in the brainstem, which considers the inherent deficiencies in the dynamics of the SCC. A negative feedback loop is added to the integrator to prevent noise accumulation in the integrator’s output. Head translation in space and tilt relative to gravity are sensed by the otolith organs in the inner ear. Here, a neural signal based on gravito-inertial acceleration is obtained, which does not distinguish between tilt and translation. Additional sensory clues about head orientation relative to gravity, provided by the SCC, help to solve the gravito-inertial ambiguity, by conveying a signal to a gravity direction (i.e., head tilt) estimator in the cerebellum, which integrates head rotation velocity over time. The gravity direction estimator delivers an estimate of the gravity vector, which can then be subtracted from the gravito-inertial acceleration to calculate inertia and estimate head linear acceleration (i.e., head translation). A somatogravic feedback loop is added to the gravity direction estimator to prevent error accumulation due to the integration of a noisy signal and bring the gravity estimate towards the gravito-inertial acceleration. Finally, a rotational feedback loop conveying a virtual angular velocity signal (i.e., the cross-vector product of the estimated gravity direction and the gravito-inertial acceleration) is additionally added to the velocity storage integrator to prevent inaccuracies in angular velocity estimation, which in turn would promote an incorrect estimation of gravity direction. The nodulus and uvula seem to be responsible for the estimation of gravity direction and feeding this signal into the rotational feedback loop. In persistent apogeotropic CPN, a lesion (cross) involving the vestibulo-cerebellar pathway that relays the estimated gravity to rotational feedback creates a bias of the estimated gravity direction toward the nose along the naso-occipital axis of the head while in ear-down positions. The persistent mismatch between the actual and estimated gravities forces the rotational feedback loop to bring the estimate of gravity direction toward the actual one, thus generating apogeotropic nystagmus. In persistent geotropic CPN, the mechanism is similar and only the direction of bias reverses (i.e., away from the nose). In paroxysmal (downwardly beating and apogeotropic) CPN, dysfunction of the nodulus and uvula generates a prominent post-rotatory response by the SCC, which causes an erroneous estimation of angular and gravity direction estimation (circles line). SCC semicircular canal, CPN central positional nystagmus. From Lemos and Strupp (Lemos and Strupp 2022).
Clinical presentation
The characteristics of the clinical presentation are variable — in terms of onset/offset (abrupt, gradual), chronicity (episodic, chronic) and intensity — which is unsurprising given the variety of diagnoses in which CPN has been observed (see below).
Physical examination
Macdonald and colleagues (Macdonald et al. 2017) state that in patients with central positional nystagmus (CPN), 58.6% exhibit additional ocular motor abnormalities (beyond the CPN itself), and 48.8% exhibit brainstem and/or cerebellar findings.
Ocular motor examination
Central positional nystagmus (CPN) can be subtle, so this is best discerned on infrared video oculography.
The positions in which CPN has been reported are usually the Dix-Hallpike maneuvers, head hanging, and side-lying.
Regarding the time course, CPN has been reported as paroxysmal or persistent, though these can be seen in one and the same patient.
Regarding the vector of nystagmus, it is noteworthy that CPN often is found in a single plane, though sometimes it can reverse direction in that plane (Choi et al. 2015). CPN can also occur in multiple planes in the same patient (Macdonald et al. 2017).
Macdonald and colleagues (Macdonald et al. 2017) conducted a systematic literature review of 28 studies (total of 82 patients). They identified the following patterns in CPN:
- 37% of patients had positional horizontal nystagmus.
- 29% of patients had positional down beat nystagmus.
- 2% of patients had positional torsional nystagmus.
- 2% of patients had positional up beat nystagmus.
- 30% of patients had positional nystagmus that included some combination of horizontal, vertical and torsional components.
Macdonald and colleagues further commented that the CPN was paroxysmal (lasting <60 seconds), occurring in:
- 85% of patients on straight head hanging.
- 64% of patients on Dix-Hallpike maneuvers.
Macdonald and colleagues (Macdonald et al. 2017), drawing on several other authors (Büttner et al. 2009; Buttner et al. 1999; von Brevern et al. 2015), provide the following Table to emphasize the features that can help distinguish nystagmus found in benign paroxysmal positional vertigo (BPPV) from central positional nystagmus (CPN).
|
Features |
Benign paroxysmal positional vertigo (BPPV) |
Central positional nystagmus (CPN) |
|
Latency following precipitating positioning maneuver |
1 – 5 seconds (shorter in horizontal canal BPPV depending on acceleration of head turn and cupulolithiasis) |
0 – 5 seconds |
|
Duration of nystagmus |
5 – 60 seconds (longer in cupulolithiasis) |
5 to >60 seconds |
|
Direction of nystagmus |
During stimulation in the plane of the affected canal torsional/vertical for posterior canal BPPV and anterior canal BPPV; horizontal for horizontal canal BPPV |
Pure vertical; pure torsional, not attributable to the stimulated canal plane |
|
Fatiguability |
Typical for posterior canal BPPV and anterior canal BPPV, rare in horizontal canal BPPV |
Possible |
|
Course of nystagmus and vertigo in an attack |
Crescendo-decrescendo typical, not common in horizontal canal BPPV |
Crescendo-decrescendo possible |
|
Vertigo (symptom) |
Typical |
Typical, with exceptions |
|
Nausea and vomiting |
Rare on single precipitating maneuvers (associated with intense nystagmus, not uncommon after several maneuvers) |
Frequent on single precipitating maneuvers (not necessarily) associated with strong nystagmus intensity |
|
Natural course of the condition |
Spontaneous recovery within several weeks in 70% – 80% |
Spontaneous recovery rare |
|
Associated neurological signs and symptoms |
None |
Often cerebellar and brainstem ocular motor signs |
|
Brain imaging |
Normal |
Lesions of the dorsal vermis and/or dorsolateral to the fourth ventricle |
|
Repositioning therapy |
Positional nystagmus disappears after appropriate positional therapy |
Refractory to repositioning therapy |
Table : Comparison of features of nystagmus found in benign paroxysmal positional vertigo (BPPV) and central positional vertigo (CPN) (Macdonald et al. 2017).
To get a sense of the frequency with which particular ocular motor features occur in CPN, Macdonald and colleagues (Macdonald et al. 2017) report data in the Table below.
|
Criterion |
Frequency |
Instances reported |
|
The direction of nystagmus is not attributable to the stimulated canal plane |
72.2% |
97 |
|
Pure vertical nystagmus |
31.3% |
144 |
|
Pure torsional nystagmus |
2.1% |
144 |
|
Direction-reversing nystagmus while the position is maintained |
3.7% |
82 |
|
Enhancement with fixation or reduced ocular fixation index |
91.7% |
12 |
|
Persists for at least 1 minute or as long as the precipitating head position is maintained |
40.7% |
81 |
|
Commences with no latency or within 3 seconds of assuming the provoking position |
94.7% |
38 |
|
Doe not fatigue with repetitive positioning |
82.1% |
28 |
|
Additional ocular motor abnormalities |
58.6% |
70 |
|
Additional brainstem or cerebellar symptoms and/or abnormalities |
48.8% |
82 |
|
Positional nystagmus does not resolve with repeated canalith repositioning maneuvers |
100% |
16 |
|
Prominent nausea or vomiting on positioning |
83.3% |
18 |
|
Prominent vertigo on positioning |
94.5% |
55 |
Table : Frequency of the typical indicators of central positional nystagmus (Macdonald et al. 2017).
Polensek and Tusa (Polensek and Tusa 2010) conducted a retrospective review of 26 patients with migraine associated vertigo (MAV) presenting while symptomatic, and they reported that 100% of these patients exhibited positional nystagmus on infrared oculography (noting that 19% of patients also exhibited spontaneous nystagmus).
A larger study by Young and colleagues (Young et al. 2021) included 101 adult patients with definite (n=27) or probable (n=74) vestibular migraine (migraine associated vertigo) and reported that positional nystagmus was observed in 100% in the supine position, 58% in the right lateral position and 92% in the left lateral position.
These findings regarding positional nystagmus in migraine associated vertigo are significant given the high prevalence of MAV in vestibular clinics.
Testing: general
For cases in which CPN is suspected, audio-vestibular testing (see below) does not “confirm” the diagnosis; rather, its purpose is to exclude peripheral (otologic) etiologies.
Testing: auditory
Auditory findings have been reported in individual cases of CPN (Cobb and Friedman 2006), but as of this writing there were no published studies systematically analyzing audiometric findings in CPN patients.
Testing: vestibular
As of this writing there were no published studies systematically analyzing vestibular test results (beyond instrumented ocular motor testing) in CPN patients.
Imaging
Brain imaging is always reasonable when CPN is suspected, with MRI (without and with contrast) being the modality of choice.
The systematic literature review by Macdonald and colleagues (Macdonald et al. 2017) observed that of 82 patients with CPN, radiologic abnormalities were reported in the following locations:
- 74% of patients had radiologic abnormalities reported in the cerebellum.
- 15% of patients had radiologic involvement reported in the 4th ventricle.
- 9% of patients had radiologic abnormalities reported in the brainstem alone.
Histopathology
Macdonald and colleagues (Macdonald et al. 2017) review studies that include some autopsy data.
Differential diagnosis
The study by Bertholon and colleagues (Bertholon et al. 2006) that prospectively studied 100 patients with positional nystagmus concluded that 12 (12%) had CPN. Of those 12 patients, the pathologies identified included cerebellar atrophy (in 2 out of 12), cerebellar infarct (in 1 out of 12), migraine associated vertigo (in 1 out of 12), multiple system atrophy (in 1 out of 12) and “various neurologic disorders,” not further specified (in 7 out of 12).
Combining imaging data with data from autopsy, Macdonald and colleagues (Macdonald et al. 2017) report the following findings, of which most pertain to the area of neuroanatomical involvement, and only a few (e.g., multiple system atrophy, CANVAS) refer to specific diagnoses:
- Cerebellum (n=61)
- Unspecified (n=20)
- Cerebellar vermis (n=15)
- Cerebellopontine angle (n=9)
- Nodulus (n=8)
- Superior cerebellar peduncle (n=3)
- Diffuse cerebellar involvement (n=6)
- Multiple system atrophy (n=3)
- Anti-epileptic toxicity (n=2)
- CANVAS (n=2)
- Brainstem (n=7)
- Isolated pontine lesion (n=1)
- Isolated medullary lesion (n=6)
- Floor of 4th ventricle (n=12)
- Diffuse cerebral disease (n=5)
Reported etiologies include:
- Migraine disorders, including:
- Vestibular migraine (Polensek and Tusa 2010; Young et al. 2021).
- Familial hemiplegic migraine type 1 (Suzuki et al. 2016; Yabe et al. 2008).
- Genetically-mediated cerebellar diseases, such as:
- Some of the spinocerebellar ataxias (SCA), such as SCA type 6 (Yu-Wai-Man et al. 2009) and SCA type 31 (Yabe et al. 2015).
- Episodic ataxia, type 1 (Jorge et al. 2022) and type 2 (Ling et al. 2020; Muth et al. 2021).
- Stroke (Lemos et al. 2019), usually in the cerebellum (Lee et al. 2017), particularly when involving the nodulus (Kim et al. 2012; Nam et al. 2009) or vermis (Sakata et al. 1991).
- Autoimmune-mediated, including:
- Paraneoplastic syndromes (Eggers et al. 2012; Wray et al. 2011).
- Multiple sclerosis (Anagnostou et al. 2006; Anagnostou et al. 2008; Lemos et al. 2019; Thomsen et al. 1978).
- Autoimmune (non-paraneoplastic, non-focal), such as in association with anti-GAD (glutamic acid decarboxylase) antibodies (Martins et al. 2018).
- Drug toxicity, such as from amiodarone (Arbusow et al. 1998), lamotrigine (Oh et al. 2006) or pregabalin (Choi et al. 2014).
If one starts with a known neurological diagnosis, it is noteworthy that CPN has been reported in:
- Nearly 100% of patients with migraine associated vertigo (MAV) (Polensek and Tusa 2010; Young et al. 2021).
- 49% of focal cerebellar lesions (Lee et al. 2017).
- 15% of patients with diffuse cerebellar disease (of various etiologies) (Feil et al. 2019).
- Supratentorial and infratentorial tumors (Cawthorne and Hinchcliffe 1961; Nylén 1939; Thomsen et al. 1978).
Benign paroxysmal positional vertigo (BPPV) can present with unusual forms of nystagmus, in which case it is sometimes referred to as “atypical BPPV.” However, when a case provisionally diagnosed as atypical BPPV fails to respond to reasonable attempts at treatment, it is sensible to consider the possibility of CPN (Bhattacharyya et al. 2017; Kim et al. 2022; Male et al. 2025; von Brevern et al. 2015).
Management
Management depends on the underlying diagnosis (if identifiable). Lemos (Lemos 2026) reviews selected etiologies, including:
- CPN attributable to an intracranial tumor may improve with resection (Chan et al. 1991; Drachman et al. 1977) and radiation (Barber 1984; Gregorius et al. 1976; Shoman and Longridge 2007).
- CPN attributable to infratentorial stroke occasionally improves with clonazepam (Oh et al. 2022).
- CPN attributable to an acute multiple sclerosis plaque in various brainstem locations may respond to methylprednisolone (Anagnostou et al. 2006; Anagnostou et al. 2008).
- CPN as part of a paraneoplastic syndrome may improve with treatment of the underlying malignancy (Wray et al. 2011).
- CPN due to familial hemiplegic migraine type 1 (FHM1) sometimes responds to acetazolamide (Suzuki et al. 2016), but not reliably (Yabe et al. 2008).
- CPN associated with episodic ataxia type 1 (EA1) may respond to acetazolamide (Jorge et al. 2022).
- CPN may improve by discontinuing the offending drug when due to toxicity from amiodarone (Arbusow et al. 1998), lamotrigine (Oh et al. 2006) or pregabalin (Choi et al. 2014).
Prognosis
Prognosis depends on the underlying diagnosis (if identifiable).
References
Anagnostou E, Mandellos D, Limbitaki G, Papadimitriou A, Anastasopoulos D (2006) Positional nystagmus and vertigo due to a solitary brachium conjunctivum plaque. J Neurol Neurosurg Psychiatry 77: 790-2. doi: 10.1136/jnnp.2005.084624
Anagnostou E, Varaki K, Anastasopoulos D (2008) A minute demyelinating lesion causing acute positional vertigo. J Neurol Sci 266: 187-9. doi: 10.1016/j.jns.2007.09.013
Arbusow V, Strupp M, Brandt T (1998) Amiodarone-induced severe prolonged head-positional vertigo and vomiting. Neurology 51: 917. doi: 10.1212/wnl.51.3.917
Barber HO (1984) Positional nystagmus. Otolaryngol Head Neck Surg 92: 649-55. doi: 10.1177/019459988409200611
Bertholon P, Tringali S, Faye MB, Antoine JC, Martin C (2006) Prospective study of positional nystagmus in 100 consecutive patients. Ann Otol Rhinol Laryngol 115: 587-94. doi: 10.1177/000348940611500804
Bhattacharyya N, Gubbels SP, Schwartz SR, Edlow JA, El-Kashlan H, Fife T, Holmberg JM, Mahoney K, Hollingsworth DB, Roberts R, Seidman MD, Steiner RW, Do BT, Voelker CC, Waguespack RW, Corrigan MD (2017) Clinical Practice Guideline: Benign Paroxysmal Positional Vertigo (Update). Otolaryngol Head Neck Surg 156: S1-S47. doi: 10.1177/0194599816689667
Büttner U, Brandt T, Helmchen C (2009) The direction of nystagmus is important for the diagnosis of central paroxysmal positioning nystagmus (cPPV). Neuro-Ophthalmology 21: 97-104. doi: 10.1076/noph.21.2.97.3919
Buttner U, Helmchen C, Brandt T (1999) Diagnostic criteria for central versus peripheral positioning nystagmus and vertigo: a review. Acta Otolaryngol 119: 1-5. doi: 10.1080/00016489950181855
Cawthorne T, Hinchcliffe R (1961) Positional nystagmus of the central type as evidence of subtentorial metastases. Brain 84: 415-26. doi: 10.1093/brain/84.3.415
Chan T, Logan P, Eustace P (1991) Intermittent downbeat nystagmus secondary to vermian arachnoid cyst with associated obstructive hydrocephalus. J Clin Neuroophthalmol 11: 293-6.
Choi JY, Glasauer S, Kim JH, Zee DS, Kim JS (2018a) Characteristics and mechanism of apogeotropic central positional nystagmus. Brain 141: 762-775. doi: 10.1093/brain/awx381
Choi JY, Kim JH, Kim HJ, Glasauer S, Kim JS (2015) Central paroxysmal positional nystagmus: Characteristics and possible mechanisms. Neurology 84: 2238-46. doi: 10.1212/WNL.0000000000001640
Choi JY, Park YM, Woo YS, Kim SU, Jung JM, Kwon DY (2014) Perverted head-shaking and positional downbeat nystagmus in pregabalin intoxication. J Neurol Sci 337: 243-4. doi: 10.1016/j.jns.2013.12.007
Choi SY, Jang JY, Oh EH, Choi JH, Park JY, Lee SH, Choi KD (2018b) Persistent geotropic positional nystagmus in unilateral cerebellar lesions. Neurology 91: e1053-e1057. doi: 10.1212/WNL.0000000000006167
Cobb FE, Friedman LB (2006) Positional nystagmus of central origin. J Am Acad Audiol 17: 85-92. doi: 10.3766/jaaa.17.2.2
Drachman DA, Diamond ER, Hart CW (1977) Posturally-evoked vomiting; Association with posterior fossa lesions. Ann Otol Rhinol Laryngol 86: 97-101. doi: 10.1177/000348947708600117
Eggers SD, Pittock SJ, Shepard NT, Habermann TM, Neff BA, Klebig RR (2012) Positional periodic alternating vertical nystagmus with PCA-Tr antibodies in Hodgkin lymphoma. Neurology 78: 1800-2. doi: 10.1212/WNL.0b013e3182583085
Eggers SDZ, Bisdorff A, von Brevern M, Zee DS, Kim JS, Perez-Fernandez N, Welgampola MS, Della Santina CC, Newman-Toker DE (2019) Classification of vestibular signs and examination techniques: Nystagmus and nystagmus-like movements. J Vestib Res 29: 57-87. doi: 10.3233/VES-190658
Feil K, Strobl R, Schindler A, Krafczyk S, Goldschagg N, Frenzel C, Glaser M, Schoberl F, Zwergal A, Strupp M (2019) What Is Behind Cerebellar Vertigo and Dizziness? Cerebellum 18: 320-332. doi: 10.1007/s12311-018-0992-8
Glasauer S, Dieterich M, Brandt T (2001) Central positional nystagmus simulated by a mathematical ocular motor model of otolith-dependent modification of Listing’s plane. J Neurophysiol 86: 1546-54. doi: 10.1152/jn.2001.86.4.1546
Gregorius FK, Crandall PH, Baloh RW (1976) Positional vertigo with cerebellar astrocytoma. Surg Neurol 6: 283-6.
Jorge A, Melancia D, Figueiredo C, Galego O, Oliveira J, Martins AI, Lemos J (2022) Downbeat Nystagmus in Episodic Ataxia Type 1 Associated with a Novel KCNA1 Mutation. Mov Disord 37: 430-432. doi: 10.1002/mds.28843
Kim HA, Yi HA, Lee H (2012) Apogeotropic central positional nystagmus as a sole sign of nodular infarction. Neurol Sci 33: 1189-91. doi: 10.1007/s10072-011-0884-x
Kim JM, Lee SH, Kim HJ, Kim JS (2022) Less talked variants of benign paroxysmal positional vertigo. J Neurol Sci 442: 120440. doi: 10.1016/j.jns.2022.120440
Lee SU, Choi JY, Kim HJ, Park JJ, Zee DS, Kim JS (2017) Impaired Tilt Suppression of Post-Rotatory Nystagmus and Cross-Coupled Head-Shaking Nystagmus in Cerebellar Lesions: Image Mapping Study. Cerebellum 16: 95-102. doi: 10.1007/s12311-016-0772-2
Lee SU, Kim HJ, Lee ES, Choi JH, Choi JY, Kim JS (2021) Central positional nystagmus in inferior cerebellar peduncle lesions: a case series. J Neurol 268: 2851-2857. doi: 10.1007/s00415-021-10435-7
Lemos J (2026) Treatment of Central Positional Nystagmus. In: Ravat P, Kattah JC, Shaikh AG (eds) Advances in Translational Neuroscience of Vestibular and Eye Movement Disorders. Springer Nature Switzerland, Cham, pp 265-278
Lemos J, Martins AI, Duque C, Pimentel S, Nunes C, Goncalves AF (2019) Positional Testing in Acute Vestibular Syndrome: a Transversal and Longitudinal Study. Otol Neurotol 40: e119-e129. doi: 10.1097/MAO.0000000000002067
Lemos J, Strupp M (2022) Central positional nystagmus: an update. Journal of Neurology 269: 1851-1860. doi: 10.1007/s00415-021-10852-8
Ling X, Kim HJ, Lee JH, Choi JY, Yang X, Kim JS (2020) Positioning Velocity Matters in Central Paroxysmal Positional Vertigo: Implication for the Mechanism. Front Neurol 11: 591602. doi: 10.3389/fneur.2020.591602
Macdonald NK, Kaski D, Saman Y, Al-Shaikh Sulaiman A, Anwer A, Bamiou DE (2017) Central Positional Nystagmus: A Systematic Literature Review. Front Neurol 8: 141. doi: 10.3389/fneur.2017.00141
Male AJ, Korres G, Koohi N, Kaski D (2025) Rethinking positional nystagmus: beyond BPPV. Journal of Neurology 272: 615. doi: 10.1007/s00415-025-13335-2
Martins AI, Carvalho JN, Amorim AM, Geraldo A, Eggenberger E, Lemos J (2018) Disabling Central Paroxysmal Positioning Upbeat Nystagmus and Vertigo Associated With the Presence of Anti-Glutamic Acid Decarboxylase Antibodies. J Neuroophthalmol 38: 32-35. doi: 10.1097/WNO.0000000000000547
Muth C, Teufel J, Schols L, Synofzik M, Franke C, Timmann D, Mansmann U, Strupp M (2021) Fampridine and Acetazolamide in EA2 and Related Familial EA: A Prospective Randomized Placebo-Controlled Trial. Neurol Clin Pract 11: e438-e446. doi: 10.1212/CPJ.0000000000001017
Nam J, Kim S, Huh Y, Kim JS (2009) Ageotropic central positional nystagmus in nodular infarction. Neurology 73: 1163. doi: 10.1212/WNL.0b013e3181bacfde
Nylén CO (1939) Statistical analysis of oto-neurological symptomatology in supra- and subtentorial tumours. Acta Oto-Laryngologica 27: 24-31. doi: 10.3109/00016483909120560
Oh EH, Kim H, Choi SY, Choi KD, Choi JH (2022) Paroxysmal central positional nystagmus responsive to clonazepam. J Neurol 269: 1028-1031. doi: 10.1007/s00415-021-10770-9
Oh SY, Kim JS, Lee YH, Lee AY, Kim J, Kim JM (2006) Downbeat, positional, and perverted head-shaking nystagmus associated with lamotrigine toxicity. J Clin Neurol 2: 283-5. doi: 10.3988/jcn.2006.2.4.283
Polensek SH, Tusa RJ (2010) Nystagmus during attacks of vestibular migraine: an aid in diagnosis. Audiol Neurootol 15: 241-6. doi: 10.1159/000255440
Sakata E, Ohtsu K, Itoh Y (1991) Positional nystagmus of benign paroxysmal type (BPPN) due to cerebellar vermis lesions. Pseudo-BPPN. Acta Otolaryngol Suppl 481: 254-7. doi: 10.3109/00016489109131394
Shoman N, Longridge N (2007) Cerebellar vermis lesions and tumours of the fourth ventricle in patients with positional and positioning vertigo and nystagmus. J Laryngol Otol 121: 166-9. doi: 10.1017/S0022215106004063
Suzuki M, Fujiwara K, Tsubuku T, Yabe I, Sasaki H, Fukuda S (2016) Time course of downbeat positioning nystagmus in familial hemiplegic migraine type 1 treated with acetazolamide. J Neurol Sci 368: 206-8. doi: 10.1016/j.jns.2016.07.020
Thomsen J, Zilstorff K, Johnsen NJ (1978) Positional nystagmus of the persistent type. ORL J Otorhinolaryngol Relat Spec 40: 86-91. doi: 10.1159/000275390
von Brevern M, Bertholon P, Brandt T, Fife T, Imai T, Nuti D, Newman-Toker D (2015) Benign paroxysmal positional vertigo: Diagnostic criteria. J Vestib Res 25: 105-17. doi: 10.3233/VES-150553
Wray SH, Dalmau J, Chen A, King S, Leigh RJ (2011) Paraneoplastic disorders of eye movements. Ann N Y Acad Sci 1233: 279-84. doi: 10.1111/j.1749-6632.2011.06113.x
Yabe I, Kitagawa M, Suzuki Y, Fujiwara K, Wada T, Tsubuku T, Takeichi N, Sakushima K, Soma H, Tsuji S, Niino M, Saitoh S, Sasaki H (2008) Downbeat positioning nystagmus is a common clinical feature despite variable phenotypes in an FHM1 family. J Neurol 255: 1541-4. doi: 10.1007/s00415-008-0970-z
Yabe I, Matsushima M, Yoshida K, Ishikawa K, Shirai S, Takahashi I, Sasaki H (2015) Rare frequency of downbeat positioning nystagmus in spinocerebellar ataxia type 31. J Neurol Sci 350: 90-2. doi: 10.1016/j.jns.2014.12.042
Young AS, Nham B, Bradshaw AP, Calic Z, Pogson JM, D’Souza M, Halmagyi GM, Welgampola MS (2021) Clinical, oculographic, and vestibular test characteristics of vestibular migraine. Cephalalgia: 3331024211006042. doi: 10.1177/03331024211006042
Yu-Wai-Man P, Gorman G, Bateman DE, Leigh RJ, Chinnery PF (2009) Vertigo and vestibular abnormalities in spinocerebellar ataxia type 6. J Neurol 256: 78-82. doi: 10.1007/s00415-009-0068-2
![]()