By Marcello Cherchi, MD PhD
For patients
“Chiari malformation” (CM) refers to several rare malformations of certain parts of the brain (usually the cerebellum, sometimes also the brainstem). Although these malformations are present from birth, the most common types may not cause symptoms until adulthood, and typical symptoms include headaches, visual problems (such as blurry vision), dizziness and difficulty swallowing. The diagnosis may be suspected from findings on a patient’s physical examination, but confirmation with a brain MRI is appropriate. Even if a patient’s brain MRI shows findings compatible with CM, it is still reasonable to search for other, more common causes of these symptoms, and if that search does not show any more likely explanation, then CM is probably the correct diagnosis. Some doctors may consider a trial of medication for certain symptoms. If a patient’s symptoms are very intrusive, or if they do not respond to medication, then referral to neurosurgery is appropriate.
For clinicians
Overview
Chiari malformations (CMs) are developmental anomalies that neuroanatomically involve structures in the posterior fossa and craniocervical junction, but can also involve other organ systems. The more common types are CM1 (58.7% of cases), CM2 (22.9% of cases) and CM4 (15.3% of cases). The remaining types (0, 0.5, 1.5, 3.5, 4, 5) are very rare, and would not usually present to an otoneurologist.
In an otoneurology practice, a patient may be referred (usually for a complaint of disequilibrium, or for findings of nystagmus noted on physical examination) who has had brain imaging performed that identified cerebellar tonsillar descent. Whether or not the tonsillar descent is sufficient to meet criteria for Chiari 1 malformation, the usual clinical query is whether the position of the cerebellar tonsils accounts for the patient’s disequilibrium, and if so, whether the patient should be referred to neurosurgery (usually suboccipital decompression, sometimes with high cervical laminectomy).
From an otoneurological perspective the unifying features of CM are oculomotor and vestibular abnormalities related to cerebellar dysfunction, as the cerebellum is either being physically impinged upon at the craniocervical junction (due to herniation into the spinal canal), or is hypoplastic. Oculomotor findings have been well described in CM1, fairly described in CM2, and not described well (or at all) for the remaining types. The most common vestibular oculomotor findings include gaze evoked nystagmus (horizontal more than vertical), spontaneous vertical nystagmus (down beat more than up beat), deficits in smooth pursuit and optokinetic responses, all of which localize to the cerebellum, and are not specific for CM. Less common vestibular oculomotor findings include periodic alternating nystagmus and see-saw nystagmus. Other (non-vestibular) oculomotor findings include strabismus, specifically acquired estotropia (usually comitant), and rarely exotropia.
Some aspects of the nystagmus (at least of CM1) may respond to pharmacologic treatment, though data supporting efficacy are weak.
Surgical decompression is sometimes attempted in management of CM1. Reports of surgical outcomes are variable. Moreover, since having a rare disease (such as CM) does not preclude getting a more common disease, even in a patient with radiologically confirmed CM we feel it is still medically reasonable to undertake a screening workup (we usually check vestibular evoked myogenic potentials, video head impulse testing, videonystagmography and rotatory chair testing) for more common causes of disequilibrium, such as vestibular neuritis. While it is true that unilateral vestibular weakness has been reported in CM1, such a deficit is much more commonly caused by vestibular neuritis, so a trial of vestibular rehabilitation therapy is reasonable. If workup for alternative diagnoses is unrevealing, and if a patient’s symptoms interfere with level of function, and if vestibular and oculomotor findings are compatible with what is known about CM, then referral to neurosurgery is reasonable.
Audiologically, some CM patients exhibit sensorineural hearing loss, which may be asymmetrical. There are case reports of this hearing loss improving after surgical decompression.
Introduction
Carmel and Markesbery (Carmel and Markesbery 1972) point out that John Cleland (1835 – 1925) was probably the first to publish (Cleland 1883) a description of posterior fossa anomalies that would later come to be known as Chiari malformations, but Dr. Cleland has not received appropriate credit for the discovery.
Dr. Hans Chiari (1851 – 1916) described three types of malformations in 1891 (Chiari 1891) and a fourth type in 1895 (Chiari 1895). Over the years investigators have described additional types (0, 0.5, 1.5, 3.5, 5).

According to Carmel and Markesbery (Carmel and Markesbery 1972), the 1894 paper by Dr. Julius Arnold (1835 – 1915) (Arnold 1894) “described multiple anomalies in a newborn infant. Much of this paper is devoted to details of visceral and lung abnormalities, and only scant attention was given to the brain stem.”
Despite the limited description in Arnold’s work, Carmel and Markesbery (Schwalbe and Gredig 1907) note that a subsequent publication by Schwalbe and Gredig (Schwalbe and Gredig 1907) referred to the second type of malformation as the “malformation of Arnold and Chiari,” so for a time this was known as an “Arnold-Chiari malformation.”
In the present discussion we will refer to these as “Chiari malformations,” abbreviated CM.
The Table below, from Russel and Wick (Russell, Wick et al. 1992), provides a very brief overview of the four main types.

Horn and colleagues (Horn, Shepard et al. 2018) analyzed a nationwide database of 520,561 reported cases of CM. Of these they identified that CM1 accounted for 305,726 (58.7%), CM2 for 119,632 (22.9%), CM3 for 15,540 (3%) and CM4 for 79,663 (15.3%). Thus the relative prevalence, in descending order, is CM1 > CM2 > CM4 > CM3.
Horn and colleagues also reported that 44.3% of cases were associated with at least one concurrent anomaly (other than the Chiari malformation itself), which can be in almost any other organ system.
Radiologic landmarks
The anatomical landmark by which cerebellar tonsillar descent is judged is the McRae line (McRae 1953) which, on a midsagittal image, connects the basion (anterior margin of the foramen magnum) to the opisthion (posterior margin of the foramen magnum).
Type 0 Chiari malformation (CM0)
Bogdanov and colleagues (Bogdanov, Faizutdinova et al. 2022) comment that, “Some investigators have used CM0 [Chiari malformation type 0] to refer to patients with and without syringomyelia with minimal or absent TH [cerebellar tonsillar herniation], radiological features of SPCF [small posterior cranial fossa], and typical clinical signs and symptoms of CM1, including occipital headache, posterior cervical pain, and cerebellomedullary dysfunction,” adding that, “investigations support classifying CM0 without syringomyelia as a classical CM1 [Chiari malformation type 1] variant because CM0 and CM1 share findings of an SPCF and caudally located obex.”
In other words, CM0 is very similar to CM1 except that the tonsillar descent does not meet criteria for CM1.
The Figure below, from Bogdanov and colleagues (Bogdanov, Faizutdinova et al. 2022), shows several examples of sagittal and axial images of CM0 cases.
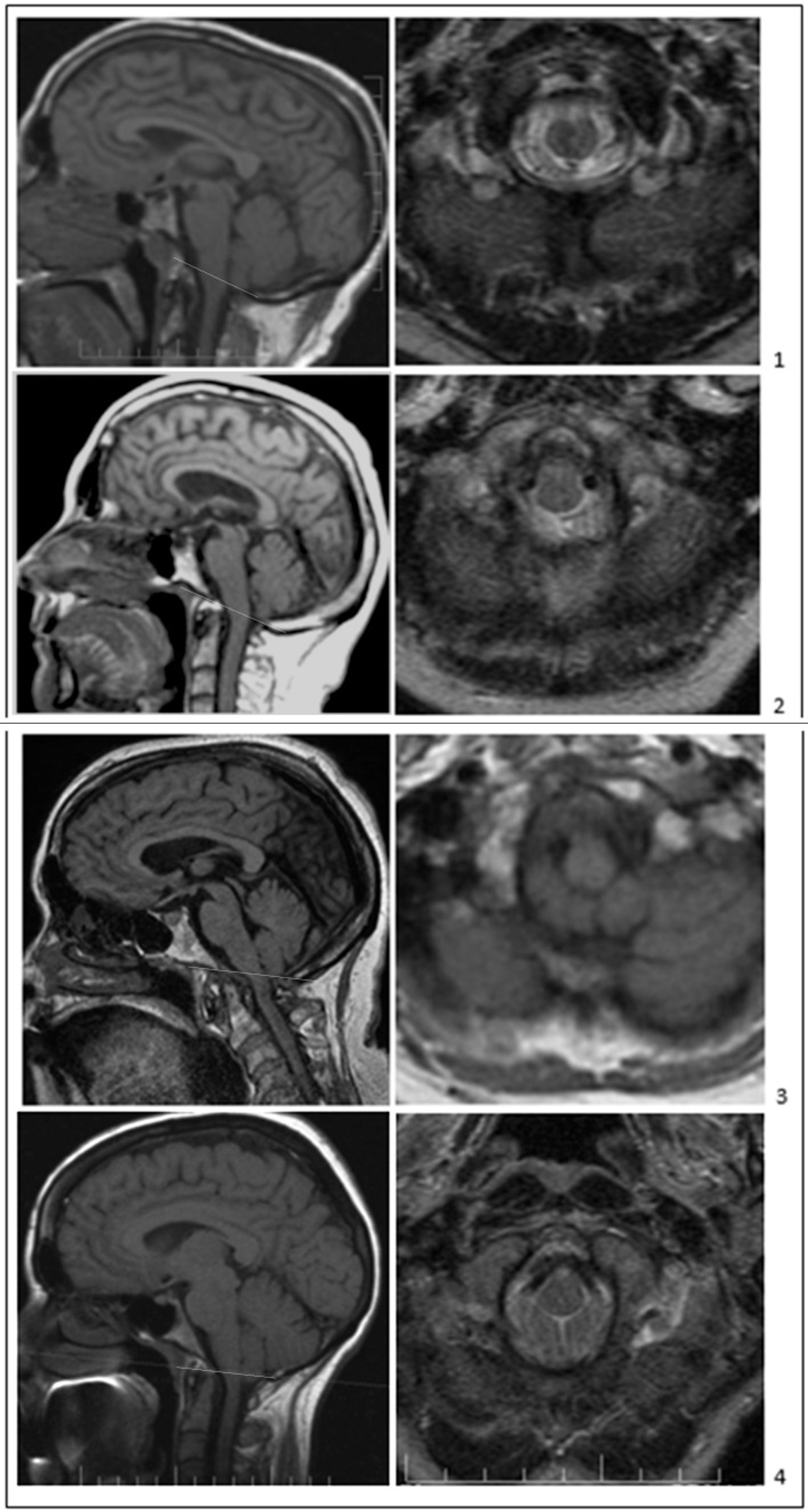
Figure : Types of CM0 without syringomyelia. The McRae line (white line) is shown on the T1-weighted sagittal images (left panels). Axial images at McRae’s line are shown (right panels) on T1-weighted (3) and T2-weighted (1,2,4) images. Adult symptomatic CM0-only patients with tonsillar herniation ≤2 mm (panels (1–3)) and borderline CM1-only patients with tonsillar herniation 3 mm (4). All patients demonstrate short bones (CL and SO < 40 mm), crowdedness of the PCF, tight foramen magnum, and CM1-like clinical manifestations: transient localized suboccipital cough-related headaches (1) or constant and transient suboccipital headaches (2–4), truncal ataxia, vertigo, dizziness, and upper motor neuron signs. The sagittal images in (1,3) (left panels) also show spinal canal narrowing at the level of the odontoid. From Bogdanov et al. (2022).
Type 0.5 Chiari malformation (CM0.5)
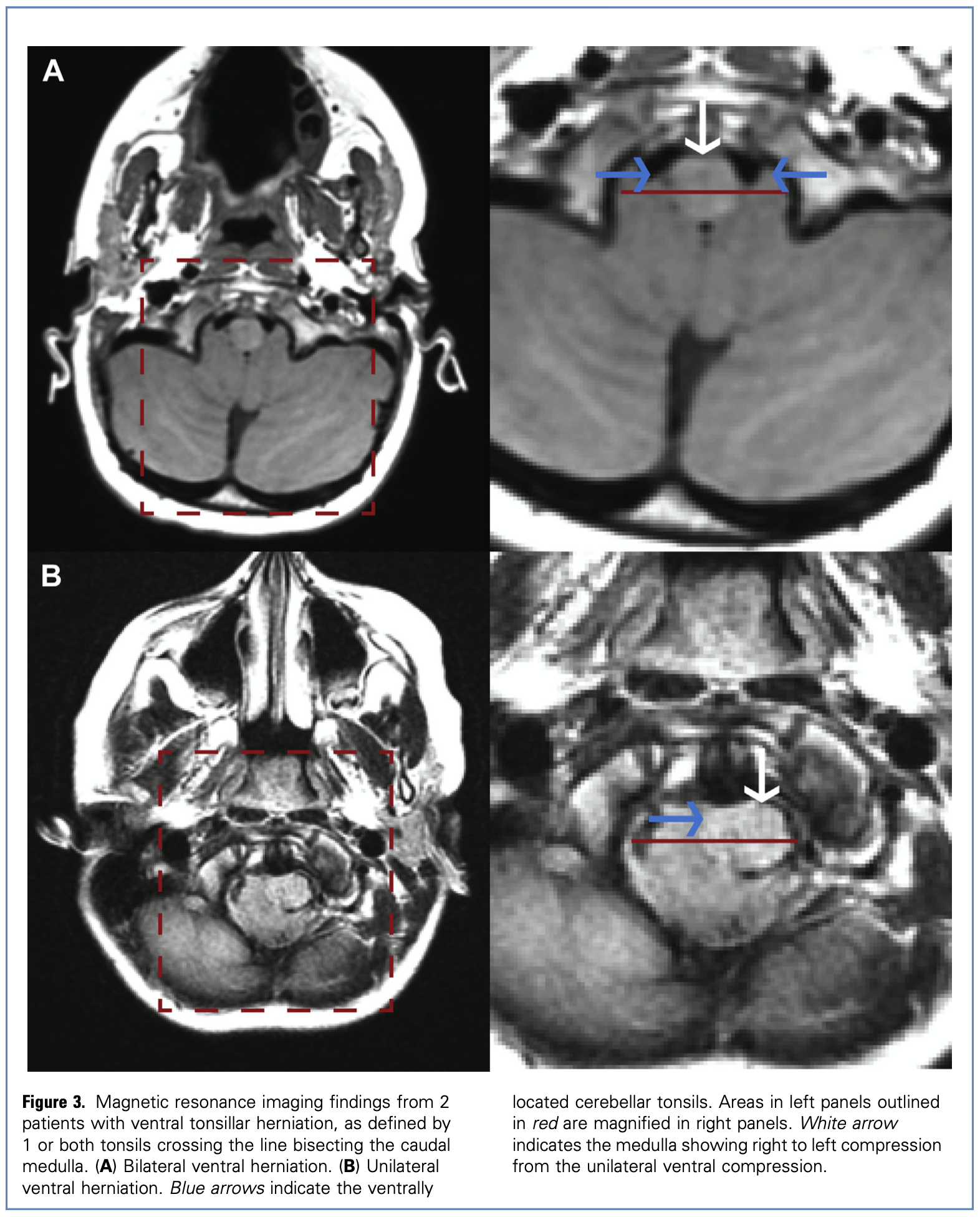
Morgenstern and colleagues (Morgenstern, Tosi et al. 2020) identified a group of patients in whom the cerebellar tonsils were not merely downwardly displaced, but also ventrally herniated, which they define as, “the tonsils crossing a line that bisects the caudal medulla at the level of the foramen magnum.”
The Figure below, from Morgenstern and colleagues (Morgenstern, Tosi et al. 2020), demonstrates axial images in which the cerebellar tonsils wrap ventrally around the medulla.
Type 1 Chiari malformation (CM1)
Chiari’s original description (Chiari 1891) was:
“The first type is the lengthening of the tonsils and the medial parts of the inferior lobes of the cerebellum to form cone-shaped processes which accompany the medulla oblongata into the vertebral canal.”
Cesmebasi and colleagues (Cesmebasi, Loukas et al. 2015) provide a more detailed list of the anatomical features associated with CM1:
Brain: Usually normal except for the cerebellar tonsils.
Ventricles/cisterns: In some cases the 4th ventricle is elongated.
Cerebellar tonsils: Caudal displacement of cerebellar tonsils more than 3 – 5 mm inferior to the plane of the foramen magnum.
Skull: Basilar skull and craniocervical junction abnormalities are found in about 50% of cases and can include underdevelopment of the occipital bone; increased size of the foramen magnum; shortened clivus.
Meninges: Slope of the tentorium cerebelli is often elevated; arachnoid mater at the level of the foramen magnum is often thickened.
Spinal canal: Commonly associated findings include a Klippel-Feil deformity (maldeveloped vertebra and/or fusion between two or more cervical vertebrae); atlantoaxial assimilation; retroflexion of the odontoid process; thickening of the ligamentum flavum; scoliosis.
Spinal cord: Syrinx is present in 50 – 75% of cases.
Barkovich and colleagues (Barkovich, Wippold et al. 1986) reviewed MRIs from 25 patients with CM1 and compared them to 200 normal controls. They reported that a cutoff point of 3 or 4 mm of cerebellar tonsillar descent had 96% sensitivity and 99.5% specificity for identifying CM1, and that a cutoff point of 5 mm of cerebellar tonsillar descent had a sensitivity of 92% and specificity of 100% for identifying CM1. Based on this study, many investigators use cerebellar tonsillar descent of ≥5 mm as a criterion for CM1.
Epidemiology of CM1
The prevalence of CM1 is estimated at 96 cases per 100,000 people, with a median age at diagnosis of 10.5 years (Morgenstern, Tosi et al. 2020).
The age of symptom onset is variable. Some investigators report that most patients with CM1 develop symptoms in the 2nd or 3rd decade of life (Goodwin and Halvorson 2012), while others report the 3rd or 4th decade of life (Pokharel and Siatkowski 2004).
Clinical presentation of CM1
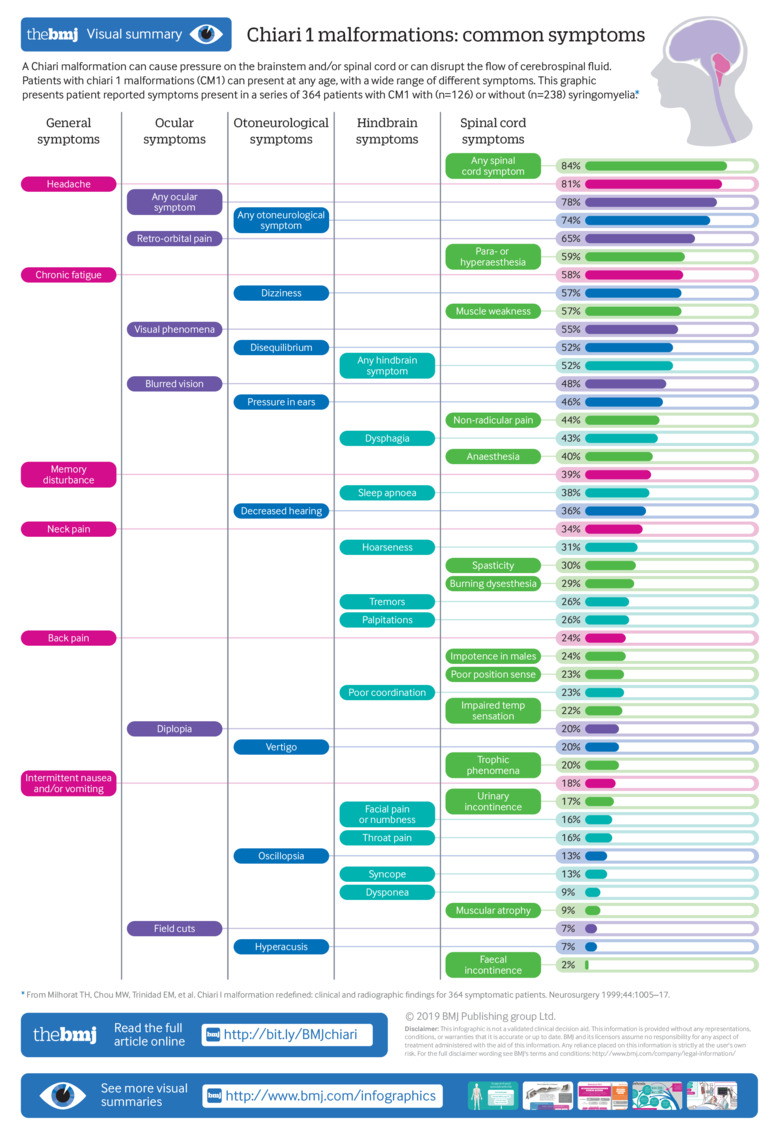
Piper and colleagues (Piper, Pike et al. 2019) took information from Milhorat and colleagues (Milhorat, Chou et al. 1999) regarding the categories and frequencies of symptoms in CM1 patients:
Audiologic findings in CM1
Kumar and colleagues (Kumar, Patni et al. 2002) reported on 77 patients with CM1. Of these patients they noted 10 had bilateral sensorineural hearing loss and 22 had unilateral sensorineural hearing loss.
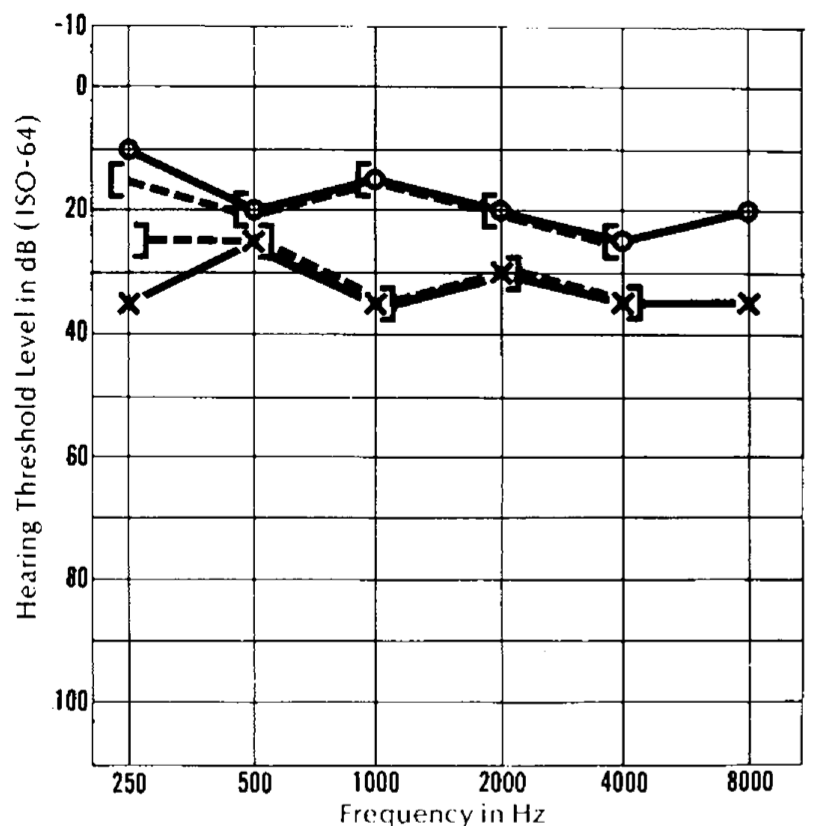
The sensorineural hearing loss can be symmetrical or asymmetrical, and the patterns variable. The Figures below show audiograms from three different patients reported by Rydell and colleagues.
 |
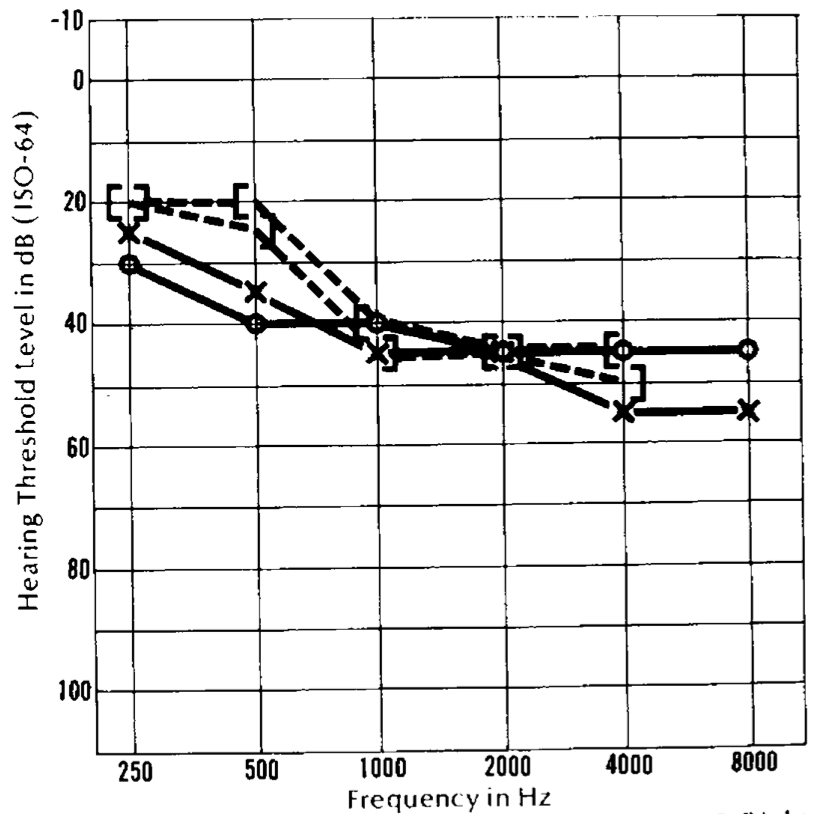 |
 |
Oculomotor findings in CM1
Regrettably, many reports may list “nystagmus” as a physical examination finding in CM1 but do not characterize it further, which limits their utility. For instance, Hida and colleagues (Hida, Iwasaki et al. 1999) that 2/16 (13%) of their CM1 patients exhibited “nystagmus,” but provided no details.
Shaikh and Ghasia (Shaikh and Ghasia 2015) comment that overall, nystagmus (of all types) is the most common oculomotor abnormality in CM1. They state that in CM1, gaze-evoked nystagmus (sometimes with rebound nystagmus) is present in 30% of patients, and down beat nystagmus is present in 4 – 6 % of patients.
Kumar and colleagues (Kumar, Patni et al. 2002) reported on 77 patients with CM1. Of these patients they noted that 27 (35%) had horizontal nystagmus, 3 (4%) had spontaneous up beat nystagmus, 4 (5%) had spontaneous down beat nystagmus, 4 (5%) had saccadic dysmetria, 3 (4%) had impaired optokinetic nystagmus, 12 (16%) had impaired smooth pursuit, and 9 (12%) patients had “positional nystagmus.”
Liebenberg and colleagues (Liebenberg, Georges et al. 2005) reviewed forty CM1 patients. They reported that 12/40 (30%) had oculomotor and vestibulo-ocular abnormalities. Of these twelve, 4/12 (33%) had “horizontal nystagmus,” 3/12 (25%) had down beat nystagmus, and 1/12 (8%) had “jerky pursuit.”
Numerous reports describe spontaneous down beat nystagmus in CM1 (Chait and Barber 1979, Faria, Spector et al. 1980, Hosford and Spector 1985, Goodwin and Halvorson 2012). This spontaneous down beat nystagmus may augment on lateral gaze (Pieh and Gottlob 2000, Goodwin and Halvorson 2012). Sometimes this down beat nystagmus is intermittent (Pedersen, Troost et al. 1980, Yee, Baloh et al. 1984).
Rarely, CM1 may produce spontaneous up beat nystagmus (Hatashita, Shimoji et al. 1980).
A number of cases of CM1 have been reported presenting primarily or exclusively with acquired esotropia (Passo, Shults et al. 1984, Lewis, Kline et al. 1996, Weeks and Hamed 1999, Biousse, Newman et al. 2000, Imes and Quinn 2001, Defoort-Dhellemmes, Denion et al. 2002, Pokharel and Siatkowski 2004, Hentschel, Yen et al. 2005, Kowal, Yahalom et al. 2006), usually comitant.
Ghasia and colleagues (Ghasia, Gulati et al. 2014) reported intermittent exotropia and gaze-evoked nystagmus depending on viewing position.
There are several reports of periodic alternating nystagmus (PAN) in CM1 (Korres, Balatsouras et al. 2001, Al-Awami, Flanders et al. 2005).
Rarely, CM1 can cause see-saw nystagmus (Zimmerman, Roach et al. 1986).
Instrumented vestibular testing in CM1: videonystagmography (VNG) or electronystagmography (ENG)
Goldschagg and colleagues (Goldschagg, Feil et al. 2017) treated ten adult CM1 patients with suboccipital decompression and unilateral tonsillectomy. They reported that 9 of these patients underwent videonystagmography (VNG) pre-operatively, and that 3/9 (33%) exhibited pathological caloric asymmetry.
Yee and colleagues (Yee, Baloh et al. 1984) reported a case of CM1 in which electronystagmography showed geotropic direction changing positional nystagmus.
Mossman and colleagues (Mossman, Bronstein et al. 1990) studied electronystagmography on one CM1 patient and observed convergence nystagmus, which is a very unusual finding.
Instrumented vestibular testing in CM1: scleral search coils
Lewis and colleagues (Lewis, Kline et al. 1996) studied five CM1 patients with scleral search coils and reported that, “All patients reported onset of horizontal diplopia due to acquired esotropia as an initial manifestation of the Arnold-Chiari I malformation.” The authors concluded that, “Acquired esotropia, often in association with other eye movement abnormalities, may be an early sign of Arnold-Chiari I malformation.”
Pieh and Gottlob (Pieh and Gottlob 2000) studied two CM1 patients with scleral search coils and reported “nystagmus of skew,” meaning they observed synchronous down beat nystagmus of one eye and up beat nystagmus of the other eye.
Instrumented vestibular testing in CM1: video head impulse testing (vHIT)
Kim and colleagues (Kim, Kim et al. 2019) studied a CM1 patient and reported that horizontal head impulses upward ocular deviation during the impulse, followed by a corrective downward saccade — in other words, perverted nystagmus.
Instrumented vestibular testing in CM1: rotatory chair testing (RCT)
Baloh and colleagues (Baloh, Beykirch et al. 1988) studied a CM1 patient with serial videonystagmography and rotatory chair testing over 3 years. They reported that on rotatory chair testing, the step velocity component showed progressive decline in the duration of post-rotatory nystagmus (in other words, progressive decrease in the time constant); slow harmonic acceleration showed no responses at 0.2 Hz (responses were present at higher frequencies); and disappearance of optokinetic nystagmus. These findings are similar to what would be seen in bilateral vestibular weakness.
Yee and colleagues (Yee, Baloh et al. 1984) reported a case of CM1 in which caloric responses were absent bilaterally, and slow harmonic acceleration showed reduced vestibulo-ocular reflex responses at low frequencies. Again, these findings are similar to what would be seen in bilateral vestibular weakness.
Summary of oculomotor findings
Russel and Wick (Russell, Wick et al. 1992) summarize possible oculomotor findings in CM1 and CM2 as follows:
“Patients typically have down-beat nystagmus caused by herniation of the cerebellar tonsils downward through the foramen magnum. The imbalance of the otolith-ocular reflexes, brought on by faulty communication between the vestibular neurons, the vertical subnuclei of the oculomotor nucleus, and the interstitial nucleus, results in vertical pendular eye movements. Loss of the flocculus inhibition due to compression of the cerebellum frequently prevents fixation suppression, causing constant shifts in fixation as the patient’s eye tries to find a fixation point. Clinical evaluation of eye movements generally reveals that the saccade system is intact, but smooth pursuits, optokinetic nystagmus (OKN), and fixation suppression are all significantly affected. It is possible, although less likely, to have down beat nystagmus associated with gaze deviation internuclear ophthalmoplegia [and] see-saw nystagmus.”
Instrumented vestibular testing in CM1: vestibular evoked myogenic potentials (VEMPs)
Goldschagg and colleagues (Goldschagg, Feil et al. 2017) treated ten adult CM1 patients with suboccipital decompression and unilateral tonsillectomy. They reported that two of the patients had abnormal cervical vestibular evoked myogenic potentials, and that these both improved postoperatively.
Instrumented vestibular testing in CM1: computerized dynamic posturography (CDP)
Goldschagg and colleagues (Goldschagg, Feil et al. 2017) treated ten adult CM1 patients with suboccipital decompression and unilateral tonsillectomy. They reported no changes in static posturography from pre-operative to postoperative performance.
Imaging in CM1
The Figure below, from Piper and colleagues (Piper, Pike et al. 2019), shows T1 (left) and T2 (right) sagittal images of an infant with CM1.
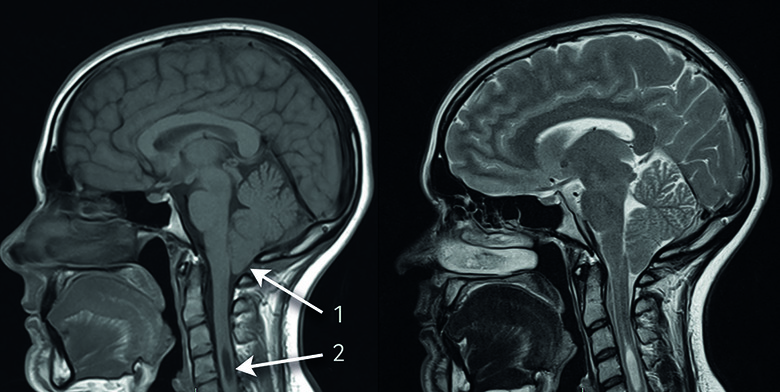
Rarely, serial imaging of a CM1 patient may show slow, progressive descent of the cerebellar tonsils. Such a case was captured by Pokharel and Siatkowski (Pokharel and Siatkowski 2004) in the Figures displayed below.
 |
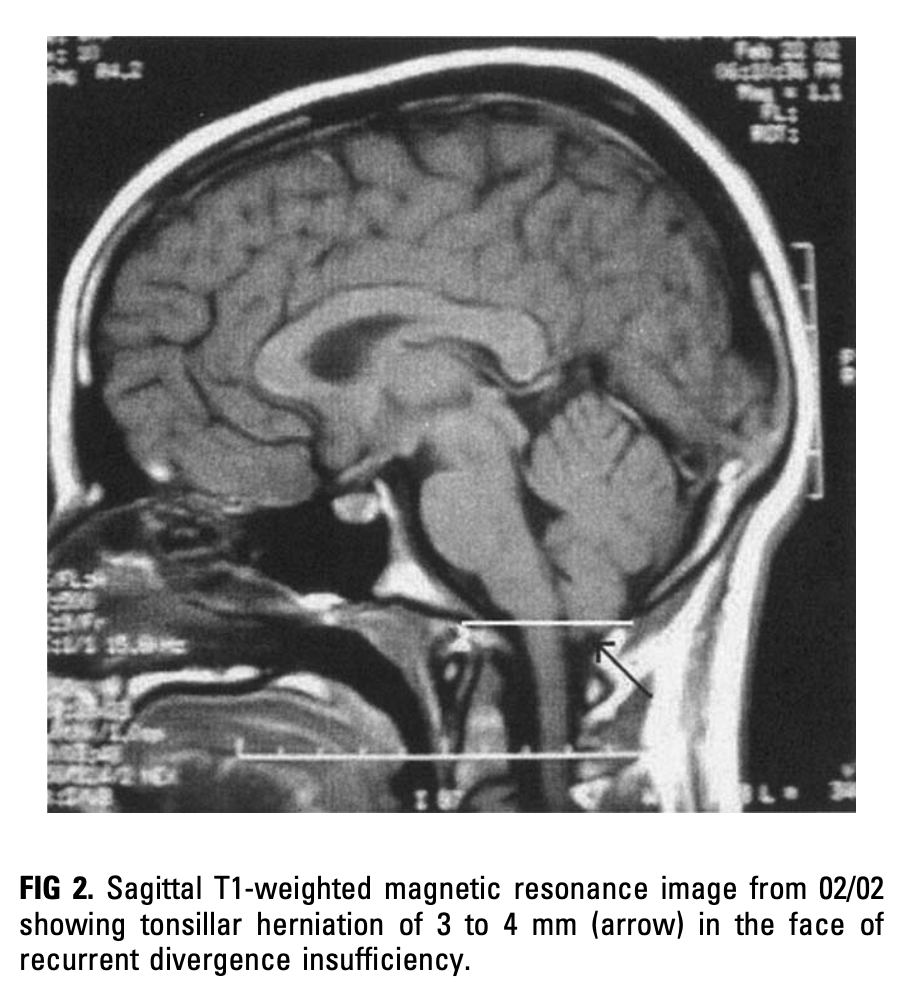 |
Differential diagnosis of CM1
The main differential diagnosis of CM1 includes other cerebellar disorders.
Because CM1 can produce intermittent symptoms of extraocular muscle abnormalities, bulbar dysfunction and proximal limb weakness, it can be mistaken for myasthenia gravis (Muthusamy, Matte et al. 2011).
Uncommonly CM1 can present with only esotropia (Passo, Shults et al. 1984, Lewis, Kline et al. 1996, Weeks and Hamed 1999, Biousse, Newman et al. 2000, Imes and Quinn 2001, Defoort-Dhellemmes, Denion et al. 2002, Pokharel and Siatkowski 2004, Hentschel, Yen et al. 2005, Kowal, Yahalom et al. 2006), usually comitant, and thus be mistaken for primary strabismus (Passo, Shults et al. 1984).
Treatment of CM1: pharmacologic
In some patients the oculomotor abnormalities in CM1 may be ameliorated pharmacologically. Options are reviewed by Shaikh and Ghasia (Shaikh and Ghasia 2015).
Several agents have been studied for the treatment of down beat nystagmus in CM1, including clonazepam (Currie and Matsuo 1986, Young and Huang 2001), baclofen (Dieterich, Straube et al. 1991), gabapentin (Averbuch-Heller, Tusa et al. 1997), 3,4‑diaminopyridine (Strupp, Schuler et al. 2003) and 4‑aminopyridine (Claassen, Spiegel et al. 2013). These studies were either small, or had unsatisfactory results.
For periodic alternating nystagmus, baclofen is the first line agent (Stahl, Plant et al. 2002, Straube, Leigh et al. 2004); memantine has also been studied (Kumar, Thomas et al. 2009).
Treatment of CM1: surgical
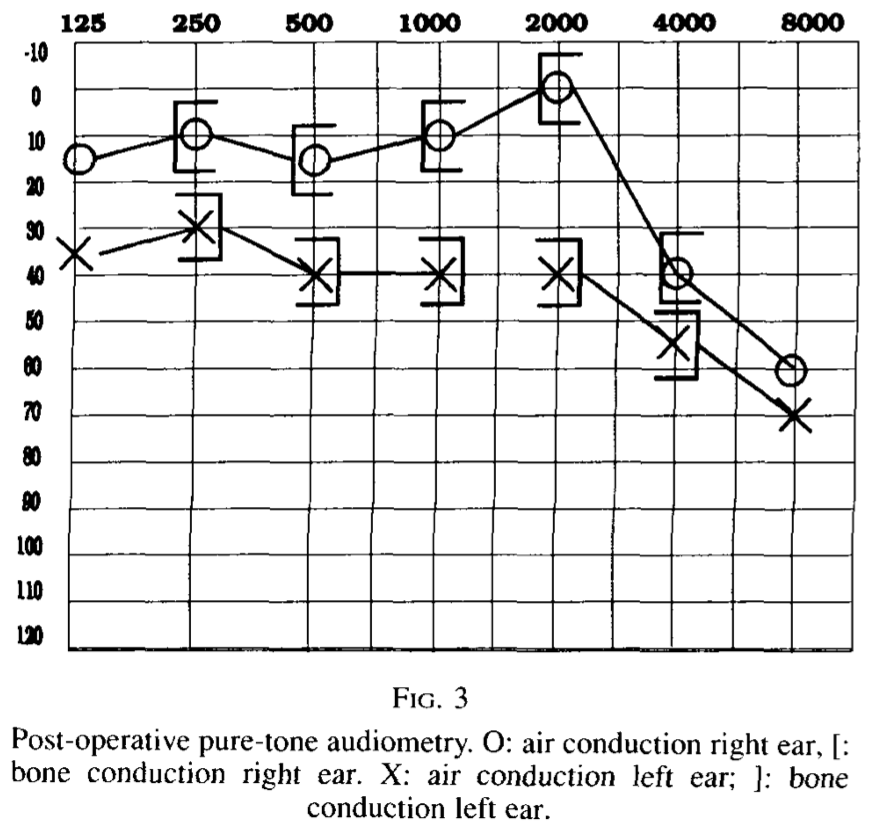
Albers and Ingels (Albers and Ingels 1993) reported some improvement in asymmetrical sensorineural hearing loss in CM1 after decompression surgery.
The Figure below, from Albers and Ingels (Albers and Ingels 1993), shows a pre-operative audiogram from a patient with CM1 who underwent posterior fossa decompression and cervical laminectomy, and a postoperative audiogram performed 1 year later. There was modest improvement in the left-sided hearing loss.
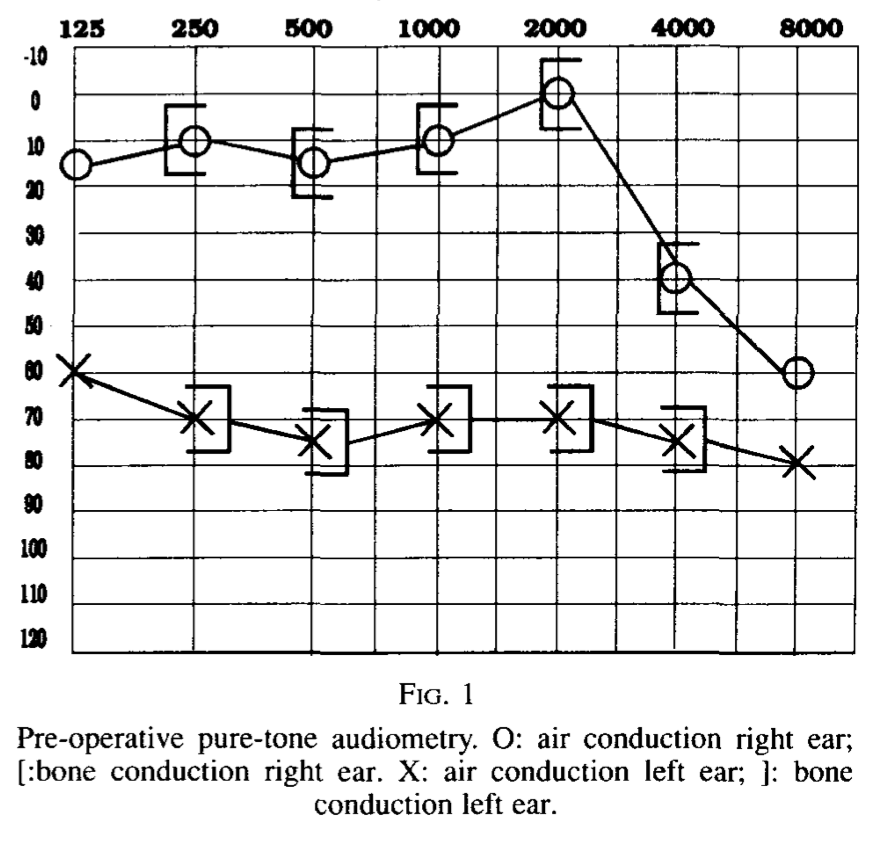 |
 |
Numerous reports describe improvement in nystagmus after decompression surgery for CM1 (Goodwin and Halvorson 2012), but often these observations are simply from case reports. For example:
- Pederson and colleagues (Pedersen, Troost et al. 1980) studied a CM1 patient with intermittent down beat nystagmus and oscillopsia and reported that after suboccipital decompression surgery, “There was a dramatic diminution of the visual complaints and downbeat nystagmus.”
- Gingold and Winfield (Gingold and Winfield 1991) reported on one CM1 patient whose oscillopsia completely resolved after suboccipital decompression and upper cervical laminectomy.
- Pieh and Gottlob (Pieh and Gottlob 2000) reported on one CM1 patient with “nystagmus of skew” that improved within 3 months of having undergone posterior fossa decompression and upper cervical laminectomy.
- Yee and colleagues (Yee, Baloh et al. 1984) reported on one CM1 patient with episodic vertical oscillopsia and spontaneous down beat nystagmus whose symptoms resolved following suboccipital decompression.
- Zimmerman and colleagues (Zimmerman, Roach et al. 1986) reported a case of CM1 with oscillopsia and see-saw nystagmus whose symptoms and nystagmus diminished (but did not resolve) after foramen magnum decompression and C1 laminectomy.
For trends in surgical outcomes we find case series more informative.
Liebenberg and colleagues (Liebenberg, Georges et al. 2005) studied twelve CM1 patients who underwent posterior fossa decompression. They reported that oculomotor and vestibulo-ocular abnormalities resolved in 8/12 (66%) of patients and improved in 1/12 (8%). Of the 8 patients whose abnormalities resolved, the mean time to complete resolution was 15.5 months (range 3 – 71 months).
Some reports of surgical intervention for CM1 are more guarded. For example, Dones colleagues (Dones, De Jesus et al. 2003) retrospectively reviewed records of 27 CM1 patients who had undergone surgery. They reported that the distribution depended on the absence/presence of a syrinx.
|
Pre-operative |
Post-operative |
Change |
|
|
Nystagmus, total |
8/27 (30%) |
2/27 (7%) |
From 8 to 2 = 6/8 (75%) |
|
Nystagmus, with syrinx |
3/16 (19%) |
0/16 (0%) |
From 3 to 0 = 0/3 (100%) |
|
Nystagmus, without syrinx |
5/11 (45%) |
2/11 (18%) |
From 5 to 2 = 3/5 (60%) |
|
Vertigo, total |
6/27 (22%) |
3/27 (11%) |
From 6 to 3 = 3/6 (50%) |
|
Vertigo, with syrinx |
0/16 (0%) |
0/16 (0%) |
— |
|
Vertigo, without syrinx |
6/11 (55%) |
3/11 (27%) |
From 6 to 3 = 3/6 (50%) |
Table : Pre-operative and post-operative nystagmus and vertigo in Chiari 1 malformation without and with syrinx. From Dones et al. (2003).
Goldschagg and colleagues (Goldschagg, Feil et al. 2017) studied ten CM1 patients immediately before surgery, immediately after surgery, and 3 months postoperatively. The surgery was suboccipital decompression with unilateral cerebellar tonsillectomy. The Table below summarizes their outcomes.
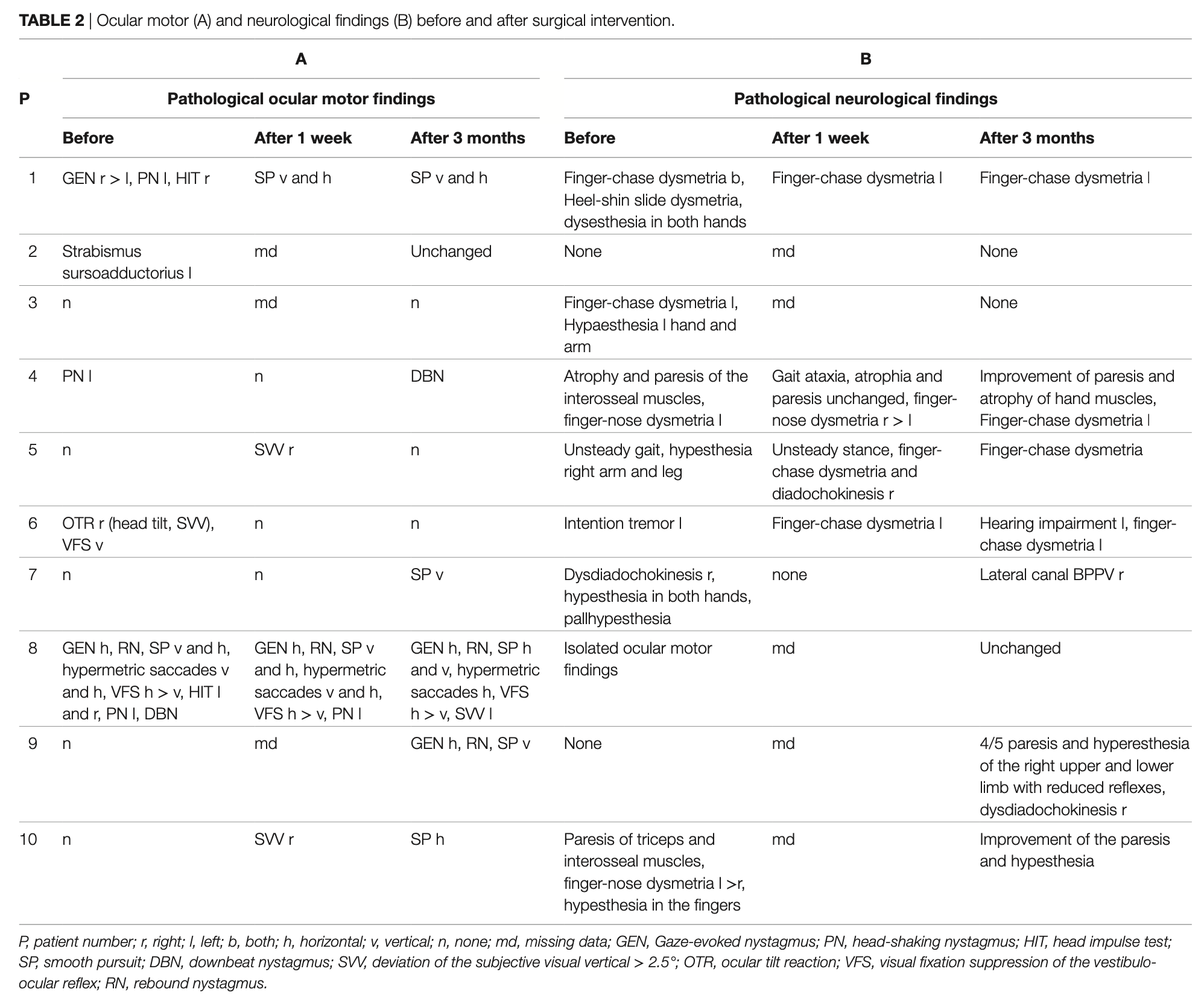
Our interpretation of these results is as follows:
- Improvement:
- Patient 6 improved: Pre-operatively there was an abnormal ocular tilt reaction, abnormal subjective visual vertical, and abnormal visual fixation suppression. At 3 months postoperatively these had all improved.
- No change:
- Patients 2, 3 and 5 were unchanged at 3 months postoperatively.
- Change (some abnormalities improved, but new ones appeared):
- Patient 1 changed: Pre-operatively had gaze evoked nystagmus, head shaking nystagmus and abnormal video head impulse testing on the right. At 3 months postoperatively there was deficient horizontal and vertical smooth pursuit.
- Patient 4 changed: Pre-operatively had head shaking nystagmus. At 3 months postoperatively there was spontaneous down beat nystagmus.
- Patient 8 changed: Pre-operatively there was bilaterally abnormal video head impulse testing, head shaking nystagmus and spontaneous down beat nystagmus. At 3 months post-operatively those features had improved, but subjective visual vertical was abnormal.
- Worsening:
- Patient 7 worsened: Pre-operatively was normal. At 3 months post-operatively there was abnormal smooth pursuit.
- Patient 9 worsened: Pre-operatively was normal. At 3 months post-operatively there was gaze evoked nystagmus, rebound nystagmus and abnormal smooth pursuit.
- Patient 10 worsened: Pre-operatively was normal. At 3 months post-operatively there was abnormal smooth pursuit.
In short, for this series of CM1 patients studied by Goldschagg and colleagues, 1/10 (10%) of patients improved, 3/10 (30%) had no change, 3/10 (30%) developed new abnormalities, and 3/10 (30%) worsened. However, note that these patients underwent surgery that involved not only suboccipital decompression, but also unilateral cerebellar tonsillectomy.
In terms of instrumented vestibular testing, Goldschagg and colleagues reported that 2/10 (20%) had abnormal asymmetry on cervical vestibular evoked myogenic potentials, and that these both improved postoperatively. They reported that 3/9 (33%) of patients had a pathological caloric asymmetry, and that this improved in 1/3 (33%) postoperatively.
Giammattei and colleagues (Giammattei, Messerer et al. 2020) performed a retrospective review of 38 adult CM1 patients followed for ≥5 years. They reported that gait impairment improved in 83% of patients.
Type 1.5 Chiari malformation (CM1.5)
Cesmebasi and colleagues (Cesmebasi, Loukas et al. 2015) state that this category is intended to “address patients with Chiari type I malformation with an added component of brainstem and fourth ventricle elongation/caudal descent. Chiari type 1.5 patients have symptoms similar to Chiari type I patients and may have a higher incidence of developing syringomyelia.”
The Figure below, from Tubbs and colleagues (Tubbs, Iskandar et al. 2004), shows two examples of what they classify as CM1.5.
Type 2 Chiari malformation (CM2)
Chiari’s original description was:
“A second type is formed by the elongation of parts of the cerebellum into the dilated vertebral canal within the elongated 4th ventricle, extending into the vertebral canal.”
Cesmebasi and colleagues (Cesmebasi, Loukas et al. 2015) list the following anatomical features of CM2:
Brain: Multiple anomalies at all levels (telencephalon, diencephalon, mesencephalon, metencephalon, myelencephalon).
-
- Telencephalon: There may be partial or complete agenesis of the corpus callosum, and partial or complete absence of the septum pellucidum. When the corpus callosum is absent there may be interdigitation of the cerebral hemispheres. The cerebral cortex may have sulcal deformities. The olfactory bulb may be partially or completely absent. The cingulate gyrus may be absent. The internal aspect of the lateral ventricles may have a granular appearance due to heterotopic gray matter bulging into them.
- Diencephalon: 75 – 90% of patients exhibit an enlarged massa intermedia that is often anteriorly displaced. The hypothalamus may be elevated. The habenular commissure and the pineal gland may be elongated.
- Mesencephalon: The midbrain is usually elongated. The quadrigeminal plate is usually shortened. Tectal beaking may be present. The cerebral aqueduct may be deformed (stretched, laterally compressed, forked, stenotic, posteriorly kinked). Cranial nerve nuclei may be hypoplastic.
- Metencephalon: The cerebellum is smaller and may upwardly herniate over the tentorium cerebelli. The cerebellum is often laterally displaced. The vermis is herniated through the foramen magnum. Cerebellar folia may be absent or dysplastic. The lateral cerebellar edges may extent anteriorly towards the brainstem and basilar artery (“cerebellar inversion”). The pons is flattened and elongated. The ventral pontine surface may be indented by a displayed vertebral artery or by the anterior rim of the foramen magnum. There may be dysplasia of the basal pontine nuclei, tegmental nuclei and cranial nerve nuclei.
- Myelencephalon: The medulla may be flattened and elongated, giving it a “trumpet-like” appearance. About 70% of patients have a protrusion caudal to the gracile and cuneate tubercles at C2 – C4 levels, sometimes termed a cervicomedullary kink, buckle, hump or spur.
- Other: The malformations tend to affect cranial and upper cervical nerves that take a vertical course. The vertebrobasilar system and its branches may be caudally displaced. The great vein of Galen may be elongated.
- Ventricles/cisterns: About 90% of patients have hydrocephalus. The lateral ventricles are often asymmetric. The frontal horns are more pointed.
Cerebellar tonsils: Herniation of the cerebellar vermis with caudal descent of the brainstem and 4th ventricle.
Skull: Craniolacunia/lückenschädel (“beaten copper” appearance of the bone). There may be scalloping of the anterior portion of the frontal bone, posteriomoedial aspect of the petrous part of the temporal bones and jugular tubercles. The scalloping of the petrous part of the petrous part of the temporal bone may shorten the internal auditory canal. The foramen magnum is usually enlarged. The posterior cranial fossa is smaller and flattened. The opisthion (mid-point on the posterior margin of the foramen magnum) may be notched; cranioschisis (congenital failure of the skull to completely close) is sometimes seen.
Meninges: Tentorium cerebelli is usually heart- or V‑shaped, widened, hypoplastic and low-lying.
Spinal canal: There may be scalloping of the odontoid process, enlargement of the cervical spinal canal, incomplete formation of the posterior arch of the atlas, and Klippel-Feil anomaly.
Spinal cord: Most patients have a myelomeningocele. About 6% of these patients also have an associated split cord malformation (diastematomyelia).
Oculomotor findings in CM2
Arnold and colleagues (Arnold, Baloh et al. 1990) studied 3 cases of CM2. All 3 patients had bilateral internuclear ophthalmoplegia, deficits in smooth pursuit, deficits in optokinetic responses and vestibulo-ocular responses. Two had abduction paresis and deficits in smooth pursuit.
Longridge and Mallinson (Longridge and Mallinson 1985) studied a 29-year-old CM2 patient. On face-to-face oculomotor examination there was subtle spontaneous right beat nystagmus; there was also right greater than left-sided gaze evoked nystagmus; up beat nystagmus on up gaze and down beat nystagmus on down gaze (in other words, both horizontal and vertical gaze evoked nystagmus), saccadic hypermetria and saccadic smooth pursuit. On dynamic visual acuity testing she went from 20/20 to worse than 20/100.
Tubbs and colleagues (Tubbs, Soleau et al. 2004) studied children with CM2 and concluded that “there is a correlation between the presence and severity of nystagmus and the severity of tectal beaking.”
Instrumented oculomotor testing in CM2: videonystagmography (VNG)
Salman and colleagues (Salman, Sharpe et al. 2005) studied saccades using videonystagmography (VNG) in 21 patients (age 8 – 19 years) with CM2 compared to 39 healthy controls. Surprisingly, they reported no differences in saccades between the CM2 and the control groups. In a subsequent publication (Salman, Sharpe et al. 2006) these investigators concluded that this normality was due to saccadic adaptation.
Salman and colleagues (Salman, Sharpe et al. 2007) studied smooth pursuit using videonystagmography in the same group of 21 patients compared to 38 healthy controls and reported that in the 8 patients with nystagmus, smooth pursuit gain was abnormal.
Instrumented oculomotor testing in CM2: electronystagmography (ENG)
Longridge and Mallinson (Longridge and Mallinson 1985) studied a 29-year-old CM2 patient with electronystagmography (ENG). They reported bilateral horizontal gaze evoked nystagmus, subtle rebound nystagmus, saccadic hypermetria, saccadic smooth pursuit, abnormal optokinetic responses and abnormal visual fixation suppression.
Imaging in CM2
The Figure below, from Piper and colleagues (Piper, Pike et al. 2019), shows CM2.
Type 3 Chiari malformation (CM3)
Chiari’s original description, as translated by Ivashchuk and colleagues (Ivashchuk, Loukas et al. 2015), was:
“Of the third type of serial changes of the cerebellum caused by chronic congenital hydrocephalus, there was only one case available to me. It demonstrated the greatest degree of displacement of the cerebellum, out of the cranial cavity through the foramen magnum into the vertebral canal, involving the displacement of nearly the entire cerebellum, which was itself hydrocephalic, into a cervical spina bifida.”
Ivashchuk and colleagues (Ivashchuk, Loukas et al. 2015) extracted the salient features of the Chiari malformation type 3 in the following Table.

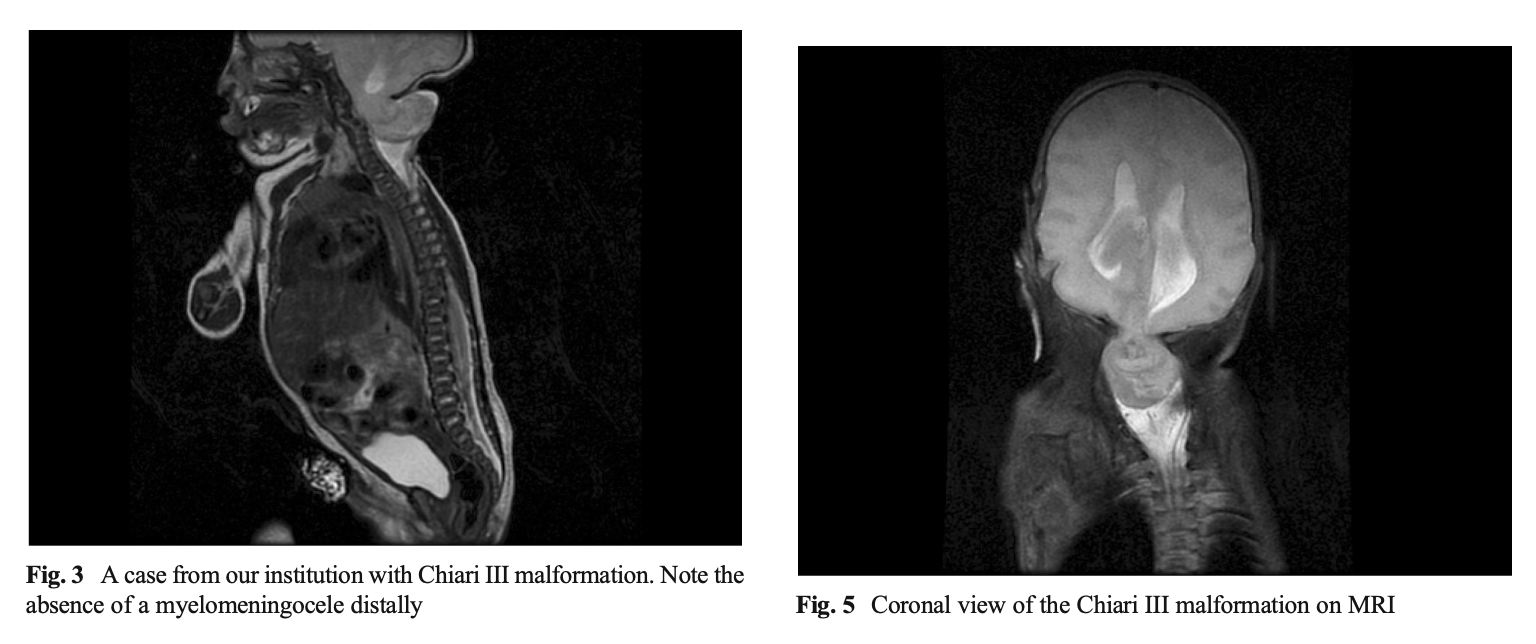
Cesmebasi and colleagues (Cesmebasi, Loukas et al. 2015) state that, “Chiari III malformation are defined as findings of a Chiari II malformation with the addition of a posterior cranial fossa encephalocele that contains cerebellar and brainstem tissue.” They list the following anatomical abnormalities associated with CM3:
Brain: In all cases the cerebellum is herniated into a low occipital or high cervical encephalocele. Some patients also have herniation of the brainstem, occipital lobes, dural venous sinuses, and subarachnoid or ventricular cerebrospinal fluid spaces. Additionally, partial or complete dysplasia of the corpus callosum is seen.
Skull: Scalloping of the dorsal clivus and petrous ridge. Occasionally the parietal bones are hypoplastic. There is usually an enlarged foramen magnum.
Spine: Generally CM3 patients have osseous deformities of the upper cervical spine. Some have posterior cervical agenesis.
Oculomotor findings in CM3
Gorgoglione and colleagues (Gorgoglione, Laera et al. 2023) reported the case of an infant with CM3 who exhibited “nystagmus,” which they did not describe in any further detail.
Imaging in CM3
The Figure below, from Ivashchuk and colleagues (Ivashchuk, Loukas et al. 2015), shows sagittal and coronal views of a patient with CM3.
Type 3.5 Chiari malformation (CM3.5)
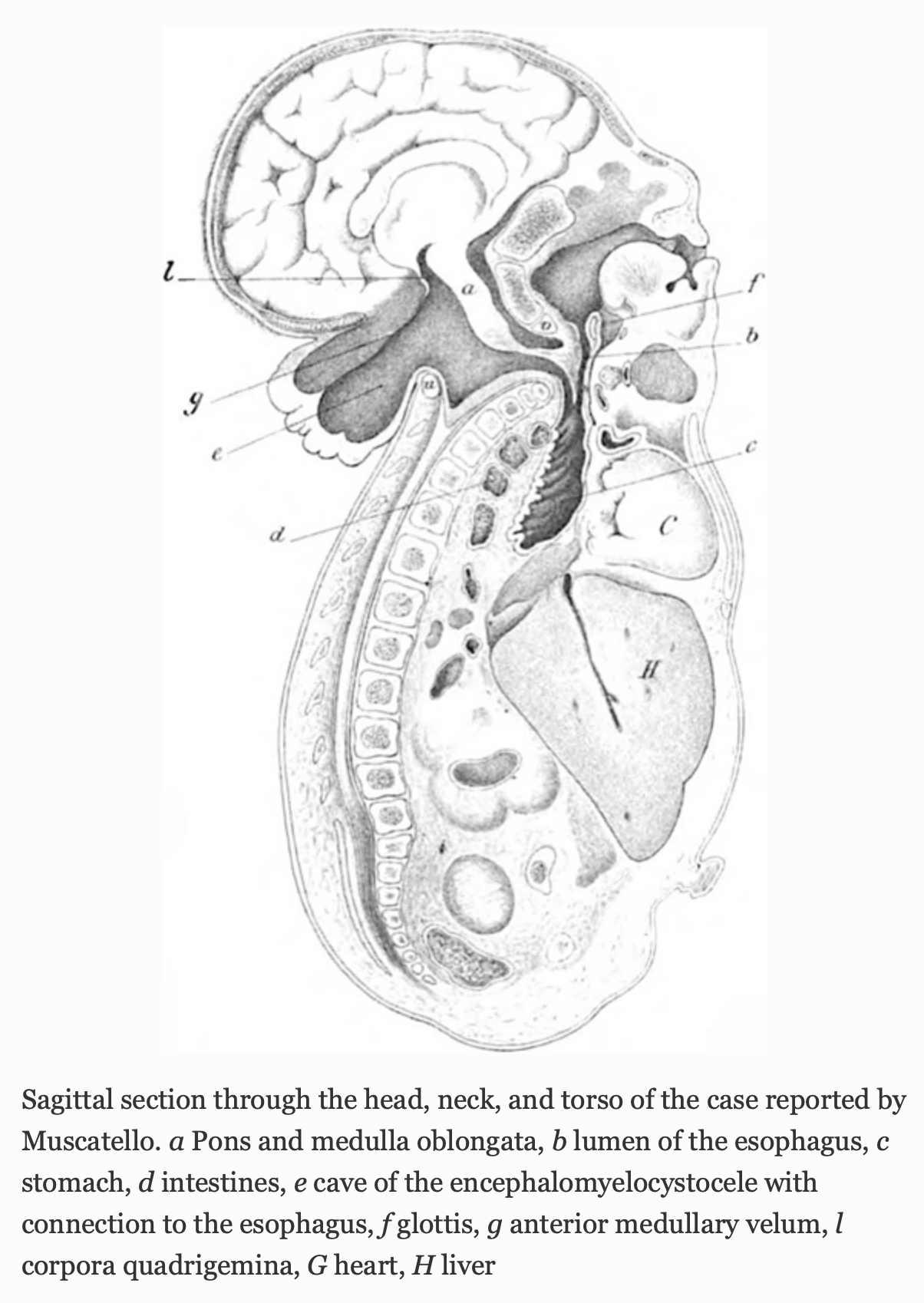
In 1894 Dr. Giuseppe Muscatello (Muscatello 1894) published a case that he described as a variant of CM3. Fisahn and colleagues (Fisahn, Shoja et al. 2016) viewed this case as unique (“the only reported case”) and proposed calling it “Chiari 3.5.”
The Figure below, from Fisahn and colleagues (Fisahn, Shoja et al. 2016), is Muscatello’s (Muscatello 1894) original illustration.
Type 4 Chiari malformation (CM4)
Chiari’s original description (Chiari 1895), as translated by Tubbs and colleagues (Tubbs, Demerdash et al. 2016), was:
Medical history: The encephalocele consists of a cylindrical, broad based, 6 cm long, and 3 cm wide skin sac. The sac has an ulcerated opening measuring about 8 cm. Through this opening, another, 4 cm wide inner sac, similarly ulcerated at the end, protrudes. That second, inner sac is made up of meninges with longitudinally oriented folds, almost of the size of a little finger. The inner sac communicates with the intracranial compartment through a 1 cm2 wide opening located just below the posterior fontanel. The outer sac is contingent with the scalp. In surgery the inner sac was disconnected and resected at the intracranial opening and closed with the excess skin overlying it. The patient passed 36 h after surgery.
Pathological-anatomical diagnosis: Meningitis cerebralis suppurativa et suppuratio in ventriculis cerebri ex ulceratione hydrencephaloceles occipitalis (horas XXXVI ante mortem amputatae). Dilatatio congenita ventriculorium cerebri. Pneumonia lobularis bilateralis. Sclerema textus adipose subcutanei.
Dissection of the central nervous system: The child’s head is disproportionally large compared to its height (47 cm), just a little more narrow. Hair shaved. Inferior to the posterior fontanel there is a skin bulge measuring 6 cm × 3 cm × 2 cm with a 6 cm long closed incision at the top. The edges are edematous and erythematous.
The soft skull is ecchymotic, pale, in the area of the bulge edematous, serous, and purulent. Circumferentially the skull measures 27 cm. The sutures are split. The sagittal suture is 0.5 cm wide. The anterior fontanel is larger measuring 4 cm in length and 2.5 cm in width. The posterior fontanel is normal in size. The skull bones are fairly rigid. In the squama ossis occiptialis, 2 cm inferior to the posterior fontanel, there is an opening of the size of the nail of a small finger. The opening is surrounded by the split of the processus sagittalis major of the pachymenix. Through that opening a hemorrhagic and necrotic mass of inner meninges and brain parenchyma are extruding. In the sinus durae matris there is dark, liquid, and also freshly coagulated blood. The sinus sagittalis major is split into two sinuses from the area of the posterior fontanel that diverge to form the sinus transversi. The tentorium cerebelli is narrow, the processus falciformis minor is absent, the posterior fossa is small. The inner surface of the skull base has deep impressiones digitatae. The foramen occipital magnum is remarkably narrow. The inner meninges are pale and infiltrated by serous and purulent liquid.
The gyri and sulci of the cerebrum appear normal. The lateral ventricles and third ventricle are enlarged and contain serous and purulent liquid. The brain matter is pale, moist, and soft. Originating from the precuneus are two processes of brain matter that contain the occipital horns and plexus choroidei of the lateral ventricles and protrude through the previously mentioned opening.
The cerebellum is only half the size of a normal cerebellum. It is 3.5 cm wide and 2 cm long. The largest diameter from the upper to the lower surface is 3 cm. It is asymmetric with the left hemisphere significantly smaller than the right. The lobi superiores, posteriores et inferiores are easily distinguished, the tonsils and flocculi are rudimentary. The inferior vermis is more prominent and the incisura marginalis posterior is absent. The pons and medulla oblongata are 14 and 11 mm long, respectively. Both are thin.
A median sagittal section of the cerebellum, pons, medulla oblongata, and upper cervical spinal cord shows unusual rolling of the lower portion of the arbor vitae resulting in a deep location of the pyramis (a), high location of the uvula (b), and location of the nodulus (c) of the inferior vermis on the upper end of the rolling of the arbor vitae. The dorsal wall of the fourth ventricle (d) is bowed due to the rolling and the velum medullare posterius (e) is very long. The entry of the tela choroidea ventriculi IV is well demarcated (f) and at the usual location. The ponticuli (g) are longer and thicker, as is the velum medullare posterius. The corpora dentata cerebelli are smaller, otherwise normally shaped. The pons and medulla are normal.
The spinal cord is of dense consistency. The inner meninges of medulla oblongata, pons, and cerebellum are not purulent.
Microscopic examination of the encephalocele occipitalis, vermis, pons, medulla oblongata, corpus dentatum of the right cerebellar hemisphere, the fifth cervical segment, fifth and ninth thoracic segments, and third segment of the spinal cord is performed after solidification with liquor mülleri.
The inner sac of the encephalocele has two layers. The outer layer is highly vascularized and hemorrhagic as are the inner meninges while the inner layer is of sclerotic brain matter. The latter makes up the previously mentioned longitudinally oriented folds. The inner surface of the sac is lined with cylindrical epithelium.
The vermis, pons, and medulla oblongata are examined using the right half of the section. The vermis has normal texture. Its cortex has an external granular layer, a molecular layer, and an inner granular layer. There are numerous Purkinje cells. The medulla is mostly black on copper hematoxylin stain. The pons and medulla oblongata are normal. Only the pyramids are devoid of medullary substance. The fourth ventricle is lined with normal epithelium. The elongated velum medullare posterius and taenia plexus chorioidei ventriculi IV are sclerotic.
The corpus dentatum cerbelli has prominent folding and numerous multipolar ganglion cells. The medulla spinalis has a mild dilatation of the central canal lined with epithelium and the pyramidal tract is devoid of medullary substance.
Tubbs and colleagues (Tubbs, Demerdash et al. 2016) translated sections of this paper and extracted the salient features listed in the Table below.

The Figure below, from Singh and colleagues (Singh, Arora et al. 2018), shows a CM4.

Type 5 Chiari malformation (CM5)
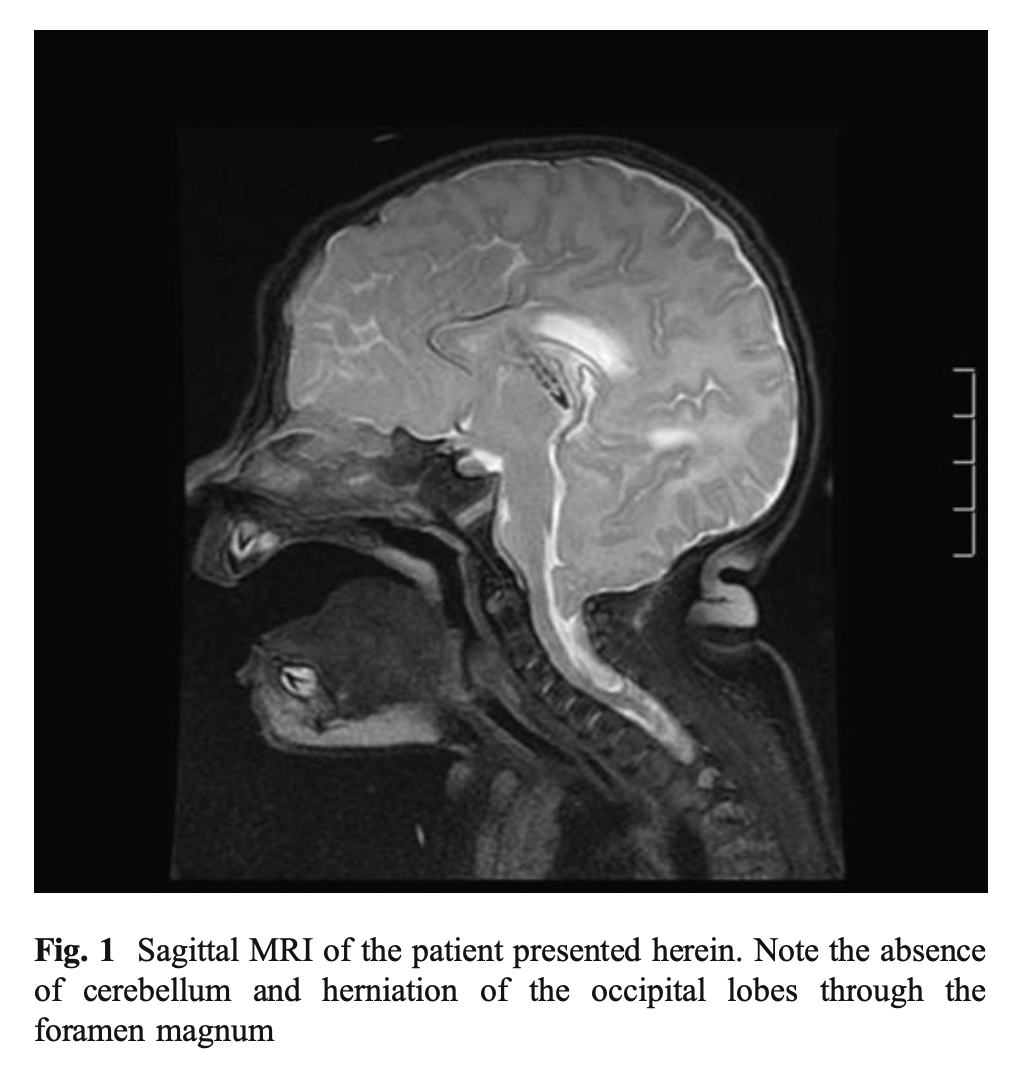
Tubbs and colleagues (Tubbs, Muhleman et al. 2012) reported the case of an infant with cerebellar agenesis and herniation of the occipital lobe through the foramen magnum. They proposed designating this “Chiari 5 malformation.”
The Figure below, from Tubbs and colleagues (Tubbs, Muhleman et al. 2012), shows herniation of the occipital lobes through the foramen magnum, and absence of the cerebellum.
References
Al-Awami A, Flanders ME, Andermann F, Polomeno RC (2005) Resolution of periodic alternating nystagmus after decompression for Chiari malformation. Can J Ophthalmol 40: 778-80. doi: 10.1016/S0008-4182(05)80101-2
Albers FW, Ingels KJ (1993) Otoneurological manifestations in Chiari-I malformation. J Laryngol Otol 107: 441-3. doi: 10.1017/s0022215100123370
Arnold AC, Baloh RW, Yee RD, Hepler RS (1990) Internuclear ophthalmoplegia in the Chiari type II malformation. Neurology 40: 1850-4. doi: 10.1212/wnl.40.12.1850
Arnold J (1894) Myelocyste, Transposition von Gewebskeimen und Sympodie. Beitr Path Anat 16: 1-28.
Averbuch-Heller L, Tusa RJ, Fuhry L, Rottach KG, Ganser GL, Heide W, Buttner U, Leigh RJ (1997) A double-blind controlled study of gabapentin and baclofen as treatment for acquired nystagmus. Ann Neurol 41: 818-25. doi: 10.1002/ana.410410620
Baloh RW, Beykirch K, Tauchi P, Yee RD, Honrubia V (1988) Ultralow vestibuloocular reflex time constants. Ann Neurol 23: 32-7. doi: 10.1002/ana.410230107
Barkovich AJ, Wippold FJ, Sherman JL, Citrin CM (1986) Significance of cerebellar tonsillar position on MR. AJNR Am J Neuroradiol 7: 795-9.
Biousse V, Newman NJ, Petermann SH, Lambert SR (2000) Isolated comitant esotropia and Chiari I malformation. Am J Ophthalmol 130: 216-20. doi: 10.1016/s0002-9394(00)00457-8
Bogdanov EI, Faizutdinova AT, Heiss JD (2022) The Small Posterior Cranial Fossa Syndrome and Chiari Malformation Type 0. J Clin Med 11. doi: 10.3390/jcm11185472
Carmel PW, Markesbery WR (1972) Early descriptions of the Arnold-Chiari malformation. The contribution of John Cleland. J Neurosurg 37: 543-7. doi: 10.3171/jns.1972.37.5.0543
Cesmebasi A, Loukas M, Hogan E, Kralovic S, Tubbs RS, Cohen-Gadol AA (2015) The Chiari malformations: a review with emphasis on anatomical traits. Clin Anat 28: 184-94. doi: 10.1002/ca.22442
Chait GE, Barber HO (1979) Arnold-Chiari malformation–some otoneurological features. J Otolaryngol 8: 65-70.
Chiari H (1891) Ueber Veränderungen des Kleinhirns infolge von Hydrocephalie des Grosshirns [Regarding changes of the cerebellum from cerebral hydrocephalus]. Deutsche medicinische Wochenschrift 17: 1172-1175. doi: 10.1055/s-0029-1206803
Chiari H (1895) Über Veränderungen des Kleinhirns, des Pons und der Medulla oblongata in Folge von congenitaler Hydrcephalie des Grosshirns. Denkschriften der Akademie der Wissenschaften in Wien 63: 71-116.
Claassen J, Spiegel R, Kalla R, Faldon M, Kennard C, Danchaivijitr C, Bardins S, Rettinger N, Schneider E, Brandt T, Jahn K, Teufel J, Strupp M, Bronstein A (2013) A randomised double-blind, cross-over trial of 4-aminopyridine for downbeat nystagmus–effects on slowphase eye velocity, postural stability, locomotion and symptoms. J Neurol Neurosurg Psychiatry 84: 1392-9. doi: 10.1136/jnnp-2012-304736
Cleland J (1883) Contribution to the Study of Spina Bifida, Encephalocele, and Anencephalus. J Anat Physiol 17: 257-92.
Currie JN, Matsuo V (1986) The use of clonazepam in the treatment of nystagmus-induced oscillopsia. Ophthalmology 93: 924-32. doi: 10.1016/s0161-6420(86)33640-6
Defoort-Dhellemmes S, Denion E, Arndt CF, Bouvet-Drumare I, Hache JC, Dhellemmes P (2002) Resolution of acute acquired comitant esotropia after suboccipital decompression for Chiari I malformation. Am J Ophthalmol 133: 723-5. doi: 10.1016/s0002-9394(02)01354-5
Dieterich M, Straube A, Brandt T, Paulus W, Buttner U (1991) The effects of baclofen and cholinergic drugs on upbeat and downbeat nystagmus. J Neurol Neurosurg Psychiatry 54: 627-32. doi: 10.1136/jnnp.54.7.627
Dones J, De Jesus O, Colen CB, Toledo MM, Delgado M (2003) Clinical outcomes in patients with Chiari I malformation: a review of 27 cases. Surg Neurol 60: 142-7; discussion 147-8. doi: 10.1016/s0090-3019(03)00131-9
Faria MA, Jr., Spector RH, Tindall GT (1980) Downbeat nystagmus as the salient manifestation of the Arnold-Chiari malformation. Surg Neurol 13: 333-6.
Fisahn C, Shoja MM, Turgut M, Oskouian RJ, Oakes WJ, Tubbs RS (2016) The Chiari 3.5 malformation: a review of the only reported case. Childs Nerv Syst 32: 2317-2319. doi: 10.1007/s00381-016-3255-3
Ghasia FF, Gulati D, Westbrook EL, Shaikh AG (2014) Viewing condition dependence of the gaze-evoked nystagmus in Arnold Chiari type 1 malformation. J Neurol Sci 339: 134-9. doi: 10.1016/j.jns.2014.01.045
Giammattei L, Messerer M, Daniel RT, Aghakhani N, Parker F (2020) Long-term outcome of surgical treatment of Chiari malformation without syringomyelia. J Neurosurg Sci 64: 364-368. doi: 10.23736/S0390-5616.17.04063-2
Gingold SI, Winfield JA (1991) Oscillopsia and primary cerebellar ectopia: case report and review of the literature. Neurosurgery 29: 932-6.
Goldschagg N, Feil K, Ihl F, Krafczyk S, Kunz M, Tonn JC, Strupp M, Peraud A (2017) Decompression in Chiari Malformation: Clinical, Ocular Motor, Cerebellar, and Vestibular Outcome. Front Neurol 8: 292. doi: 10.3389/fneur.2017.00292
Goodwin D, Halvorson AR (2012) Chiari I malformation presenting as downbeat nystagmus: clinical presentation, diagnosis, and management. Optometry 83: 80-6.
Gorgoglione ML, Laera R, Curcio A, Caruso G, Delia G, Germano AF (2023) Treatment of Chiari III Malformation in Infant with 4K 3D ORBEYE Exoscope. World Neurosurg 171: 144. doi: 10.1016/j.wneu.2023.01.020
Hatashita S, Shimoji T, Shimizu A, Ishizawa A, Tanaka M (1980) [A case of up-beat nystagmus associated with Chiari malformation type I (author’s transl)]. No Shinkei Geka 8: 989-94.
Hentschel SJ, Yen KG, Lang FF (2005) Chiari I malformation and acute acquired comitant esotropia: case report and review of the literature. J Neurosurg 102: 407-12. doi: 10.3171/ped.2005.102.4.0407
Hida K, Iwasaki Y, Koyanagi I, Abe H (1999) Pediatric syringomyelia with chiari malformation: its clinical characteristics and surgical outcomes. Surg Neurol 51: 383-90; discussion 390-1. doi: 10.1016/s0090-3019(98)00088-3
Horn SR, Shepard N, Vasquez-Montes D, Bortz CA, Segreto FA, De La Garza Ramos R, Goodwin CR, Passias PG (2018) Chiari malformation clusters describe differing presence of concurrent anomalies based on Chiari type. J Clin Neurosci 58: 165-171. doi: 10.1016/j.jocn.2018.06.045
Hosford DA, Spector RH (1985) Arnold-Chiari malformation in a geriatric patient. South Med J 78: 618-21. doi: 10.1097/00007611-198505000-00028
Imes RK, Quinn TA (2001) Acute comitant esotropia in Chiari 1 malformation. Ophthalmology 108: 834. doi: 10.1016/s0161-6420(00)00439-5
Ivashchuk G, Loukas M, Blount JP, Tubbs RS, Oakes WJ (2015) Chiari III malformation: a comprehensive review of this enigmatic anomaly. Childs Nerv Syst 31: 2035-40. doi: 10.1007/s00381-015-2853-9
Kim SH, Kim HJ, Kim JS (2019) Perverted Downward Corrective Saccades During Horizontal Head Impulses in Chiari Malformation. Cerebellum 18: 333-339. doi: 10.1007/s12311-018-1000-z
Korres S, Balatsouras DG, Zournas C, Economou C, Gatsonis SD, Adamopoulos G (2001) Periodic alternating nystagmus associated with Arnold-Chiari malformation. J Laryngol Otol 115: 1001-4. doi: 10.1258/0022215011909602
Kowal L, Yahalom C, Shuey NH (2006) Chiari 1 malformation presenting as strabismus. Binocul Vis Strabismus Q 21: 18-26.
Kumar A, Patni AH, Charbel F (2002) The Chiari I malformation and the neurotologist. Otol Neurotol 23: 727-35. doi: 10.1097/00129492-200209000-00021
Kumar A, Thomas S, McLean R, Proudlock FA, Roberts E, Boggild M, Gottlob I (2009) Treatment of acquired periodic alternating nystagmus with memantine: a case report. Clin Neuropharmacol 32: 109-10. doi: 10.1097/WNF.0b013e3181873697
Lewis AR, Kline LB, Sharpe JA (1996) Acquired esotropia due to Arnold-Chiari I malformation. J Neuroophthalmol 16: 49-54.
Liebenberg WA, Georges H, Demetriades AK, Hardwidge C (2005) Does posterior fossa decompression improve oculomotor and vestibulo-ocular manifestations in Chiari 1 malformation? Acta Neurochir (Wien) 147: 1239-40; discussion 1240. doi: 10.1007/s00701-005-0612-5
Longridge NS, Mallinson AI (1985) Arnold-Chiari malformation and the otolaryngologist: place of magnetic resonance imaging and electronystagmography. Laryngoscope 95: 335-9.
McRae DL (1953) Bony abnormalities in the region of the foramen magnum: correlation of the anatomic and neurologic findings. Acta radiol 40: 335-54. doi: 10.3109/00016925309176595
Milhorat TH, Chou MW, Trinidad EM, Kula RW, Mandell M, Wolpert C, Speer MC (1999) Chiari I malformation redefined: clinical and radiographic findings for 364 symptomatic patients. Neurosurgery 44: 1005-17. doi: 10.1097/00006123-199905000-00042
Morgenstern PF, Tosi U, Uribe-Cardenas R, Greenfield JP (2020) Ventrolateral Tonsillar Position Defines Novel Chiari 0.5 Classification. World Neurosurg 136: 444-453. doi: 10.1016/j.wneu.2020.01.147
Mossman SS, Bronstein AM, Gresty MA, Kendall B, Rudge P (1990) Convergence nystagmus associated with Arnold-Chiari malformation. Arch Neurol 47: 357-9. doi: 10.1001/archneur.1990.00530030139030
Muscatello G (1894) Ueber die angeborenen Spalten des Schädels und der Wirbelsäule [About the hereditary clefts of the skull and the spine]. Archiv für klinische Chirurgie 47: 162-301.
Muthusamy P, Matte G, Kosmorsky G, Chemali KR (2011) Chiari type I malformation: a mimicker of myasthenia gravis. Neurologist 17: 86-8. doi: 10.1097/NRL.0b013e3181f83870
Passo M, Shults WT, Talbot T, Palmer EA (1984) Acquired esotropia. A manifestation of Chiari I malformation. J Clin Neuroophthalmol 4: 151-4. doi: 10.3109/01658108409034894
Pedersen RA, Troost BT, Abel LA, Zorub D (1980) Intermittent downbeat nystagmus and oscillopsia reversed by suboccipital craniectomy. Neurology 30: 1239-42. doi: 10.1212/wnl.30.11.1239
Pieh C, Gottlob I (2000) Arnold-Chiari malformation and nystagmus of skew. J Neurol Neurosurg Psychiatry 69: 124-6. doi: 10.1136/jnnp.69.1.124
Piper RJ, Pike M, Harrington R, Magdum SA (2019) Chiari malformations: principles of diagnosis and management. BMJ 365: l1159. doi: 10.1136/bmj.l1159
Pokharel D, Siatkowski RM (2004) Progressive cerebellar tonsillar herniation with recurrent divergence insufficiency esotropia. J AAPOS 8: 286-7. doi: 10.1016/j.jaapos.2004.01.014
Russell GE, Wick B, Tang RA (1992) Arnold-Chiari malformation. Optom Vis Sci 69: 242-7.
Salman MS, Sharpe JA, Eizenman M, Lillakas L, To T, Westall C, Steinbach MJ, Dennis M (2005) Saccades in children with spina bifida and Chiari type II malformation. Neurology 64: 2098-101. doi: 10.1212/01.WNL.0000166034.71337.5E
Salman MS, Sharpe JA, Eizenman M, Lillakas L, To T, Westall C, Steinbach MJ, Dennis M (2006) Saccadic adaptation in Chiari type II malformation. Can J Neurol Sci 33: 372-8. doi: 10.1017/s0317167100005321
Salman MS, Sharpe JA, Lillakas L, Steinbach MJ, Dennis M (2007) Smooth ocular pursuit in Chiari type II malformation. Dev Med Child Neurol 49: 289-93. doi: 10.1111/j.1469-8749.2007.00289.x
Schwalbe E, Gredig M (1907) Ueber Entwicklungsstörungen des Kleinhirns, Hirnstamms und Halsmarks bei Spina bifida (Arnold’sche und Chiari’sche Missbildung) [On developmental disorders of the cerebellum, brainstem and cervical spinal cord in spina bifida (Arnold’s and Chiari’s malformations)]. Beitr Path Anat 40: 132-194.
Shaikh AG, Ghasia FF (2015) Neuro-ophthalmology of type 1 Chiari malformation. Expert Rev Ophthalmol 10: 351-357. doi: 10.1586/17469899.2015.1057505
Singh R, Arora R, Kumar R (2018) Clinical Notes on Chiari Malformation. J Craniofac Surg 29: e417-e421. doi: 10.1097/SCS.0000000000004424
Stahl JS, Plant GT, Leigh RJ (2002) Medical treatment of nystagmus and its visual consequences. J R Soc Med 95: 235-7. doi: 10.1258/jrsm.95.5.235
Straube A, Leigh RJ, Bronstein A, Heide W, Riordan-Eva P, Tijssen CC, Dehaene I, Straumann D (2004) EFNS task force–therapy of nystagmus and oscillopsia. Eur J Neurol 11: 83-9. doi: 10.1046/j.1468-1331.2003.00754.x
Strupp M, Schuler O, Krafczyk S, Jahn K, Schautzer F, Buttner U, Brandt T (2003) Treatment of downbeat nystagmus with 3,4-diaminopyridine: a placebo-controlled study. Neurology 61: 165-70. doi: 10.1212/01.wnl.0000078893.41040.56
Tubbs RS, Demerdash A, Vahedi P, Griessenauer CJ, Oakes WJ (2016) Chiari IV malformation: correcting an over one century long historical error. Child’s Nervous System 32: 1175-1179. doi: 10.1007/s00381-015-2765-8
Tubbs RS, Iskandar BJ, Bartolucci AA, Oakes WJ (2004a) A critical analysis of the Chiari 1.5 malformation. J Neurosurg 101: 179-83. doi: 10.3171/ped.2004.101.2.0179
Tubbs RS, Muhleman M, Loukas M, Oakes WJ (2012) A new form of herniation: the Chiari V malformation. Childs Nerv Syst 28: 305-7. doi: 10.1007/s00381-011-1616-5
Tubbs RS, Soleau S, Custis J, Wellons JC, Blount JP, Oakes WJ (2004b) Degree of tectal beaking correlates to the presence of nystagmus in children with Chiari II malformation. Childs Nerv Syst 20: 459-61. doi: 10.1007/s00381-004-0948-9
Weeks CL, Hamed LM (1999) Treatment of acute comitant esotropia in Chiari I malformation. Ophthalmology 106: 2368-71. doi: 10.1016/S0161-6420(99)90541-9
Yee RD, Baloh RW, Honrubia V (1984) Episodic vertical oscillopsia and downbeat nystagmus in a Chiari malformation. Arch Ophthalmol 102: 723-5. doi: 10.1001/archopht.1984.01040030579023
Young YH, Huang TW (2001) Role of clonazepam in the treatment of idiopathic downbeat nystagmus. Laryngoscope 111: 1490-3. doi: 10.1097/00005537-200108000-00029
Zimmerman CF, Roach ES, Troost BT (1986) See-saw nystagmus associated with Chiari malformation. Arch Neurol 43: 299-300. doi: 10.1001/archneur.1986.00520030085024
![]()