By Marcello Cherchi, MD PhD
For patients
It is unusual for brain problems to cause deafness. In rare cases, disease affecting the auditory cortex on both sides may cause deafness; such cases are usually due to stroke, and in such the onset of bilateral deafness is abrupt. A hearing test confirms this hearing loss. A brain MRI may help identify the underlying cause. Treatment depends on the underlying cause. Patients usually do not recover significant hearing.
For clinicians
Overview
The afferent auditory system decussates early in its course (beginning at the dorsal and ventral cochlear nuclei), and multiple times during its ascent to the auditory cortex located in the superior temporal gyrus of each temporal lobe. Consequently, a unilateral lesion of the auditory cortex does not cause hearing loss. Bilateral lesions of the auditory cortex are very unusual, but such cases can result in cortical deafness. Of these rare cases, the most common cause is stroke, and in such cases the symptom onset is abrupt. Most patients will complain of hearing loss Physical examination and audiometry will show sensorineural hearing loss. Middle latency responses and late latency responses may be abnormal or absent. Imaging (preferably MRI) should be performed to characterize the lesions. Treatment depends on the etiology. Prognosis is poor; recovery is usually minimal.
Introduction: review of anatomy
Anatomically the cochlea exhibits tonotopic mapping, with type 1 hair cells mediating the highest frequencies at the base of the cochlea, and those mediating the lowest frequencies at its apex (Pickles 2015), as shown in the Figure below, from Li and colleagues (Li et al. 2021).
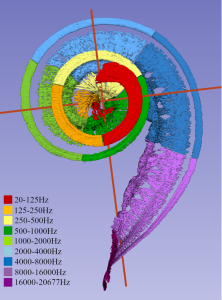
This tonotopic mapping is preserved throughout the entire course of afferent auditory signals, from the spiral ganglion all the way up to the auditory cortex, as illustrated in the Figure below, from Saenz and Langers (Saenz and Langers 2014). Primary auditory cortex is located in the superior temporal gyrus of each temporal lobe, which is irrigated by the middle cerebral artery.

Auditory signals travel from the spiral ganglion through the auditory component of the cochleovestibular nerve and synapse in the dorsal and ventral cochlear nuclei in the brainstem. From there, some fibers decussate immediately via commissural pathways through the trapezoid body, while other fibers ascend. There are multiple other decussations during this ascent through the afferent auditory system, including at the levels of the nuclei of the lateral lemniscus (via the commissure of Probst), the inferior colliculus (via the commissure of the inferior colliculus) and the auditory cortex (via the corpus callosum). Thus, by the time auditory signals have reached primary auditory cortex, there is significant bilaterality of encoding. For this reason, a lesion of one primary auditory cortex does not cause deafness, though it can cause a variety of other acoustic perceptual abnormalities.
Epidemiology
Cortical deafness is rare because of the combination of lesions required.
Pathophysiological mechanism of disease
Like any area of the central nervous system, primary auditory cortex is vulnerable to all manner of lesions, including infarction, demyelination and tumor.
Because of the multiple decussations mentioned earlier, a unilateral lesion of the auditory cortex does not cause deafness. However, bilateral lesions can, and do, cause cortical deafness. It is unusual to suffer lesions in bilateral auditory cortices, consequently cortical deafness is quite rare (Brody et al. 2013). In my first 15 years of subspecialty practice, I encountered only one convincing case of cortical deafness.
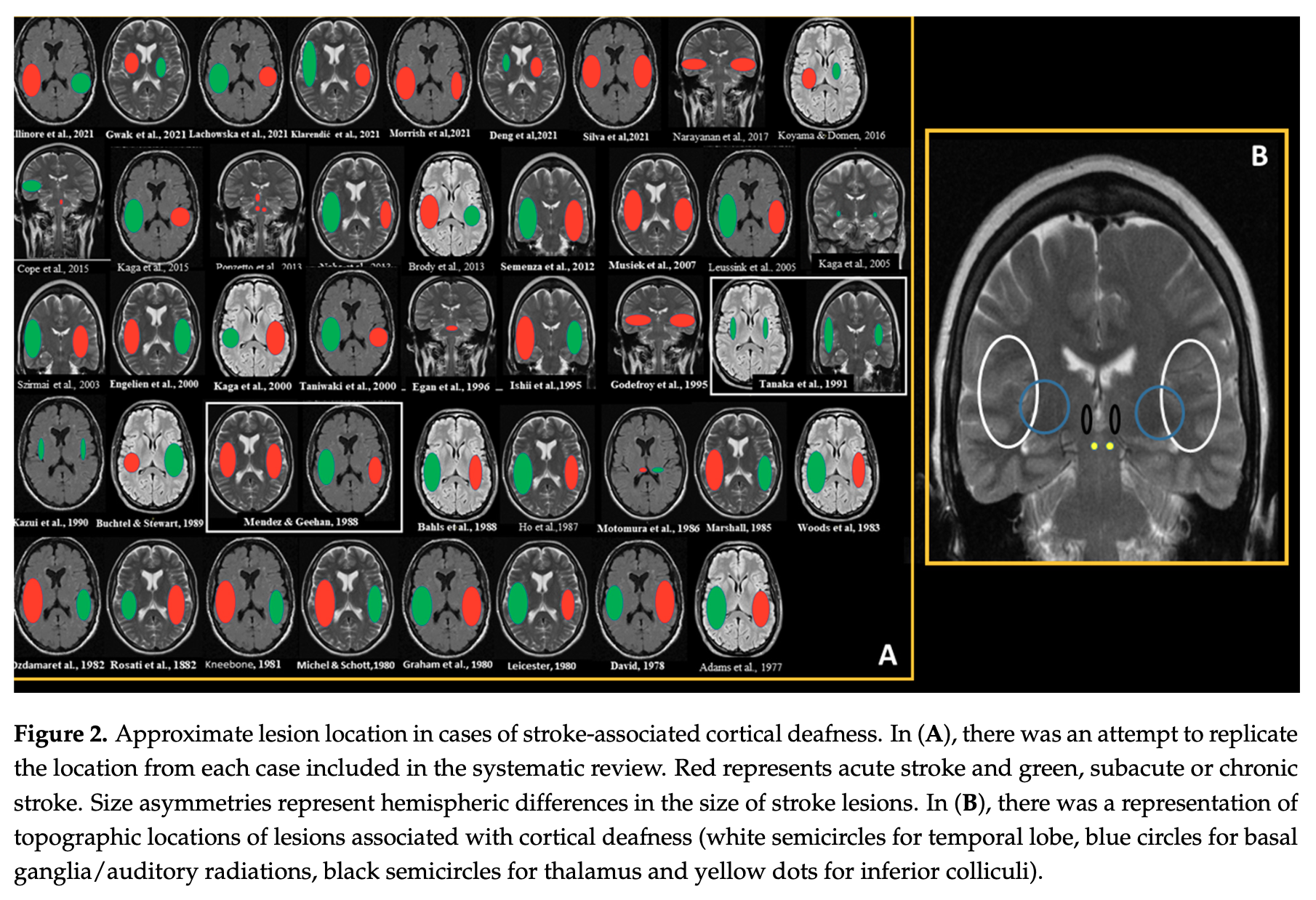
Although cortical deafness is rare, the most common cause is bilateral temporal lobe infarctions, which is documented by numerous case reports in the literature (Adams et al. 1977; Bahls et al. 1988; Brody et al. 2013; Graham et al. 1980; Kaga et al. 2015; Kneebone and Burns 1981; Lachowska et al. 2021; Michel et al. 1980; Narayanan et al. 2017; Silva et al. 2020). In some cases the infarctions may be subcortical (Earnest et al. 1977; Tanaka et al. 1991) rather than cortical.
The location of such infarctions can be variable. Silva and colleagues (Silva et al. 2021) reviewed 46 cases of infarct-related cortical deafness and summarized their findings in the Figure below.
Less common causes of cortical deafness include meningitis (Sasidharan et al. 2020) and multiple sclerosis (Tabira et al. 1981). There is one report of Creutzfeldt-Jacob disease presenting with cortical deafness (Tobias et al. 1994). One report describes hearing loss following head trauma (Singh et al. 1997), but in this case the trauma caused a subdural hematoma and other significant intracranial lesions.
Clinical presentation
Most patients present with a complaint of hearing loss. Some patients will deny hearing loss, suggesting a type of auditory agnosia analogous to the visual agnosia experienced in Anton syndrome (cortically blind patients who deny vision loss).
Physical examination
Patients have sensorineural hearing loss on examination.
Ocular motor examination
Unless the patient has suffered additional lesions outside of the auditory cortices, ocular motor examination should be normal.
Testing: auditory
In most cases of cortical deafness, audiometry shows profound bilateral sensorineural hearing loss, as demonstrated in the audiogram in the Figure below, from Narayanan and colleagues (Narayanan et al. 2017).

Testing: brainstem auditory evoked responses (BAERs)
Some reported cases in which electrophysiological testing was performed report absence of middle latency evoked responses (Ozdamar et al. 1982; Vedder et al. 1988) or late latency responses (Sasidharan et al. 2020).
Imaging
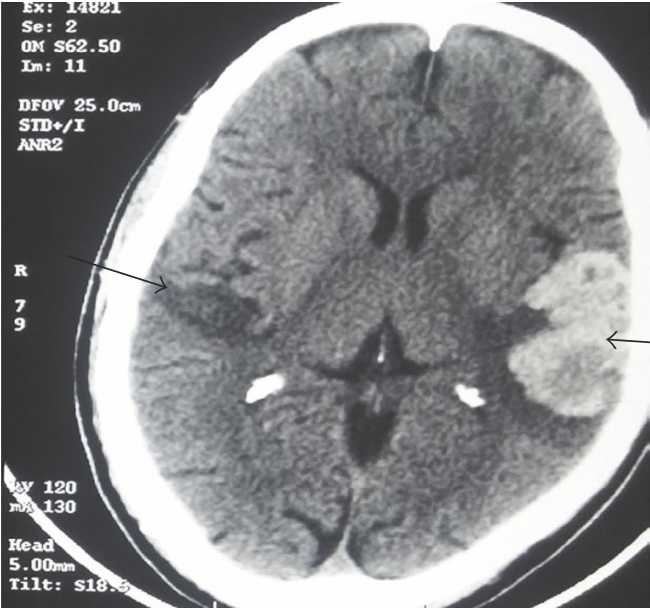
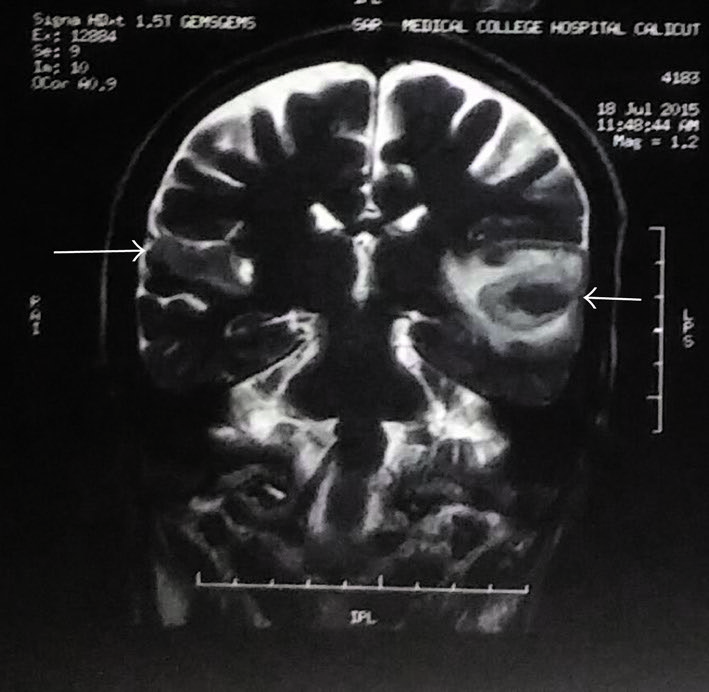
The Figures below, from Narayanan and colleagues (Narayanan et al. 2017), show bilateral temporal lobe infarctions with left-sided hemorrhagic transformations.
 |
 |
Differential diagnosis
In a patient with abrupt-onset total bilateral hearing loss, cortical deafness (from infarction) is a consideration. If the underlying etiology is a slower process, such as multiple sclerosis or tumor, then the onset of hearing loss may occur over a longer period, and could perhaps resemble diseases such as autoimmune inner ear disease.
Treatment
Treatment involves management of the underlying disease, if possible. Recovery is usually poor.
Prognosis
Prognosis for recovery of any hearing is poor. Brody and colleagues (Brody et al. 2013) state that, “Presentation and recovery of hearing are dependent on the extent of the initial lesions. The majority of patients can expect improvements in pure-tone auditory thresholds over time; however patients should be counseled that recovery of the ability to understand speech is unlikely.”
References
Adams AE, Rosenberger K, Winter H, Zollner C (1977) A case of cortical deafness. Arch Psychiatr Nervenkr (1970) 224: 213-20. doi: 10.1007/BF00348003
Bahls FH, Chatrian GE, Mesher RA, Sumi SM, Ruff RL (1988) A case of persistent cortical deafness: clinical, neurophysiologic, and neuropathologic observations. Neurology 38: 1490-3. doi: 10.1212/wnl.38.9.1490
Brody RM, Nicholas BD, Wolf MJ, Marcinkevich PB, Artz GJ (2013) Cortical deafness: a case report and review of the literature. Otol Neurotol 34: 1226-9. doi: 10.1097/MAO.0b013e31829763c4
Earnest MP, Monroe PA, Yarnell PR (1977) Cortical deafness: demonstration of the pathologic anatomy by CT scan. Neurology 27: 1172-5. doi: 10.1212/wnl.27.12.1172
Graham J, Greenwood R, Lecky B (1980) Cortical deafness–a case report and review of the literature. J Neurol Sci 48: 35-49. doi: 10.1016/0022-510x(80)90148-3
Kaga K, Shinjo Y, Enomoto C, Shindo M (2015) A case of cortical deafness and loss of vestibular and somatosensory sensations caused by cerebrovascular lesions in bilateral primary auditory cortices, auditory radiations, and postcentral gyruses – complete loss of hearing despite normal DPOAE and ABR. Acta Otolaryngol 135: 389-94. doi: 10.3109/00016489.2014.980914
Kneebone CS, Burns RJ (1981) A case of cortical deafness. Clin Exp Neurol 18: 91-7.
Lachowska M, Pastuszka A, Sokolowsk J, Szczudlik P, Niemczyk K (2021) Cortical Deafness Due to Ischaemic Strokes in Both Temporal Lobes. J Audiol Otol 25: 163-170. doi: 10.7874/jao.2020.00269
Li H, Helpard L, Ekeroot J, Rohani SA, Zhu N, Rask-Andersen H, Ladak HM, Agrawal S (2021) Three-dimensional tonotopic mapping of the human cochlea based on synchrotron radiation phase-contrast imaging. Sci Rep 11: 4437. doi: 10.1038/s41598-021-83225-w
Michel F, Peronnet F, Schott B (1980) A case of cortical deafness: clinical and electrophysiological data. Brain Lang 10: 367-77. doi: 10.1016/0093-934x(80)90062-0
Narayanan S, Majeed KA, Subramaniam G, Narayanan A, Navaf KM (2017) A Case of Cortical Deafness due to Bilateral Heschl Gyrus Infarct. Case Rep Med 2017: 6816748. doi: 10.1155/2017/6816748
Ozdamar O, Kraus N, Curry F (1982) Auditory brain stem and middle latency responses in a patient with cortical deafness. Electroencephalogr Clin Neurophysiol 53: 224-30. doi: 10.1016/0013-4694(82)90027-x
Pickles JO (2015) Auditory pathways: anatomy and physiology. Handb Clin Neurol 129: 3-25. doi: 10.1016/B978-0-444-62630-1.00001-9
Saenz M, Langers DR (2014) Tonotopic mapping of human auditory cortex. Hear Res 307: 42-52. doi: 10.1016/j.heares.2013.07.016
Sasidharan M, Itty JE, Hinduja G, Hasna S, Priya D (2020) Cortical deafness as a sequela to meningitis: a single case study. Intractable Rare Dis Res 9: 247-250. doi: 10.5582/irdr.2020.03072
Silva G, Goncalves R, Taveira I, Mouzinho M, Osorio R, Nzwalo H (2021) Stroke-Associated Cortical Deafness: A Systematic Review of Clinical and Radiological Characteristics. Brain Sci 11. doi: 10.3390/brainsci11111383
Silva J, Sousa M, Mestre S, Nzwalo I, Nzwalo H (2020) Cortical deafness of following bilateral temporal lobe stroke. J Stroke Cerebrovasc Dis 29: 104827. doi: 10.1016/j.jstrokecerebrovasdis.2020.104827
Singh M, Ahluwalia H, Lal KK, Chauhan SP (1997) Cortical deafness following head injury -a case report. Indian J Otolaryngol Head Neck Surg 49: 81-4. doi: 10.1007/BF03021334
Tabira T, Tsuji S, Nagashima T, Nakajima T, Kuroiwa Y (1981) Cortical deafness in multiple sclerosis. J Neurol Neurosurg Psychiatry 44: 433-6. doi: 10.1136/jnnp.44.5.433
Tanaka Y, Kamo T, Yoshida M, Yamadori A (1991) ‘So-called’ cortical deafness. Clinical, neurophysiological and radiological observations. Brain 114 ( Pt 6): 2385-401. doi: 10.1093/brain/114.6.2385
Tobias E, Mann C, Bone I, de Silva R, Ironside J (1994) A case of Creutzfeldt-Jakob disease presenting with cortical deafness. J Neurol Neurosurg Psychiatry 57: 872-3. doi: 10.1136/jnnp.57.7.872
Vedder JS, Barrs DM, Fifer RC (1988) The use of middle latency response in the diagnosis of cortical deafness. Otolaryngol Head Neck Surg 98: 333-7. doi: 10.1177/019459988809800412
![]()