By Marcello Cherchi, MD PhD
For patients
The phrase, “large cochlear aqueduct,” refers to the anatomical configuration of a tiny connection between the inner ear and the inside of the skull. This is rare, and it is occasionally observed on certain brain imaging studies. Investigators disagree on whether this finding is significant, and whether it explains any symptoms or diseases. If on an imaging study you are found to have a “large cochlear aqueduct,” then consultation with an otolaryngologist is reasonable.
For clinicians
Overview
Literature suggests that the normative range of sizes and configurations of the cochlear aqueduct is variable, bringing into question what constitutes a “large” cochlear aqueduct (CA). Even if a high resolution temporal bone CT is interpreted a showing a large CA, it is presently unclear whether this explains any symptoms, and whether it predicts surgical complications during cochlear implantation. If a “large” CA is incidentally discovered on imaging, then referral to an otolaryngologist is reasonable.
Introduction
When a neuro-otologic surgeon is placing a cochlear implant, some patients exhibit an unexpectedly brisk flow of cerebrospinal fluid — colloquially referred to as “gushers” and “oozers” (Schuknecht and Reisser 1988). In the interest of being able to predict (and avoid) this surgical complication, investigators have explored whether the underlying cause might be an (inappropriately) large cochlear aqueduct (CA).
Some investigators have also postulated that large CAs may be the etiology of otopathologies such as sensorineural hearing loss and perilymphatic fistula (Mukherji, Baggett et al. 1998).
However, a large CA is an unusual finding. Some authors have expressed skepticism about its existence (Jackler and Hwang 1993, Daly and Donnelly 1996), let alone whether it causes or explains any disease.
Anatomy
The cochlear aqueduct (CA) is a small canal in the temporal bone connecting the cochlea to the subarachnoid space (Bianchin, Polizzi et al. 2016). The CA contains the cochlear canaliculus (Migirov and Kronenberg 2005) and perilymphatic (periotic) duct (Mukherji, Baggett et al. 1998, Bianchin, Polizzi et al. 2016), which connects the perilymphatic compartment of the inner ear (scala tympani) with the cerebrospinal fluid space of the posterior cranial fossa (Jackler and Hwang 1993, Mukherji, Baggett et al. 1998).
Multiple authors (Mukherji, Baggett et al. 1998, Migirov and Kronenberg 2005, Bianchin, Polizzi et al. 2016), citing Jackler and Hwang (Jackler and Hwang 1993), recognize four segments of the CA, proceeding from lateral to medial:
- The lateral (cochlear) orifice/aperture, also called the medial aperture, which opens into the scala tympani at the basal turn of the cochlea, adjacent to the round window membrane.
- The otic capsule segment, which runs medially through the temporal bone. In normal individuals, the narrowest portion (isthmus) of the cochlear aqueduct lies within the otic capsule segment (Migirov and Kronenberg 2005).
- The petrous apex segment.
- The medial (cranial) orifice/aperture, which opens into the subarachnoid space in the posterior cranial fossa, adjacent to the pars nervosa of the jugular foramen.
Histologically, “Unlike the vestibular aqueduct, the cochlear aqueduct does not contain a true epithelium-lined duct” , but rather is “filled with a loose mesh of connective tissue that, although permeable to fluid, limits the patency” of the CA (Bianchin, Polizzi et al. 2016).
Migirof and Kronenberg (Migirov and Kronenberg 2005) studied high-resolution CT images of 488 normal patients. They reported the cochlear aqueducts to be “asymmetric… in 10.6% of the images,” suggesting that 89.4% of individuals have symmetric CAs.
Embryology
In the search for possible explanations of large CA, some authors begin by focusing on the embryonic development.
“The embryology of the CA [cochlear aqueduct] differs substantially from that of the other components of the inner ear, both in timing and tissue of origin… The CA… is a secondary formation created by enclosure of a soft tissue mesh that is entrapped in a channel within the developing otic capsule” (Jackler and Hwang 1993).
More specifically, there are “separate embryological origins of the cochlear aqueduct and membranous labyrinth. The membranous labyrinth is derived from the otic vesicle. The perilymphatic duct initially appears as an outpouching of the subarachnoid space. The cochlear aqueduct forms in the mesoderm surrounding the perilymphatic duct and gradually ossified during embryogenesis” (Schuknecht and Reisser 1988).
In view of the embryonic development, some authors hypothesize that a large CA reflects developmental arrest. “The development of the periotic duct and its adjacent structures in the human fetus occur over a 6-week period (20 – 26 weeks). At 22 weeks of gestation, the petrous apex ossifies and forms the medial wall of the CA [cochlear aqueduct], and the progressive otic capsule ossification leads to the formation of the lateral wall of the CA. Subsequently, the width of the CA diminishes with gestational age. Therefore, an enlarged CA may result from the developmental arrest of the primitive CA” (Kim, Sim et al. 2013).
Post-natally the CA appears to change to some degree. “During the neonatal period the CA [cochlear aqueduct] is shorter, straighter, wider, and more free of arachnoid mesh than in adults” (Jackler and Hwang 1993). However, as far as the narrowest point of the CA is concerned, the temporal bone study by Gopen and colleagues showed that, “there were no significant changes in this diameter with increasing [post-natal] age” (Gopen, Rosowski et al. 1997).
Hydraulics
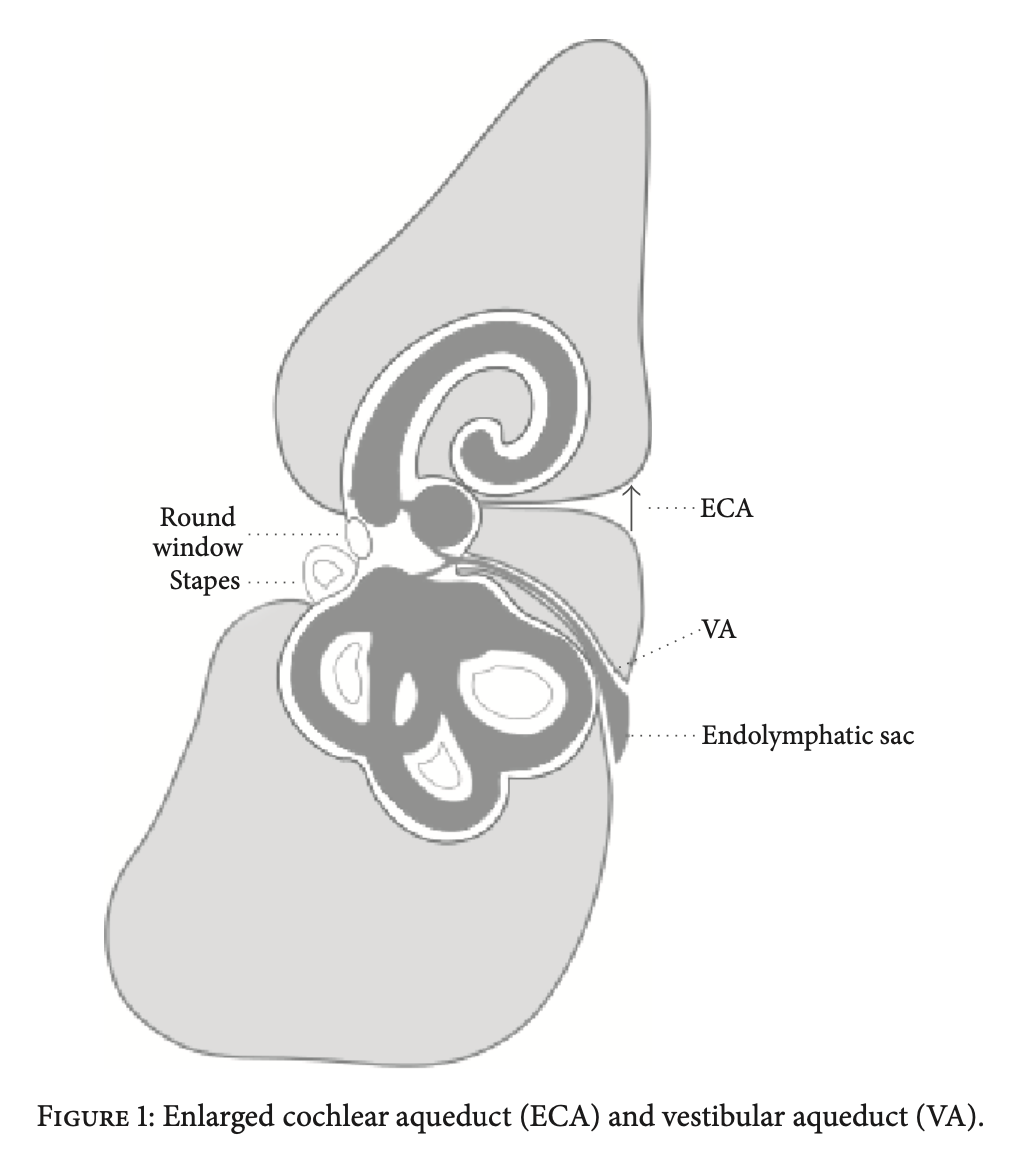
The Figure below, from Bianchin and colleagues (Bianchin, Polizzi et al. 2016), is a schematic depiction of the relative dispositions of the cochlear aqueduct and nearby structures.
The Figure below, from Gopen and colleagues (Gopen, Rosowski et al. 1997), schematically depicts the hydraulic arrangement of the stapes (in the oval window), the cochlea, the round window and the cochlear aqueduct.
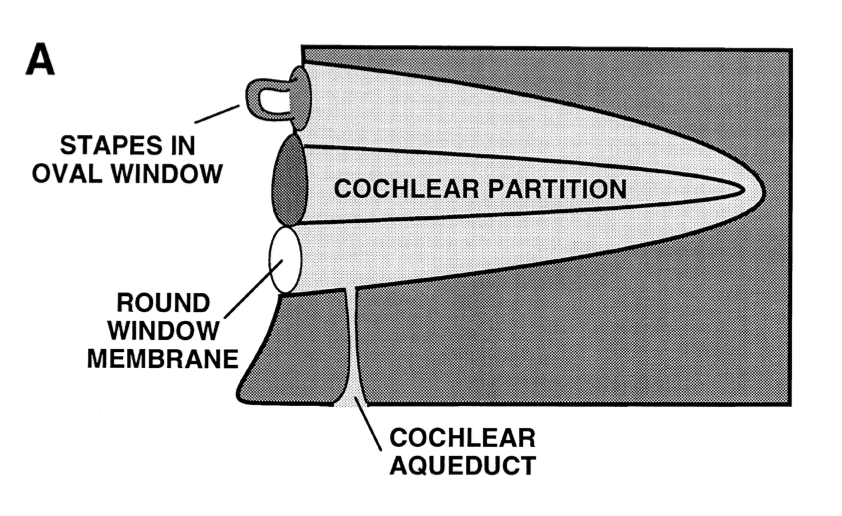
The series of 101 temporal bones studied by Gopen and colleagues revealed that, “The narrowest point of the [cochlear] aqueduct as defined by its bony wall was found to lie 200 – 300 micrometers from its cochlear end. The mean value (±1 SD) for this smallest diameter was 138 (±58) micrometers” (Gopen, Rosowski et al. 1997).
Allen (Allen 1987) used the Poiseuille-Hagen equation to model the flow of fluid through the cochlear aqueduct.
Jackler and Hwang commented that, “Allen has argued that because fluid flow through a tube varies with the fourth power of its diameter, even slight enlargements of the CA [cochlear aqueduct] may have a substantial impact on perilymphatic flows into the vestibule” (Jackler and Hwang 1993). Stimmer similarly commented that, “The flow of liquids through tubes is a linear function of pressure, viscosity and length of the tube and in fourth degree power function of the radius of the lumen. Therefore, the radius, i.e. the diameter is the most critical factor for the flow rate; small variations in size of the CA [cochlear aqueduct] cause large variations in flow rate” (Stimmer 2011).
However, Jackler and Hwang also critiqued this model, noting that, “This simple model of fluid flow through a tube, while conceptually useful, does not take into account the obstructing effect of the membranous mesh that fills the aqueduct. Deficiencies in these membrane baffles theoretically may result in increased flows through even a normal-sized aqueduct” (Jackler and Hwang 1993).
Significance of patency/occlusion of the cochlear aqueduct
Gopen and colleagues (Gopen, Rosowski et al. 1997) studied 101 temporal bones from patients aged 0 – 100 years and observed four categories of CA:
- Patent lumen throughout.
- Lumen filled with loose connective tissue.
- Lumen occluded by bone.
- Obliteration of the aqueduct.
Gopen and colleagues found the following distribution of these categories by decade of life to be as follows:
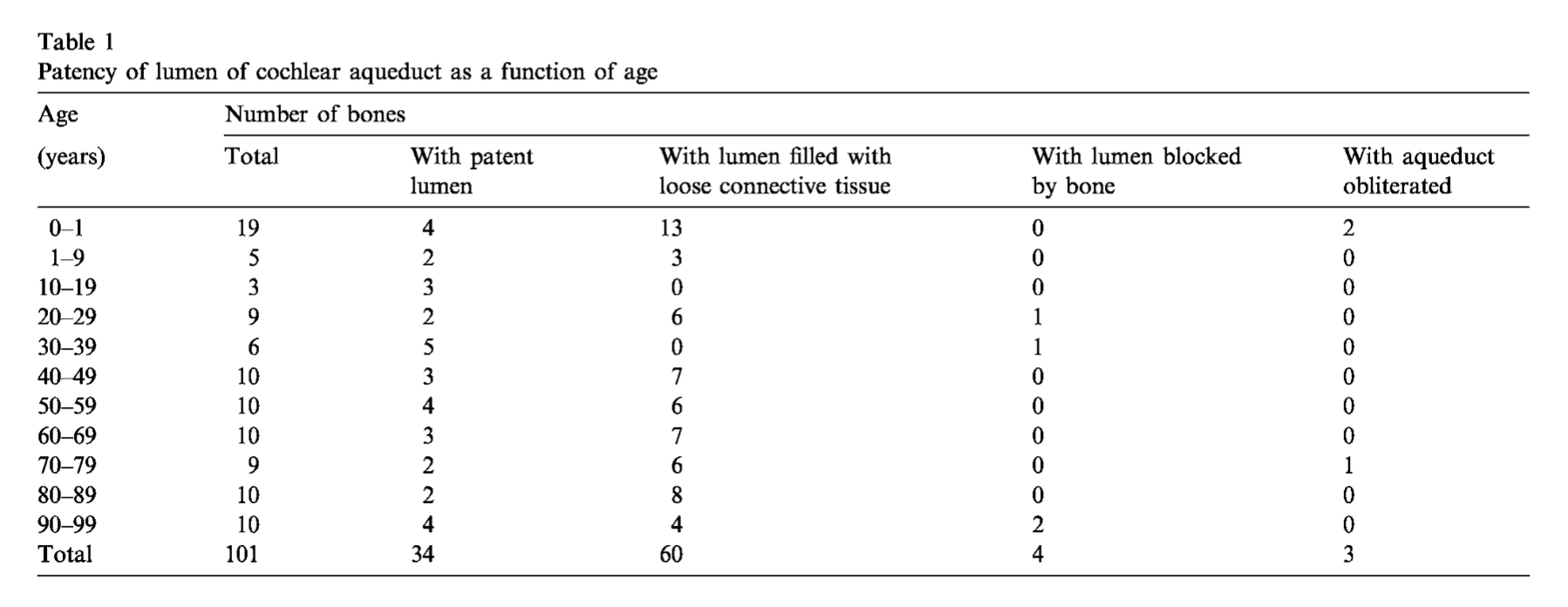
Overall, Gopen and colleagues found that, “The distribution of patency of the lumen in 101 normal temporal bones was: patent lumen throughout aqueduct 34%, lumen filled with loose connective tissue 59%, lumen blocked by bone 4%, and obliteration of the aqueduct 3%,” and they specifically commented that, “Normal ears may have cochlear aqueducts that are completely occluded or obliterated” (Gopen, Rosowski et al. 1997). These findings led Gopen and colleagues to conclude that, “occlusion of the cochlear aqueduct in the human causes no apparent ill effect on the normal inner ear” (Gopen, Rosowski et al. 1997).
Stimmer (Stimmer 2011) studied 400 high resolution temporal bone CTs of patients aged 1 – 94 years (average 43 years) and reported, “a diameter of more thatn 1 mm in the otic capsule portion could not be found in any of the 400 cases we examined,” and “we did not find a single CA [cochlear aqueduct] that was completely visible in all segments with a diameter of more than 1 mm in the otic capsule segment,” and therefore, “we concluded that not a single case of pathologic CA enlargement could be found using highly developed multislice CT -technique. According to these findings CA-enlargement could not be used as an explanation for clinical inner ear deficits.”
Risk factors
Kim and colleagues (Kim, Sim et al. 2013) studied 35 patients (70 ears) to determine whether the presence of a large vestibular aqueduct correlates in any way with a large cochlear aqueduct. They reported that, “The findings indicated that the mean CA [cochlear aqueduct] size was significantly larger in patients with EVA [enlarged vestibular aqueduct],” yet, “linear regression analysis revealed no statistically significant correlation between CA and VA [vestibular aqueduct] size.” In other words, patients with large vestibular aqueducts also have large cochlear aqueducts, but beyond simply being “large,” there is otherwise no correlation between the degree of oversizing of the two structures.
Imaging
High resolution temporal bone CT is the imaging modality of choice for diagnosing and characterizing the cochlear aqueduct.
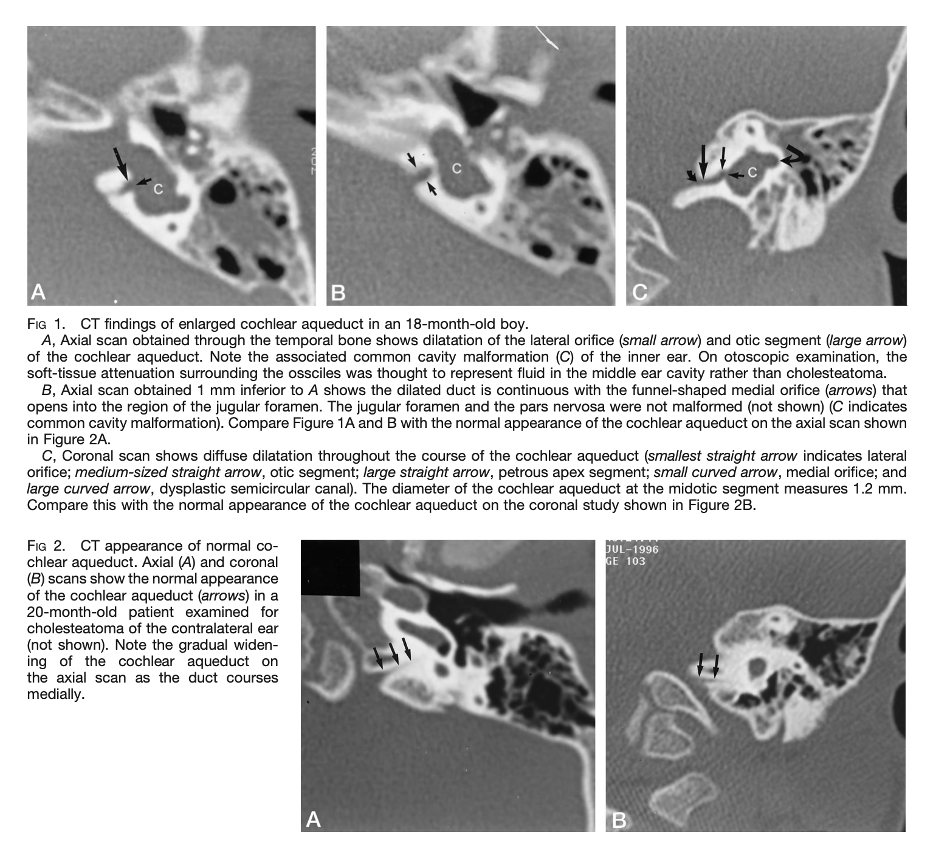
The Figure below, from Mukherji and colleagues (Mukherji, Baggett et al. 1998), compares images of a large cochlear aqueduct (top three panels) with those of a normal cochlear aqueduct (bottom 2 panels).
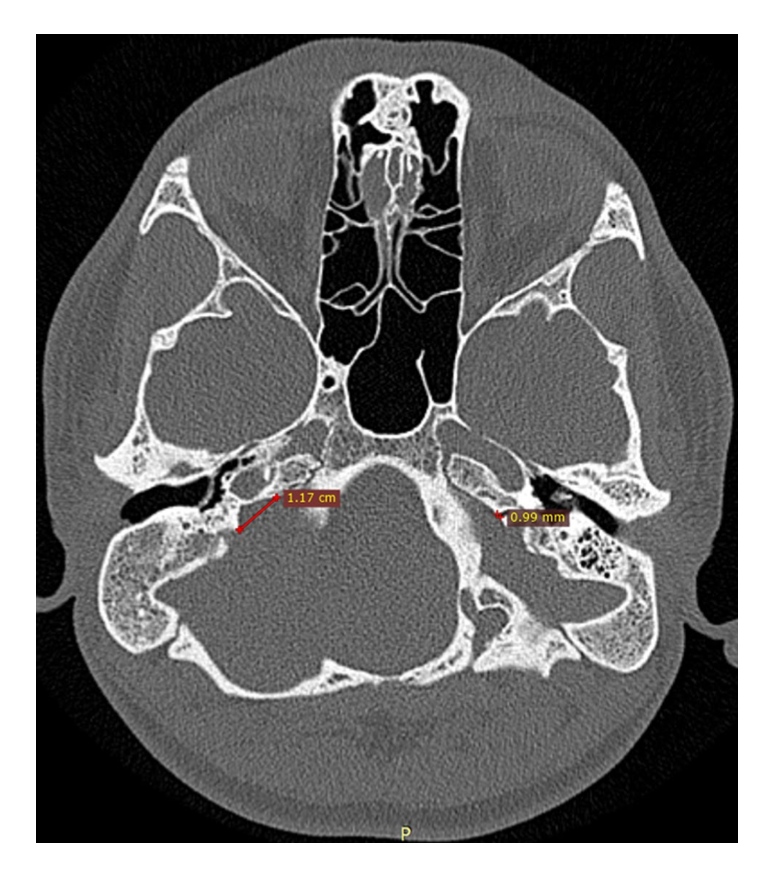
The Figure below, from Satar and colleagues (Satar, Genc et al. 2021), shows a high resolution axial CT demonstrating significant asymmetry between the cochlear aqueducts, with the patient’s right CA being much larger than the left.
References
Allen GW (1987) Fluid flow in the cochlear aqueduct and cochlea-hydrodynamic considerations in perilymph fistula, stapes gusher, and secondary endolymphatic hydrops. Am J Otol 8: 319-22.
Bianchin G, Polizzi V, Formigoni P, Russo C, Tribi L (2016) Cerebrospinal Fluid Leak in Cochlear Implantation: Enlarged Cochlear versus Enlarged Vestibular Aqueduct (Common Cavity Excluded). Int J Otolaryngol 2016: 6591684. doi: 10.1155/2016/6591684
Daly CA, Donnelly MJ (1996) The enlarged cochlear aqueduct–a radiological flying saucer? Clin Radiol 51: 821. doi: 10.1016/s0009-9260(96)80019-5
Gopen Q, Rosowski JJ, Merchant SN (1997) Anatomy of the normal human cochlear aqueduct with functional implications. Hear Res 107: 9-22. doi: 10.1016/s0378-5955(97)00017-8
Jackler RK, Hwang PH (1993) Enlargement of the cochlear aqueduct: fact or fiction? Otolaryngol Head Neck Surg 109: 14-25. doi: 10.1177/019459989310900104
Kim BG, Sim NS, Kim SH, Kim UK, Kim S, Choi JY (2013) Enlarged cochlear aqueducts: a potential route for CSF gushers in patients with enlarged vestibular aqueducts. Otol Neurotol 34: 1660-5. doi: 10.1097/MAO.0b013e3182a036e4
Migirov L, Kronenberg J (2005) Radiology of the cochlear aqueduct. Ann Otol Rhinol Laryngol 114: 863-6. doi: 10.1177/000348940511401110
Mukherji SK, Baggett HC, Alley J, Carrasco VH (1998) Enlarged cochlear aqueduct. AJNR Am J Neuroradiol 19: 330-2.
Satar B, Genc H, Meral SC (2021) Why did we encounter gusher in a stapes surgery case? Was it enlarged medial aperture of the cochlear aqueduct? Surg Radiol Anat 43: 225-229. doi: 10.1007/s00276-020-02602-8
Schuknecht HF, Reisser C (1988) The morphologic basis for perilymphatic gushers and oozers. Adv Otorhinolaryngol 39: 1-12. doi: 10.1159/000415649
Stimmer H (2011) Enlargement of the cochlear aqueduct: does it exist? Eur Arch Otorhinolaryngol 268: 1655-61. doi: 10.1007/s00405-011-1527-9
![]()