By Marcello Cherchi, MD PhD
For patients
Systemic lupus erythematosus (SLE), sometimes referred to as simply “lupus,” is an autoimmune disease whose cause is unknown. SLE can affect any part of the body. Occasionally it can cause hearing loss, tinnitus, ear fullness or disequilibrium. If you are known to have lupus and develop such symptoms, your doctor may suggest checking some tests of hearing and balance function to figure out whether those symptoms are really due to lupus, or due to more common causes.
For clinicians
Overview
Systemic lupus erythematosus (SLE), sometimes referred to as simply “lupus,” is an autoimmune disease of uncertain etiology that can involve nearly any organ system, and thus can have an extremely broad range of clinical presentations. SLE is more common in non-white populations and is much more common in women. SLE probably becomes manifest through a combination of genetic susceptibility and environmental/epigenetic factors. When SLE involves the central nervous system and specifically affects auditory or vestibular function, the mechanism of disease may involve deposition of immune complexes and cytotoxic damage. SLE patients may develop hearing loss that is usually sensorineural, typically involves the higher rather than the lower frequencies, and can be unilateral, asymmetrically bilateral, or symmetrically bilateral; the onset may be abrupt or gradual. SLE patients can develop other aural symptoms as well, such as tinnitus and aural fullness. While less common, SLE patients can also develop disequilibrium and ocular motor disorders.
Introduction
Systemic lupus erythematosus (SLE), sometimes referred to as simply “lupus,” is an autoimmune disease of uncertain etiology that can involve nearly any organ system. Its remarkable clinical heterogeneity has led to reasonable questions regarding whether this is truly a single disease (Rivas-Larrauri and Yamazaki-Nakashimada 2016).
Over the centuries the term “lupus” has been applied to what were probably a broad range of diseases, but in the early 19th century the work of several scholars, including Moritz Kaposi, James H. Sequiera, H. Balean and William Osler, led to the understanding of this as an autoimmune-mediated clinical entity with variable presentations (Felten et al. 2022).
Epidemiology
The incidence and prevalence of SLE has been difficult to determine for a variety of reasons; “Reports of the global incidence and prevalence of SLE vary widely, owing to inherent variation in population demographics, environmental exposures and socioeconomic factors. Differences in study design and case definitions also contribute to inconsistent reporting” (Barber et al. 2021), and reported “variability may reflect true differences across populations, or result from methodological differences of studies” (Danchenko et al. 2006).
Epidemiological studies generally report SLE to be more common in non-white racial groups (Danchenko et al. 2006; Pons-Estel et al. 2017) and more common in women (Pons-Estel et al. 2017). Although SLE occurs worldwide (Pons-Estel et al. 2017), there is a paucity of good quality epidemiologic data for most countries (Tian et al. 2023). Nevertheless, there appears to be considerable variability in geographic distribution, and considerable disagreement between studies; for example, Danchenko and colleagues report that, “There is a trend towards higher incidence and prevalence of SLE in Europe and Australia compared to the U.S.A. In Europe, the highest prevalence was reported in Sweden, Iceland and Spain” (Danchenko et al. 2006), while Rees and colleagues report North America to have the highest disease burden (Rees et al. 2017).
In the United States, analysis of data from the Centers for Disease Control suggests that in 2002 – 2009 the pooled incidence rate per 100,000 person-years was 5.1 (95% confidence interval 4.6 to 5.6) (Izmirly et al. 2021a), and the pooled prevalence rate was 72.8 per 100,000 person-years (95% confidence interval 65.3 – 81.0) (Izmirly et al. 2021b).
Genetics
The genetics of SLE are still being explored (Croker and Kimberly 2005; Goulielmos et al. 2018; Hess and Farhey 1994, 1995; James 2014; Lewkonia 1992; Marion and Postlethwaite 2014; Relle et al. 2015; Sestak et al. 2011; Sestak et al. 2007; Shen and Tsao 2004; Yim and Wakeland 2001), and similar to a number of other autoimmune diseases, it seems likely that SLE becomes manifest when there is a confluence of genetic susceptibility and environmental/epigenetic factors.
Pathophysiological mechanism of disease
The etiology of SLE is unknown. The ability of SLE to involve the central nervous system probably results from, “multiple contributors such as various immune effectors and the brain-intrinsic neuroimmune interfaces that are breached by the immune effectors. The postulated neuroimmune interfaces include the blood-brain barrier, blood-cerebrospinal fluid barrier, meningeal barrier, and glymphatic system,” such that “the underlying pathophysiology can be classified as vasculitis and vasculopathy, APS [anti-phospholipid syndrome], demyelinating syndrome, or autoimmune antibody-mediated encephalitis” (Ota et al. 2022).
As far as auditory disorders are concerned, many authors implicate the deposition of immune complexes (Sperling et al. 1998), while others suggest a range of mechanisms; for instance, it has been speculated that, “The main mechanisms involved in inner ear damage include the autoimmune response, deposition of immune complexes in the vessels and, to a lesser extent, cytotoxic damage” (Di Stadio and Ralli 2017).
As far as vestibular symptoms concerned, there are scattered case reports and case series of patients with cerebellar syndromes. Among all patients with systemic lupus erythematosus, cerebellar ataxia appears to be rare; al-Arfaj and colleagues (al-Arfaj and Naddaf 1995) report 1 out of 150 patients (0.7%); Singh and colleagues (Singh et al. 1988) report 3 out of 350 patients (0.9%); Cha and Kim (Cha and Kim 2020) report a rate of 2%. Lupus-related cerebellar ataxia can occur at any age; Liao and colleagues (Liao et al. 2003) reported a case of an 11-year-old male whose presenting symptoms were disequilibrium and unsteady gait.
The mechanism of lupus-related cerebellar ataxia is unclear. Most authors postulate an autoimmune cerebellitis (Kristoff et al. 2022). Some authors suggest cerebellar strokes secondary to lupus vasculopathy (Ghosh et al. 2014), while others dispute this (Yaginuma et al. 2000). There is a report of cerebellar ataxia secondary to lupus-related hydrocephalus (Ahmed et al. 2017).
Clinical presentation: auditory alone
Abbasi and Yazdi (Abbasi et al. 2013) presented a case-control study of 45 SLE patients, in whom reported symptoms included hearing loss in 5 (11%), tinnitus in 3 (7%) and ear fullness in 1 (2%). They also performed audiometry on these patients and calculated that the relative risk for sensorineural hearing loss in SLE patients compared to controls was 3.7 (95% confidence interval 2.3 – 4.9).
Gomides and colleagues (Gomides et al. 2007) reported on a series of 45 female SLE patients and found that 25 (56%) reported some auditory symptom (hearing loss, ear fullness, tinnitus), and sensorineural hearing loss was identified in 7 (16%).
Sperling and colleagues (Sperling et al. 1998) organized a self-directed questionnaire study of 84 SLE patients and found that 26 (31%) reported aural symptoms. Of these 84 patients, this included 13 (15%) who reported unilateral hearing loss with or without tinnitus, and 14 (17%) who reported bilateral hearing loss with or without tinnitus.
Caldarelli and colleagues (Caldarelli et al. 1986) reported the case of a 51-year-old woman who presented with sudden sensorineural hearing loss on one side, and then three weeks later, on the other side, and subsequent workup led to a diagnosis of SLE.
Clinical presentation: vestibular alone
Lupus-related cerebellar ataxia can present acutely (Appenzeller et al. 2008; Chattopadhyay et al. 2011; Sy et al. 2021; Yaginuma et al. 2000) or subacutely (Iwasaki et al. 2012; Manto et al. 1996) and can be rapidly progressive (Casciato et al. 2018). Cerebellar ataxia can occur in a patient with pre-existing manifestations of systemic lupus erythematosus, but rarely can be the initial manifestation (Kutlubaev et al. 2020).
Clinical presentation: auditory and vestibular
Maciaszczyk and colleagues (Maciaszczyk et al. 2011) studied 35 unselected consecutive SLE patients and compared them to 30 healthy controls. Symptoms reported by the SLE patients included disequilibrium in 71%, tinnitus in 40%, hyperacusis in 17%, hearing loss in 17% and ear fullness in 3%.
Batuecas-Caletrio and colleagues (Batuecas-Caletrio et al. 2013) studied 89 SLE patients. Of these, 8 (9%) reported episodic vertigo.
Karatas and colleagues (Karatas et al. 2007) studied 28 SLE patients and found that reported symptoms included tinnitus in 9 (32%), hearing loss in 2 (7%) and disequilibrium in 8 (29%).
The Table below summarizes the auditory and vestibular symptoms reported by Karatas and colleagues (Karatas et al. 2007).
|
Tinnitus |
Hearing loss |
Vertigo |
Asymptomatic |
|
|
Tinnitus alone |
2 |
|||
|
Hearing loss alone |
||||
|
Vertigo alone |
3 |
|||
|
Tinnitus + hearing loss + vertigo |
2 |
1 |
2 |
|
|
Tinnitus + hearing loss |
3 |
1 |
||
|
Tinnitus + vertigo |
2 |
3 |
||
|
Asymptomatic |
9 |
|||
|
Totals |
9 (32%) |
2 (7%) |
8 (29%) |
9 (32%) |
The temporal progression of hearing loss is variable. In some cases the hearing loss presents acutely (Caldarelli et al. 1986; Riera et al. 2020), while in others it can progress over months (Khalidi et al. 2008).
Ocular motor examination
In rare cases, SLE can present with ocular motor abnormalities that are detectable on face-to-face examination.
Cakir and colleagues report a case of a 45-year-old woman who presented with acute-onset disequilibrium, ataxia and diplopia, and was found to have internuclear ophthalmoplegia (Cakir et al. 2014). Galindo and colleagues (Galindo et al. 1998) conducted a retrospective study of 268 SLE patients and identified internuclear ophthalmoplegia in four (1.5%).
Kunavarapu and colleagues (Kunavarapu et al. 2001) reported a case presenting with one-and-a-half syndrome.
Liao and colleagues (Liao et al. 2003) presented the case of an 11-year-old male whose presenting examination showed “bilateral vertical nystagmus.”
Testing: auditory
Abbasi and Yazdi (Abbasi et al. 2013) presented a case-control study of 45 SLE patients in whom they performed audiometry. They calculated that the relative risk for sensorineural hearing loss in SLE patients compared to controls was 3.7 (95% confidence interval 2.3 – 4.9).
Bowman and colleagues (Bowman et al. 1986) prospectively studied 30 patients who had been hospitalized for an SLE exacerbation, and “found an 8% incidence of substantial, previously undetected hearing loss without attributable cause.”
Gomides and colleagues (Gomides et al. 2007) reported on a series of 45 female SLE patients and found that 25 (56%) reported some auditory symptom (hearing loss, ear fullness, tinnitus), and sensorineural hearing loss was identified in 7 (16%).
Jimenez-Alonso and colleagues (Jimenez-Alonso et al. 2002) studied 91 consecutive SLE patients and found that 14 (15%) exhibited sensorineural hearing loss, “affecting mainly the high and middle frequencies,” which was bilateral in 9 and unilateral in 5.
Kastanioudakis and colleagues (Kastanioudakis et al. 2002) compared 43 SLE patients and 50 age-matched healthy controls. They found hearing loss in 9 (23%) of SLE patients, of whom 8 had sensorineural hearing loss and 1 had conductive hearing loss. Of the 9 patients with sensorineural hearing loss, the loss was bilateral and symmetrical in 1, bilateral but asymmetrical in four, and unilateral in 3. In the 1 patient with conductive hearing loss, the loss was unilateral.
Andonopoulos and colleagues (Andonopoulos et al. 1995) compared 40 unselected consecutive SLE patients and compared them to 65 age-matched healthy controls, and divided them into age groups of 30-39, 40-49, 50-59 and 60-69 years. They found that, “A statistically significant decrease in hearing acuity at low frequencies (125-500 Hz) was found in the patients aged 16-59 compared with the controls, whereas in the first group of young patients (16-29) a similar decrease was observed in the high frequencies as well,” and concluded that, “With regard to the high frequencies, our results suggest that young SLE patients exhibit a “premature aging” of the inner ear which eventually stabilizes. On the contrary, at low frequencies there is a regular decrease in acuity in all groups (except for the elderly patients: 60-69), which may indicate a subclinical vestibular hydrops.”
Maciaszczyk and colleagues (Maciaszczyk et al. 2011) studied 35 unselected consecutive SLE patients and compared them to 30 healthy controls. They reported that, “Longer ABR [auditory brainstem evoked response] latency averages were observed in the group of SLE patients compared to control.”
Testing: vestibular
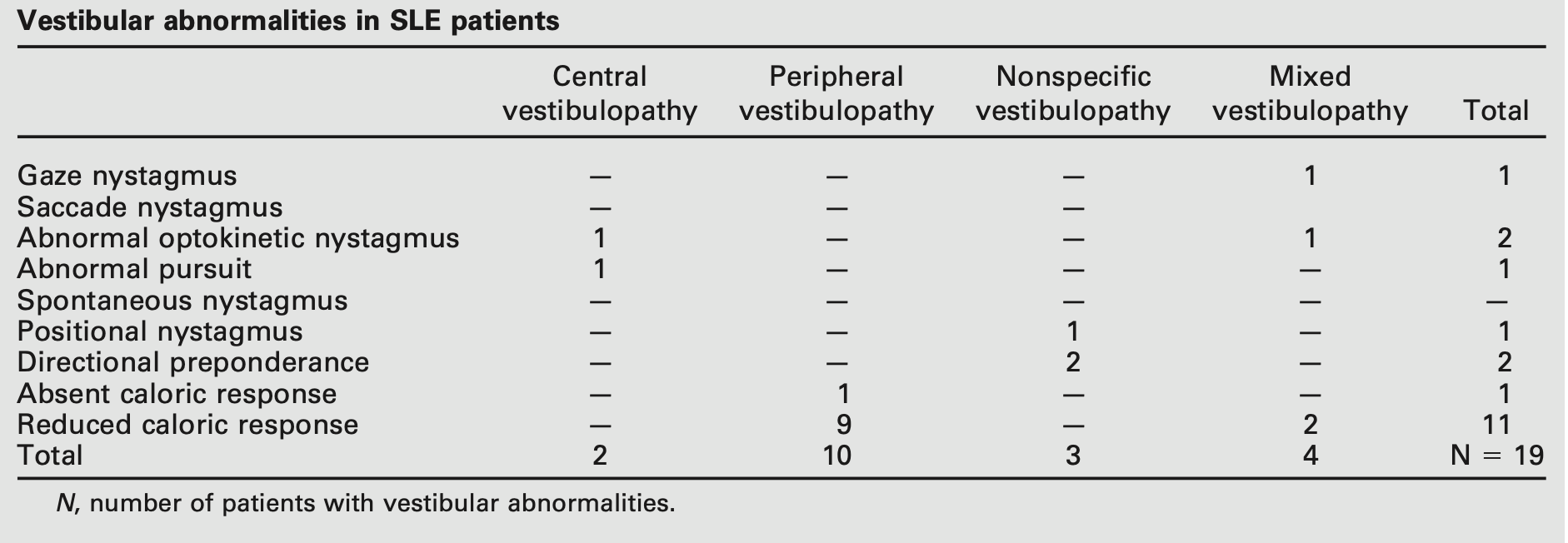
Karatas and colleagues (Karatas et al. 2007) studied electronystagmography in 28 SLE patients and reported abnormalities in 50% of them.
The Table below shows the specific electronystagmography abnormalities reported by Karatas and colleagues (Karatas et al. 2007).
Testing: other
Of the studies that compare audiologic findings with other clinical features or serum markers of disease activity, most find no clear correlation (Andonopoulos et al. 1995; Batuecas-Caletrio et al. 2013; Bowman et al. 1986). In contrast, Sperling and colleagues (Sperling et al. 1998) report some correlation with average creatinine and C3 complement levels.
Imaging
In SLE patients with ocular motor abnormalities, brain MRI may identify discrete brainstem lesions (Kunavarapu et al. 2001).
Differential diagnosis
Since SLE can affect essentially every organ system, its differential diagnosis is extraordinarily broad.
It is rare for an otoneurologist or neuro-otologist to be the first clinician to make a diagnosis of lupus. The more common scenario is that a patient who already carries a diagnosis of SLE is referred for a complaint of disequilibrium or hearing loss, and the usual clinical query is whether the symptom is attributable to the already established diagnosis of SLE, or whether it is due to independently occurring disease.
In this circumstance it is medically reasonable to undertake a screening otovestibular workup to identify whether another treatable condition (separate from SLE) is present. If no other disease is identified, then management of otovestibular symptoms will be management of the SLE itself.
Treatment
The management of SLE patients is usually undertaken by a rheumatologist.
In lupus patients with cerebellar ataxia, management often includes steroids (al-Arfaj and Naddaf 1995; Chattopadhyay et al. 2011; Iwasaki et al. 2012; Manto et al. 1996; Sy et al. 2021; Yaginuma et al. 2000), usually intravenous (Kristoff et al. 2022), less commonly oral (Ghosh et al. 2014). Other immunomodulators (Sy et al. 2021) such as azathioprine (al-Arfaj and Naddaf 1995) or mycophenolate mofetil (Ghosh et al. 2014), are sometimes considered.
Prognosis
Prognosis for hearing loss in SLE patients is poor, particularly when it presents acutely (Riera et al. 2020).
References
Abbasi M, Yazdi Z, Kazemifar AM, Bakhsh ZZ (2013) Hearing loss in patients with systemic lupus erythematosus. Glob J Health Sci 5: 102-6. doi: 10.5539/gjhs.v5n5p102
Ahmed HMA, El-Gohary R, Fayed F, El-Gendy H (2017) Cerebellar ataxia and obstructive hydrocephalus, rare neurologic presentations in patients with systemic lupus erythematosus. Rheumatol Int 37: 1917-1930. doi: 10.1007/s00296-017-3773-7
al-Arfaj HF, Naddaf HO (1995) Cerebellar atrophy in systemic lupus erythematosus. Lupus 4: 412-4. doi: 10.1177/096120339500400513
Andonopoulos AP, Naxakis S, Goumas P, Lygatsikas C (1995) Sensorineural hearing disorders in systemic lupus erythematosus. A controlled study. Clin Exp Rheumatol 13: 137-41.
Appenzeller S, Cendes F, Costallat LT (2008) Cerebellar ataxia in systemic lupus erythematosus. Lupus 17: 1122-6. doi: 10.1177/0961203308093071
Barber MRW, Drenkard C, Falasinnu T, Hoi A, Mak A, Kow NY, Svenungsson E, Peterson J, Clarke AE, Ramsey-Goldman R (2021) Global epidemiology of systemic lupus erythematosus. Nat Rev Rheumatol 17: 515-532. doi: 10.1038/s41584-021-00668-1
Batuecas-Caletrio A, del Pino-Montes J, Cordero-Civantos C, Calle-Cabanillas MI, Lopez-Escamez JA (2013) Hearing and vestibular disorders in patients with systemic lupus erythematosus. Lupus 22: 437-42. doi: 10.1177/0961203313477223
Bowman CA, Linthicum FH, Jr., Nelson RA, Mikami K, Quismorio F (1986) Sensorineural hearing loss associated with systemic lupus erythematosus. Otolaryngol Head Neck Surg 94: 197-204. doi: 10.1177/019459988609400212
Cakir A, Duzgun E, Demir S, Aydin A (2014) Jerky seesaw nystagmus with internuclear ophthalmoplegia as the presenting finding in systemic lupus erythematosus. J Fr Ophtalmol 37: e157-9. doi: 10.1016/j.jfo.2014.04.016
Caldarelli DD, Rejowski JE, Corey JP (1986) Sensorineural hearing loss in lupus erythematosus. Am J Otol 7: 210-3.
Casciato S, Mascia A, Quarato PP, D’Aniello A, Scoppetta C, Di Gennaro G (2018) Subacute cerebellar ataxia as presenting symptom of systemic lupus erythematosus. Eur Rev Med Pharmacol Sci 22: 7401-7403. doi: 10.26355/eurrev_201811_16279
Cha JM, Kim HS (2020) Functional Improvement after Taking Rehabilitation Program in Cerebellar Ataxia in a Patient with Systemic Lupus Erythematosus: a Case Report. Brain Neurorehabil 13: e11. doi: 10.12786/bn.2020.13.e11
Chattopadhyay P, Dhua D, Philips CA, Saha S (2011) Acute cerebellar ataxia in lupus. Lupus 20: 1312-5. doi: 10.1177/0961203311403346
Croker JA, Kimberly RP (2005) Genetics of susceptibility and severity in systemic lupus erythematosus. Curr Opin Rheumatol 17: 529-37. doi: 10.1097/01.bor.0000169360.15701.27
Danchenko N, Satia JA, Anthony MS (2006) Epidemiology of systemic lupus erythematosus: a comparison of worldwide disease burden. Lupus 15: 308-18. doi: 10.1191/0961203306lu2305xx
Di Stadio A, Ralli M (2017) Systemic Lupus Erythematosus and hearing disorders: Literature review and meta-analysis of clinical and temporal bone findings. J Int Med Res 45: 1470-1480. doi: 10.1177/0300060516688600
Felten R, Lipsker D, Sibilia J, Chasset F, Arnaud L (2022) The history of lupus throughout the ages. J Am Acad Dermatol 87: 1361-1369. doi: 10.1016/j.jaad.2020.04.150
Galindo M, Pablos JL, Gómez-Reino JJ (1998) Internuclear ophthalmoplegia in systemic lupus erythematosus. Semin Arthritis Rheum 28: 179-86. doi: 10.1016/s0049-0172(98)80034-6
Ghosh K, Chatterjee A, Ghosh S, Chakraborty S (2014) Cerebellar ataxia in a young patient: A rare path to lupus. J Neurosci Rural Pract 5: S75-6. doi: 10.4103/0976-3147.145212
Gomides AP, do Rosario EJ, Borges HM, Gomides HH, de Padua PM, Sampaio-Barros PD (2007) Sensorineural dysacusis in patients with systemic lupus erythematosus. Lupus 16: 987-90. doi: 10.1177/0961203307084160
Goulielmos GN, Zervou MI, Vazgiourakis VM, Ghodke-Puranik Y, Garyfallos A, Niewold TB (2018) The genetics and molecular pathogenesis of systemic lupus erythematosus (SLE) in populations of different ancestry. Gene 668: 59-72. doi: 10.1016/j.gene.2018.05.041
Hess EV, Farhey Y (1994) Epidemiology, genetics, etiology, and environment relationships of systemic lupus erythematosus. Curr Opin Rheumatol 6: 474-80. doi: 10.1097/00002281-199409000-00004
Hess EV, Farhey Y (1995) Etiology, environmental relationships, epidemiology, and genetics of systemic lupus erythematosus. Curr Opin Rheumatol 7: 371-5. doi: 10.1097/00002281-199509000-00002
Iwasaki Y, Okamoto A, Shoda H, Takahashi Y, Fujio K, Kawahata K, Yamamoto K (2012) Subacute cerebellar ataxia and atrophy developed in a young woman with systemic lupus erythematosus whose cerebrospinal fluid was positive for antineuronal cell antibody. Lupus 21: 324-8. doi: 10.1177/0961203311418270
Izmirly PM, Ferucci ED, Somers EC, Wang L, Lim SS, Drenkard C, Dall’Era M, McCune WJ, Gordon C, Helmick C, Parton H (2021a) Incidence rates of systemic lupus erythematosus in the USA: estimates from a meta-analysis of the Centers for Disease Control and Prevention national lupus registries. Lupus Sci Med 8. doi: 10.1136/lupus-2021-000614
Izmirly PM, Parton H, Wang L, McCune WJ, Lim SS, Drenkard C, Ferucci ED, Dall’Era M, Gordon C, Helmick CG, Somers EC (2021b) Prevalence of Systemic Lupus Erythematosus in the United States: Estimates From a Meta-Analysis of the Centers for Disease Control and Prevention National Lupus Registries. Arthritis Rheumatol 73: 991-996. doi: 10.1002/art.41632
James JA (2014) Clinical perspectives on lupus genetics: advances and opportunities. Rheum Dis Clin North Am 40: 413-32, vii. doi: 10.1016/j.rdc.2014.04.002
Jimenez-Alonso J, Gutierrez-Cabello F, Castillo JL, Sabio JM, Hidalgo-Tenorio C, Leon L, Grupo Lupus Virgen de las N (2002) Ear involvement in systemic lupus erythematosus patients: a comparative study. J Laryngol Otol 116:103-7. J Laryngol Otol 116: 746. doi: 10.1258/002221502760238127
Karatas E, Onat AM, Durucu C, Baglam T, Kanlikama M, Altunoren O, Buyukhatipoglu H (2007) Audiovestibular disturbance in patients with systemic lupus erythematosus. Otolaryngol Head Neck Surg 136: 82-6. doi: 10.1016/j.otohns.2006.06.1255
Kastanioudakis I, Ziavra N, Voulgari PV, Exarchakos G, Skevas A, Drosos AA (2002) Ear involvement in systemic lupus erythematosus patients: a comparative study. J Laryngol Otol 116: 103-7. doi: 10.1258/0022215021910032
Khalidi NA, Rebello R, Robertson DD (2008) Sensorineural hearing loss in systemic lupus erythematosus: case report and literature review. J Laryngol Otol 122: 1371-6. doi: 10.1017/S0022215108001783
Kristoff TJ, Shoskes A, Claytor B (2022) Lupus Cerebellitis Presenting With Ataxia: A Case Report. Neurohospitalist 12: 117-120. doi: 10.1177/19418744211021221
Kunavarapu C, Kesavan RB, Pevil-Ulysee M, Mohan SS (2001) Systemic lupus erythematosus presenting as “one-and-a-half syndrome”. J Rheumatol 28: 874-5.
Kutlubaev MA, Idrisova RF, Zakirova EN, Hardy TA (2020) Cerebellar ataxia as a first manifestation of systemic lupus erythematosus. Acta Neurol Belg 120: 1241-1243. doi: 10.1007/s13760-020-01376-5
Lewkonia RM (1992) The clinical genetics of lupus. Lupus 1: 55-62. doi: 10.1177/096120339200100202
Liao CH, Yang YH, Chiang BL (2003) Systemic lupus erythematosus with presentation as vertigo and vertical nystagmus: report of one case. Acta Paediatr Taiwan 44: 158-60.
Maciaszczyk K, Durko T, Waszczykowska E, Erkiert-Polguj A, Pajor A (2011) Auditory function in patients with systemic lupus erythematosus. Auris Nasus Larynx 38: 26-32. doi: 10.1016/j.anl.2010.04.008
Manto MU, Rondeaux P, Jacquy J, Hildebrand JG (1996) Subacute pancerebellar syndrome associated with systemic lupus erythematosus. Clin Neurol Neurosurg 98: 157-60. doi: 10.1016/0303-8467(96)00013-3
Marion TN, Postlethwaite AE (2014) Chance, genetics, and the heterogeneity of disease and pathogenesis in systemic lupus erythematosus. Semin Immunopathol 36: 495-517. doi: 10.1007/s00281-014-0440-x
Ota Y, Srinivasan A, Capizzano AA, Bapuraj JR, Kim J, Kurokawa R, Baba A, Moritani T (2022) Central Nervous System Systemic Lupus Erythematosus: Pathophysiologic, Clinical, and Imaging Features. Radiographics 42: 212-232. doi: 10.1148/rg.210045
Pons-Estel GJ, Ugarte-Gil MF, Alarcon GS (2017) Epidemiology of systemic lupus erythematosus. Expert Rev Clin Immunol 13: 799-814. doi: 10.1080/1744666X.2017.1327352
Rees F, Doherty M, Grainge MJ, Lanyon P, Zhang W (2017) The worldwide incidence and prevalence of systemic lupus erythematosus: a systematic review of epidemiological studies. Rheumatology (Oxford) 56: 1945-1961. doi: 10.1093/rheumatology/kex260
Relle M, Weinmann-Menke J, Scorletti E, Cavagna L, Schwarting A (2015) Genetics and novel aspects of therapies in systemic lupus erythematosus. Autoimmun Rev 14: 1005-18. doi: 10.1016/j.autrev.2015.07.003
Riera JL, Del RMM, Musuruana JL, Cavallasca JA (2020) Sudden Sensorineural Hearing Loss in Systemic Lupus Erythematosus and Antiphospholipid Syndrome: A Clinical Review. Curr Rheumatol Rev 16: 84-91. doi: 10.2174/1573397115666191016101741
Rivas-Larrauri F, Yamazaki-Nakashimada MA (2016) Systemic lupus erythematosus: Is it one disease? Reumatol Clin 12: 274-81. doi: 10.1016/j.reuma.2016.01.005
Sestak AL, Fürnrohr BG, Harley JB, Merrill JT, Namjou B (2011) The genetics of systemic lupus erythematosus and implications for targeted therapy. Ann Rheum Dis 70 Suppl 1: i37-43. doi: 10.1136/ard.2010.138057
Sestak AL, Nath SK, Sawalha AH, Harley JB (2007) Current status of lupus genetics. Arthritis Res Ther 9: 210. doi: 10.1186/ar2176
Shen N, Tsao BP (2004) Current advances in the human lupus genetics. Curr Rheumatol Rep 6: 391-8. doi: 10.1007/s11926-004-0014-3
Singh RR, Prasad K, Kumar A, Misra A, Padmakumar K, Malaviya AN (1988) Cerebellar ataxia in systemic lupus erythematosus: three case reports. Ann Rheum Dis 47: 954-6. doi: 10.1136/ard.47.11.954
Sperling NM, Tehrani K, Liebling A, Ginzler E (1998) Aural symptoms and hearing loss in patients with lupus. Otolaryngol Head Neck Surg 118: 762-5. doi: 10.1016/S0194-5998(98)70265-7
Sy MCC, Reyes NGD, Zamora GT, Fernandez MLL (2021) Cerebellar ataxia as a primary manifestation of neuropsychiatric systemic lupus erythematosus. BMJ Case Rep 14. doi: 10.1136/bcr-2020-236825
Tian J, Zhang D, Yao X, Huang Y, Lu Q (2023) Global epidemiology of systemic lupus erythematosus: a comprehensive systematic analysis and modelling study. Ann Rheum Dis 82: 351-356. doi: 10.1136/ard-2022-223035
Yaginuma M, Suenaga M, Shiono Y, Sakamoto M (2000) Acute cerebellar ataxia of a patient with SLE. Clin Neurol Neurosurg 102: 37-9. doi: 10.1016/s0303-8467(99)00078-5
Yim YS, Wakeland EK (2001) The genetics of lupus. Curr Opin Nephrol Hypertens 10: 437-43. doi: 10.1097/00041552-200105000-00022
![]()