By Marcello Cherchi, MD PhD
For patients
In simple terms, the phrase “multifactorial disequilibrium” refers to a situation in which there are multiple medical problems causing disequilibrium. In some cases it may be fairly clear at the outset that multiple medical problems are responsible for this symptom. In other cases it may not be so obvious, and consultation with several medical specialists may be required. Your doctor may consider checking several tests of balance function, and/or referring you to one or more specialists. The specific treatment(s) will depend on which diseases are discovered to be contributing to the symptom of disequilibrium.
For clinicians
Overview
The phrase “multifactorial disequilibrium” (or “multifactorial dizziness”) refers to the symptom of disequilibrium to which multiple etiologies are contributory. Humans maintain equilibrium through a system that involves multiple inputs (vestibular, visual, proprioceptive), multiple processing steps, and multiple outputs (ocular motor, somatomotor). This “many-to-many” arrangement has both a disadvantage (multiple vulnerabilities) and an advantage (redundancy). Redundancy can compensate for vulnerability up to a point, but beyond that threshold, compensation is no longer adequate, and the symptom of disequilibrium may become manifest. The clinical suspicion for multifactorial disequilibrium sometimes requires corroboration from consultation with several subspecialties. However, an otoneurologist or neuro-otologist should still evaluate these patients for discrete vestibular problems (such as benign paroxysmal positional vertigo, vestibular neuritis, etc.). Treatment and prognosis of multifactorial disequilibrium depends on which underlying pathologies are contributory. Vestibular rehabilitation therapy may be helpful.
Introduction
The phrase “multifactorial disequilibrium” (or “multifactorial dizziness”) refers to the symptom of disequilibrium to which multiple etiologies are contributory. This idea has acquired an unfavorable reputation in neurology because it is sometimes viewed as a refuge for when the diagnosis is unknown.
We agree that the diagnosis of multifactorial disequilibrium should not be a substitute for a reasonable attempt at using “Occam’s razor” (the principle of parsimony) to seek a single underlying cause of this symptom. However, this heuristic (the principle of parsimony) is only that: it is a reasonable starting assumption, but it is not always correct. The reality is that clinicians do encounter cases of “one-symptom-from-many-diagnoses,” and medical diagnostic thinking must accommodate this reality.
What is the “multi” in multifactorial disequilibrium?
In order to clarify the idea of multifactorial disequilibrium it is helpful to take a high-level view of the organization by which the vestibular system maintains equilibrium. Neurological training encourages a “circuitry” conception of neurophysiology. We offer the Figure below as an illustration of this.

Many aspects of this Figure will probably be familiar to trained physicians, while others may be less well known. We will highlight several points.
Overall: Many-to-many arrangement of systems
This is a system in which there are multiple sensory inputs, multiple motor outputs, and multiple processing steps that get from the inputs to the outputs. In other words, this is a many-to-many arrangement of systems.
Input: don’t forget auditory input
For didactic purposes textbooks usually state that the vestibular system takes into account three sensory modalities as its inputs: (1) inner ear vestibular function; (2) vision; (3) the proprioceptive component of somatic sensation. These three are emphasized because they do, indeed, constitute the main inputs.
However, that view is also an oversimplification, because the brain takes into account other inputs as well, such as hearing. While not as well developed as in bats, humans actually do have a limited echolocation ability (Flanagin et al. 2017; Schornich et al. 2012; Schornich et al. 2013; Thaler and Goodale 2016; Tirado et al. 2021; Wallmeier et al. 2013; Wallmeier et al. 2015). Auditory input enhances a person’s spatial awareness and perception of movement (Campos et al. 2018). Hearing loss is correlated with worsening of balance; conversely, hearing augmentation with hearing aids (Negahban et al. 2017; Rumalla et al. 2015) or cochlear implants (Mazaheryazdi et al. 2017; Shayman et al. 2017; Weaver et al. 2017) can improve a person’s balance — though it remains unclear whether hearing amplification influences the clinically meaningful outcome of falls.
Processing input: sensory integration
The brain does not only collect raw data from the various sensory input modalities. These inputs need to be interpreted and processed, and that processing may involve up-weighting or down-weighting a particular input, prioritizing the relevance of the inputs, arriving at an understanding of the environment (see “spatial awareness,” below) and eventually combining those processed inputs into a coherent picture of a person’s orientation in, and movement through, space. This interpretation, processing, and forming a coherent picture of one’s environment, position and movement is referred to as sensory integration (Peterka 2002; Peterka and Loughlin 2004). Sensory integration is probably also an important mechanism by which the brain “works around” sensory deficits (more on this below).
Processing input: spatial awareness
Spatial awareness (Ishikawa 2021) has been an underappreciated aspect of the way a person develops and maintains a mental “map” of an environment. While this may be simply one component of a broader set of cognitive capacities, it likely influences a person’s ability to negotiate an environment, and in this sense is relevant to “vestibular” function.
Orchestration of output: internal modeling
At a high but non-conscious level, many of our motor functions — including the motor activities involved in maintenance of balance and ambulation — require us to make predictions about how our actions affect the world around us, and to “test” those predictions in real time. Computational biologists refer to this predictive task as internal modeling. This idea was originally proposed by Francis and Wonham (Francis and Wonham 1976), and further developed by researchers such as Wolpert and colleagues (Wolpert and Ghahramani 2000; Wolpert et al. 1995).
An accessible example of internal modeling comes from what happens when you want to take a bottle of milk out of the refrigerator. If you see that the bottle of milk is full, you make a prediction that you will need to exert a certain amount of force to lift it up. If you believe that the milk bottle is empty when it is actually full, then your prediction of required force will be mistaken; the amount of force that you exert to lift the bottle will be greater than what is needed, and you will bump the milk bottle against the top of the refrigerator. The Figure below, from Wolpert and Ghahramani (Wolpert and Ghahramani 2000), illustrates this concept.
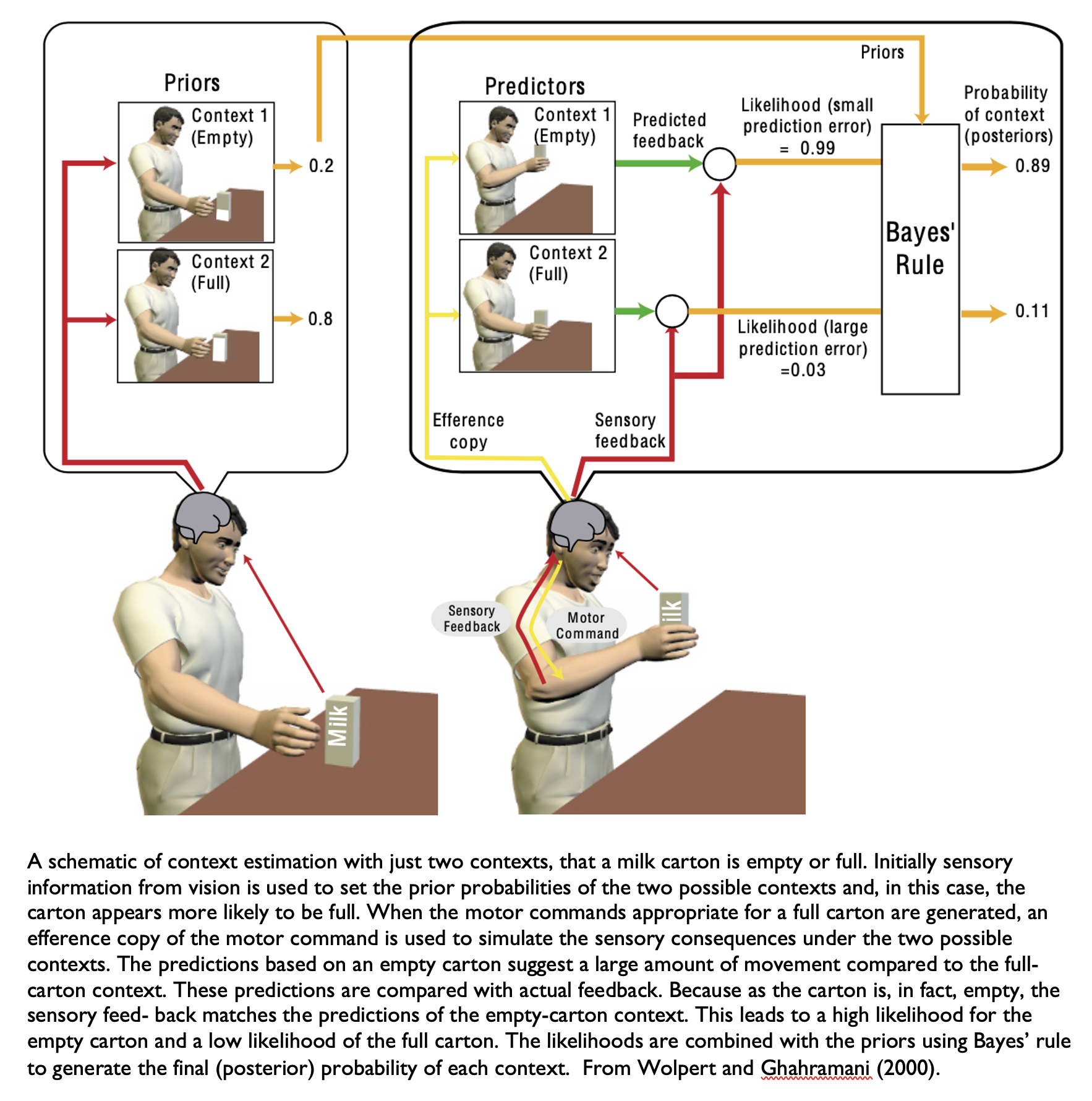
The ability to develop internal models is hard-wired, though the specific models themselves are built on data accumulated from real-world experience.
The underlying neuroanatomy of internal modeling is complex, and just beginning to be studied. The Figure below, from Scott (Scott 2012), illustrates one proposal.
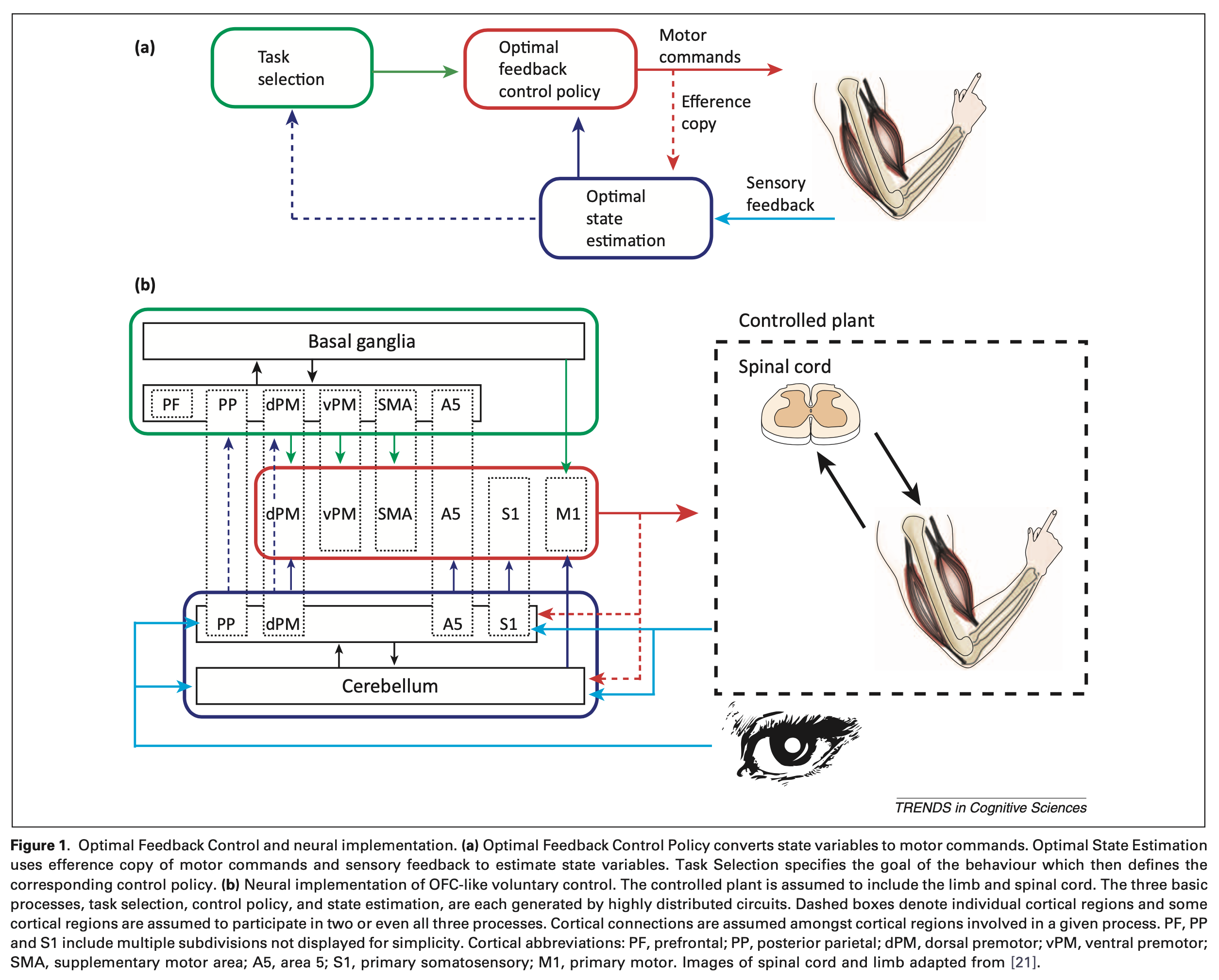
Consequences of the many-to-many arrangement
We return to the “many-to-many” arrangement of this system, meaning that there are multiple sensory inputs and multiple motor outputs. This characteristic has advantages and drawbacks.
An obvious drawback to this system is that there are many components, each of which is susceptible to a variety of pathology. In other words, the fact that there are multiple components broadens its vulnerability.
In contrast, the multiplicity (of inputs and outputs) means that there is redundancy, which confers resilience against damage to any one modality. There is evidence that when confronted with diminished or unreliable input in one sensory modality, the brain may increase reliance on other modalities — a process sometimes referred to as “sensory reweighting” (Asslander and Peterka 2014; Carver et al. 2006) or “sensory substitution” (Sadeghi et al. 2012). For example, there is evidence that patients with vestibular loss begin to upweight their reliance on auditory cues for spatial awareness (Vitkovic et al. 2016).
Returning to the “multi” in multifactorial disequilibrium
The property of redundancy (in both sensory input and motor output) can partially offset the inherent vulnerability of this system — up to a certain point.
This is the point at which multisensory disequilibrium becomes relevant. While redundancy, sensory reweighting/substitution, and other strategies can compensate for sensory and motor deficits to a point, beyond that threshold such compensation is no longer sufficient for adequate maintenance of equilibrium.
The relevance of multifactorial disequilibrium should now be clearer. A particular medical problem may interfere with only one component of the many-to-many arrangement described earlier, and the problem may be modest enough that, in isolation, the deficit that it imposes does not manifest as a clinically meaningful functional limitation that reaches a patient’s awareness in the form of a disturbance of equilibrium.
But a human can be afflicted with multiple medical problems, of which each may interfere with a different component of the many-to-many arrangement. While each individual deficit by itself may not be sufficient to provoke disequilibrium, in aggregate, these deficits may start to interfere with the patient’s equilibrium. Clinicians can envision plenty of circumstances in which this may occur.
An easy and common example would be poorly controlled diabetes mellitus. Diabetes can eventually lead to diabetic retinopathy, though this visual deficit in isolation may not cause disequilibrium. Diabetes can also lead to diabetic peripheral neuropathy, though this somatosensory deficit in isolation may not cause disequilibrium. Diabetes can also lead to diabetic autonomic neuropathy and problems such as orthostatic intolerance, though in isolation this may not cause disequilibrium. However, if all three deficits are present simultaneously, then in aggregate they may cause disequilibrium. This is a circumstance in which one underlying disease (diabetes) causes multiple problems (retinopathy, peripheral neuropathy, autonomic neuropathy), each of which can contribute to disequilibrium.
There are also many examples of independently occurring diseases ultimately leading to multifactorial disequilibrium. This is more likely to happen with common diseases, simply because there is a higher likelihood that these will overlap (co-occur) in a single person. For example, a person who performs heavy labor outdoors may be susceptible to developing cataracts and arthritis. This person may also indulge in alcohol too frequently. In this scenario there may be a visual deficit (from cataracts), a deficit in orchestration of output (alcoholic cerebellar degeneration) and a mechanical deficit (arthritis involving the spine, hips, knees and ankles). Any one of these deficits in isolation may not have caused sufficient functional impairment for the person to seek care, but in aggregate they may cause disequilibrium that brings the person to medical attention.
Clinical presentation
One can imagine innumerable combinations and permutations of deficits in the “circuitry” of equilibrium described above, so there is no single presentation characteristic of multifactorial disequilibrium.
Physical examination and ocular motor examination
Findings on examination will depend on which pathologies are contributing to multifactorial disequilibrium.
Testing: vestibular
Undertaking a screening otovestibular workup is nearly always reasonable for patients in whom multifactorial disequilibrium is suspected. Identification of an objective vestibular deficit can corroborate the suspicion for at least that component of the problem.
Testing: other
Although patients with suspected multifactorial disequilibrium may initially be referred to otoneurology, the “multi” characteristic of this problem means that other specialists (and perhaps specialized testing) will be required, depending on the clinical scenario. For example, it may be appropriate to evaluate other sensory modalities, such as assessment of vision (consultation with ophthalmology) or somatosensory input (consultation with a specialist in neuropathy for possible electromyography and nerve conduction velocities). It may also be appropriate to evaluate motor output (consultation with neuromuscular neurology), check for basal ganglionic problems (consultation with a movement disorders specialist), or evaluate for mechanical limitations (consultation with an orthopedist or rheumatologist).
Differential diagnosis
We have been repeating phrases such as, “each individual deficit in isolation may not cause disequilibrium, but in aggregate, multiple deficits can.” In other words, we have been assuming that in patients with multifactorial disequilibrium, each individual deficit is modest.
However, one should bear in mind that even patients who truly have multifactorial disequilibrium are still vulnerable to more pronounced individual pathologies, and it is important to evaluate for these. For example, if the diabetic discussed earlier develops benign paroxysmal positional vertigo, or if the laborer discussed earlier develops vestibular neuritis, then these should be recognized and treated.
Treatment
Treatment of multifactorial disequilibrium will depend almost entirely on the pathologies identified.
If multiple deficits are found, but each is relatively modest, one should still consider a trial of vestibular rehabilitation therapy. Vestibular physical therapists are very skilled at training patients in strategies of sensory substitution, and diminishing fall risk. Occupational therapy consultation may also be appropriate.
Prognosis
The prognosis of multifactorial disequilibrium depends on the specific pathologies identified.
References
Asslander L, Peterka RJ (2014) Sensory reweighting dynamics in human postural control. J Neurophysiol 111: 1852-64. doi: 10.1152/jn.00669.2013
Campos J, Ramkhalawansingh R, Pichora-Fuller MK (2018) Hearing, self-motion perception, mobility, and aging. Hear Res 369: 42-55. doi: 10.1016/j.heares.2018.03.025
Carver S, Kiemel T, Jeka JJ (2006) Modeling the dynamics of sensory reweighting. Biol Cybern 95: 123-34. doi: 10.1007/s00422-006-0069-5
Flanagin VL, Schornich S, Schranner M, Hummel N, Wallmeier L, Wahlberg M, Stephan T, Wiegrebe L (2017) Human Exploration of Enclosed Spaces through Echolocation. J Neurosci 37: 1614-1627. doi: 10.1523/JNEUROSCI.1566-12.2016
Francis BA, Wonham WM (1976) The internal model principle of control theory. Automatica 12: 457-465. doi: 10.1016/0005-1098(76)90006-6
Ishikawa T (2021) Spatial thinking, cognitive mapping, and spatial awareness. Cogn Process 22: 89-96. doi: 10.1007/s10339-021-01046-1
Mazaheryazdi M, Moossavi A, Sarrafzadah J, Talebian S, Jalaie S (2017) Study of the effects of hearing on static and dynamic postural function in children using cochlear implants. Int J Pediatr Otorhinolaryngol 100: 18-22. doi: 10.1016/j.ijporl.2017.06.002
Negahban H, Bavarsad Cheshmeh Ali M, Nassadj G (2017) Effect of hearing aids on static balance function in elderly with hearing loss. Gait Posture 58: 126-129. doi: 10.1016/j.gaitpost.2017.07.112
Peterka RJ (2002) Sensorimotor integration in human postural control. J Neurophysiol 88: 1097-118. doi: 10.1152/jn.2002.88.3.1097
Peterka RJ, Loughlin PJ (2004) Dynamic regulation of sensorimotor integration in human postural control. J Neurophysiol 91: 410-23. doi: 10.1152/jn.00516.2003
Rumalla K, Karim AM, Hullar TE (2015) The effect of hearing aids on postural stability. Laryngoscope 125: 720-3. doi: 10.1002/lary.24974
Sadeghi SG, Minor LB, Cullen KE (2012) Neural correlates of sensory substitution in vestibular pathways following complete vestibular loss. J Neurosci 32: 14685-95. doi: 10.1523/JNEUROSCI.2493-12.2012
Schornich S, Nagy A, Wiegrebe L (2012) Discovering your inner bat: echo-acoustic target ranging in humans. J Assoc Res Otolaryngol 13: 673-82. doi: 10.1007/s10162-012-0338-z
Schornich S, Wallmeier L, Gessele N, Nagy A, Schranner M, Kish D, Wiegrebe L (2013) Psychophysics of human echolocation. Adv Exp Med Biol 787: 311-9. doi: 10.1007/978-1-4614-1590-9_35
Scott SH (2012) The computational and neural basis of voluntary motor control and planning. Trends Cogn Sci 16: 541-9. doi: 10.1016/j.tics.2012.09.008
Shayman CS, Earhart GM, Hullar TE (2017) Improvements in Gait With Hearing Aids and Cochlear Implants. Otol Neurotol 38: 484-486. doi: 10.1097/MAO.0000000000001360
Thaler L, Goodale MA (2016) Echolocation in humans: an overview. Wiley Interdiscip Rev Cogn Sci 7: 382-393. doi: 10.1002/wcs.1408
Tirado C, Gerdfeldter B, Karnekull SC, Nilsson ME (2021) Comparing Echo-Detection and Echo-Localization in Sighted Individuals. Perception 50: 308-327. doi: 10.1177/03010066211000617
Vitkovic J, Le C, Lee SL, Clark RA (2016) The Contribution of Hearing and Hearing Loss to Balance Control. Audiol Neurootol 21: 195-202. doi: 10.1159/000445100
Wallmeier L, Gessele N, Wiegrebe L (2013) Echolocation versus echo suppression in humans. Proc Biol Sci 280: 20131428. doi: 10.1098/rspb.2013.1428
Wallmeier L, Kish D, Wiegrebe L, Flanagin VL (2015) Aural localization of silent objects by active human biosonar: neural representations of virtual echo-acoustic space. Eur J Neurosci 41: 533-45. doi: 10.1111/ejn.12843
Weaver TS, Shayman CS, Hullar TE (2017) The Effect of Hearing Aids and Cochlear Implants on Balance During Gait. Otol Neurotol 38: 1327-1332. doi: 10.1097/MAO.0000000000001551
Wolpert DM, Ghahramani Z (2000) Computational principles of movement neuroscience. Nat Neurosci 3 Suppl: 1212-7. doi: 10.1038/81497
Wolpert DM, Ghahramani Z, Jordan MI (1995) An internal model for sensorimotor integration. Science 269: 1880-2. doi: 10.1126/science.7569931
![]()