By Marcello Cherchi, MD PhD
For patients
Some cancers of the head and neck are treated with radiation therapy, and this radiation can sometimes affect inner ear structures, resulting in symptoms such as hearing loss, tinnitus, and disequilibrium. In this circumstance the doctor may check tests of hearing and balance function, and depending on those results, the patient may be referred to an audiologist and/or a vestibular physical therapist.
For clinicians
Overview
The management of some malignancies of the head and neck may include radiation therapy. Even with good planning, the temporal bone, and thus structures relevant to audiovestibular function, often fall within the radiation field. Radiation induces both short-term effects (perhaps through the mechanism of vasculitis) and long-term effects (by damaging DNA and thus disrupting cellular function, and probably through ischemia due to radiation-caused or radiation-accelerated atherosclerosis). Auditory effects — usually manifesting as hearing loss — may become apparent within months, and tends to be progressive (beyond what would be expected from age alone). Vestibular symptoms may present after some delay, sometimes years later. Audiometry will generally show predominantly sensorineural hearing loss, typically greater in the higher frequencies. Vestibular studies may reveal reduced or absent caloric responses, and delayed or absent vestibular evoked myogenic potentials responses. The audiologic and vestibular deficits from radiation therapy are irreversible. Patients with auditory deficits should be referred to audiology regarding amplification (for hearing loss) and/or masking (for tinnitus). In patients with disequilibrium it is medically reasonable to attempt a course of vestibular physical therapy, particularly if workup reveals vestibular weakness.
Introduction
Treatment with ionizing radiation plays an important role in the management of certain neoplasms of the head and neck. In some instances radiation therapy is purposefully directed at neoplasms affecting audiovestibular structures, such as the treatment of a vestibular schwannoma with stereotactic radiosurgery (SRS) or fractionated stereotactic radiotherapy (FSRT). Neoplasms elsewhere in the head and neck may require fractionated radiotherapy (FRT) or intensity-modulated radiation therapy (IMRT) (Bhandare et al. 2009).
Radiation therapy probably works through multiple mechanisms.
“Deposition of radiation energy in tissue results in cell injury and death. Most of radiation’s tissue effect is thought to be a result of the damage to DNA. This occurs both directly and indirectly. The latter, the predominant mechanism, involves ionization of surrounding water molecules to form free radicals, which, in turn, result in double- strand breaks in DNA. This injury may result in cell death during mitosis, induction of apoptosis (i.e., programmed cell death), recovery, and cell cycle arrest or terminal differentiation through activation of repair pathways that may also play a role in tumor suppression (e.g., activation of p53). The radio-response of a tissue depends on the inherent sensitivity of the cells, the kinetics of cell population, total dose, dose per fraction, and time-dose fractionation. Cells with fast turnover rate or higher mitotic activity exhibit more sensitivity to radiation, subjecting to cell death in attempting subsequent mitosis. This is the basis for therapeutic RT (i.e., relatively greater damage to highly reproductive tumor cells). Further, RT fractionation offers the potential for greater differential sparing of normal tissues and killing of tumor cells” (Bhandare et al. 2009).
Radiation therapy is carefully planned to maximally target neoplastic tissue, while minimizing the effects on healthy tissues. However, even with optimal planning, “In treatment for malignancies of the brain, nasopharynx, paranasal sinuses, and several other sites in the head and neck, the temporal bone is included within the field receiving radiation” (Hoistad et al. 1998) and thus audiovestibular structures may still lie in the path of the delivered radiotherapy.
The Figure below, from Kong and colleagues (Kong et al. 2016), illustrates radiation isodose lines in planning carbon ion radiotherapy for a patient with locally recurrent nasopharyngeal carcinoma. Even with careful planning, even though the neoplasm in question is centimeters away from structures relevant to audiovestibular function, the temporal bone still receives some radiation exposure.
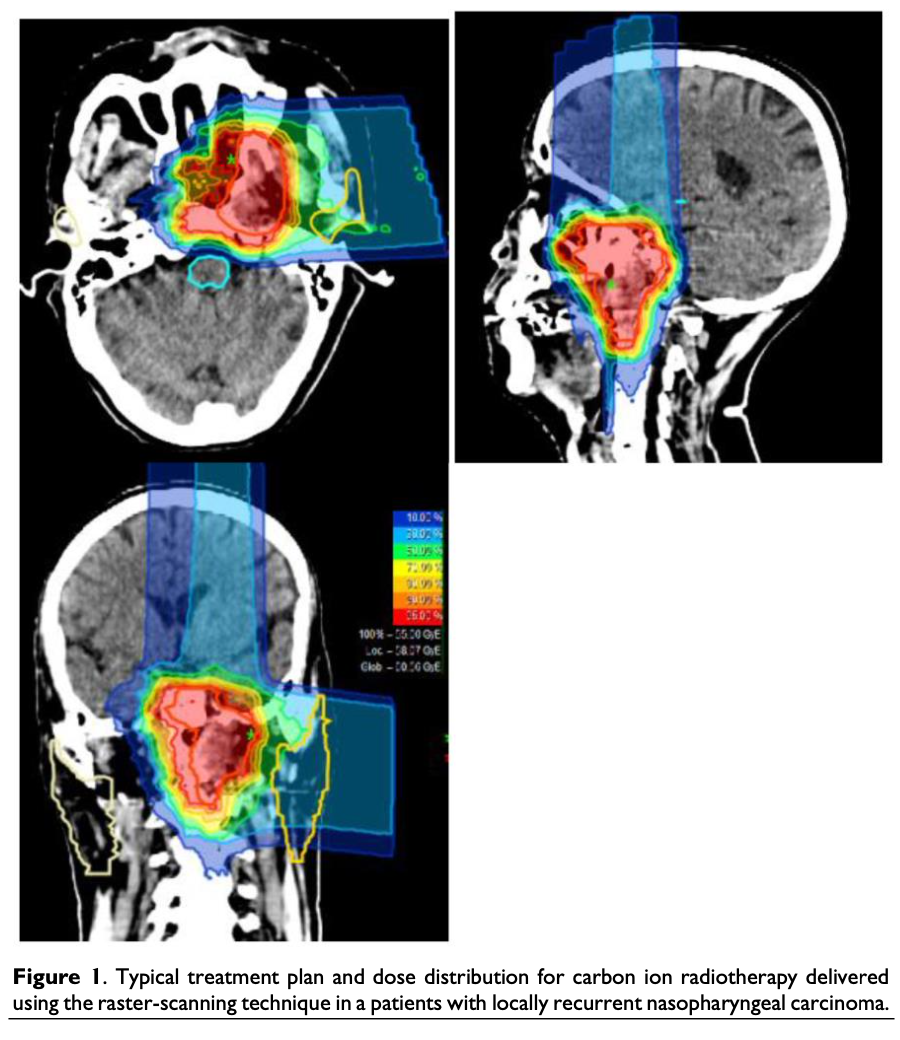
Studies have shown that, “The entire auditory-vestibular system is vulnerable to RT [radiotherapy] injury. Nearly half of all patients that have undergone RT for head and neck tumors demonstrate evidence of auditory or vestibular system pathology” (Bhandare et al. 2009).
Unlike some other aspects of oncologic care, there is no national-level consortium specifically tasked with the study of audiovestibular outcomes of cancer treatment. The LENT‑SOMA (“Late Effects Normal Tissue Task Force – Subjective, Objective, Management, Analytic scales”) framework was a proposal for tracking outcomes in multiple organ systems ([No authors] 1995), including the ear, but was never validated, and has been criticized as inadequately granular for many purposes. Consequently, the study of audiovestibular effects of cancer treatment have drawn on case reports and case series.
The audiologic effects of radiation therapy have been studied more thoroughly than the vestibular effects. As Frisch and colleagues state, “The scientific understanding of the effects of radiation on the vestibular apparatus is lacking compared to our understanding of its association with sensorineural hearing loss” (Frisch et al. 2018).
Epidemiology
Taking into account the caveats cited above, Bhandare and colleagues (Bhandare et al. 2009), citing data from Kwong and colleagues (Kwong et al. 1996), states that some degree of permanent hearing loss from radiation for treatment of head and neck cancers may be as high as 54%.
Many treatment regimens for head and neck cancers include platinum-based agents such as cisplatin, which has established ototoxicity. Most studies (Lambert et al. 2016), though not all (Kwong et al. 1996) conclude that the combination of radiation therapy and cisplatin has a synergistic effect with respect to adversely impacting hearing.
Bhandare and colleagues (Bhandare et al. 2009), citing data from Singh and Slevin (Singh and Slevin 1991), state that “vestibular dysfunction occurs in roughly 25% to 30% of patients treated with RT [radiation therapy] to the temporal bone.” Drawing on data from several other studies (Chao et al. 1998; Gabriele et al. 1992; Johannesen et al. 2002), Bhandare and colleagues state that, “Caloric weakness has been reported in 9% to 36%” of patients treated with radiation involving the temporal bone.
Pathophysiological mechanism of disease
The mechanism by which radiation disrupts labyrinthine function is incompletely understood, however:
“The unique nature of the inner ear cell lines leads to unique manifestations of RT injury… Hair cells and the stria vascularis have been implicated as the two major sites of inner ear radiation toxicity” (Bhandare et al. 2009).
Bhandare and colleagues (Bhandare et al. 2009), citing data from Leach (Leach 1965), comment that:
“Histologic studies have shown that the greatest damage to inner ear is the result of injury to the vessels of stria vascularis… An autopsy study after a high dose of radiation demonstrated absence of the organ of Corti, macula of the utricle and the cristae of the semicircular canals” (Bhandare et al. 2009).
Radiation can have different effects over time. It has been suggested that some of the short-term effects (typically within hours to days) of radiation may be attributable to vasculitis (Borsanyi and Blanchard 1962), while the longer-term effects are due to cellular dysfunction consequent to DNA damage from radiation.
Radiation is also known to provoke or accelerate atherosclerosis (Venkatesulu et al. 2018), and these effects may not become apparent for years. Most studies of this topic pertain to coronary artery disease and carotid artery disease. However, it is plausible that radiation has similar effects on smaller arteries, such as the labyrinthine artery which is the only blood supply to the inner ear. Thus, labyrinthine ischemia (secondary to radiation-caused or radiation-accelerated atherosclerosis) is an additional mechanism by which radiation may have long term effects on the audiologic and vestibular systems.
The longer-term effects of radiation are generally more difficult to study; as Gamble and colleagues comment, “The most obvious reason that we do not see many late sequelae of radiation of the ear is that most of these patients have a malignancy which causes their demise before enough time has elapsed for these changes to develop” (Gamble et al. 1968).
Gamble and colleagues conducted temporal bone studies of irradiated guinea pigs and humans, and generalized that, “the vestibular apparatus may be more resistant to the effects of radiation than the cochlea” (Gamble et al. 1968).
Clinical presentation: audiologic
Hearing loss has been reported to begin as early as 3 months after completing radiation therapy (Ho et al. 1999; Wang et al. 2004). Bhandare and colleagues (Bhandare et al. 2009), citing data of patients treated for nasopharyngeal carcinoma from Ho and colleagues (Ho et al. 1999) and Kwong and colleagues (Kwong et al. 1996), state that the latency from completion of radiation therapy to onset of hearing loss is 1.5 – 2 years. The risk of progressive hearing loss (attributable to radiation and not to age) likely persists to at least 3 – 4 years (Grau and Overgaard 1996), and observation of trends suggests that the trajectory of hearing loss likely extends further (Ho et al. 1999).
Clinical presentation: vestibular
Details are scanty regarding symptoms of disequilibrium in patients who have received radiation therapy in the head and neck. Often the descriptions are limited to the level of case reporting; for instance, Leach states, “Vestibular symptoms were noticed even sooner, usually within a month or two but delayed for two and a half years in one patient who then suffered severe loss of balance” (Leach 1965).
Young and colleagues (Young et al. 2004) studied 113 nasopharyngeal carcinoma (NPC) patients who had received radiation therapy and complained of post-irradiation “vertigo.” Of these patients, 67 were men and 46 were women; their mean age at onset of NPC was 46±8 years; the onset of “vertigo” occurred at a mean age of 56±8 years, with an interval ranging from 1 to 27 years (mean 10 years). Given this interval, they concluded that, “Vertigo is a late complication in irradiated NPC survivors.”
Testing: in general
The degrees of affectation of audiologic and vestibular function may be different from each other. Jonahessen and colleagues studied audiometry and caloric testing in 33 patients who received unilateral radiation for brain tumor and reported that, “There was no correlation between the degree of reduced caloric response and sensorineural hearing loss” (Johannesen et al. 2002).
Testing: auditory
Hearing loss from radiation tends to affect the high frequencies more than the low frequencies (Kwong et al. 1996). As we will discuss below, the hearing loss tends to be progressive.
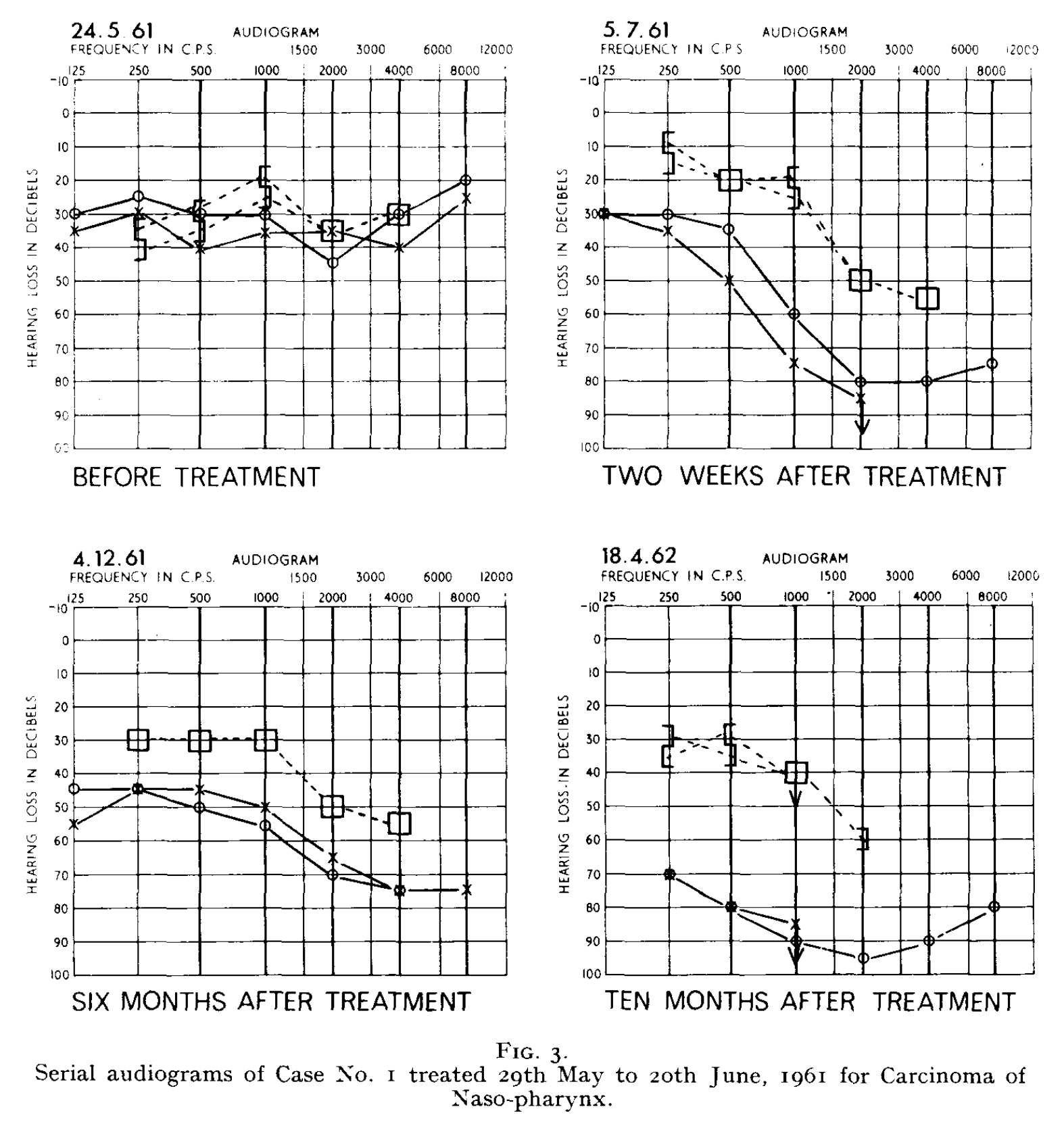
The Figure below, from Leach (Leach 1965), shows serial audiometry from prior to radiation therapy to 10 months after radiation therapy in a patient treated for nasopharyngeal carcinoma.
Testing: vestibular
Johanessen and colleagues studied 33 patients who received unilateral radiation for brain tumor and reported that on electronystagmography, “Three patients [9%] had significantly reduced response to caloric stimulation of the vestibular inner ear of the irradiated side” (Johannesen et al. 2002).
Young and colleagues (Young et al. 2004) studied 113 nasopharyngeal carcinoma (NPC) patients who had received radiation therapy and complained of post-irradiation “vertigo.” Of these patients, 67 were men and 46 were women; their mean age at onset of NPC was 46±8 years; the onset of “vertigo” occurred at a mean age of 56±8 years, with an interval ranging from 1 to 27 years (mean 10 years). They performed electronystagmography on these patients and reported that:
“Electronystagmography disclosed multidirection nystagmus in three patients and unidirection nystagmus in nine patients. The former consisted of three patients with radiation necrosis of the brain, whereas the latter included sudden deafness in five, intracranial relapse in one, and peripheral labyrinthine lesion in three. Abnormal eye tracking test was disclosed in 58 patients (51%) and abnormal optokinetic nystagmus test in 48 patients (42%). Excluding 5 patients who refused to perform caloric testing, 45 patients (42%) displayed absent ice-water caloric response in one or both ears, 39 patients (36%) had reduced caloric responses, and 24 patients (22%) had bilateral normal caloric responses” (Young et al. 2004).
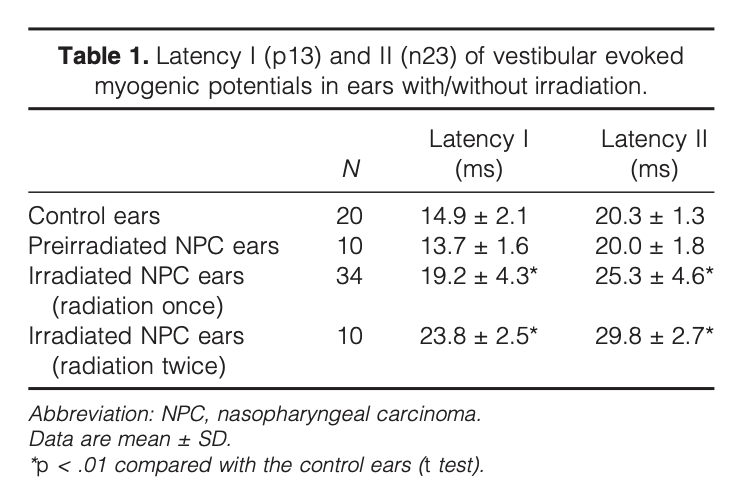
Wu and colleagues (Wu et al. 2003) compared cervical vestibular evoked myogenic potentials (VEMPs) in two groups of nasopharyngeal carcinoma (NPC) patients (one group had received radiation therapy once, the other group had received radiation therapy twice) and healthy control subjects. They reported abnormalities in cervical vestibular evoked myogenic potentials responses, ranging from delayed latencies (61%) to complete absence (5%). Interestingly, the delay in cervical vestibular evoked myogenic potentials increased after a second radiation treatment, as shown in the Table below. They speculated that, “Delayed VEMPs in NPC survivors are possibly due to radiation-induced otitis media, a brainstem lesion, or neck fibrosis” (Wu et al. 2003).
Radiation can cause otitis media, and otitis media can in turn cause conductive hearing loss, which could alter VEMP responses. This means that there may be different contributors to VEMP abnormalities in patients who had received radiation, some from middle ear dysfunction (from otitis media) and some from damage to the labyrinth, vestibular nerve or vestibular nuclei. In order to disentangle these effects, Yang and colleagues (Yang and Young 2004) compared cervical vestibular evoked myogenic potentials (VEMPs) in fourteen “radiation-induced otitis media (ROM)” nasopharyngeal carcinoma patients and 14 age- and gender-matched patients with chronic otitis media (“COM”) who did not receive radiation therapy and did not have nasopharyngeal carcinoma. They reported that, “Most (89%) COM ears revealed normal VEMPs, and most (61%) ROM ears demonstrated delayed VEMPs, indicating that ROM is different from COM because of the larger affected areas such as retrolabyrinthine or brainstem involvement.”
Histopathology
Treatment of various head and neck cancers involves not only radiation but also chemotherapy (such as cisplatin) which is known to have significant ototoxicity, and in studies of such cases it is difficult or impossible to distinguish audiovestibular effects of the two treatment modalities.
Very few temporal bone studies have been published regarding patients who have received radiation therapy alone (i.e., with no chemotherapy). Hoistad and colleagues (Hoistad et al. 1998) reported on 9 temporal bones from 5 patients who had received radiation therapy alone for various head and neck cancers. They reported:
“Histologic changes in the five patients (9 temporal bones) irradiated included one patient with a tympanic membrane thickened in the posterosuperior quadrant. Ossicles were normal except in one patient who had surgical changes caused by lateral resection of the temporal bone and mastoidectomy; this patient was missing all ossicles except the stapedial footplate. Another patient showed resorptive changes in the incus. Two bones demonstrated atrophy of the annular ligament but no obvious subluxation of the stapedial footplate. Mucosa of the middle ear was thickened in every irradiated temporal bone except one, which had been irradiated 9 to 10 months previously. One temporal bone demonstrated emptying of lacunae, fibrosis, and probable osteoradionecrosis near the internal carotid artery. Every irradiated mastoid cavity had fluid present and thickened mucosa. All of these temporal bones showed reactive compensatory fibrosis involving the mastoid cavity and near the facial nerve recess. In three of the five patients who had been irradiated, the round window membrane was thickened. We noted serous fluid in the utricle and saccule. The semicircular canals were normal. There was no endolymphatic or saccular hydrops. Atrophy of the stria vascularis and strial ligament was noted more commonly in the apical turns. All irradiated temporal bones showed decreased spiral ganglion cells (Fig. 3B). Outer hair cells were absent throughout all turns, and inner hair cells were present only sporadically in apical turns” (Hoistad et al. 1998).
Schuknecht and colleagues (Schuknecht and Karmody 1966) focused attention on the effect of radiation on the osseous structures in the temporal bone, but commented that effect on soft tissues of the inner ear include labyrinthitis.
Treatment
It is believed that the cell-death-related damage from radiation is permanent, so until regenerative therapies emerge, there is no direct treatment for the auditory and vestibular deficits arising from radiation therapy.
Patients with auditory deficits may consult with audiology regarding amplification (for hearing loss) and/or masking (for tinnitus).
If a patient has vestibular symptoms it is medically reasonable to attempt a course of vestibular physical therapy. This should be especially encouraged if workup reveals vestibular weakness.
Prognosis
In general, hearing loss following radiation therapy is progressive, and beyond what would be expected for age.
The Figures below, from Ho and colleagues (Ho et al. 1999), show the increasing proportion of ears with hearing loss up to 4.5 years after radiation therapy.
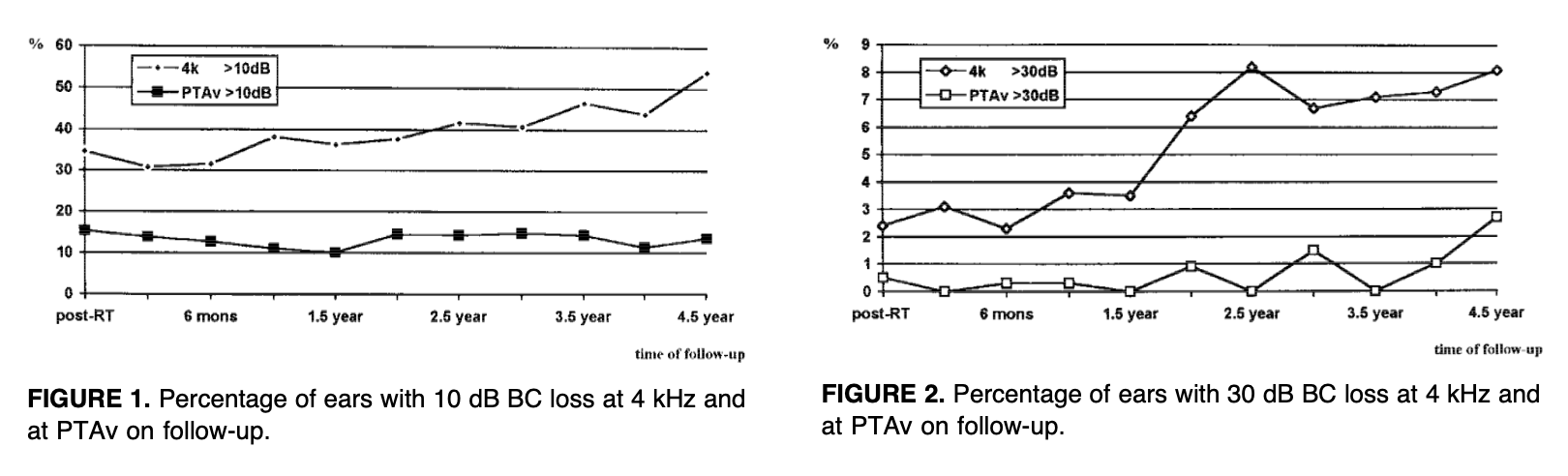
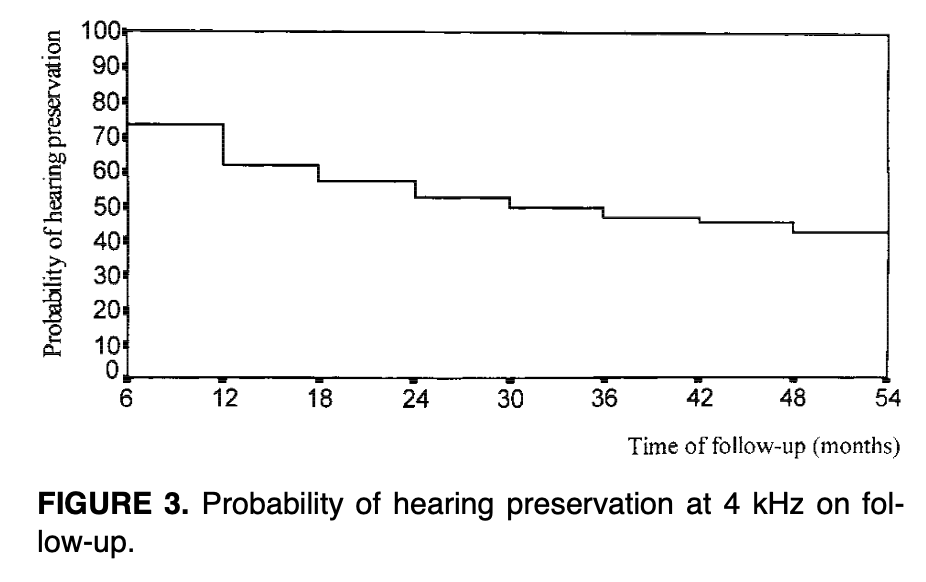
Conversely, the Figure below, from Ho and colleagues (Ho et al. 1999), shows what is effectively a Kaplan-Meier plot illustrating the progressively decreasing likelihood of hearing preservation at 4 kHz up to 54 months following radiation therapy.
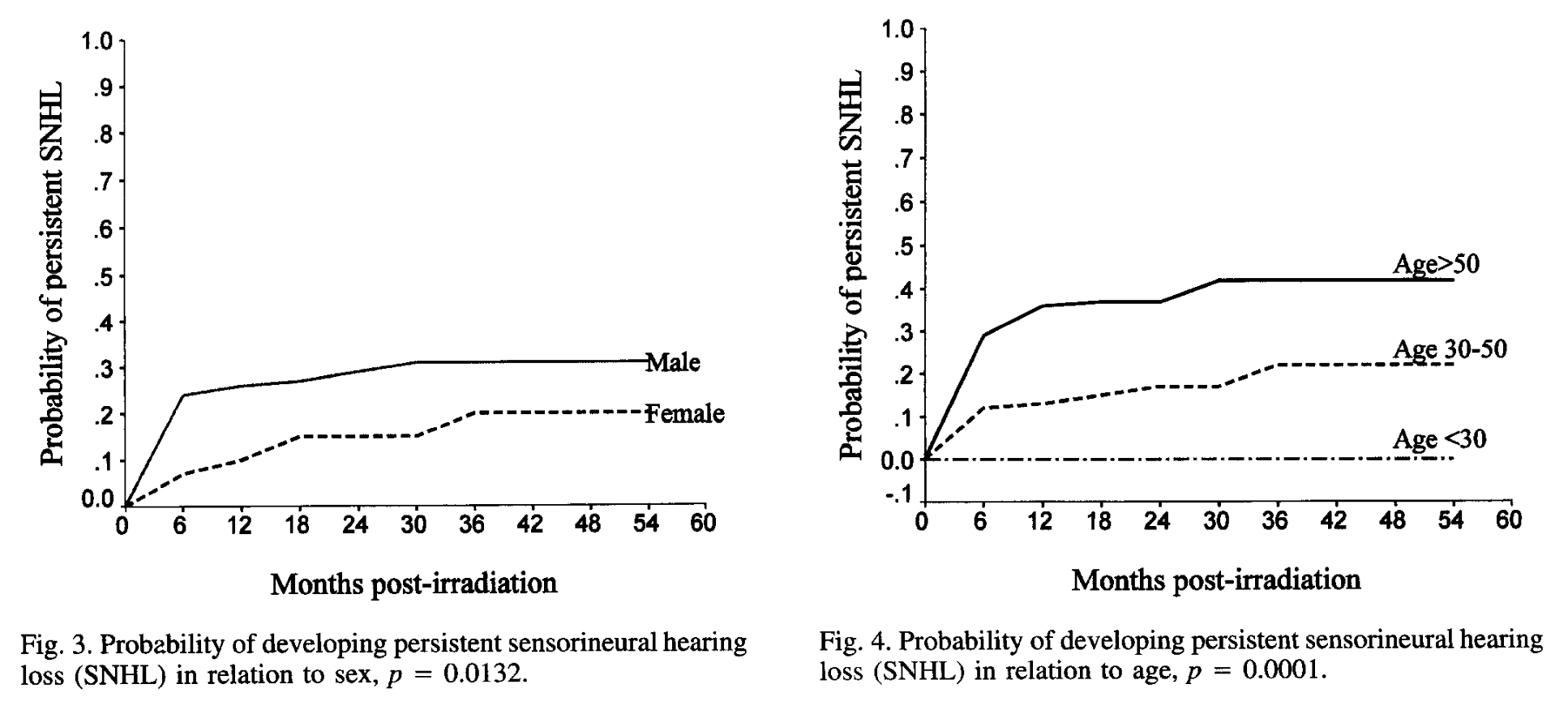
The Figures below, from Kwong and colleagues (Kwong et al. 1996), show that the likelihood of persistent sensorineural hearing loss increases with time after radiation therapy, is higher in males, and is worse in higher age ranges.
It is unknown whether vestibular deficits from radiation therapy are progressive.
References
[No authors] (1995) LENT SOMA tables. Radiother Oncol 35: 17-60.
Bhandare N, Mendenhall WM, Antonelli PJ (2009) Radiation effects on the auditory and vestibular systems. Otolaryngol Clin North Am 42: 623-34. doi: 10.1016/j.otc.2009.04.002
Borsanyi SJ, Blanchard CL (1962) Ionizing radiation and the ear. JAMA 181: 958-61. doi: 10.1001/jama.1962.03050370026006
Chao WY, Tseng HZ, Tsai ST (1998) Caloric response and postural control in patients with nasopharyngeal carcinoma after radiotherapy. Clin Otolaryngol Allied Sci 23: 439-41.
Frisch CD, Beadle BM, Gidley PW (2018) Radiation Effects on the Temporal Bone. In: Gidley PW, DeMonte F (eds) Temporal Bone Cancer. Springer International Publishing, Cham, pp 379-402
Gabriele P, Orecchia R, Magnano M, Albera R, Sannazzari GL (1992) Vestibular apparatus disorders after external radiation therapy for head and neck cancers. Radiother Oncol 25: 25-30.
Gamble JE, Peterson EA, Chandler JR (1968) Radiation effects on the inner ear. Arch Otolaryngol 88: 156-61. doi: 10.1001/archotol.1968.00770010158008
Grau C, Overgaard J (1996) Postirradiation sensorineural hearing loss: a common but ignored late radiation complication. Int J Radiat Oncol Biol Phys 36: 515-7. doi: 10.1016/s0360-3016(96)00346-x
Ho WK, Wei WI, Kwong DL, Sham JS, Tai PT, Yuen AP, Au DK (1999) Long-term sensorineural hearing deficit following radiotherapy in patients suffering from nasopharyngeal carcinoma: A prospective study. Head Neck 21: 547-53. doi: 10.1002/(sici)1097-0347(199909)21:6<547::aid-hed8>3.0.co;2-y
Hoistad DL, Ondrey FG, Mutlu C, Schachern PA, Paparella MM, Adams GL (1998) Histopathology of human temporal bone after cis-platinum, radiation, or both. Otolaryngol Head Neck Surg 118: 825-32. doi: 10.1016/S0194-5998(98)70276-1
Johannesen TB, Rasmussen K, Winther FO, Halvorsen U, Lote K (2002) Late radiation effects on hearing, vestibular function, and taste in brain tumor patients. Int J Radiat Oncol Biol Phys 53: 86-90.
Kong L, Hu J, Guan X, Gao J, Lu R, Lu JJ (2016) Phase I/II Trial Evaluating Carbon Ion Radiotherapy for Salvaging Treatment of Locally Recurrent Nasopharyngeal Carcinoma. J Cancer 7: 774-83. doi: 10.7150/jca.14399
Kwong DL, Wei WI, Sham JS, Ho WK, Yuen PW, Chua DT, Au DK, Wu PM, Choy DT (1996) Sensorineural hearing loss in patients treated for nasopharyngeal carcinoma: a prospective study of the effect of radiation and cisplatin treatment. Int J Radiat Oncol Biol Phys 36: 281-9. doi: 10.1016/s0360-3016(96)00302-1
Lambert EM, Gunn GB, Gidley PW (2016) Effects of radiation on the temporal bone in patients with head and neck cancer. Head Neck 38: 1428-35. doi: 10.1002/hed.24267
Leach W (1965) Irradiation of the ear. J Laryngol Otol 79: 870-80. doi: 10.1017/s0022215100064495
Schuknecht HF, Karmody CS (1966) Radionecrosis of the temporal bone. Laryngoscope 76: 1416-28. doi: 10.1288/00005537-196608000-00010
Singh IP, Slevin NJ (1991) Late audiovestibular consequences of radical radiotherapy to the parotid. Clin Oncol (R Coll Radiol) 3: 217-9.
Venkatesulu BP, Mahadevan LS, Aliru ML, Yang X, Bodd MH, Singh PK, Yusuf SW, Abe JI, Krishnan S (2018) Radiation-Induced Endothelial Vascular Injury: A Review of Possible Mechanisms. JACC Basic Transl Sci 3: 563-572. doi: 10.1016/j.jacbts.2018.01.014
Wang LF, Kuo WR, Ho KY, Lee KW, Lin CS (2004) A long-term study on hearing status in patients with nasopharyngeal carcinoma after radiotherapy. Otol Neurotol 25: 168-73. doi: 10.1097/00129492-200403000-00015
Wu CC, Young YH, Ko JY (2003) Effect of irradiation on vestibular evoked myogenic potentials in nasopharyngeal carcinoma survivors. Head Neck 25: 482-487. doi: PMID: 12784240
Yang TL, Young YH (2004) Radiation-induced otitis media—study of a new test, vestibular-evoked myogenic potential. Int J Radiat Oncol Biol Phys 60: 295-301. doi: PMID: 15337568
Young YH, Ko JY, Sheen TS (2004) Postirradiation vertigo in nasopharyngeal carcinoma survivors. Otol Neurotol 25: 366-70.
![]()