By Marcello Cherchi, MD PhD
For patients
Sarcoidosis is an immune disease that can affect different parts of the body, such as the lungs, skin, liver, and other organs, and is usually managed by a doctor specializing in the affected organ system (such as a lung doctor, skin doctor, etc.). When sarcoid affects the brain, spinal cord or nerves, it is called neurosarcoidosis. Some patients with neurosarcoidosis have problems with hearing and balance. If your doctor suspects neurosarcoidosis, then they may check tests or hearing and/or balance, or may suggest that you get an MRI of the brain.
For clinicians
Overview
Sarcoidosis is an idiopathic immune disorder in which caseating granulomas appear in various organ systems. The disease is rare, and only about 5% of cases involve the central nervous system (neurosarcoidosis). The etiology is unknown. Of patients with neurosarcoidosis, almost one third of patients have auditory and/or vestibular symptoms. The auditory manifestations include hearing loss and tinnitus; the hearing loss may be bilateral (usually asymmetrical) or unilateral, and is predominantly sensorineural; onset may be abrupt or gradual. The vestibular symptoms may be similarly abrupt or gradual in onset; instrumented vestibular testing usually identifies vestibular weakness (unilateral or bilateral). The auditory and vestibular symptoms may improve (though generally do not completely resolve) spontaneously; some clinicians elect treatment with immunosuppressants (such as corticosteroids), less commonly with immunomodulatory agents. For a neurosarcoidosis patient with otovestibular symptoms, an auditory and vestibular workup is reasonable; if brain imaging has not yet been performed, then MRI of the brain and internal auditory canals without and with contrast is reasonable. If otovestibular workup identifies vestibular weakness, then a trial of vestibular rehabilitation therapy is appropriate.
Introduction
The disease that eventually became known as sarcoidosis had been eponymously designated Besnier-Boeck-Schaumann disease, in recognition of the original descriptions by French dermatologist Ernest Henri Besnier (1831 – 1909) (Besnier 1889) (Beeson 1929), Norwegian dermatologist Caesar Peter Moller Boeck (1845 – 1917) ([No authors] 1970) (Boeck 1899) and Swedish dermatologist Jorgen Nielsen Schaumann (1879 – 1953) (Schaumann 2009 [1916]).
Epidemiology
Sarcoidosis is rare, with an estimated annual incidence of 11 to 36 cases per 100,000 people (Colvin 2006). Of all patients with sarcoidosis, about 5% develop neurosarcoidosis (Babin et al. 1984; von Brevern et al. 1997), which is to say around 5.5 to 18 per one million people develop neurosarcoidosis. In the United States, the disease is significantly more common among African Americans than among Caucasians (Imran et al. 2015).
Genetics
It is unclear whether genetic susceptibility factors differ between neurosarcoidosis and other forms of sarcoidosis (Hardin et al. 2022).
Pathophysiological mechanism of disease
Sarcoidosis is a systemic disease that can cause caseating granulomas in nearly every organ system. The etiology of sarcoidosis is unknown. As far as neurosarcoidosis is concerned, some investigators conclude that the auditory and vestibular symptoms are mostly attributable to vestibulocochlear nerve neuropathy (Colvin 2006), others propose a posterior fossa granulomatous meningitis (Hybels and Rice 1976), while others have documented labyrinthine involvement (Babin et al. 1984).
Clinical presentation: overall
Hooper and Holden (Hooper and Holden 1970) credit Colover (Colover 1948) with having been the first to describe vestibulocochlear nerve involvement by sarcoidosis.
Chen and McLeod (Chen and McLeod 1989) reviewed the records of 285 patients diagnosed with sarcoidosis, and identified 14 (4.9%) with neurosarcoidosis. Of the 14 patients with neurosarcoidosis, two (29%) had “deafness/vertigo.”
Babin and colleagues report that “deafness and vertigo occur as the fourth most common manifestation of neural sarcoidosis” (Babin et al. 1984).
Clinical presentation: auditory
The cadence of hearing loss in neurosarcoidosis is variable. Colvin (Colvin 2006) reported that of 31 neurosarcoidosis patients with hearing loss, the loss was sudden in 15 (48%), “rapidly progressive” in 13 (42%) and “slowly progressive” in 3 (10%).
It has been reported that the subjective hearing loss in neurosarcoidosis can fluctuate (Brihaye and Halama 1993; Hybels and Rice 1976).
Colvin (Colvin 2006) studied 49 cases of neurosarcoidosis with audiologic symptoms, and reported that 30 (61%) complained of tinnitus; of these 30 patients the tinnitus was unilateral in 11 (37%) and bilateral in 6 (20%).
Clinical presentation: vestibular
The cadence of disequilibrium in neurosarcoidosis is variable, ranging from apoplectic onset, to gradual progression over several months. O’Reilly and Burrows (O’Reilly and Burrows 1995) reported a case in which neurosarcoidosis manifested with “sudden… vertigo.” Szmulewicz and Waterston reported the case of a “6-week history of progressive vertigo” (Szmulewicz and Waterston 2012) Imran and colleagues reported a case in which “the vertigo had developed gradually over two months” (Imran et al. 2015). Smith and colleagues reported a case presenting with “a 4-month history of progressive gait imbalance” (Smith et al. 2011).
Ocular motor examination
O’Reilly and Burrows (O’Reilly and Burrows 1995) described a neurosarcoidosis patient who developed “sudden… vertigo” who on examination had spontaneous horizontal non-fatiguing nystagmus, suggestive of unilateral vestibular weakness.
Sidiropoulos and colleagues (Sidiropoulos et al. 2014) described the case of a 49-year-old lady who over the course of 3 years had gradually developed gait disturbances, truncal ataxia and dysarthria. Physical examination showed bilateral rotatory nystagmus, dysmetric saccades and palatal tremor. MRI showed an enhancing lesion in the right cerebellar peduncle extending into the pons, with hypertrophic olivary degeneration. Paratracheal lymph node biopsy confirmed sarcoidosis. This was a case in which neurosarcoidosis interrupted the Guillian-Mollaret triangle, manifesting with oculopalatal tremor, and thus is quite different from sarcoidosis involving the vestibulocochlear nerve.
Testing: auditory
The hearing loss in neurosarcoidosis tends to be greater in the high frequencies on audiometry. The hearing loss is predominantly sensorineural (94%); a minority of patients (6%) have mixed hearing loss (Colvin 2006). Colvin (Colvin 2006) reported that of 44 patients with hearing loss from neurosarcoidosis, the hearing loss was bilateral in 33 (75%) and unilateral in 11 (25%); of 24 patients with bilateral hearing loss, the loss was asymmetrical in 18 (75%) and symmetrical in 6 (25%).
The Figure below, from Babin and colleagues (Babin et al. 1984), displays a typical audiogram from a patient with deafness attributed to neurosarcoidosis (proven at autopsy).
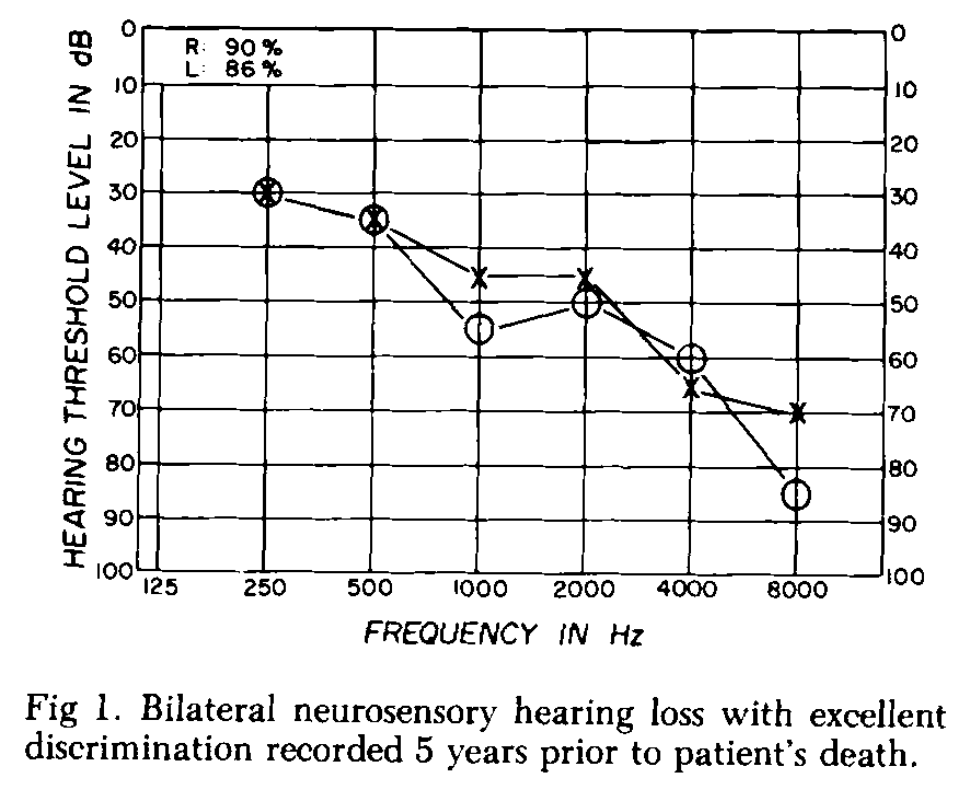
Testing: vestibular
Colvin reported that vestibular testing abnormalities were found in 23/24 (96%) of patients (Colvin 2006), with details shown in the following Table.
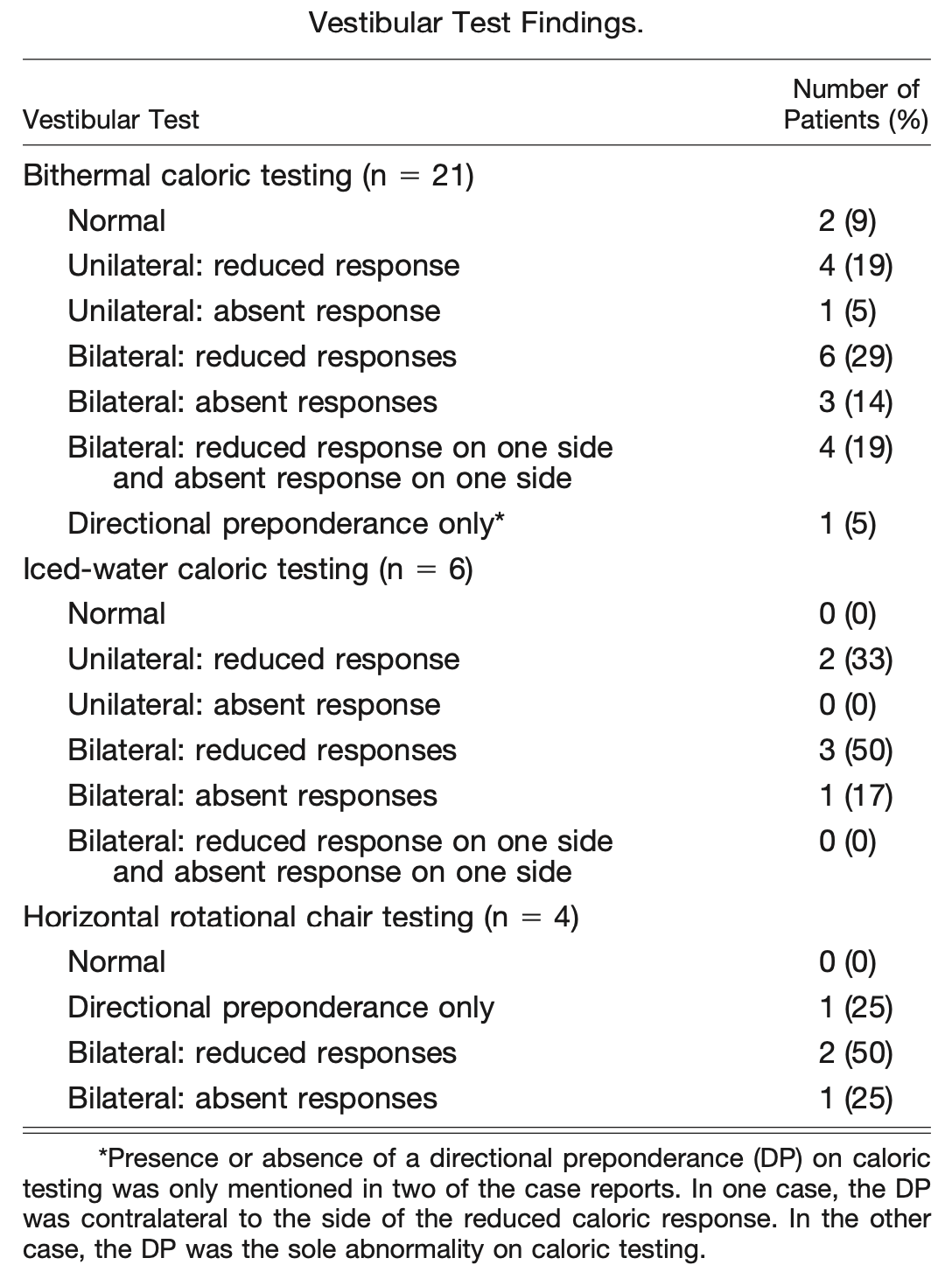
Testing: other
Tissue biopsy is definitive.
Imaging
MRI of the brain without and with contrast is the imaging modality of choice to evaluate for neurosarcoidosis (Imran et al. 2015).
Histopathology
Babin and colleagues studied the temporal bones of a 32-year-old man who had been deaf since age 27 years and who had systemic sarcoidosis, including neurosarcoidosis. They described the following labyrinthine findings (Babin et al. 1984):
“A large portion of the crista of the superior semicircular canal had undergone degeneration characterized by eosinophilic coagulation. While the crista of the lateral semicircular canal was spared, a similar process had completely destroyed the posterior crista and maculae of the utricle and saccule. The latter was ruptured. These structures did not exhibit any inflammatory infiltrates. Both the perilymphatic and endolymphatic spaces of the labyrinth were variably eosinophilic. Few of the structures of the cochlea escaped destruction. The limbus and tectorial membrane remained intact. The fluid within all three scalae had an eosinic hue. A very few inflammatory round cells were present. The organ of Corti had almost completely degenerated in all turns with the Reissner membrane adherent to it. The stria vascularis had also undergone degeneration. In some areas, often nearest the spiral vein, it was simply atrophic; in other areas, often close to the Reissner membrane, it was hypertrophic with loss of the usual layered architecture and notably devascularized. The number of cells in the spiral ganglion was only slightly reduced in contrast to the marked destruction elsewhere in the inner ear. There was a distinct, but not striking, infiltration of round cells in the modiolus and bony spiral lamina without apparent loss of primary afferent nerve fibers. Examination of all areas of the internal auditory canal (lAC) revealed a striking lymphocytic infiltrate. In most sections, this took the form of an intraneural perivascular infiltration. This infiltration extended the entire length of the lAC and involved all four nerves contained within it. In the cochlear nerve it was continuous with the infiltration within the modiolus. In both the cochlear and vestibular nerves, loss of nerve fibers was apparent” (Babin et al. 1984).
Differential diagnosis
Neurosarcoidosis can manifest with abrupt onset hearing loss, tinnitus and disequilibrium (Schick et al. 2001), thereby mimicking idiopathic labyrinthitis.
Since the hearing loss in neurosarcoidosis can fluctuate (Brihaye and Halama 1993; Hybels and Rice 1976) and be asymmetrical (Colvin 2006), the differential diagnosis includes Ménière’s disease.
Szmulewicz and Waterston (Szmulewicz and Waterston 2012) described the case of a 57-year-old lady who presented with acute onset right ear symptoms (fullness, hearing loss, tinnitus) and disequilibrium; on examination, head impulse testing showed a reduced vestibulo-ocular reflex on the right. Four weeks later she had another acute vertiginous episode and was found to have a bilaterally positive head impulse test. Lymph node biopsy confirmed sarcoidosis, but this presentation would be compatible with bilateral sequential vestibular lesions (labyrinthitis on the right, followed by vestibular neuritis on the left).
A patient with neurosarcoidosis involving the vestibular system is still vulnerable to developing more mundane vestibular pathology. For example, von Brevern and colleagues (von Brevern, Lempert, Bronstein and Kocen 1997) report the case of a 30-year-old biopsy-proven neurosarcoid patient found to have bilateral vestibular weakness, who subsequently also developed bilateral posterior canal benign paroxysmal positional vertigo.
Treatment
Agari and colleagues (Agari et al. 2007) reported the case of a 69-year-old woman with bilateral sensorineural hearing loss and bilaterally absent caloric responses attributed to biopsy-proven sarcoidosis. She was treated with prednisolone, and after 2 months, “follow-up examination showed that her vestibular dysfunction and hearing loss had improved significantly.”
Gristwood (Gristwood 1958) documented improvement in audiologic and vestibular function (caloric responses) over 3.5 months of treatment with prednisolone, as shown in the Figure below.
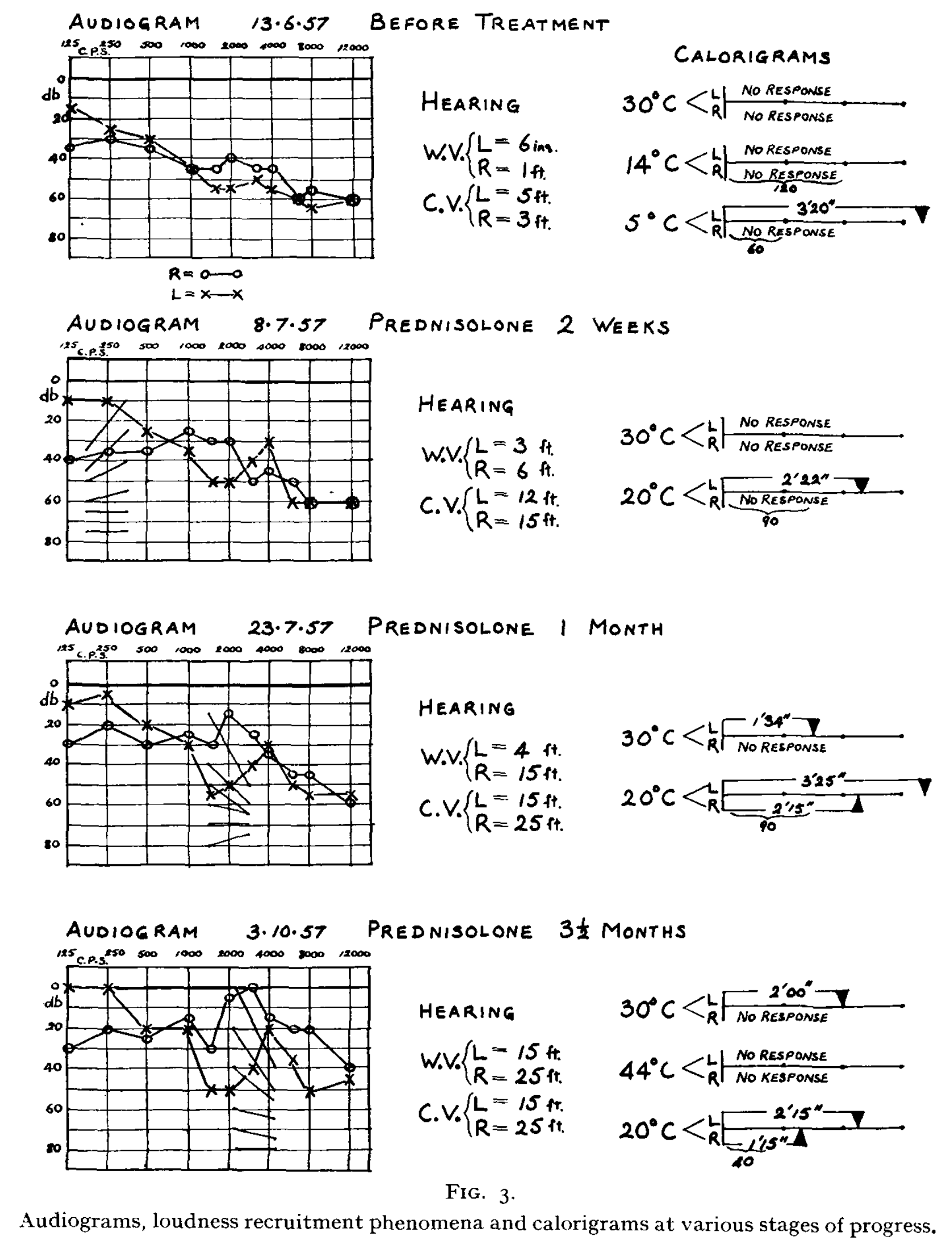
Imran and colleagues report on a neurosarcoidosis patient with disequilibrium that did not respond to prednisone, but did improve with methotrexate (Imran et al. 2015).
Other, less studied immunomodulatory agents have been tried for neurosarcoidosis, such as mycophenolate mofetil, azathioprine, cyclophosphamide, cyclosporine, cladribine and infliximab, though none has been scrutinized in a randomized controlled trial (Imran et al. 2015).
Prognosis
In some cases the auditory and vestibular symptoms attributed to neurosarcoidosis improve spontaneously, though they generally do not completely resolve.
Final comments
Neurosarcoidosis is rare, and thus should be low on the differential diagnosis of patients with new onset auditory and/or vestibular symptoms. However, in a patient already diagnosed with sarcoidosis, the index of suspicion should be much higher, given that 5% of sarcoid patients develop neurosarcoidosis, and of those, about a third develop auditory and/or vestibular symptoms.
Given the differential diagnosis described earlier, workup should include auditory testing (audiometry, otoacoustic emissions) and vestibular testing (cervical vestibular evoked myogenic potentials, ocular vestibular evoked myogenic potentials, video head impulse testing, videonystagmography and rotatory chair testing). If the hearing loss fluctuates, then electrocochleography is also reasonable. If brain imaging has not yet been performed, then an MRI of the brain and internal auditory canals without and with contrast should be considered. The clinician should keep in mind that neurosarcoidosis patients can develop more common otovestibular problems, such as benign paroxysmal positional vertigo.
By the time a sarcoidosis patient is referred to otoneurology, the disease is usually already being managed by a practitioner specializing in the organ system originally affected by the disease (e.g., pulmonology, dermatology). If otovestibular workup identifies vestibular weakness, then a trial of vestibular rehabilitation therapy is reasonable.
References
[No authors] (1970) Caesar Peter Moeller Boeck (1845-1917). Boeck’s sarcoidosis. JAMA 211: 1537-8. doi: 10.1001/jama.1970.03170090053013
Agari D, Koide R, Kashiyama T, Yoshida H, Naito R, Bandoh M (2007) Neurosarcoidosis: a treatable cause of vestibular dysfunction. Lancet 369: 878. doi: 10.1016/s0140-6736(07)60417-6
Babin RW, Liu C, Aschenbrener C (1984) Histopathology of neurosensory deafness in sarcoidosis. Ann Otol Rhinol Laryngol 93: 389-93. doi: 10.1177/000348948409300421
Beeson BB (1929) Ernest Besnier 1831-1909. Archives of Dermatology and Syphilology 20: 95-99. doi: 10.1001/archderm.1929.01440010103014
Besnier EH (1889) Lupus pernio de la face. Ann Dermatol Syph 10: 333-336.
Boeck CPM (1899) Multiple benign sarcoid of the skin. J Cutan Gen Urin Dis 17: 543-550.
Brihaye P, Halama AR (1993) Fluctuating hearing loss in sarcoidosis. Acta Otorhinolaryngol Belg 47: 23-6.
Chen RC, McLeod JG (1989) Neurological complications of sarcoidosis. Clin Exp Neurol 26: 99-112.
Colover J (1948) Sarcoidosis with involvement of the nervous system. Brain 71: 451-75. doi: 10.1093/brain/71.4.451
Colvin IB (2006) Audiovestibular manifestations of sarcoidosis: a review of the literature. Laryngoscope 116: 75-82. doi: 10.1097/01.mlg.0000184580.52723.9f
Gristwood RE (1958) Nerve deafness associated with sarcoidosis. J Laryngol Otol 72: 479-91.
Hardin A, Dawkins B, Pezant N, Rasmussen A, Montgomery C (2022) Genetics of neurosarcoidosis. J Neuroimmunol 372: 577957. doi: 10.1016/j.jneuroim.2022.577957
Hooper R, Holden H (1970) Acoustic and vestibular problems in sarcoidosis. Arch Otolaryngol 92: 386-91.
Hybels RL, Rice DH (1976) Neuro-otologic manifestations of sarcoidosis. Laryngoscope 86: 1873-8.
Imran TF, Nizami S, Eyzner I, Mirani N, Hossain T, Fede R, Capitle E (2015) Vertigo as a predominant manifestation of neurosarcoidosis. Case Rep Med 2015: 397046. doi: 10.1155/2015/397046
O’Reilly BJ, Burrows EH (1995) VIIIth cranial nerve involvement in sarcoidosis. J Laryngol Otol 109: 1089-93. doi: 10.1017/s0022215100132104
Schaumann JN (2009 [1916]) Recherches sur le lupus pernio et ses relations avec les sarcoïdes cutanées et sous-cutanées. Nordiskt Medicinskt Arkiv 49: 1-81. doi: 10.1111/j.0954-6820.1916.tb00318.x
Schick B, Brors D, Koch O, Schafers M, Kahle G (2001) Magnetic resonance imaging in patients with sudden hearing loss, tinnitus and vertigo. Otol Neurotol 22: 808-12. doi: 10.1097/00129492-200111000-00016
Sidiropoulos C, Sripathi N, Nasrallah K, Mitsias P (2014) Oculopalatal tremor, facial myokymia and truncal ataxia in a patient with neurosarcoidosis. J Clin Neurosci 21: 2255-6. doi: 10.1016/j.jocn.2014.01.025
Smith JH, Stovall KC, Coons S, Fife TD (2011) Bilateral vestibular hypofunction in neurosarcoidosis: a case report. Ear Nose Throat J 90: E1-3.
Szmulewicz DJ, Waterston JA (2012) Two patients with audiovestibular sarcoidosis. J Clin Neurosci 19: 158-61. doi: 10.1016/j.jocn.2011.07.020
von Brevern M, Lempert T, Bronstein AM, Kocen R (1997) Selective vestibular damage in neurosarcoidosis. Ann Neurol 42: 117-20. doi: 10.1002/ana.410420119
![]()