By Marcello Cherchi, MD PhD
Overview
In a broad sense the phrase “vestibular system” refers to the integration of multiple sensory inputs (inner ear, vision, somatic sensation) and orchestration of multiple outputs (ocular motor, somatomotor) for purposes of movement (such as locomotion) and maintaining posture. Given the multiple inputs and multiple outputs involved, it is understandable that the vestibular system (in this broad sense) involves a broad range of neuroanatomical pathways.
In a narrower sense, “vestibular system” can refer to the process by which type 1 vestibular hair cells in the inner ear’s labyrinth transduce acceleration (whether rotational or translational) into electrochemical signals.
Introduction
The phrase “vestibular system” can have several meanings. In a narrow sense, “vestibular system” refers to the function of the vestibular labyrinth (the inner ear’s “gyroscope” function).
In a broader sense, “vestibular system” refers to the arrangement by which multiple sensory inputs (labyrinthine, visual, somatosensory, auditory) are integrated into a coherent perception of orientation and movement, from which motor responses (somatomotor, ocular motor) are orchestrated and executed in order to maintain a desired posture or execute a desired movement. In this section we will discuss the vestibular system in this broader sense.
Multiple inputs, processing, multiple outputs
Neurologists tend to apply a “circuitry” approach when thinking about anatomy, physiology and pathology. A simplified circuitry-based schematic of the vestibular system is shown in the Figure below.

This schematic is an oversimplification in many respects. For example, it does not depict feedback loops, nor does it depict the feedforward circuits (efference copies) by which the system compares planned motor output with actual motor output.
Type 1 vestibular hair cells
The type 1 vestibular hair cells are the “building blocks” of the labyrinth’s gyroscopic capabilities. These cells function as accelerometers. They accomplish this task through mechano-electrical transduction, which is to say that they convert (transduce) the kinetic (mechanical) energy from acceleration into a neurochemical (electrical) signal.
The Figure below, from Soto and colleagues (Soto et al. 2013), summarizes the sequence through which mechano-electrical transduction (“MET”) occurs.
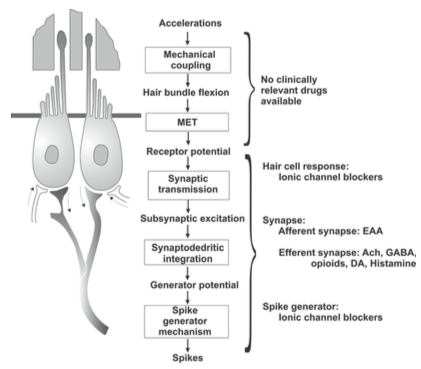
At baseline, when no acceleration is occurring, each type 1 vestibular hair cell has a tonic (baseline) rate of firing. These cells are morphologically polarized in such a fashion as to make them preferentially sensitive to acceleration in a particular direction; the rate of firing will increase (above the tonic rate) in response to acceleration in that direction, and will decrease (below the tonic rate) in response to acceleration in the opposite direction, as shown in the Figure below, from Schunke and colleagues (Schünke et al. 2007).
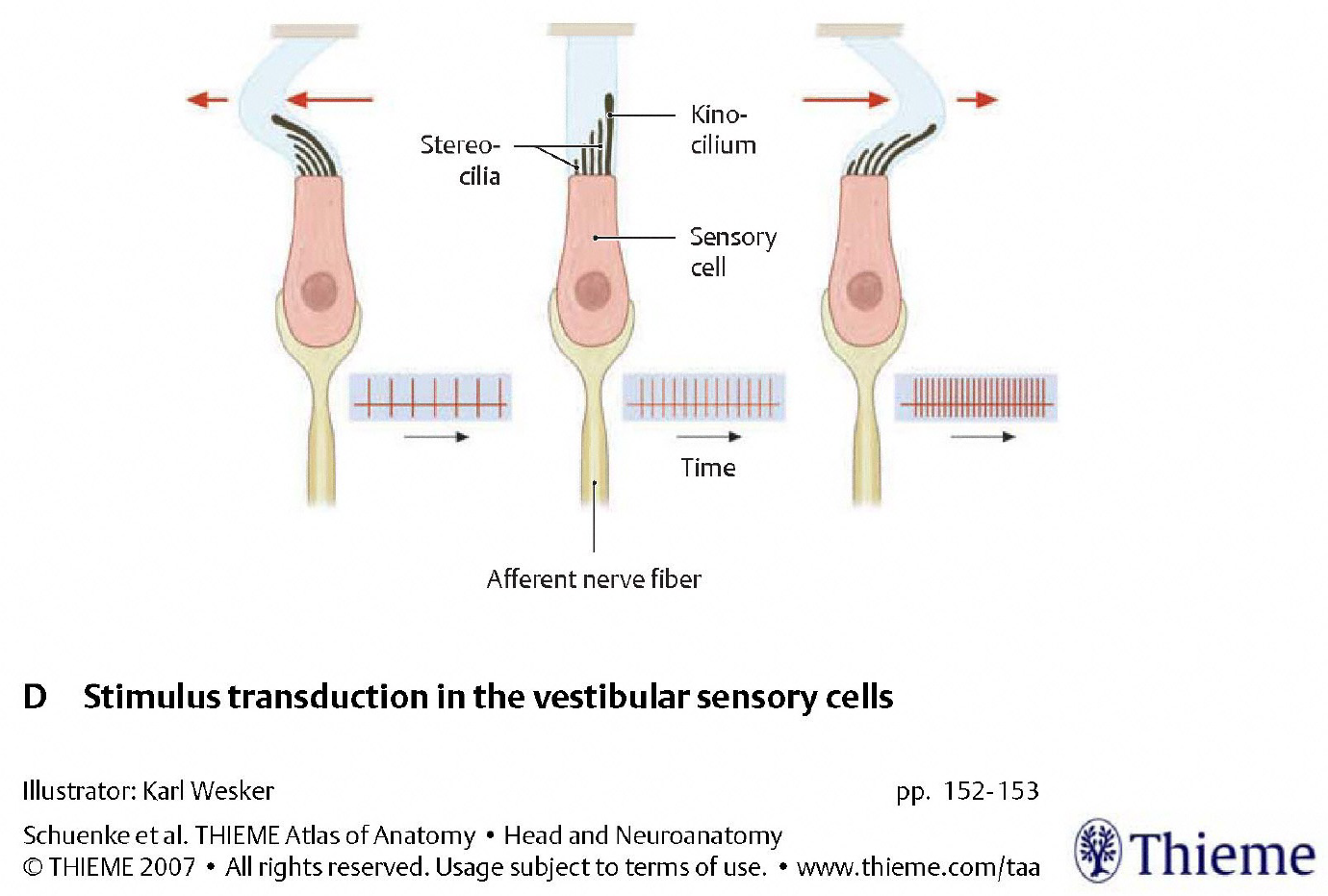
The morphologic polarization is due to the cellular architecture — specifically, the arrangement of the “hairs” (kinocilium and stereocilia) on each hair cell. Acceleration will deflect these hairs (and we will discuss the mechanics of this shortly); when the deflection is towards the kinocilium one set of chemical processes occurs; when the deflection is away from the kinocilium, a different set of chemical processes occurs.
Vestibular end-organelles: semicircular canals and otolith organs
There is one labyrinth on each side of the head, embedded in the temporal bone. Each labyrinth contains five discrete end-organelles, divided into two types:
- Three organelles are the semicircular canals, which detect rotational (angular) acceleration. These are:
- Horizontal semicircular canal, also called the lateral semicircular canal.
- Anterior semicircular canal, also called the superior semicircular canal.
- Posterior semicircular canal, sometimes called the inferior semicircular canal.
- Two organelles are the otolith organs, which detect translational (linear) acceleration. These are:
- Utricle.
- Saccule.
The arrangement of these five organelles within the labyrinth is depicted in the Figure below, from Schunke and colleagues (Schünke et al. 2007).
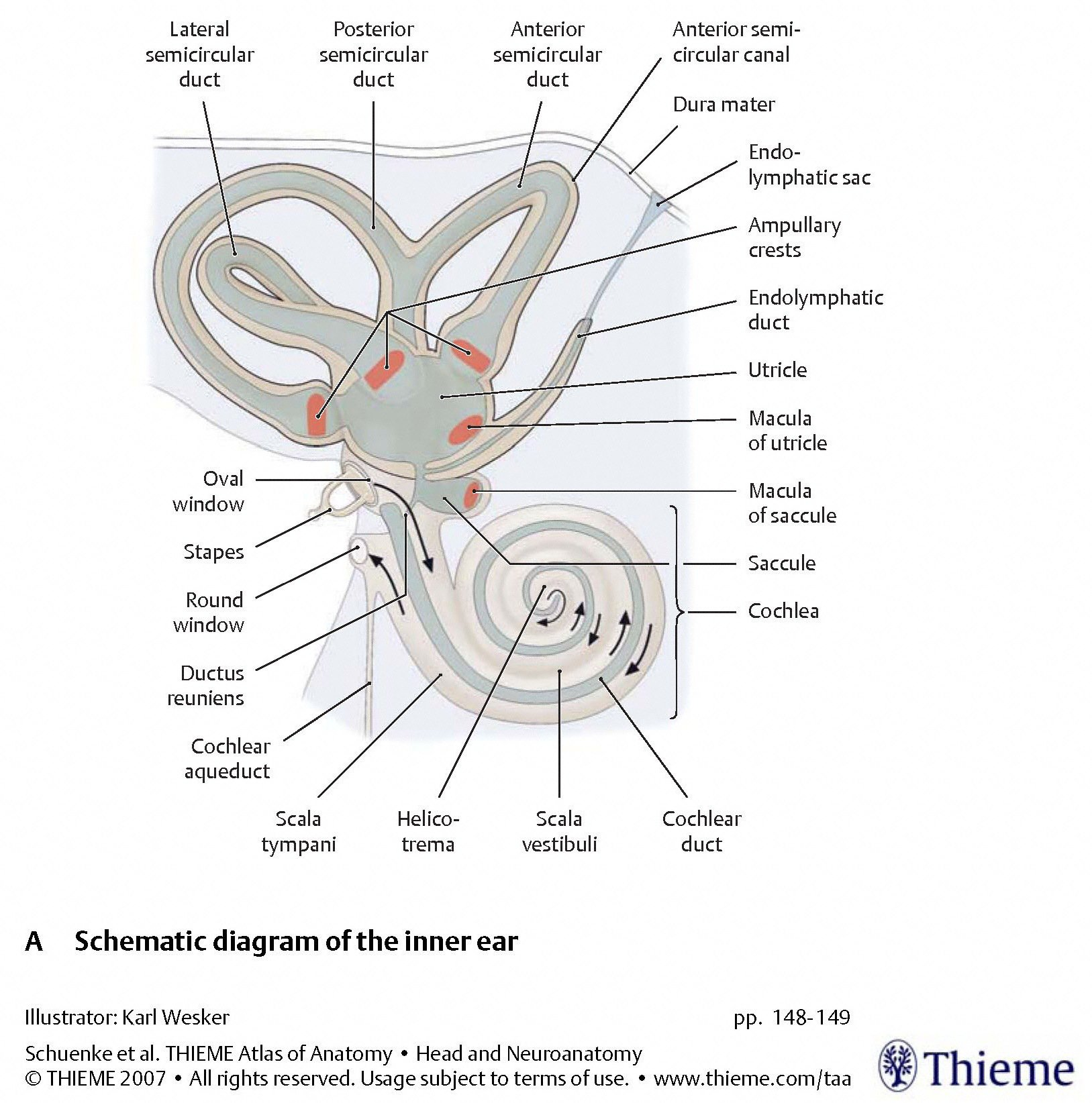
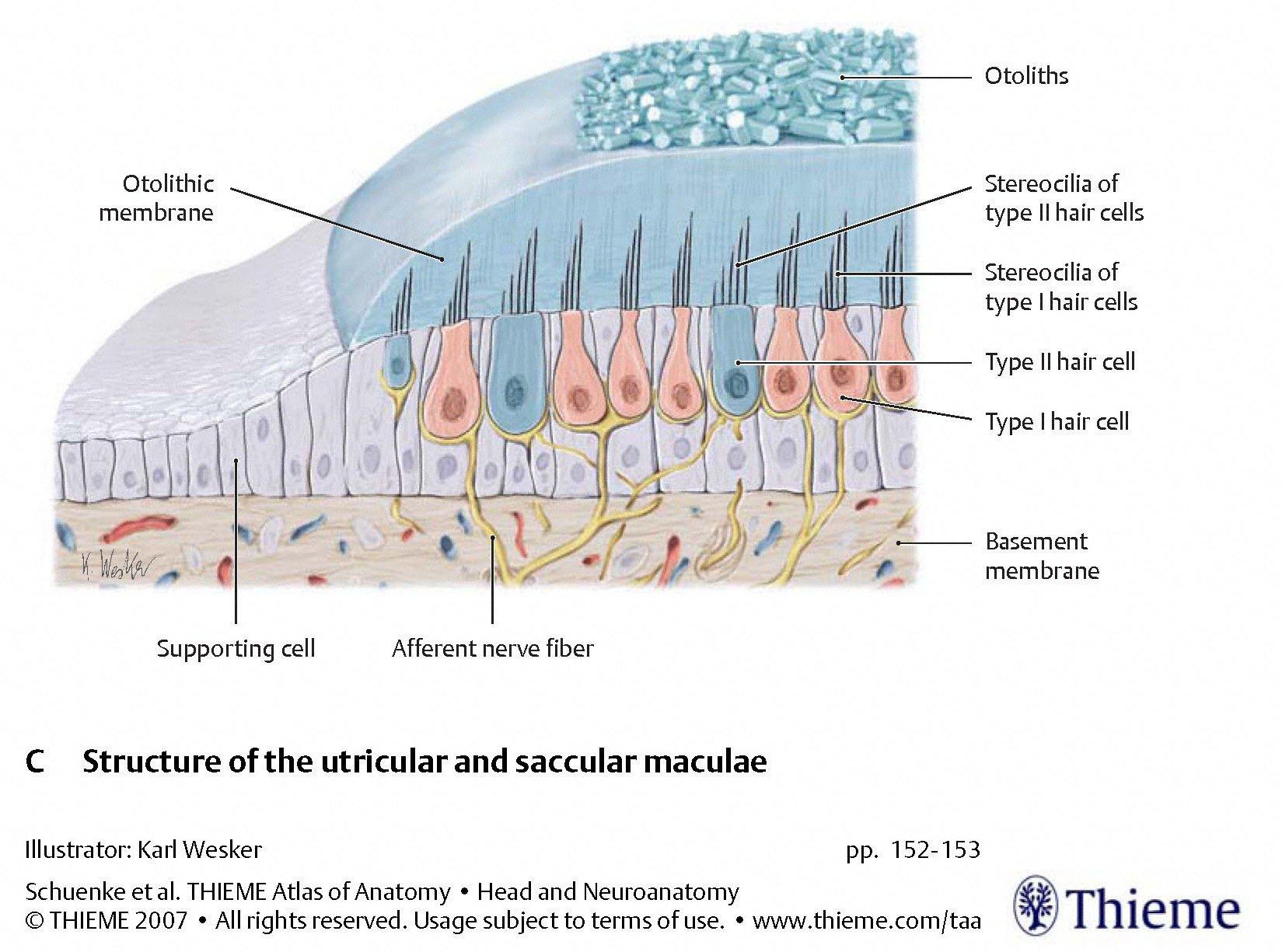
The two types of organelles have different architectures, as displayed in the Figures below, from Schunke and colleagues (Schünke et al. 2007).
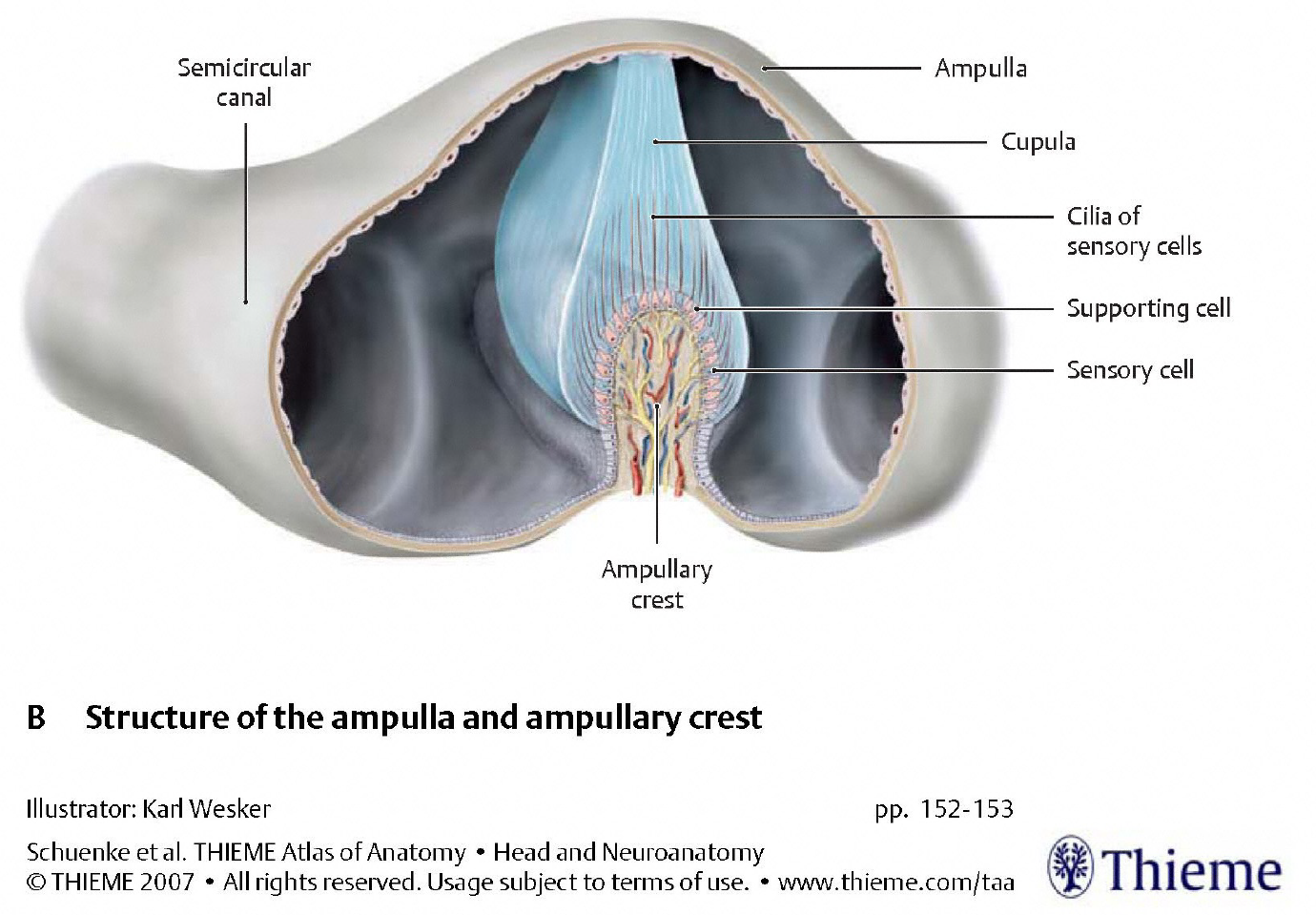 |
 |
In the semicircular canals, type 1 vestibular hair cells in the ampullary crest extend their hairs (stereocilia and kinocilia) into the gelatinous substance of the cupula. During rotational acceleration the inertia of the endolymph in the semicircular canal will deflect the cupula, which will in turn deflect the hairs of the hair cells embedded within it, as shown in the Figure below.
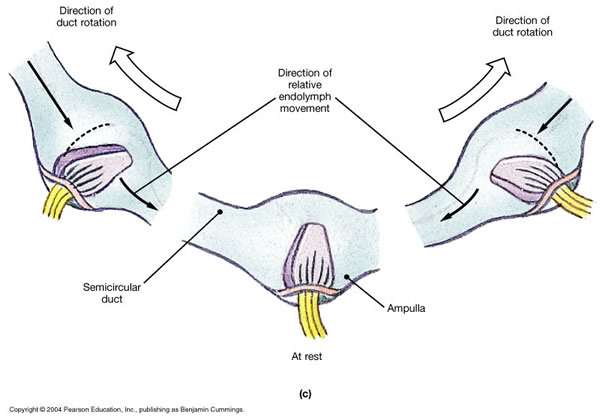
The direction of endolymphatic flow (ampullopetal/utriculofugal versus ampullofugal/utriculopetal) has different effects (excitatory or inhibitory) in different semicircular canals. Specifically:
- In the vertical semicircular canals (superior and posterior), ampullofugal/utriculopetal flow (i.e., flow away from the ampulla, toward the utricle) is excitatory.
- In the horizontal semicircular canals, ampullopetal/utriculofugal flow (i.e., flow away from the utricle toward the ampulla) is excitatory.
This is illustrated in the Figure below from Hornibrook (Hornibrook 2004).
In the otolith organs, type 1 vestibular hair cells in the macula extend their hairs (stereocilia and kinocilia) into the gelatinous substance of the otolith membrane. Otoliths embedded on the surface of the otolith membrane have a relatively high specific gravity. During linear acceleration the inertia that the otoliths impart to the otolith membrane cause the otolith membrane to move (relative to the underlying hair cells) and exert a shearing force on the hairs, as shown in the Figure below.
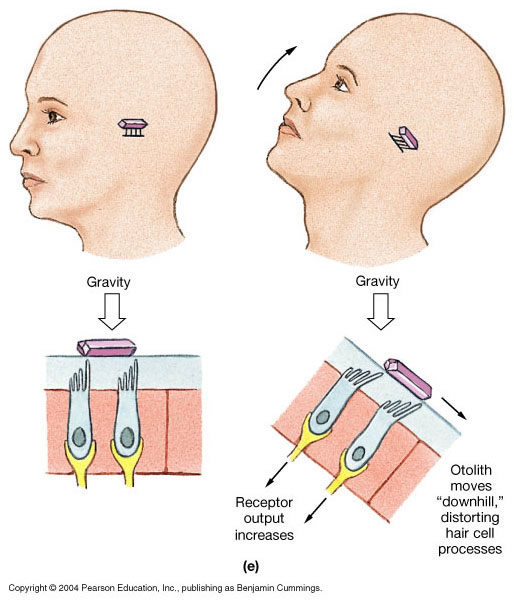
Note that a linear accelerometer cannot distinguish acceleration due to linear movement (such as when a vehicle begins to move) from acceleration due to gravity, as both forces will have the same physical effect.
This is illustrated by the Figure below. In the top panel, an individual with head upright begins to move backwards, and the linear acceleration (due to the person’s movement) deflects the hairs of the type 1 vestibular hair cells. In the lower right panel, the individual is stationary but with the head tilted forward, and the linear acceleration (due to gravity) deflects the hairs of the type 1 vestibular hair cells in the same manner.
Vestibular end-organelles: contralateral accelerometers are paired in a push-pull relationship
As mentioned earlier, each type 1 vestibular hair cell is morphologically polarized so as to be preferentially sensitive to acceleration in a particular direction. In each end-organelle, collections of type 1 vestibular hair cells are arranged so that the entire zone is preferentially sensitive to acceleration in a particular direction.
A given end-organelle on one side of the head is paired with a contralateral end-organelle such that their preferential directional sensitivities are diametrically opposed. This arrangement means that pairs of end-organelles function in a “push-pull” relationship, in the sense that acceleration (whether rotational or translational) in a given direction will maximally stimulate one end-organelle, and maximally inhibit the corresponding contralateral end-organelle.
Citing multiple sources (Blanks et al. 1975; Della Santina et al. 2005; Ifediba et al. 2007; Sato et al. 1993), Bradshaw and colleagues (Bradshaw et al. 2010) point out that “The semicircular canals are not orthogonal and not planar.” Furthermore, there is considerable inter-individual variability in canal orientation, diameter, cross-sectional area and other parameters (Cortés-Domínguez et al. 2019). Finally, and rather counter-intuitively, the plane in whose rotation the maximal stimulus from a given semicircular canal is elicited is not necessarily completely co-planar with the plane best fit to the anatomy of that canal (Ifediba et al. 2007).
Nevertheless, the three semicircular canals in each ear are sufficiently orthogonal (Della Santina et al. 2005) to depict them as such for didactic purposes, as shown in the Figure below.
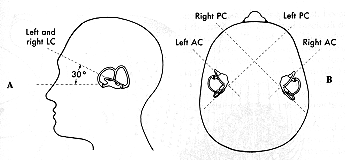
Semicircular canals that are co-planar are functionally paired. Specifically:
- The horizontal semicircular canals are functionally paired.
- The right anterior canal is functionally paired with the left posterior canal.
- The left anterior canal is functionally paired with the right posterior canal.
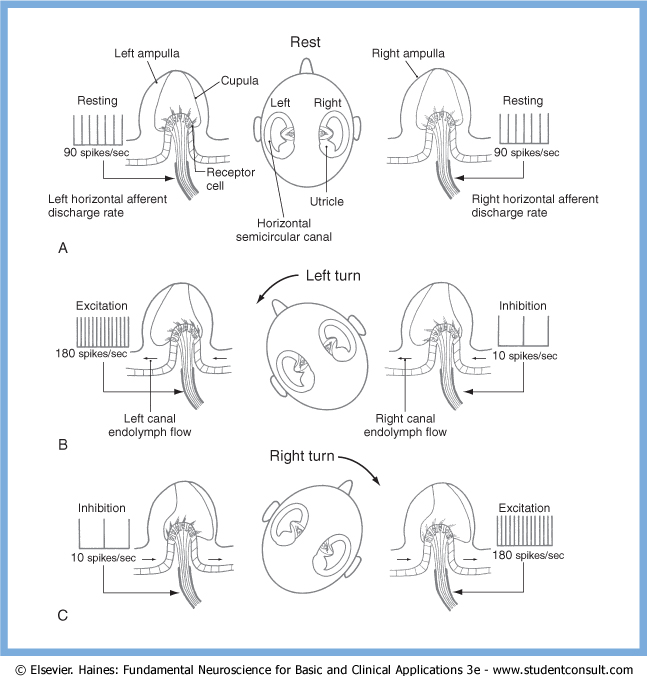
As an example of a functional pairing of semicircular canals, The Figure below, from Haines (Haines 2006), illustrates the push-pull relationship between the horizontal semicircular canals. Rotation towards one side will trigger excitation of the ipsiversive horizontal semicircular canal, and inhibition of the contraversive horizontal semicircular canal.
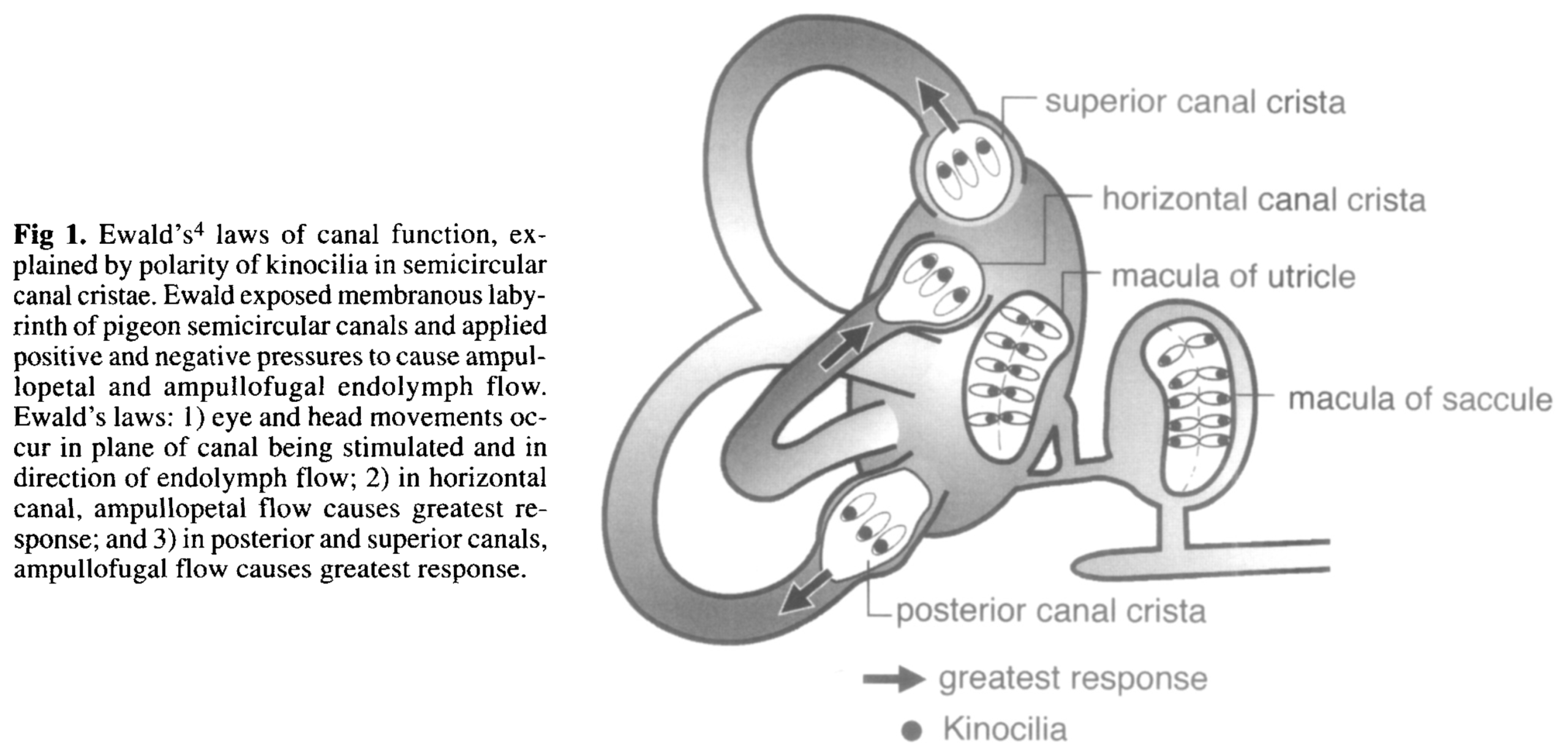
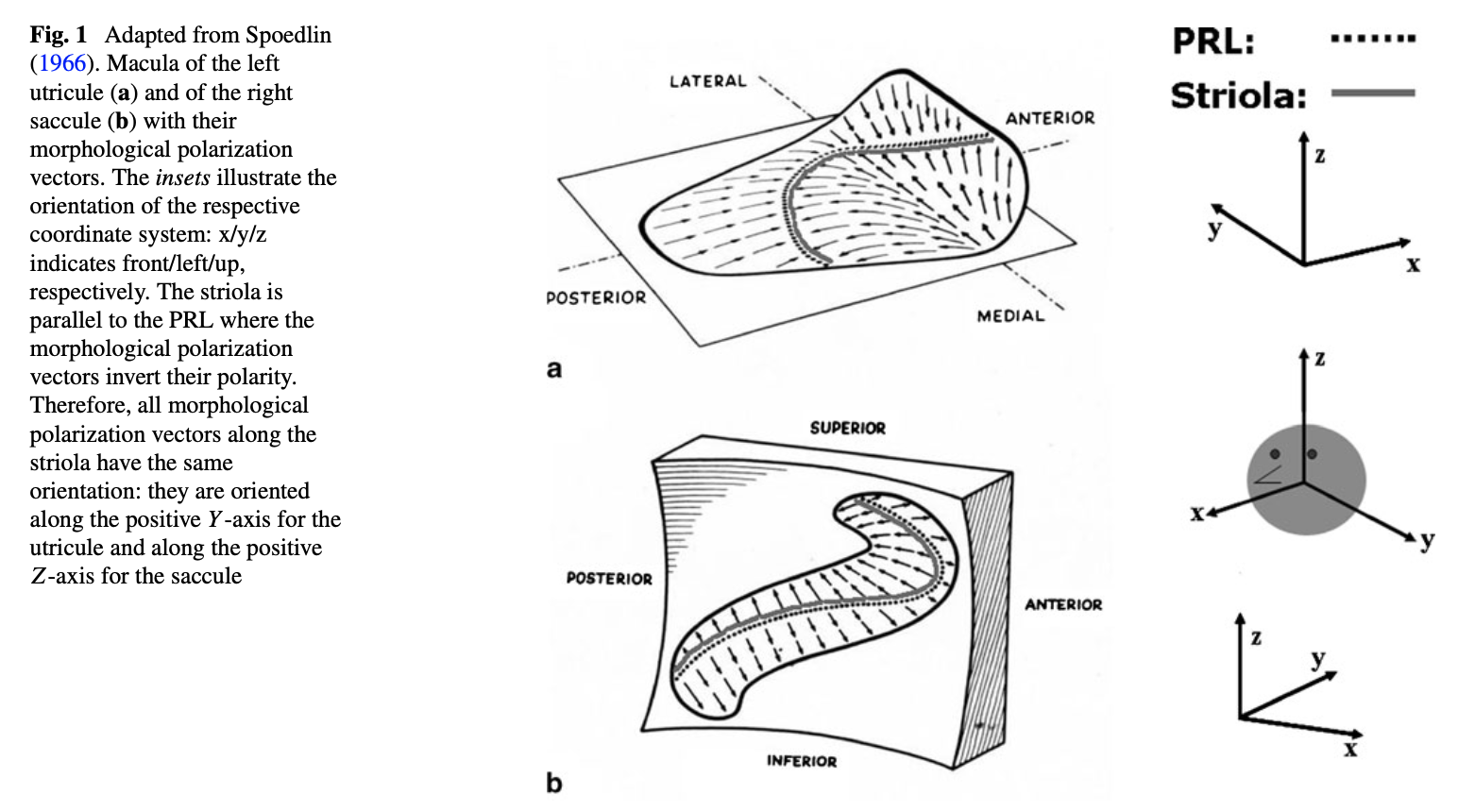
The otolith organs are also paired, though their morphology is less obvious than that of the semicircular canals, as shown in the Figure below from Dimiccoli and colleagues (Dimiccoli et al. 2013).
The co-planar arrangements of the various zones of the otolith organs are depicted in the Figure below, from Miller (Miller 1962).
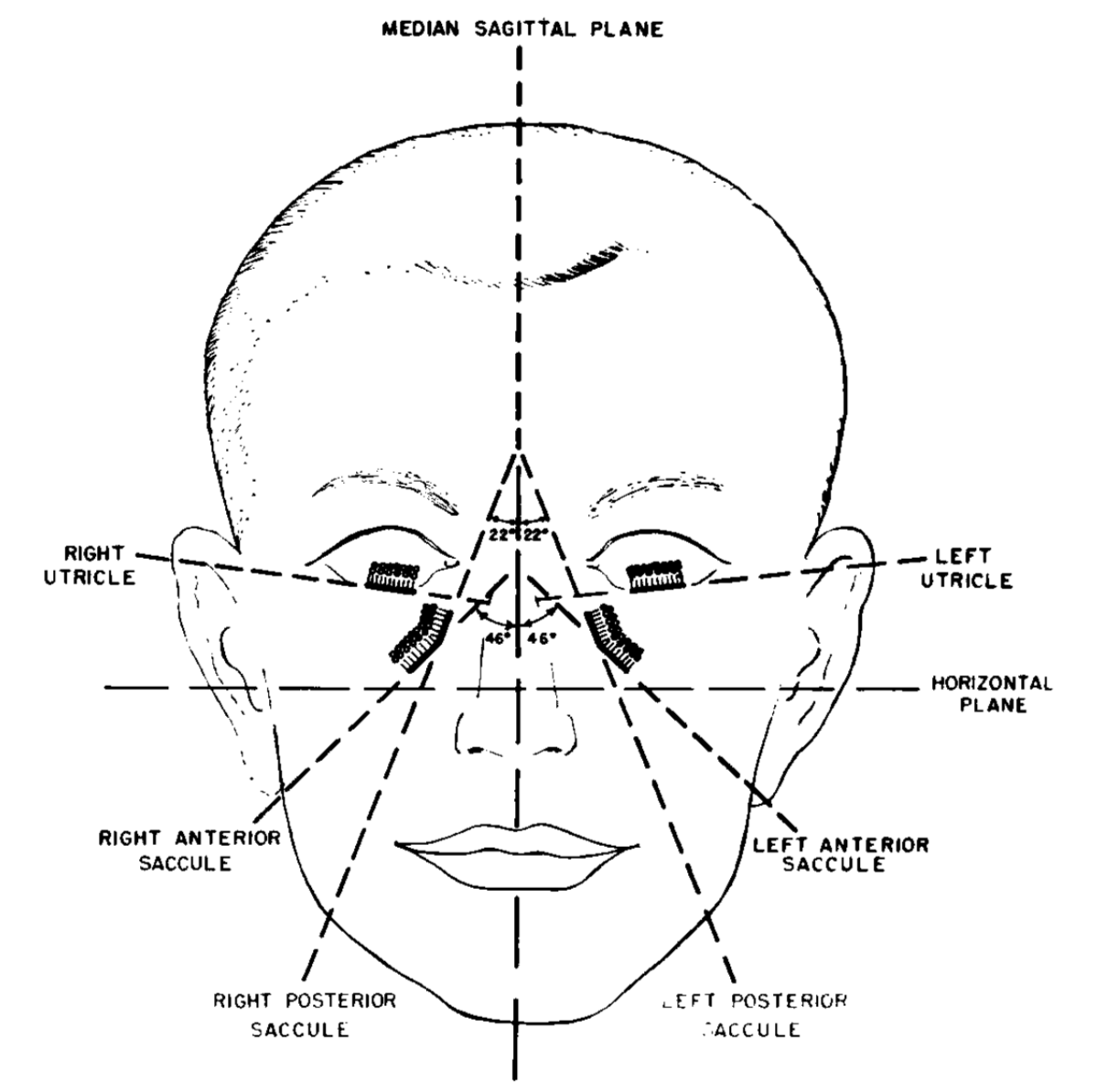
The saccule detects linear acceleration primarily along the rostro-caudal axis (up and down, “bob”), and partly along the anterior-posterior axis (forward and backward, “surge”).
The utricle detects linear acceleration primarily along the inter-aural axis (side to side, “heave”), and partly along the anterior-posterior axis (forward and backward, “surge”).
As an example of the functional pairing of otolith organs, the Figure below, from Haines (Haines 2006), illustrates the push-pull relationship between the utricles. In a person oriented upright with respect to gravity, tilting the head towards one side will cause inhibition of the ipsiversive utricle, and excitation of the contraversive utricle.
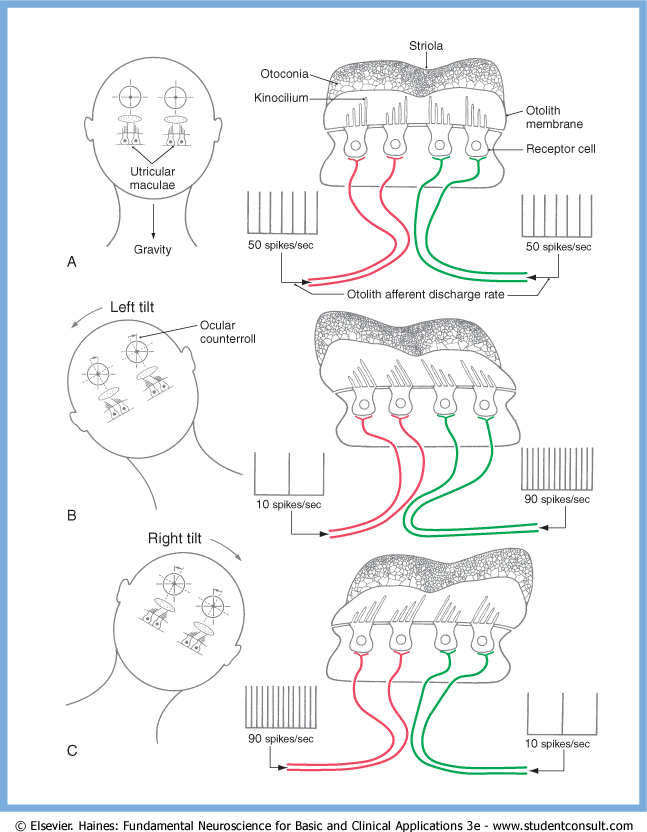
Neurochemistry of type 1 vestibular hair cells
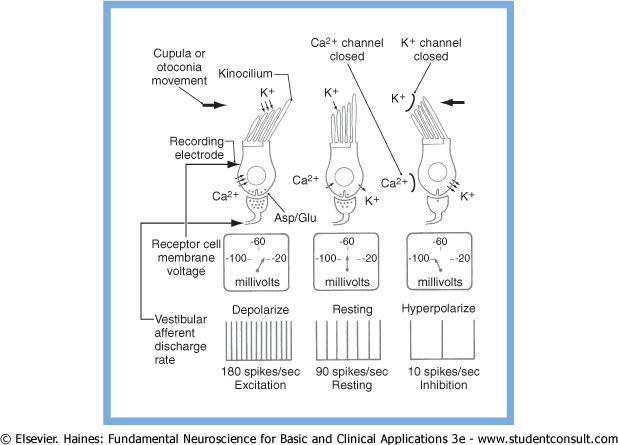
At rest, the type 1 vestibular hair cells generate a tonic rate of firing in the first order neuron.
Deflection of the stereocilia towards the kinocilium results in opening of K+ channels at the apices of the stereocilia, causing K+ to enter the cell from the endolymph, depolarizing the cell membrane. This, in turn, causes voltage-gated Ca2+ channels at the base of the cilia to open, further depolarizing the cell membrane. The influx of Ca2+ causes release of synaptic vesicles (containing aspartate or glutamate) into the synaptic cleft, thereby increasing the rate of firing of the first-order neuron.
Deflection of the stereocilia away from the kinocilium causes K+ channels at the base of the stereocilia to open, allowing K+ to exit the cell into the interstitial space, thereby hyperpolarizing the cell membrane and decreasing the rate of firing of the first order neuron.
These ion channel activities are summarized in the Figure below, from Haines (Haines 2006).
Pathways through the vestibular nerve
The first order neurons in the vestibular system are located in the vestibular (Scarpa’s) ganglion. These are bipolar neurons that receive input from type 1 vestibular hair cells, and project their axons through the vestibular nerve, which has superior and inferior divisions. The distribution of afferent fibers among the divisions of the vestibular nerve is illustrated in the Figure below, from Curthoys (Curthoys 2017).
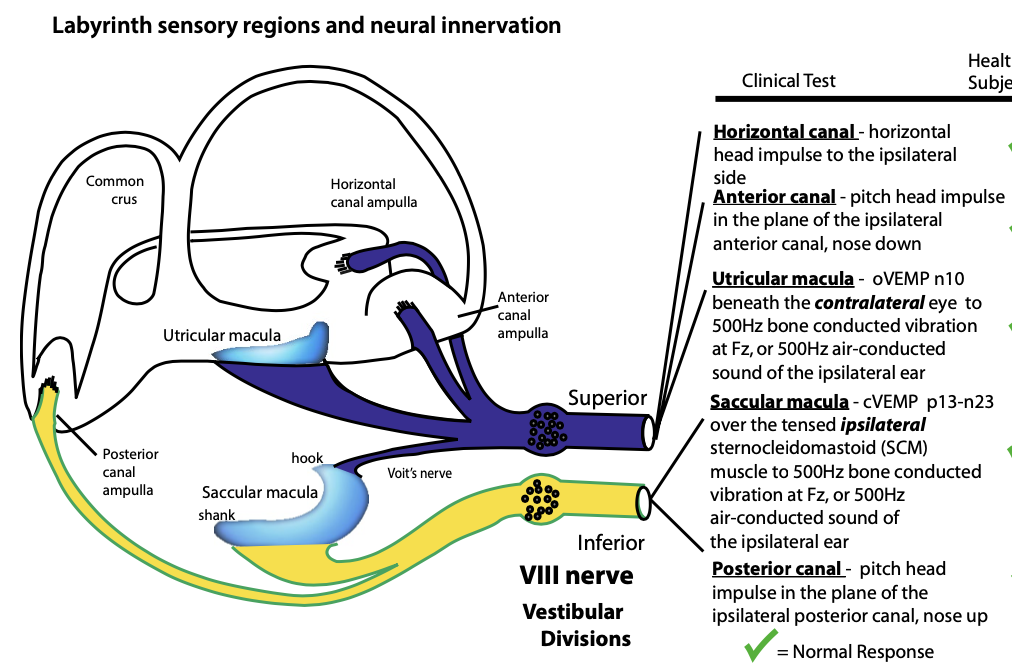
The neuroanatomy is somewhat more complex than what is shown here, due to the presence of other pathways and anastomoses (Cherchi and Yacovino 2021).
Afferent vestibular projections
After traversing the vestibular nerve, most projections from first order vestibular neurons reach targets in the vestibular nuclei.
A minority of fibers project directly (without synapsing) through the brainstem via the juxtarestiform body into the cerebellum (Haines 2004). In fact, the vestibular labyrinth is the only sensory organ in the body that sends direct primary afferent projections to the cerebellar cortex and nuclei (Haines 2006).
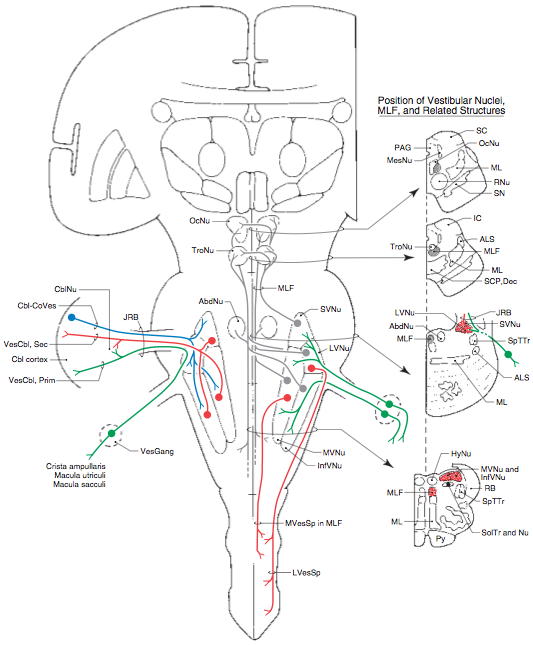
Vestibular nuclei
The majority of first order vestibular neurons project to vestibular nuclei in the brainstem (Baloh 1998). The distribution is as follows (Haines 2006):
- Afferents from the semicircular canals arrive:
- Mostly in the superior and medial vestibular nuclei
- Less in the lateral and inferior vestibular nuclei
- Afferents from the otolith organs (utricle and saccule) arrive:
- Mostly in the lateral, medial and inferior vestibular nuclei.
- Some afferents from the saccule arrive in “cell group Y”
These afferent projections from first order vestibular neurons are depicted in the Figure below from Haines (Haines 2004).
It should be noted that the vestibular nuclei receive projections from multiple other structures as well, including:
- Cerebral cortex
- Interstitial nucleus of Cajal
- Nucleus prepositus hypoglossi
- Central cervical nucleus
- Dorsal spino-cerebellar tract
- Several areas of the cerebellum
Some of these projections (from extra-vestibular structures) are depicted in the Figures below, from Niewenhuys (Nieuwenhuys et al. 2008).
 |
 |
Efferent projections from the vestibular nuclei
Efferent projections from the vestibular nuclei project broadly, with major targets in (Haines 2006):
- Oculomotor nuclei
- Vestibulocerebellum
- Contralateral vestibular nuclei
- Spinal cord
- Reticular formation
- Thalamus
The Figure below, from Schunke and colleagues (Schünke et al. 2007), illustrates some of these pathways.
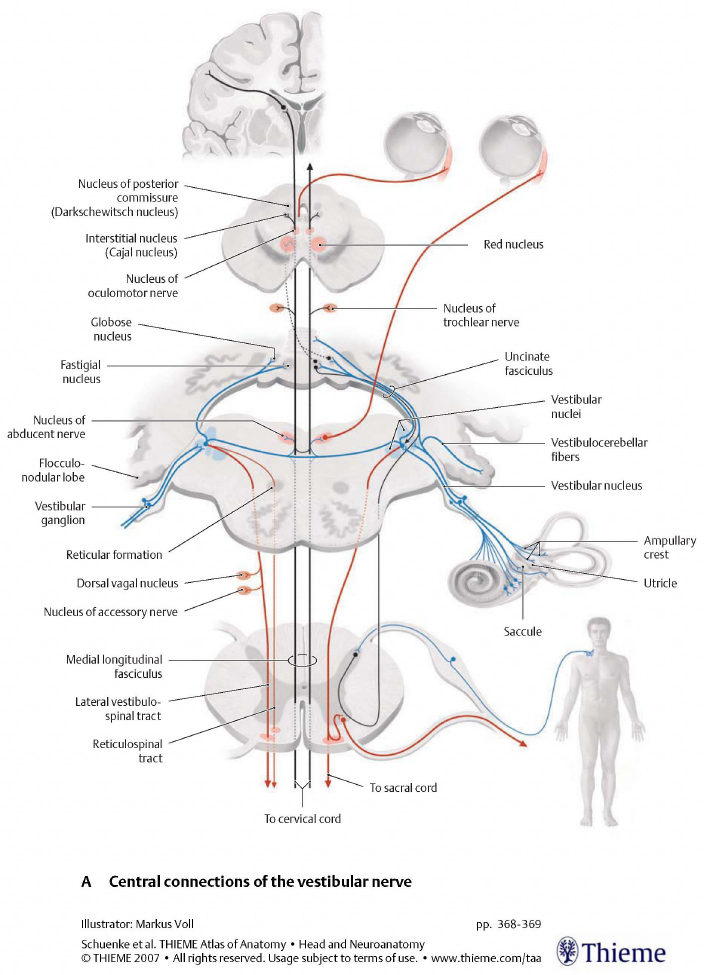
Efferent projections from vestibular nuclei to oculomotor nuclei
The vestibular nuclei have substantial projections to the ocular motor nuclei. Among other functions, these projections mediate the vestibulo-ocular reflexes that stabilize the eyes (help maintain foveation of a target) despite the perturbations from head movements.
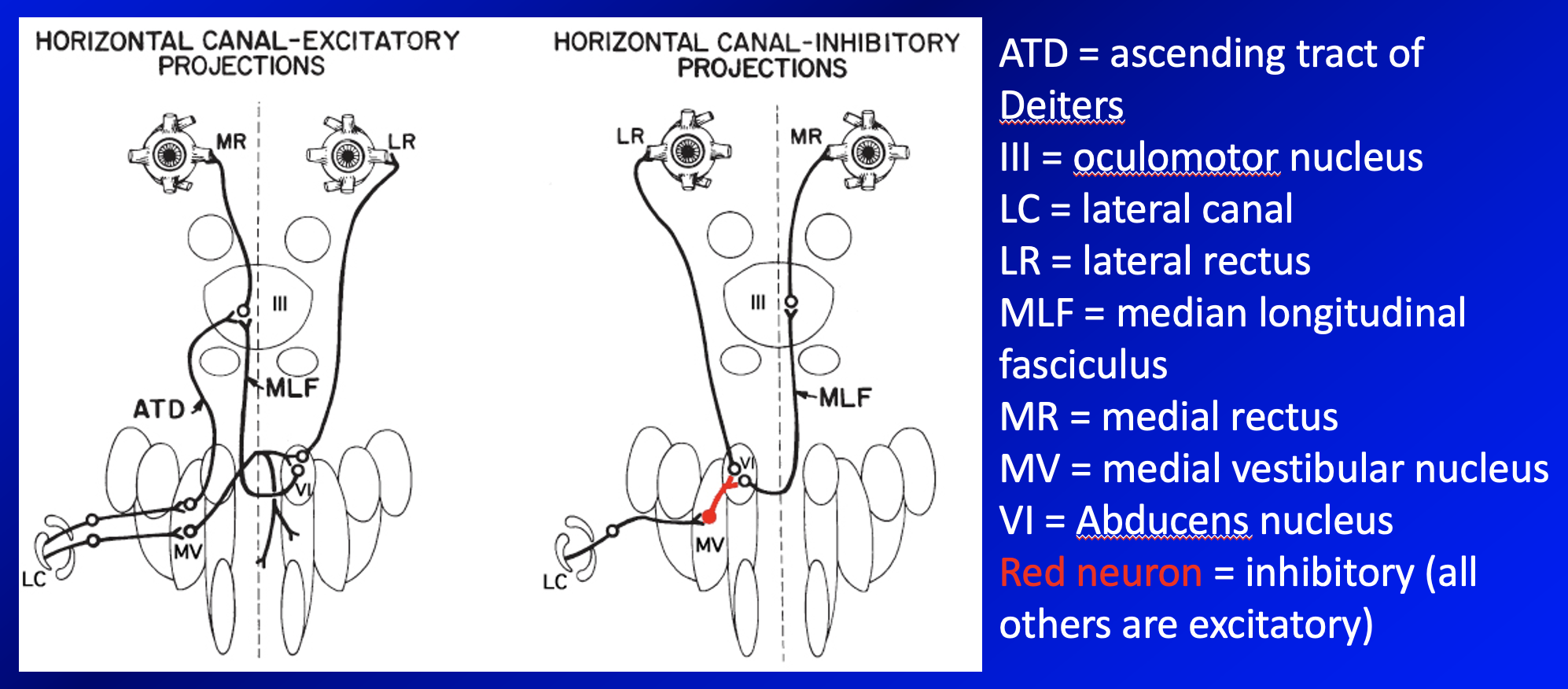
An example of such projections is shown in the Figure below, from Leigh and Zee (Leigh and Zee 2015), which shows the circuitry connecting activity in the horizontal semicircular canals with horizontal eye movements.
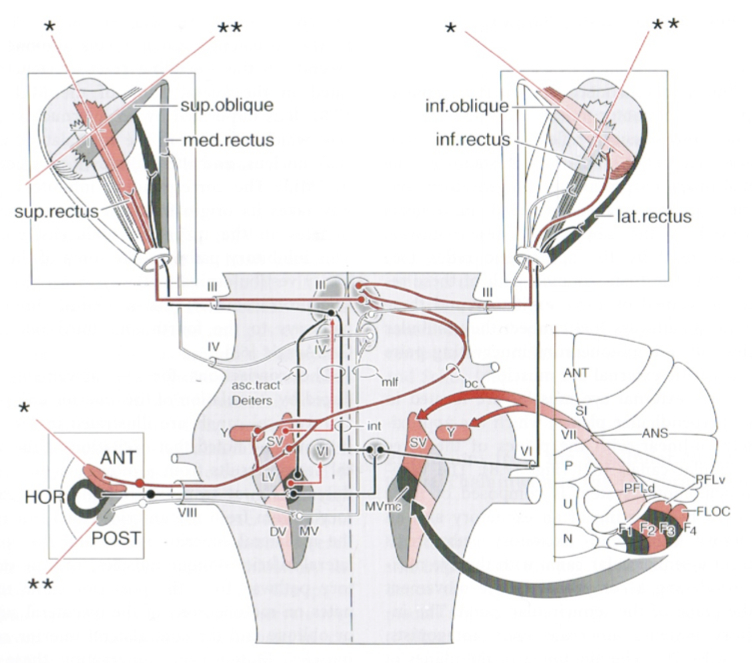
Comparable circuitry mediates eye movements in response to activity from the anterior and posterior semicircular canals, as shown in the Figure below from Niewenhuys (Nieuwenhuys et al. 2008).
Vestibular information contributes not only to the vestibulo-ocular reflex, but to other eye movements as well. Slow eye movements (such as smooth pursuit, optokinetic responses, and the slow phase of saccades) rely heavily on vestibular input (either directly from the vestibular nuclei, or indirectly via the cerebellum). General schematics of some of these pathways are depicted in the Figures below, from Haines (Haines 2006).
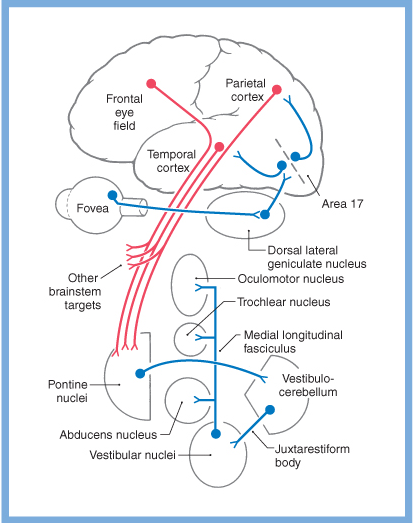 |
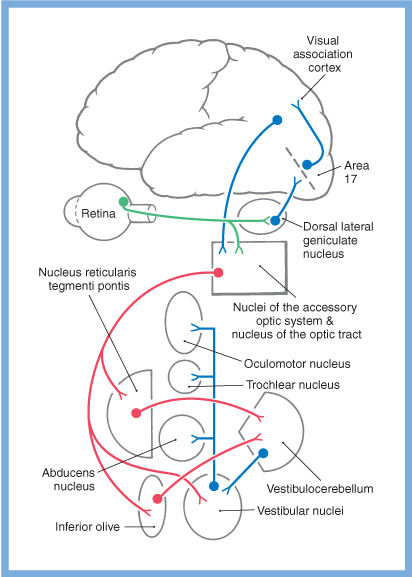 |
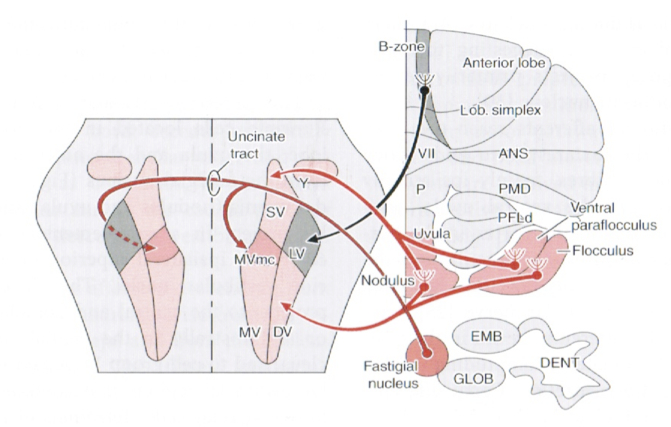
Efferent projections from vestibular nuclei to cerebellum
The vestibular nuclei also have substantial projections to various areas of the cerebellum, including (Haines 2006):
- Primary vestibulocerebellar fibers project to the:
- Dentate nucleus
- Nodulus
- Uvula
- Flocculus
- Secondary vestibulocerebellar fibers project to the:
- Flocculonodular lobe
- Uvula
- Parts of the paraflocculus
- Fastigial nuclei
- Dentate nuclei
Some of these projections are depicted in the Figure below, from Niewenhuys and colleagues (Nieuwenhuys et al. 2008).
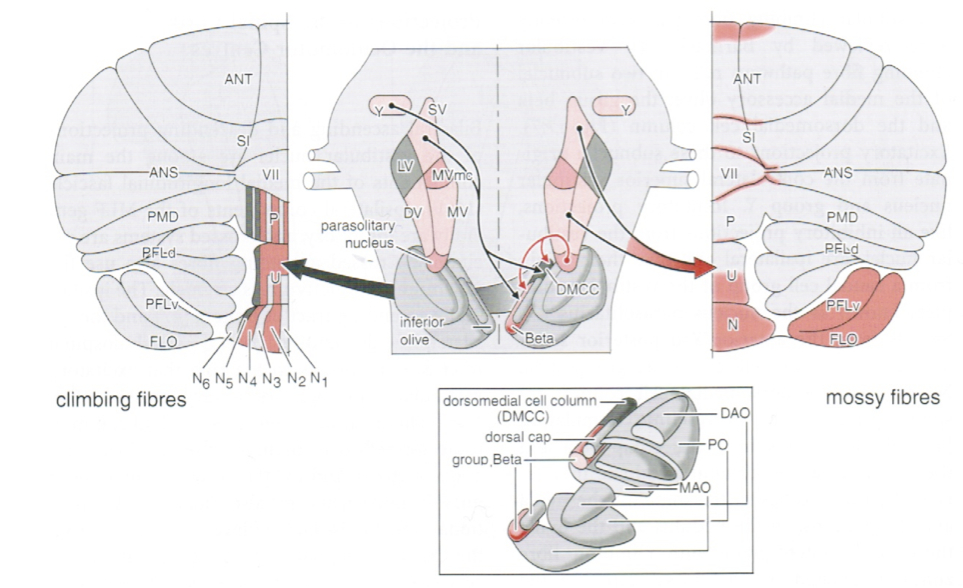
Efferent projections from vestibular nuclei to contralateral vestibular nuclei
The vestibular nuclei on one side have commissural projections to the contralateral vestibular nuclei, some of which are excitatory, others of which are inhibitory (Nieuwenhuys et al. 2008). These commissural projections appear to serve at least two purposes:
- They probably comprise the neural substrate for the velocity storage mechanism (Baloh and Kerber 2011; Bhansali et al. 1993).
- They probably facilitate central vestibular compensation during recovery from a vestibular injury (Haines 2006).
These commissural projections are illustrated in the Figure below, from Haines (Haines 2006).
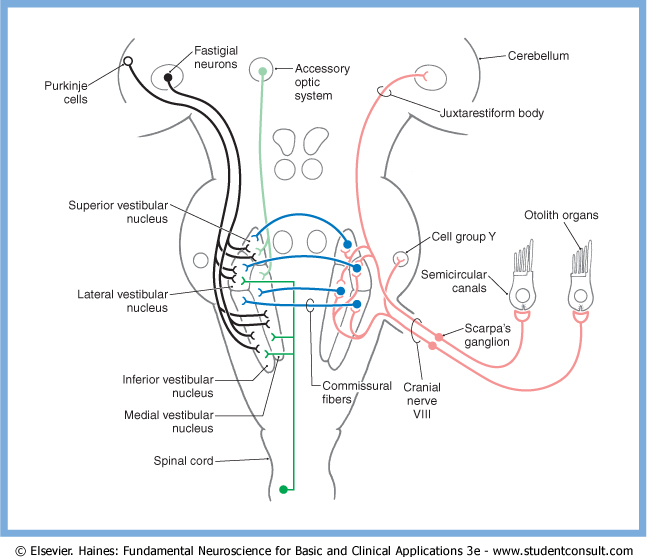
Efferent projections from vestibular nuclei to spinal cord
Projections from the vestibular nuclei reach the spinal cord via the medial and lateral vestibulospinal tracts, as shown in the Figure below from Haines (Haines 2006). These pathways influence muscle tone and mediate reflexive postural adjustments of the head, trunk and limbs. These projections contain both excitatory and inhibitory projections (Nieuwenhuys et al. 2008).

Neurons mostly in the lateral and inferior vestibular nuclei project through the lateral vestibulospinal tract, and terminate on alpha and gamma motor neurons in the ipsilateral spinal cord, releasing acetylcholine or glutamate.
Neurons in the medial vestibular nuclei (less in the inferior and lateral vestibular nuclei) project through the medial vestibulospinal tract. Some of these efferent projections remain ipsilateral, while others decussate. These projections provide both excitatory and inhibitory influence on cervical flexor and extensor motor neurons, thereby mediating the vestibulo-collic reflexes.
Efferent projections from vestibular nuclei to thalamus
Information about position and motion reaches consciousness through the vestibulo-thalamo-cortical network. Efferent projections from the superior, lateral and inferior vestibular nuclei on one side project bilaterally to two areas in the thalamus: the ventral posterolateral nucleus (VPL), and the ventral posteroinferior nucleus (VPI). Efferent projections from these vestibular nuclei on one side project ipsilaterally to two areas in the temporal lobe (in the base of the intraparietal sulcus and the base of the central sulcus) that comprise the vestibular cortex, which mediates conscious perception of vestibular stimuli. Some of these pathways are depicted in the Figure below, from Haines (Haines 2006).

Type 2 vestibular hair cells
In the auditory system, type 1 cochlear hair cells perform mechano-electrical transduction from the kinetic energy of oscillating pressure (i.e., sound) into afferent neurochemical signals. Type 2 cochlear hair cells, controlled by descending auditory pathways, modulate the activity of type 1 hair cells, functioning as a type of auditory equalizer.
The auditory and vestibular systems are phylogenetically related (Manley 2000), so it is unsurprising that the vestibular system also has type 2 vestibular hair cells, controlled by descending vestibular pathways.
Descending vestibular pathways originate primarily from the vestibular nuclei, but there also appear to be contributions from the reticular formation and perhaps other areas. These pathways project to the labyrinth, where they synapse mostly on type 2 vestibular hair cells, and less on type 1 vestibular hair cells. The Figure below provides a schematic of this system.
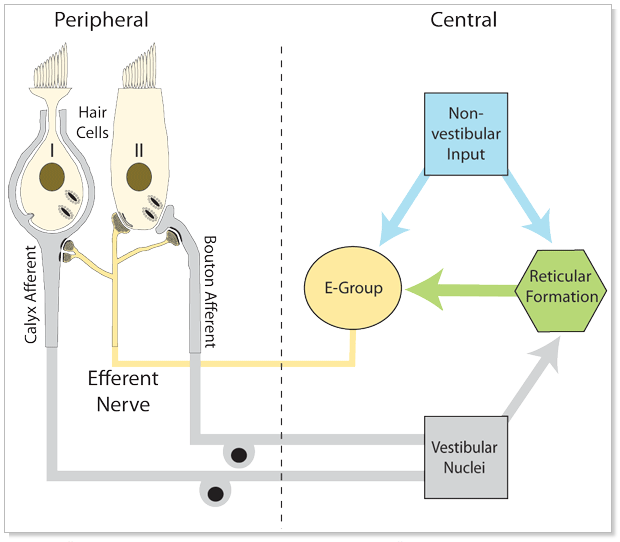
In some species, the type 2 vestibular hair cells can modulate the activity of type 1 vestibular hair cells in “both directions” (i.e., either augment or diminish the level of activity), analogous to the function of type 2 cochlear hair cells. The type 2 vestibular hair cells are incompletely understood in mammals, but they appear to serve primarily or exclusively an excitatory function (Schneider et al. 2021).
Neurotransmitters in the vestibular system
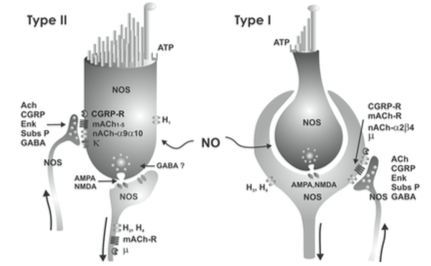
A broad array of neurotransmitters is used in the vestibular system, some of which are depicted in the Figures below, from Soto and colleagues (Soto et al. 2013).
 |
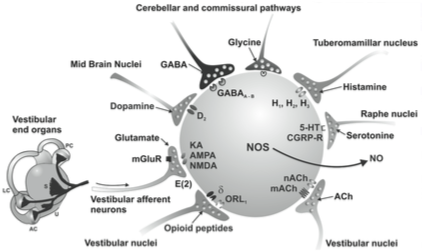 |
A non-exhaustive list of neurotransmitters in the vestibular system is provided below.
- Cerebellar cortico-vestibular fibers, project to several areas in the vestibular system and, like all output from the cerebellum, are inhibitory and release GABA (Haines 2004).
- Hypothalamic projections to the medial vestibular nucleus release histamine (Haines 2004).
- Other projections to the vestibular nuclei release dynorphin (Haines 2004).
- Vestibular projections to the cerebellum are mostly cholinergic (Nieuwenhuys et al. 2008).
- The nucleus prepositus hypoglossi sends projections to several nuclei in the superior olive where they release GABA and acetylcholine (Nieuwenhuys et al. 2008).
- Efferent projections from the vestibular nuclei through the lateral vestibulospinal tract that terminate on alpha and gamma motor neurons release glutamate and acetylcholine (Haines 2006) (Haines 2006, p. 362).
- Among descending vestibular pathways:
- Animal studies show that these terminate in the labyrinths and release acetyltransferase and cGRP (Nieuwenhuys et al. 2008).
- These have a predominantly excitatory influence of the afferent activity in the vestibular nerve and extend the dynamic range of the afferents during the large accelerations accompanying voluntary head movements (Nieuwenhuys et al. 2008).
References
Baloh RW (1998) Dizziness, hearing loss, and tinnitus. F.A. Davis Co., Philadelphia
Baloh RW, Kerber KA (2011) Clinical neurophysiology of the vestibular system, 4th edn. Oxford University Press, New York
Bhansali SA, Stockwell CW, Bojrab DI (1993) Oscillopsia in patients with loss of vestibular function. Otolaryngol Head Neck Surg 109: 120-5.
Blanks RH, Curthoys IS, Markham CH (1975) Planar relationships of the semicircular canals in man. Acta Otolaryngol 80: 185-96.
Bradshaw AP, Curthoys IS, Todd MJ, Magnussen JS, Taubman DS, Aw ST, Halmagyi GM (2010) A mathematical model of human semicircular canal geometry: a new basis for interpreting vestibular physiology. J Assoc Res Otolaryngol 11: 145-59. doi: 10.1007/s10162-009-0195-6
Cherchi M, Yacovino DA (2021) Histology and neuroanatomy suggest a unified mechanism to explain the distribution of lesion patterns in acute vestibular neuropathy. Experimental Brain Research 239: 1395–1399. doi: 10.1007/s00221-021-06094-9
Cortés-Domínguez I, Fernández-Seara MA, Perez-Fernandez N, Burguete J (2019) Systematic Method for Morphological Reconstruction of the Semicircular Canals Using a Fully Automatic Skeletonization Process. Applied Sciences 9. doi: 10.3390/app9224904
Curthoys IS (2017) The new vestibular stimuli: sound and vibration-anatomical, physiological and clinical evidence. Exp Brain Res 235: 957-972. doi: 10.1007/s00221-017-4874-y
Della Santina CC, Potyagaylo V, Migliaccio AA, Minor LB, Carey JP (2005) Orientation of human semicircular canals measured by three-dimensional multiplanar CT reconstruction. J Assoc Res Otolaryngol 6: 191-206. doi: 10.1007/s10162-005-0003-x
Dimiccoli M, Girard B, Berthoz A, Bennequin D (2013) Striola magica. A functional explanation of otolith geometry. J Comput Neurosci 35: 125-54. doi: 10.1007/s10827-013-0444-x
Haines DE (2004) Neuroanatomy: An Atlas of Structures, Sections, and Systems, 6th edn. Lippincott, Williams and Wilkins
Haines DE (2006) Fundamental neuroscience, 3rd edn. Churchill Livingstone, Philadelphia
Ifediba MA, Rajguru SM, Hullar TE, Rabbitt RD (2007) The role of 3-canal biomechanics in angular motion transduction by the human vestibular labyrinth. Ann Biomed Eng 35: 1247-63. doi: 10.1007/s10439-007-9277-y
Leigh RJ, Zee DS (2015) The neurology of eye movements, 5th edn. Oxford University Press, Oxford ; New York
Manley GA (2000) Cochlear mechanisms from a phylogenetic viewpoint. Proc Natl Acad Sci U S A 97: 11736-43. doi: 10.1073/pnas.97.22.11736
Miller EF, 2nd (1962) Counterrolling of the human eyes produced by head tilt with respect to gravity. Acta Otolaryngol 54: 479-501.
Nieuwenhuys R, Voogd J, Huijzen Cv (2008) The human central nervous system, 4th edn. Springer, New York
Sato H, Sando I, Takahashi H, Fujita S (1993) Torsion of the human semicircular canals and its influence on their angular relationships. Acta Otolaryngol 113: 171-5. doi: 10.3109/00016489309135787
Schneider GT, Lee C, Sinha AK, Jordan PM, Holt JC (2021) The mammalian efferent vestibular system utilizes cholinergic mechanisms to excite primary vestibular afferents. Sci Rep 11: 1231. doi: 10.1038/s41598-020-80367-1
Schünke M, Ross LM, Lamperti ED, Schulte E, Schumacher U (2007) Head and neuroanatomy. Thieme, Stuttgart ; New York
Soto E, Vega R, Sesena E (2013) Neuropharmacological basis of vestibular system disorder treatment. J Vestib Res 23: 119-37. doi: 10.3233/VES-130494
![]()