By Marcello Cherchi, MD PhD
For patients
The vestibulo-ocular reflex (VOR) is a function that keeps your eyes steady on a target even when your head moves. Many diseases can interfere with the proper functioning of the VOR, and some of these diseases can make a person feel disequilibrium. Your doctor may perform one or more bedside tests to screen for a problem with the VOR, but more definitive assessment may require testing with special equipment.
For clinicians
Practical summary
The vestibulo-ocular reflex (VOR) maintains an image stable on the retina despite rotational or translational movements of the head.
Physiology and neuroanatomy
The function of the vestibulo-ocular reflex (VOR) is to stabilize an image on the retina despite rotational and/or translational movements of the head (Rinaudo et al. 2019). The VOR consists of a the rotational VOR (which detects angular acceleration) and the translational VOR (which detects linear acceleration). The rotational VOR (rVOR) can be assessed to some degree with bedside testing, and in greater detail with instrumented otovestibular testing. The translational VOR (tVOR) is more difficult to assess, and usually requires instrumentation in a special research laboratory.
The Figure below, from Zee (Zee 2006), illustrates rotational and translational movements.
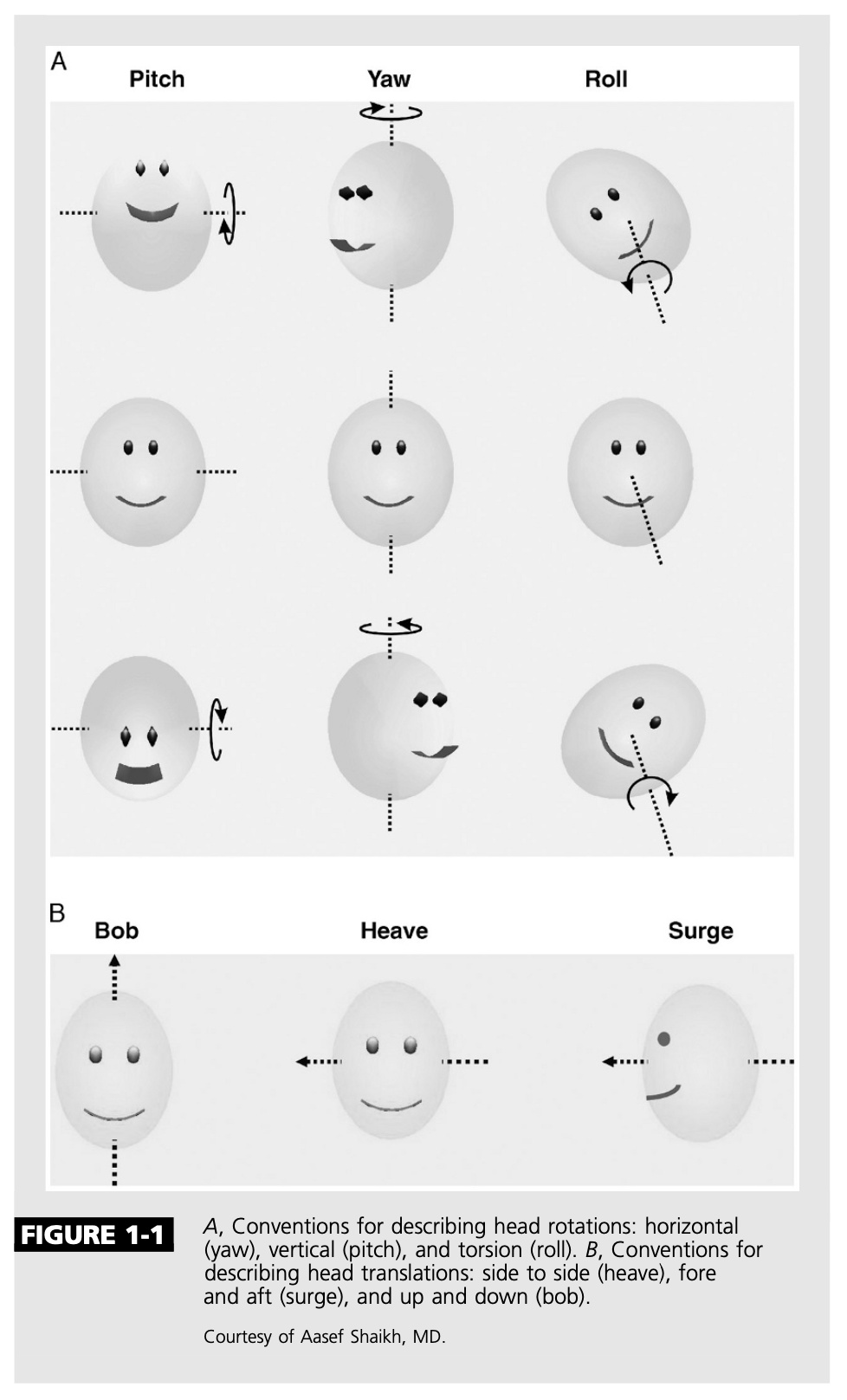
In tabular format:
|
Rotation around axis (angular movement) |
Translation along axis (linear movement) |
|
|
When acceleration around this axis occurs, the ocular motor responses are driven by the canal-ocular reflex |
When acceleration along this axis occurs, the ocular motor responses are driven by the otolith-ocular reflex |
|
|
Inter-aural axis (“left and right”) |
Pitch |
Heave |
|
Rostro-caudal axis (“up and down”) |
Yaw |
Bob |
|
Naso-occipital axis (“fore and aft”) |
Roll |
Surge |
The rotational vestibulo-ocular reflex (rVOR)
Ideally the rotational VOR (rVOR) should rotate the eyes within the orbits by an amount that is equal in magnitude, but opposite in direction, to that of the head, thereby offsetting the head movement and maintaining the point of regard stable on the fovea (in other words, the gain of eye rotation to head rotation is 1.0). The rVOR responds to pitch (rotation around the inter-aural axis), yaw (rotation around the rostro-caudal axis) and roll (rotation around the naso-occipital axis).
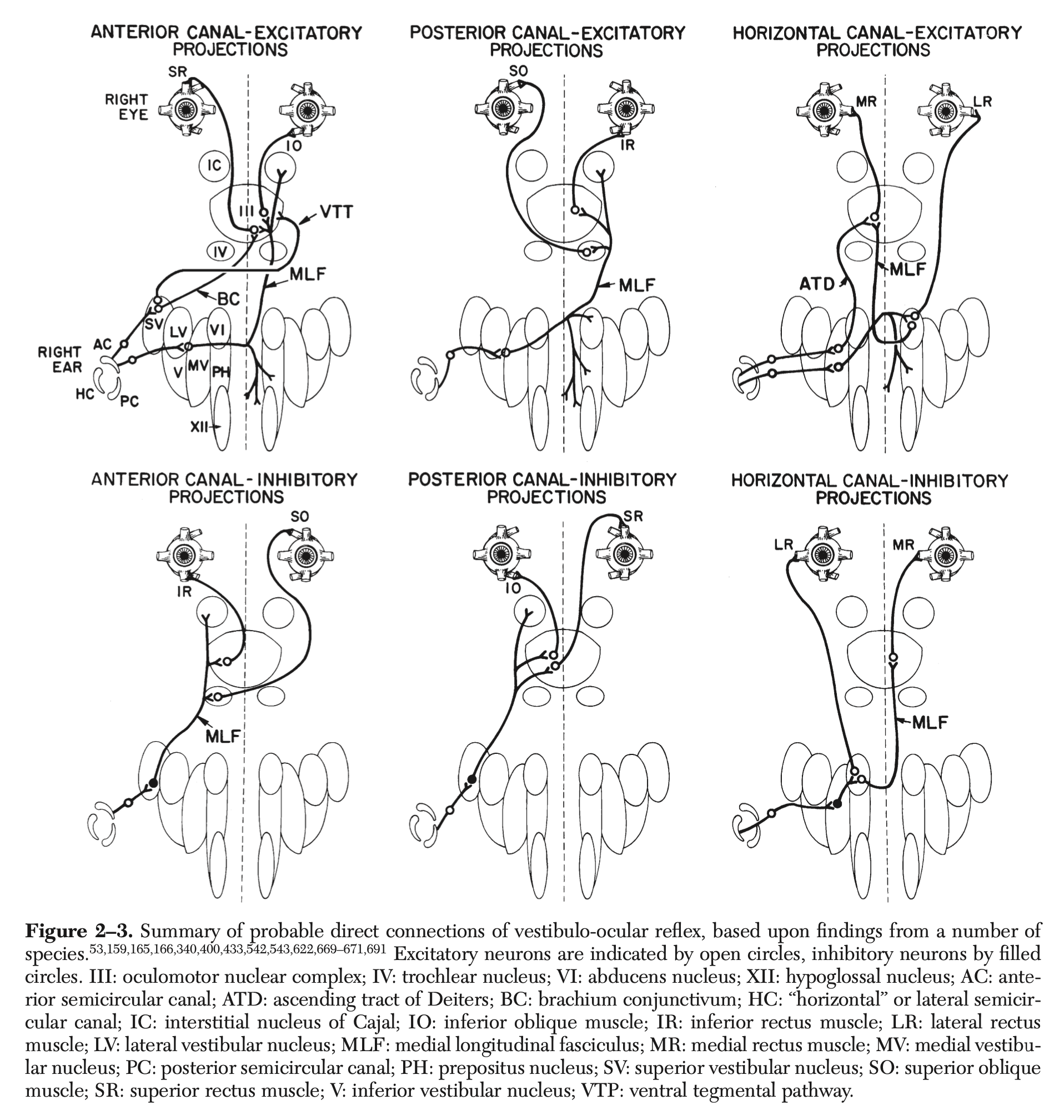
The neuroanatomy of the rVOR is well understood (Bronstein, Patel, Arshad 2015). During rotational acceleration of the head towards one side there is excitation of the ipsilateral lateral semicircular canal, and inhibition of the contralateral lateral semicircular canal. Afferent vestibular excitation from a given lateral semicircular canal passes to the vestibular (Scarpa’s) ganglion, through the superior division of the vestibular nerve and into the ipsilateral medial vestibular nucleus (MVN) situated in the mid dorsal portion of the lower pons and upper medulla. From the MVN there are some projections through the ascending tract of Dieters to the ipsilateral oculomotor nucleus which stimulates contraction of the medial rectus of the ipsilateral eye; there are other projections to the contralateral abducens nucleus which in turn stimulates contraction of the lateral rectus of the contralateral eye. In addition, from the MVN, there are projections to the ipsilateral abducens nucleus; from the abducens nucleus there are projections that inhibit the ipsilateral lateral rectus, and other projections to the contralateral oculomotor nucleus from which projections inhibit the contralateral medial rectus. Afferent vestibular inhibition from a given lateral semicircular canal will have effects opposite that described above.
The VOR can be assessed for any pair of coplanar semicircular canals; the relevant neuroanatomy is different for each canal (Bronstein, Patel, Arshad 2015). The Figure below, from Leigh and Zee (Leigh and Zee 2015), illustrates the neuroanatomy of all rotational vestibular ocular reflexes.
The VOR can adapt by altering its gain (Watanabe, Hattori, Koizuka 2003). The most common circumstance in which such adaptation occurs is when a patient dons refractive lenses (spectacles or contact lenses) to correct for hyperopia (farsightedness) or myopia (nearsightedness) (Cannon et al. 1985). Such adaptive changes in the rVOR gain occur relatively quickly and can be durable even after the refraction is discontinued (Mahfuz et al. 2020). This phenomenon applies mostly to spectacles, because contact lenses move with the eyeball and thus do not induce adaptive changes to the rVOR (Eggers and Zee 2003).
Because of this, if a patient habitually uses lenses (whether spectacles or contact lenses) for visual correction, then they should also wear them during testing (bedside or instrumented) of the rotational vestibulo-ocular reflex, otherwise one may obtain false positive results.
What tests can assess the rotational vestibulo-ocular reflex?
On bedside testing, the middle frequency range of the vestibular tuning spectrum can be evaluated with the ophthalmoscope test and with dynamic visual acuity testing, while the high frequency range can be tested with head impulse testing.
On instrumented otovestibular testing, the low frequency range of the vestibular tuning spectrum can be evaluated with caloric testing, the middle frequency range can be evaluated with rotatory chair testing, and the high frequency can be evaluated with video head impulse testing.
The translational vestibulo-ocular reflex (tVOR)
The translational vestibulo-ocular reflex (tVOR), also called the linear VOR or otolith-ocular reflex, maintains an image stable on the retina despite translational movements of the head — mostly heave (translation along the inter-aural axis) and bob (translation along the rostro-caudal axis), less surge (translation along the naso-occipital axis).
The translational VOR (tVOR) responds to translational movements, specifically linear acceleration, along the inter-aural (heave) axis (Crane et al. 2003; King et al. 2019; Ramat, Straumann, Zee 2005; Ramat and Zee 2002, 2003; Tian, Crane, Demer 2003; Tian et al. 2002; Walker, Shelhamer, Zee 2004), naso-occipital (surge) axis (Seidman, Paige, Tomko 1999; Semrau, Wei, Angelaki 2006; Tian, Mokuno, Demer 2006), rostro-caudal (bob) axis (Bush and Miles 1996; Cheng and Walker 2016; Kutz et al. 2020; Liao et al. 2008; Liao et al. 2009) or oblique axes (Tomko and Paige 1992). The tVOR is mediated by the otolith system, which feeds into the neural integrator (Hegemann et al. 2000) and can be modulated by viewing distance (Busettini, Miles, Schwarz 1991; Busettini et al. 1994; Cheng and Walker 2016; King et al. 2019; Paige et al. 1998; Schwarz and Miles 1991; Wei and Angelaki 2004) and other visual influences (Dits, King, van der Steen 2013; Gianna, Gresty, Bronstein 2000; Hashiba et al. 1995; Liao et al. 2011; Paige et al. 1998), and by somatosensory inputs (Kutz et al. 2020). The tVOR can be adversely affected by various pathologies such as unilateral vestibular weakness (Crane et al. 2005; Lempert et al. 1998; Lempert, Gresty, Bronstein 1999; Tian, Ishiyama, Demer 2007), bilateral vestibular weakness (Lempert, Gresty, Bronstein 1999), cerebellar disorders (Anastasopoulos et al. 1998; Baloh, Yue, Demer 1995; Crane, Tian, Demer 2000) and skew deviation (Schlenker et al. 2009). Many natural head movements comprise a mix of rotational and translational movements, thereby activating rVOR and tVOR simultaneously (Anastasopoulos et al. 1996; Angelaki, Green, Dickman 2001; Branoner and Straka 2018; Crane and Demer 1997, 1999; Crane, Tian, Demer 2000; Koizuka et al. 2000; Laurens, Straumann, Hess 2010; McCrea and Chen-Huang 1999; Meng et al. 2005; Paige et al. 1998; Ramaioli et al. 2019; Schneider et al. 2014).
What tests can assess the translational vestibulo-ocular reflex?
The tVOR is more difficult to study than the rVOR. Accurate study requires specialized equipment largely restricted to research laboratories, such as human centrifuges, Moog platforms, parallel swings, and linear accelerating sleds.
Although the ocular motor responses of the tVOR are harder to study, it is still possible to assess the otolith system using cervical vestibular evoked myogenic potentials and ocular vestibular evoked myogenic potentials.
Diseases that affect the vestibulo-ocular reflex
Any lesion affecting any of the neuroanatomical pathways described earlier can cause vestibular weakness and thereby influence the vestibulo-ocular reflex — and here we use the term “lesion” loosely, since not only focal lesions, but also systemic issues (such as pharmacologic vestibular suppression) can cause such affectation.
It is less common for the vestibulo-ocular reflex to be abnormally elevated (i.e., the gain is >1). Examples include some cerebellar ataxias (Zee et al. 1976) and nitrous oxide toxicity (Hamilton et al. 1986).
References
Anastasopoulos D, Gianna CC, Bronstein AM, Gresty MA (1996) Interaction of linear and angular vestibulo-ocular reflexes of human subjects in response to transient motion. Exp Brain Res 110: 465-72. doi: 10.1007/bf00229146
Anastasopoulos D, Haslwanter T, Fetter M, Dichgans J (1998) Smooth pursuit eye movements and otolith-ocular responses are differently impaired in cerebellar ataxia. Brain 121 ( Pt 8): 1497-505. doi: 10.1093/brain/121.8.1497
Angelaki DE, Green AM, Dickman JD (2001) Differential sensorimotor processing of vestibulo-ocular signals during rotation and translation. J Neurosci 21: 3968-85. doi: 10.1523/jneurosci.21-11-03968.2001
Baloh RW, Yue Q, Demer JL (1995) The linear vestibulo-ocular reflex in normal subjects and patients with vestibular and cerebellar lesions. J Vestib Res 5: 349-61.
Branoner F, Straka H (2018) Semicircular Canal Influences on the Developmental Tuning of the Translational Vestibulo-Ocular Reflex. Front Neurol 9: 404. doi: 10.3389/fneur.2018.00404
Bronstein AM, Patel M, Arshad Q (2015) A brief review of the clinical anatomy of the vestibular-ocular connections-how much do we know? Eye (Lond) 29: 163-70. doi: 10.1038/eye.2014.262
Busettini C, Miles FA, Schwarz U (1991) Ocular responses to translation and their dependence on viewing distance. II. Motion of the scene. J Neurophysiol 66: 865-78. doi: 10.1152/jn.1991.66.3.865
Busettini C, Miles FA, Schwarz U, Carl JR (1994) Human ocular responses to translation of the observer and of the scene: dependence on viewing distance. Exp Brain Res 100: 484-94. doi: 10.1007/bf02738407
Bush GA, Miles FA (1996) Short-latency compensatory eye movements associated with a brief period of free fall. Exp Brain Res 108: 337-40. doi: 10.1007/bf00228107
Cannon SC, Leigh RJ, Zee DS, Abel LA (1985) The effect of the rotational magnification of corrective spectacles on the quantitative evaluation of the VOR. Acta Otolaryngol 100: 81-8. doi: 10.3109/00016488509108591
Cheng RC, Walker MF (2016) Vertical head translation impairs dynamic visual acuity during near viewing. J Vestib Res 26: 417-423. doi: 10.3233/ves-160596
Crane BT, Demer JL (1997) Human gaze stabilization during natural activities: translation, rotation, magnification, and target distance effects. J Neurophysiol 78: 2129-44. doi: 10.1152/jn.1997.78.4.2129
Crane BT, Demer JL (1999) A linear canal-otolith interaction model to describe the human vestibulo-ocular reflex. Biol Cybern 81: 109-18. doi: 10.1007/s004220050548
Crane BT, Tian J, Wiest G, Demer JL (2003) Initiation of the human heave linear vestibulo-ocular reflex. Exp Brain Res 148: 247-55. doi: 10.1007/s00221-002-1301-8
Crane BT, Tian JR, Demer JL (2000) Initial vestibulo-ocular reflex during transient angular and linear acceleration in human cerebellar dysfunction. Exp Brain Res 130: 486-96. doi: 10.1007/s002219900266
Crane BT, Tian JR, Ishiyama A, Demer JL (2005) Initiation and cancellation of the human heave linear vestibulo-ocular reflex after unilateral vestibular deafferentation. Exp Brain Res 161: 519-26. doi: 10.1007/s00221-004-2089-5
Dits J, King WM, van der Steen J (2013) Scaling of compensatory eye movements during translations: virtual versus real depth. Neuroscience 246: 73-81. doi: 10.1016/j.neuroscience.2013.04.029
Eggers SD, Zee DS (2003) Evaluating the dizzy patient: bedside examination and laboratory assessment of the vestibular system. Semin Neurol 23: 47-58. doi: 10.1055/s-2003-40751
Gianna CC, Gresty MA, Bronstein AM (2000) The human linear vestibulo-ocular reflex to transient accelerations: visual modulation of suppression and enhancement. J Vestib Res 10: 227-38.
Hamilton K, Porlier G, Landolt J, Fraser W, Fowler B (1986) Effects of inert gas narcosis on the vestibular ocular reflex. Undersea Biomed Res 13: 355-9.
Hashiba M, Watanabe S, Watabe H, Matsuoka T, Baba S, Wada Y, Clarke AH, Scherer H, Sekiguchi C (1995) Imaginary gaze effects on eye movements induced by linear acceleration: involvement of vestibular induced smooth pursuit eye movement. Acta Otolaryngol Suppl 520 Pt 2: 372-6. doi: 10.3109/00016489509125274
Hegemann S, Shelhamer M, Kramer PD, Zee DS (2000) Adaptation of the phase of the human linear vestibulo-ocular reflex (LVOR) and effects on the oculomotor neural integrator. J Vestib Res 10: 239-47.
King S, Benoit C, Bandealy N, Karmali F (2019) The influence of target distance on perceptual self-motion thresholds and the vestibulo-ocular reflex during interaural translation. Prog Brain Res 248: 197-208. doi: 10.1016/bs.pbr.2019.04.037
Koizuka I, Katsumi N, Hattori K, Okada T, Kato I (2000) Effect of adaptive plasticity of linear vestibulo-ocular reflex upon angular vestibulo-ocular reflex. Auris Nasus Larynx 27: 89-93. doi: 10.1016/s0385-8146(99)00077-2
Kutz DF, Kolb FP, Glasauer S, Straka H (2020) Somatosensory Influence on Platform-Induced Translational Vestibulo-Ocular Reflex in Vertical Direction in Humans. Front Neurol 11: 332. doi: 10.3389/fneur.2020.00332
Laurens J, Straumann D, Hess BJ (2010) Processing of angular motion and gravity information through an internal model. J Neurophysiol 104: 1370-81. doi: 10.1152/jn.00143.2010
Leigh RJ, Zee DS (2015) The neurology of eye movements, 5th edn. Oxford University Press, Oxford ; New York
Lempert T, Gianna C, Brookes G, Bronstein A, Gresty M (1998) Horizontal otolith-ocular responses in humans after unilateral vestibular deafferentation. Exp Brain Res 118: 533-40. doi: 10.1007/s002210050309
Lempert T, Gresty MA, Bronstein AM (1999) Horizontal linear vestibulo-ocular reflex testing in patients with peripheral vestibular disorders. Ann N Y Acad Sci 871: 232-47. doi: 10.1111/j.1749-6632.1999.tb09188.x
Liao K, Schneider RM, Yaniglos SS, Bertolini G, Glendinning P, Sawyer RN, Jr., Reschke M, Leigh RJ (2011) Visual and vestibular determinants of the translational vestibulo-ocular reflex. Ann N Y Acad Sci 1233: 263-70. doi: 10.1111/j.1749-6632.2011.06148.x
Liao K, Walker MF, Joshi A, Reschke M, Leigh RJ (2008) Vestibulo-ocular responses to vertical translation in normal human subjects. Exp Brain Res 185: 553-62. doi: 10.1007/s00221-007-1181-z
Liao K, Walker MF, Joshi A, Reschke M, Strupp M, Leigh RJ (2009) The human vertical translational vestibulo-ocular reflex. Normal and abnormal responses. Ann N Y Acad Sci 1164: 68-75. doi: 10.1111/j.1749-6632.2008.03711.x
Mahfuz MM, Schubert MC, Figtree WVC, Migliaccio AA (2020) Retinal Image Slip Must Pass the Threshold for Human Vestibulo-Ocular Reflex Adaptation. J Assoc Res Otolaryngol 21: 277-285. doi: 10.1007/s10162-020-00751-6
McCrea RA, Chen-Huang C (1999) Signal processing related to the vestibulo-ocular reflex during combined angular rotation and linear translation of the head. Ann N Y Acad Sci 871: 65-80. doi: 10.1111/j.1749-6632.1999.tb09176.x
Meng H, Green AM, Dickman JD, Angelaki DE (2005) Pursuit–vestibular interactions in brain stem neurons during rotation and translation. J Neurophysiol 93: 3418-33. doi: 10.1152/jn.01259.2004
Paige GD, Telford L, Seidman SH, Barnes GR (1998) Human vestibuloocular reflex and its interactions with vision and fixation distance during linear and angular head movement. J Neurophysiol 80: 2391-404. doi: 10.1152/jn.1998.80.5.2391
Ramaioli C, Cuturi LF, Ramat S, Lehnen N, MacNeilage PR (2019) Vestibulo-Ocular Responses and Dynamic Visual Acuity During Horizontal Rotation and Translation. Front Neurol 10: 321. doi: 10.3389/fneur.2019.00321
Ramat S, Straumann D, Zee DS (2005) Interaural translational VOR: suppression, enhancement, and cognitive control. J Neurophysiol 94: 2391-402. doi: 10.1152/jn.01328.2004
Ramat S, Zee DS (2002) Translational VOR responses to abrupt interaural accelerations in normal humans. Ann N Y Acad Sci 956: 551-4. doi: 10.1111/j.1749-6632.2002.tb02879.x
Ramat S, Zee DS (2003) Ocular motor responses to abrupt interaural head translation in normal humans. J Neurophysiol 90: 887-902. doi: 10.1152/jn.01121.2002
Rinaudo CN, Schubert MC, Figtree WVC, Todd CJ, Migliaccio AA (2019) Human vestibulo-ocular reflex adaptation is frequency selective. J Neurophysiol 122: 984-993. doi: 10.1152/jn.00162.2019
Schlenker M, Mirabella G, Goltz HC, Kessler P, Blakeman AW, Wong AM (2009) The linear vestibulo-ocular reflex in patients with skew deviation. Invest Ophthalmol Vis Sci 50: 168-74. doi: 10.1167/iovs.08-2254
Schneider R, Liao K, Walker MF, Bronstein A, Leigh RJ (2014) Behavior of the human translational vestibulo-ocular reflex during simultaneous head translation and rotation. J Vestib Res 24: 329-33. doi: 10.3233/ves-140522
Schwarz U, Miles FA (1991) Ocular responses to translation and their dependence on viewing distance. I. Motion of the observer. J Neurophysiol 66: 851-64. doi: 10.1152/jn.1991.66.3.851
Seidman SH, Paige GD, Tomko DL (1999) Adaptive plasticity in the naso-occipital linear vestibulo-ocular reflex. Exp Brain Res 125: 485-94. doi: 10.1007/s002210050706
Semrau JA, Wei M, Angelaki DE (2006) Scaling of the fore-aft vestibulo-ocular reflex by eye position during smooth pursuit. J Neurophysiol 96: 936-40. doi: 10.1152/jn.00185.2006
Tian JR, Crane BT, Demer JL (2003) Vestibular catch-up saccades augmenting the human transient heave linear vestibulo-ocular reflex. Exp Brain Res 151: 435-45. doi: 10.1007/s00221-003-1492-7
Tian JR, Crane BT, Wiest G, Demer JL (2002) Effect of aging on the human initial interaural linear vestibulo-ocular reflex. Exp Brain Res 145: 142-9. doi: 10.1007/s00221-002-1111-z
Tian JR, Ishiyama A, Demer JL (2007) Effect of unilateral vestibular deafferentation on the initial human vestibulo-ocular reflex to surge translation. Exp Brain Res 176: 575-87. doi: 10.1007/s00221-006-0636-y
Tian JR, Mokuno E, Demer JL (2006) Vestibulo-ocular reflex to transient surge translation: complex geometric response ablated by normal aging. J Neurophysiol 95: 2042-54. doi: 10.1152/jn.00635.2005
Tomko DL, Paige GD (1992) Linear vestibuloocular reflex during motion along axes between nasooccipital and interaural. Ann N Y Acad Sci 656: 233-41. doi: 10.1111/j.1749-6632.1992.tb25212.x
Walker MF, Shelhamer M, Zee DS (2004) Eye-position dependence of torsional velocity during interaural translation, horizontal pursuit, and yaw-axis rotation in humans. Vision Res 44: 613-20. doi: 10.1016/j.visres.2003.10.009
Watanabe S, Hattori K, Koizuka I (2003) Flexibility of vestibulo-ocular reflex adaptation to modified visual input in human. Auris Nasus Larynx 30 Suppl: S29-34. doi: 10.1016/s0385-8146(02)00134-7
Wei M, Angelaki DE (2004) Viewing distance dependence of the vestibulo-ocular reflex during translation: extra-otolith influences. Vision Res 44: 933-42. doi: 10.1016/j.visres.2003.11.016
Zee DS (2006) Pathophysiology of vestibular symptoms and signs: the clinical examination. Continuum 12: 13-32.
Zee DS, Yee RD, Cogan DG, Robinson DA, Engel WK (1976) Ocular motor abnormalities in hereditary cerebellar ataxia. Brain 99: 207-34. doi: 10.1093/brain/99.2.207
![]()