By Marcello Cherchi, MD PhD
For patients
The bedside head impulse test (bHIT) is a bedside examination maneuver that assesses the neurological circuits that connect part of the inner ear’s gyroscope function to certain eye movements, and can help diagnose some causes of disequilibrium. During this test the doctor holds the sides of your head firmly and turns your head with small, quick movements to one side or the other, while observing for certain eye movements. The test is not uncomfortable. The test takes about one minute. You do not need any special preparation for this test.
For clinicians
Overview
The rotational vestibulo-ocular reflex (rVOR) maintains an image stable on the retina despite rotational movements of the head by rotating the eyes an equal amount but in the opposite direction of head movement. The high frequency range of the vestibular tuning spectrum can be evaluated with head impulse test (HIT), sometimes also called the head thrust test or the Halmagyi-Curthoys test. In this section we will refer to this as the bedside head impulse test (bHIT) in order to distinguish it from the computerized version called the video head impulse testing (vHIT). The bHIT is sensitive to both unilateral and bilateral vestibular weakness.
Introduction
There are several methods for assessing the rotational vestibulo-ocular reflex (rVOR) at the bedside, including the head impulse test, dynamic visual acuity testing and the ophthalmoscope test. The head impulse test assesses the high end of the vestibular tuning spectrum, while dynamic visual acuity testing and the ophthalmoscope test assess the middle frequency range.
The significance of the head impulse test was first recognized by Halmagyi and Curthoys (Halmagyi and Curthoys 1988) using the scleral search coil technique. However, investigators soon began exploring whether a non-instrumented version of the test had clinical value.
Physiology and neuroanatomy
The function of the vestibulo-ocular reflex (VOR) is to stabilize an image on the retina despite rotational and/or translational movements of the head (Rinaudo et al. 2019). Ideally the rotational VOR (rVOR) should rotate the eyes within the orbits by an amount that is equal in magnitude, but opposite in direction, to that of the head, thereby offsetting the head movement and maintaining the point of regard stable on the fovea (in other words, the gain of eye rotation to head rotation is 1.0).
The neuroanatomy of the rVOR is well understood (Bronstein et al. 2015). Note that the VOR can be assessed for any pair of coplanar semicircular canals; the relevant neuroanatomy is different for each canal (Bronstein et al. 2015). The bHIT is usually employed to assess the function of the lateral semicircular canals. In principle a similar technique could be used to assess the vertical canals, but in clinical practice this is rarely performed.
The bHIT assesses the high frequencies of the vestibular tuning spectrum, and this has two consequences. First, the high acceleration — on the order of 10,000 deg/sec/sec (Jorns-Haderli et al. 2007) — can saturate the signals generated by the semicircular canals. Second, because of Ewald’s second law (Ewald 1892), unilateral vestibular hypofunction is more likely to bring out an asymmetry between the function of the coplanar canals. Jorns-Haderli and colleagues explain this as follows:
“Head impulses mainly drive the short latency, oligosynaptic VOR pathways from the semicircular canals to the extraocular muscles. Polysynaptic pathways via the cerebellum are less efficient in transmitting such high acceleration vestibular stimuli. The oligosynaptic pathways show distinct non-linear properties in that the contribution of the signals from the excited semicircular canals to the ocular motor response is greater than the contribution of the signals from the inhibited semicircular canals. This principle, known as Ewald’s second law, is probably the result of a non-linear pathway, which during high accelerations is driven into inhibitory cut-off on the side of inhibited semicircular canals. In the case of unilateral peripheral vestibular hypofunction, Ewald’s second law results in an asymmetric gain of the VOR (i.e., the gain during high acceleration head rotations towards the lesioned side is lower than towards the healthy side)” (Jorns-Haderli et al. 2007).
These characteristics are what make the high frequency bHIT more likely to detect unilateral vestibular hypofunction than medium frequency bedside tests (such as dynamic visual acuity testing and the ophthalmoscope test) which are more effective at detecting bilateral vestibular weakness.
Equipment needed
No special equipment is needed to perform bedside head impulse testing.
How to perform the test
The patient is seated. The examiner stands facing the patient and instructs the patient to maintain gaze on the examiner’s nose, which should be about 50 cm from the patient’s eyes (Yip et al. 2016) on the patient’s primary position of gaze. The examiner firmly grasps both sides of the patient’s head and executes quick turns (randomly to one side or the other) consisting of very abrupt impulses (acceleration-deceleration rotations of the head around the rostro-caudal axis) that are of high acceleration (in the range of 10,000 deg/sec/sec) but small in the amplitude; authors cite different amplitudes of total excursion off the midsagittal plane, from 10-20 degrees (Jorns-Haderli et al. 2007) to 15-20 degrees (Yip et al. 2016). The examiner should repeat these impulses enough times to conclude whether the eyes get “dragged off target” by the impulses, and thus whether overt compensatory saccades (see below) are present or absent.
Despite the theoretical concern about bHIT causing cervical arterial dissections (Ullman and Edlow 2010), there is no literature to support this (Kaski and Bronstein 2012b).
What this test assesses
The bHIT as described above assesses the high frequency end of the vestibular tuning spectrum of the rotational VOR of the horizontal semicircular canals and the corresponding neuroanatomical pathways of that component of the VOR.
How to interpret the test results
In a person with an intact horizontal rVOR, during the bHIT towards a given side, the patient’s eyes will remain fixated on the examiner’s nose despite the head rotation. This is considered a normal result.
In a patient with a deficient horizontal rVOR on a given side, during the bHIT, the head’s rotation toward that side will “drag the eyes along with it,” after which (because the patient had been instructed to maintain gaze fixed on the examiner’s nose) there will be a corrective saccade away from the lesioned side, bringing the gaze back on target. This observable saccade is called an “overt compensatory saccade,” where the term “overt” refers to the fact that the saccade occurs after the head movement is complete. This is considered a “positive bHIT” on whichever side the impulse was directed towards, and this finding suggests a hypoactive high frequency horizontal rVOR on that side.
The Figure below, from Kaski and Bronstein (Kaski and Bronstein 2012a), is a top view schematic of the bHIT starting position (left panel), normal leftward bHIT (center panel) and abnormal leftward bHIT (right panel).
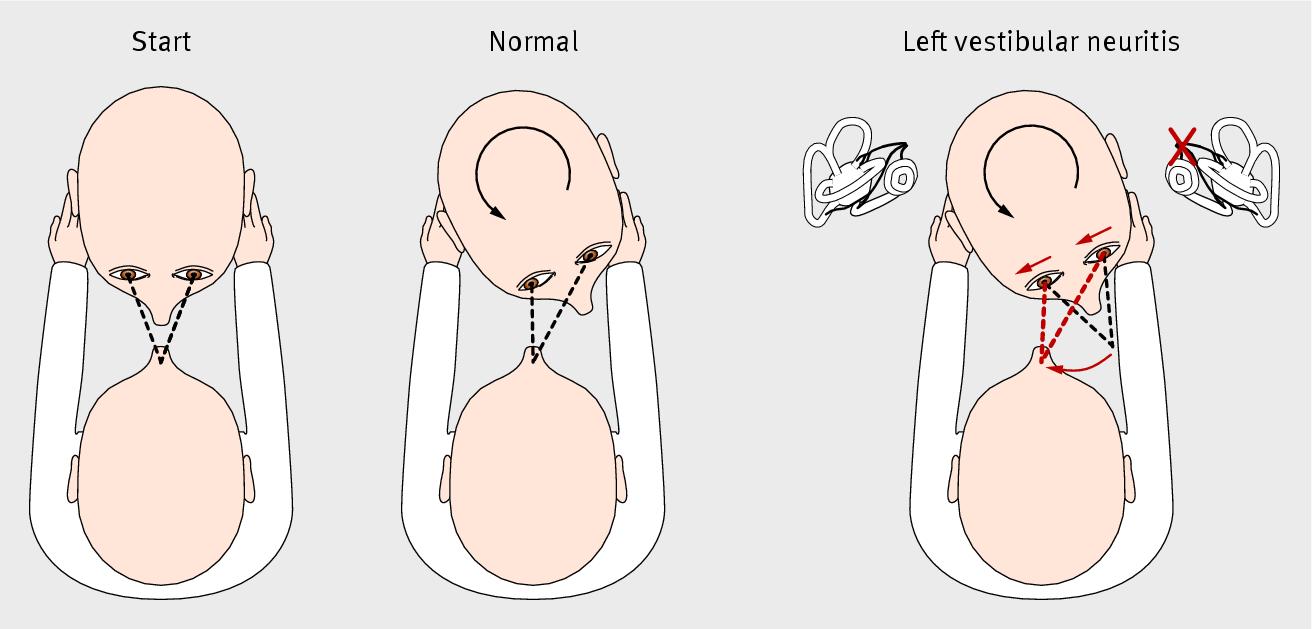
Early studies (Beynon et al. 1998; Harvey et al. 1997; Perez and Rama-Lopez 2003) criticized head impulse testing as less sensitive compared to caloric testing. However, as Jorns-Haderli and colleagues point out (Jorns-Haderli et al. 2007), this is not a fair comparison, because caloric testing assesses the very low end of the vestibular tuning frequency spectrum, whereas the head impulse test assesses the very high end of the vestibular tuning frequency spectrum — in other words, caloric testing and horizontal head impulse testing, although both involve activity of the lateral semicircular canals, they are testing different things.
Jorns-Haderli and colleagues state that the more informative comparison would be between bedside HIT (bHIT) and video HIT (vHIT). Jorns-Haderli and colleagues (Jorns-Haderli et al. 2007) conducted a study making just such a comparison, using vHIT as the criterion standard, and reported that, “On average, bHIT sensitivity was 69.9% and bHIT specificity 67.05%.” A similar study by Yip and colleagues (Yip et al. 2016) found that bHIT had a sensitivity of 66.0% and specificity of 86.2%.
Another point about bHIT is that this maneuver is not easy to perform or interpret. Jorns-Haderli and colleagues (Jorns-Haderli et al. 2007) note clear differences between “experts” and “non-experts” in the bHIT’s sensitivity and specificity. Lelli and colleagues (Lelli et al. 2019) report that final year otolaryngology residents did not perform or interpret the test adequately.
Contraindications
Although there is no literature supporting the concern that bHIT can cause or worsen cervical arterial dissection (Kaski and Bronstein 2012b; Ullman and Edlow 2010), this theoretical possibility still makes many clinicians shy of this procedure. Our own practice is to refrain from performing bHIT in patients with risk factors such as connective tissue diseases or a prior history of carotid or vertebral artery dissection.
Limitations
This test requires that the patient have sufficient cervical rotational motion. Patients with cervical vertebral fusions, or significant cervical arthritis or cervicalgia, or other limitations on cervical range of motion, may be unable to perform this test.
Pitfalls
This test may be difficult to interpret, or uninterpretable, in patients with strabismus, or in patients whose visual impairment precludes them from foveating a target (in this case, the examiner’s nose) adequately.
The diagnostic eye movements may be difficult to see by bHIT. Some clinicians have advocated recording the patient’s eye movements using the slow-motion feature of a smartphone, such as Dr. Brian Cohn (https://epmonthly.com/article/take-a-hints/) and Dr. Scott Weingart (https://emcrit.org/emcrit/iphone-hit/). This also has the advantage of being able to identify “covert compensatory saccades” (corrective saccades that occur during the movement of the head impulse). We regard this as an ingenious use for this technology. We would add that when one is interested in this level of diagnostic detail, it is probably more fruitful to do the instrumented version of head impulse testing called video head impulse testing (vHIT).
Diseases that may be diagnosed by this test
The bedside head impulse test of the horizontal rotational vestibulo-ocular reflex may help identify unilateral vestibular weakness or bilateral vestibular weakness, each of which in turn may have many etiologies. Yip and colleagues state that, “an abnormal VOR was easier to detect if the deficit was unilateral with low gain and large asymmetry between the two labyrinths, or there was bilateral severe vestibular hypofunction” (Yip et al. 2016).
Additional notes
The bHIT is one component of a broader protocol called HINTs (head impulse, nystagmus, test of skew) that has been studied in the acute vestibular syndrome for differentiating peripheral vestibular disease (usually vestibular neuritis) from central vestibular disease (usually posterior fossa stroke).
The bedside head impulse test (bHIT) has a computerized version called the video head impulse testing (vHIT) which we discuss elsewhere.
References
Beynon GJ, Jani P, Baguley DM (1998) A clinical evaluation of head impulse testing. Clin Otolaryngol Allied Sci 23: 117-22. doi: 10.1046/j.1365-2273.1998.00112.x
Bronstein AM, Patel M, Arshad Q (2015) A brief review of the clinical anatomy of the vestibular-ocular connections-how much do we know? Eye (Lond) 29: 163-70. doi: 10.1038/eye.2014.262
Ewald EJR (1892) Physiologische Untersuchungen über das Endorgan des Nervus octavus. Bergmann, Wiesbaden
Halmagyi GM, Curthoys IS (1988) A clinical sign of canal paresis. Arch Neurol 45: 737-9. doi: 10.1001/archneur.1988.00520310043015.
Harvey SA, Wood DJ, Feroah TR (1997) Relationship of the head impulse test and head-shake nystagmus in reference to caloric testing. Am J Otol 18: 207-13.
Jorns-Haderli M, Straumann D, Palla A (2007) Accuracy of the bedside head impulse test in detecting vestibular hypofunction. J Neurol Neurosurg Psychiatry 78: 1113-8. doi: 10.1136/jnnp.2006.109512
Kaski D, Bronstein AM (2012a) Making a diagnosis in patients who present with vertigo. BMJ : British Medical Journal 345: e5809. doi: 10.1136/bmj.e5809
Kaski D, Bronstein AM (2012b) Response to: Making a diagnosis in patients who present with vertigo. BMJ: e5809. doi: 10.1136/bmj.e5809
Lelli DA, Tse D, Vaccani JP (2019) Competence of final year otolaryngology residents with the bedside head impulse test. J Otolaryngol Head Neck Surg 48: 7. doi: 10.1186/s40463-019-0326-y
Perez N, Rama-Lopez J (2003) Head-impulse and caloric tests in patients with dizziness. Otol Neurotol 24: 913-7.
Rinaudo CN, Schubert MC, Figtree WVC, Todd CJ, Migliaccio AA (2019) Human vestibulo-ocular reflex adaptation is frequency selective. J Neurophysiol 122: 984-993. doi: 10.1152/jn.00162.2019
Ullman E, Edlow JA (2010) Complete Heart Block Complicating the Head Impulse Test. Archives of Neurology 67: 1272-1274. doi: 10.1001/archneurol.2010.266
Yip CW, Glaser M, Frenzel C, Bayer O, Strupp M (2016) Comparison of the Bedside Head-Impulse Test with the Video Head-Impulse Test in a Clinical Practice Setting: A Prospective Study of 500 Outpatients. Front Neurol 7: 58. doi: 10.3389/fneur.2016.00058
![]()