By Marcello Cherchi, MD PhD
For patients
Here are answers to some of the most common questions about video head impulse testing (vHIT).
|
What is this test? |
Video head impulse testing (vHIT) assesses specific eye movements that happen when you move your head quickly. |
|
What is this test looking for? |
This test tracks eye movement abnormalities that occur in certain inner ear conditions that cause disequilibrium. |
|
Is this test experimental or investigational? |
This test is approved by the Food and Drug Administration. It is neither experimental nor investigational. |
|
What happens during this test? |
During this test you wear a set of lightweight goggles containing a tiny camera. The audiologist or technologist will instruct you to keep your gaze on a target on the wall. The audiologist or technologist will rotate your head through a repetitive series of small, quick movements. |
|
Is this test uncomfortable? |
The test is not uncomfortable. |
|
How long does this test take? |
The test takes about 20 minutes. |
|
Do I have to prepare for this test? |
There are no specific preparations for this test. |
|
Are there any special instructions for what to do after the test? |
There are no special instructions for what to do after this test. |
For clinicians
Overview
The most common protocol using video head impulse testing (vHIT) assesses the rotational vestibulo-ocular reflex in the high frequency end of the vestibular tuning spectrum. This technique can be applied to any of the six semicircular canals.
Introduction
For decades, vestibular testing was only able to assess the low frequencies (through caloric testing) and low to medium frequencies (through rotatory chair testing) of the vestibulo-ocular reflex, and those tests could only assess function of the lateral semicircular canals.
This changed beginning in 1988 when Halmagyi and Curthoys (Halmagyi and Curthoys 1988) made the crucial observation that rapid, small amplitude head rotations (subsequently called “head impulses” or “head thrusts”) could identify canal paresis. Their original paper pertained only to horizontal semicircular canal function, but it was quickly realized that a similar technique applied to other rotational planes could assess any of the six semicircular canals (Hougaard and Abrahamsen 2019; MacDougall et al. 2013a, b).
Physiology and neuroanatomy
Video head impulse testing assesses two features: (1) the gain of the vestibulo-ocular reflex, and (2) the presence/absence of compensatory saccades.
Gain of the vestibulo-ocular reflex
At its core, video head impulse testing is an assessment of the gain of the rotational vestibulo-ocular reflex. But the fact that it particularly assesses the high frequency of the vestibular tuning spectrum derives from specific properties of the peripheral vestibular system, and has a specific consequence.
The specific properties of the normally functioning vestibular system are:
- The geometric arrangements of the labyrinths are such that:
- The semicircular canals come in co-planar pairs. The lateral semicircular canals are paired with each other; the right anterior semicircular canal is paired with the left posterior semicircular canal; the left anterior canal is paired with the right posterior canal.
- Rotation of the head around a given axis will necessarily induce an excitatory response in one canal, and an inhibitory response in its paired co-planar canal.
- The central vestibular system uses the difference between the excitatory and inhibitory inputs to calculate the direction and magnitude of rotational acceleration.
- There are excitatory-inhibitory asymmetries in the vestibular system, whereby:
- Excitation has a greater effect than inhibition (from Ewald’s second and third laws).
- As rotational acceleration increases, the inhibitory responses saturate more quickly than the excitatory responses.
The specific consequence is that a rapid rotational acceleration of the head will preferentially assess the function of the semicircular canal with an excitatory response. Head impulse testing leverages this characteristic of semicircular canal function.
To illustrate this, consider the Figure below, adapted from Hill (Hill 2017) and Dr. Kamran Barin, which depicts the normal physiology of a rightward head impulse test.
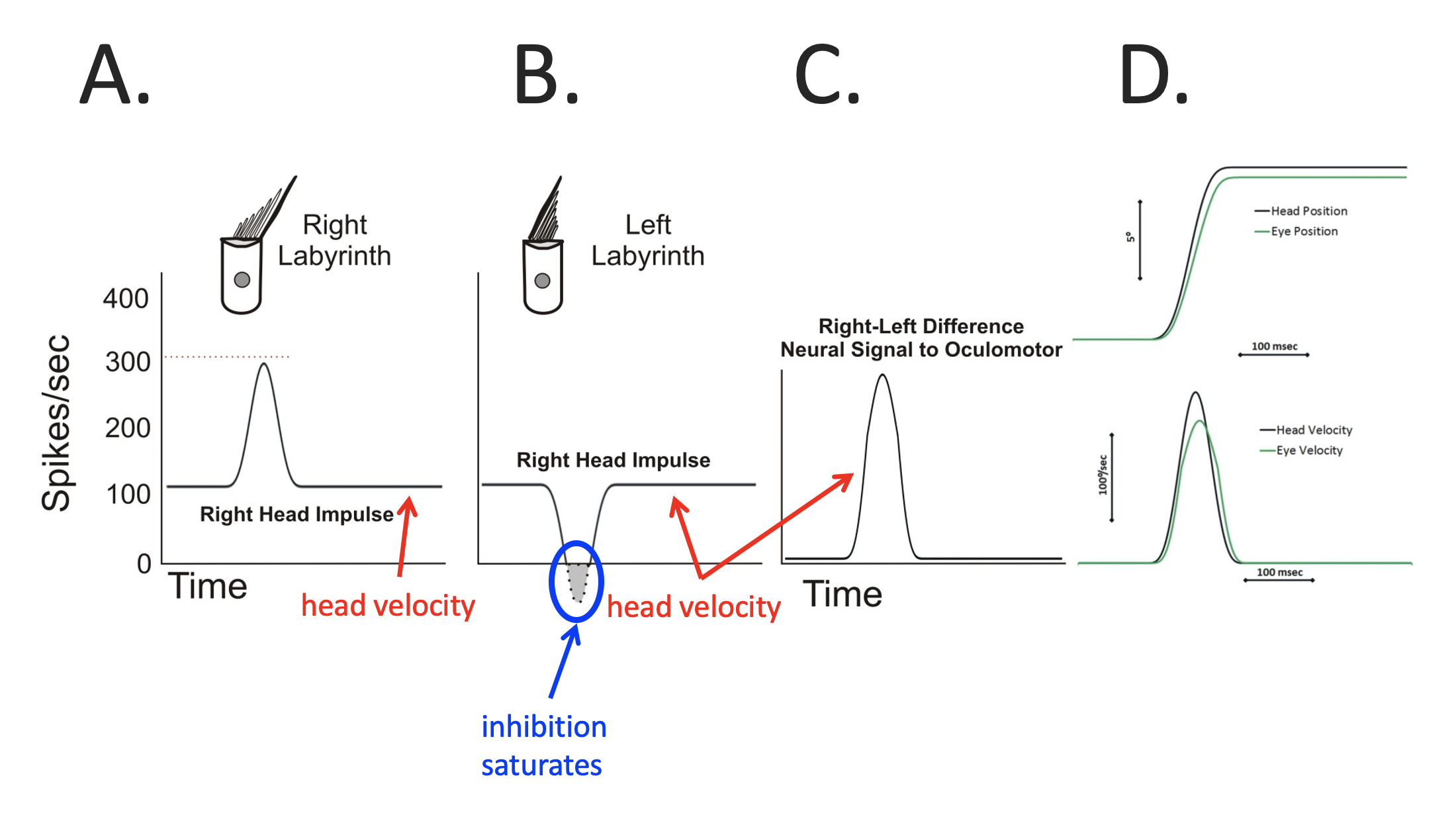
In this figure:
- Panel A shows the normal excitatory activity (in terms of spike frequency) of a right-sided horizontal semicircular canal type 1 vestibular hair in response to a rightward head impulse.
- Panel B shows normal inhibitory activity (in terms of spike frequency) of a left-sided horizontal semicircular canal type 1 vestibular hair cell in response to the rightward head impulse. Note that the inhibitory response can bring the spike frequency down to zero, but not lower (there is no “negative spike frequency”).
- Panel C shows the difference between the excitatory and inhibitory responses.
- In Panel D, the lower graph shows head velocity (the rightward impulse) and eye velocity (leftward rotation); the magnitude of the eye velocity is very close to that of the head velocity; it is slightly smaller because of the saturation of the left-sided inhibitory response.
- In Panel D, the upper graph shows the absolute magnitudes of the head and eye positions; the absolute magnitude of the eye position is slightly less than that of the head position, meaning that the head turn has only slightly dragged the eyes “off target.”
In comparison, consider the Figure below, adapted from Hill (Hill 2017) and Dr. Kamran Barin, which depicts the physiology of a rightward head impulse test in a patient with total right-sided vestibular loss.
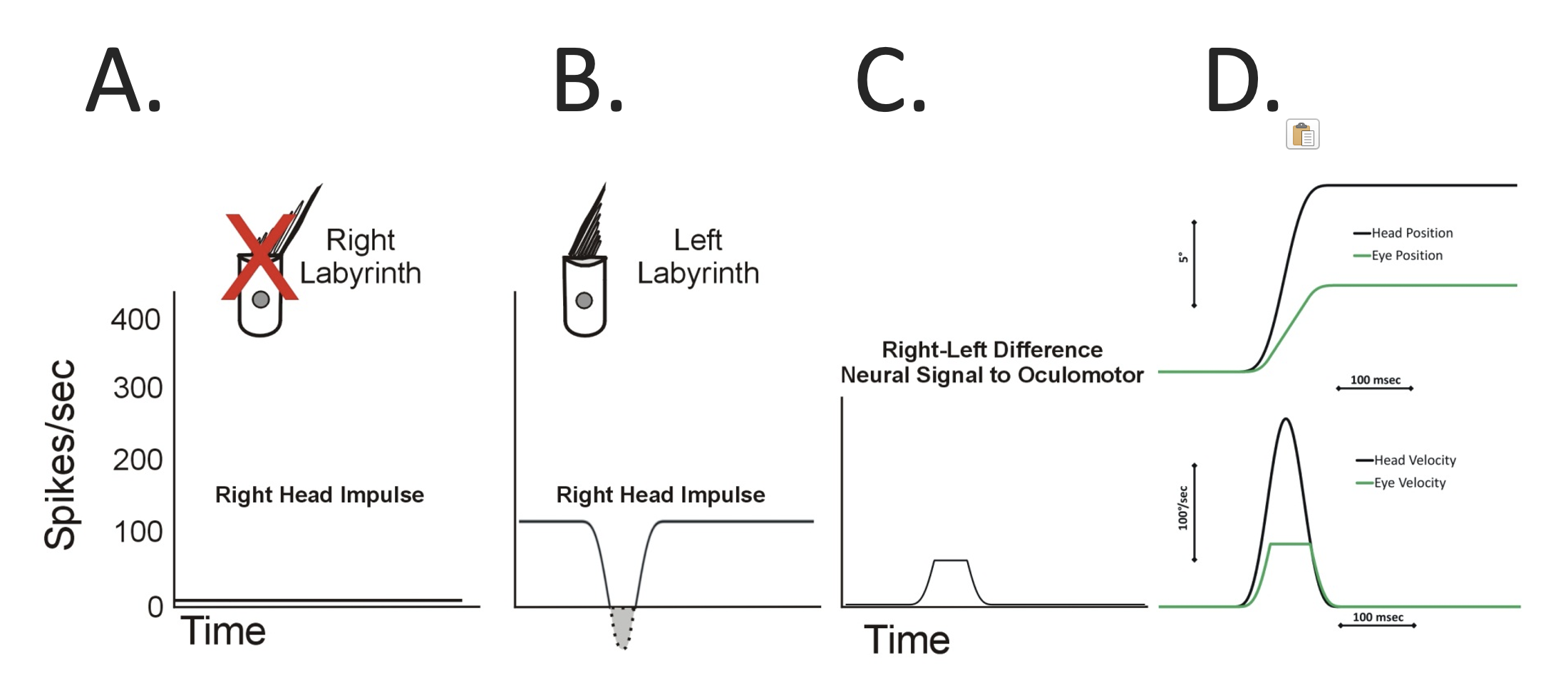
In this figure:
- Panel A shows that there is pathological absence of excitatory activity from the right-sided horizontal semicircular canal in response to the rightward head impulse.
- Panel B shows that there is normal inhibitory activity from the left-sided horizontal semicircular canal from in response to the rightward head impulse, but it is smaller, and it still saturates.
- Panel C shows the difference between the excitatory and inhibitory responses.
- In Panel D, the lower graph shows the absolute magnitude of the head and eye velocities. The magnitude of the eye velocity is much lower (than in the earlier normal example) because it only reflects the left-sided labyrinthine inhibitory response.
- In Panel D, the upper graph shows the absolute magnitude of the head and eye positions. The magnitude of the eye position is significantly less than that of the head position, meaning that the head turn as greatly dragged the eyes “off target.”
Now consider the same patient with total right-sided vestibular loss who undergoes a leftward head impulse test. This is depicted in the Figure below, adapted from Hill (Hill 2017) and Dr. Kamran Barin.
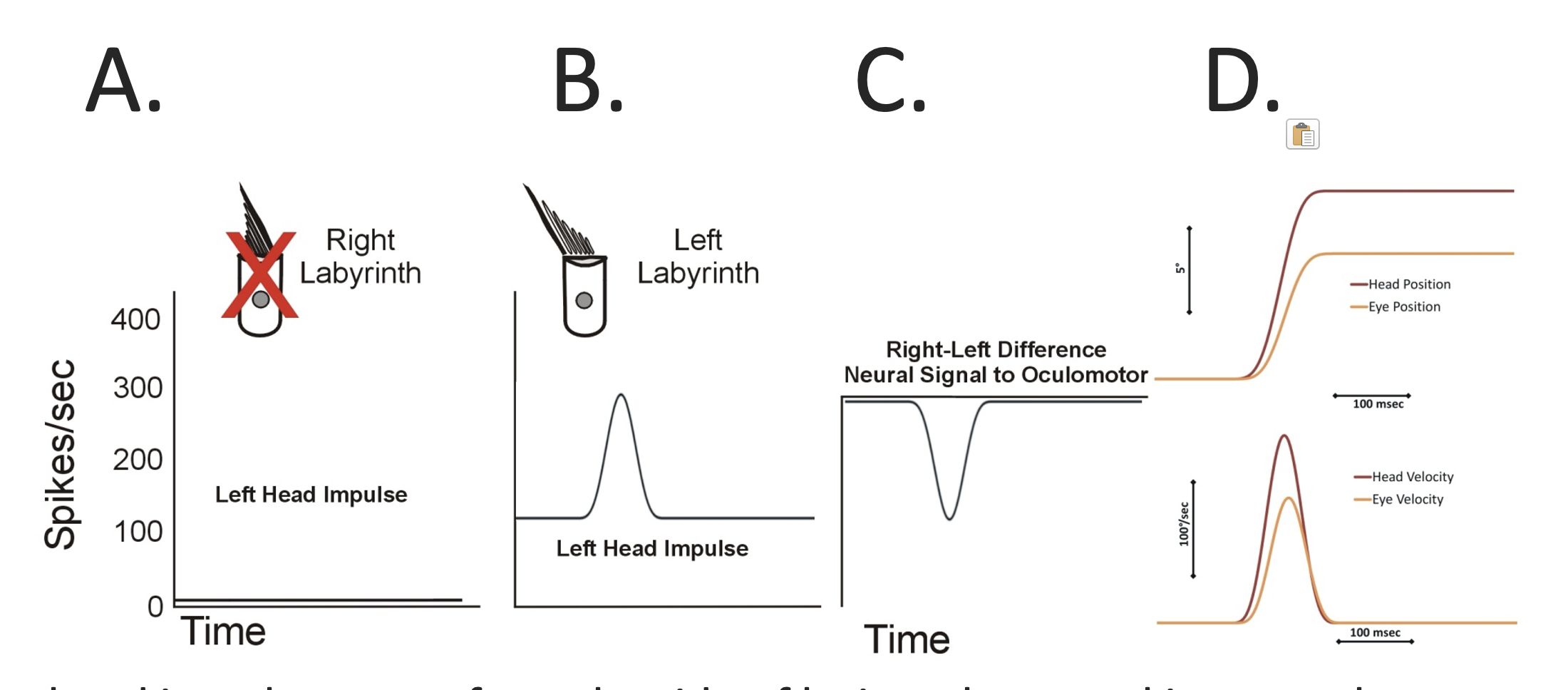
In this figure:
- Panel A shows that there is pathological absence of inhibitory activity from the right-sided horizontal semicircular canal in response to a leftward head impulse.
- Panel B shows that normal excitatory activity from the left-sided horizontal semicircular canal in response to the leftward head impulse.
- Panel C shows the difference between the excitatory and inhibitory responses.
- In Panel D, the lower graph shows the absolute magnitude of the head and eye velocities. The magnitude of the eye velocity is moderately lower (than in the earlier normal example) because it reflects only the left-sided labyrinthine excitatory response.
- In Panel D, the upper graph shows the absolute magnitude of the head and eye positions. The magnitude of the eye position is moderately less than that of the head position, meaning that the head turn has moderately dragged the eyes “off target.”
The difference between the previous two Figures illustrates how video head impulse testing leverages excitatory-inhibitory vestibular asymmetry, and preferentially assesses the canal with excitatory activity.
Compensatory saccades
For the most common protocol of video head impulse testing, the patient is instructed to maintain their gaze on an earth-stationary target. If the patient’s rotational vestibulo-ocular reflexes are intact, then head impulses will not be able to “pull the eyes off-target” — in other words, the patient will successfully continue to foveate the target despite the head impulses.
If the rotational vestibulo-ocular reflex is deficient, then the eyes will be dragged off-target by a head impulse. Because of the instructions to the patient, they will execute a saccade back to the target, variously called a “corrective saccade,” “compensatory saccade,” “refixation saccade” or “catch-up saccade.”
If these saccades are initiated after the head impulse is completed (i.e., after head velocity has returned to zero), then they are called “overt saccades,” which derives from the fact that in bedside head impulse testing, these saccades were usually observable by a clinician on face-to-face examination. In contrast, if these saccades are initiated during the head impulse (i.e., before head velocity has returned to zero), then they are called “covert saccades,” and these are usually not observable on face-to-face examination.
The Figure below, adapted from Hill (Hill 2017) and Dr. Kamran Barin, shows an idealized example of an overt saccade following a rotational vestibulo-ocular response with low gain.
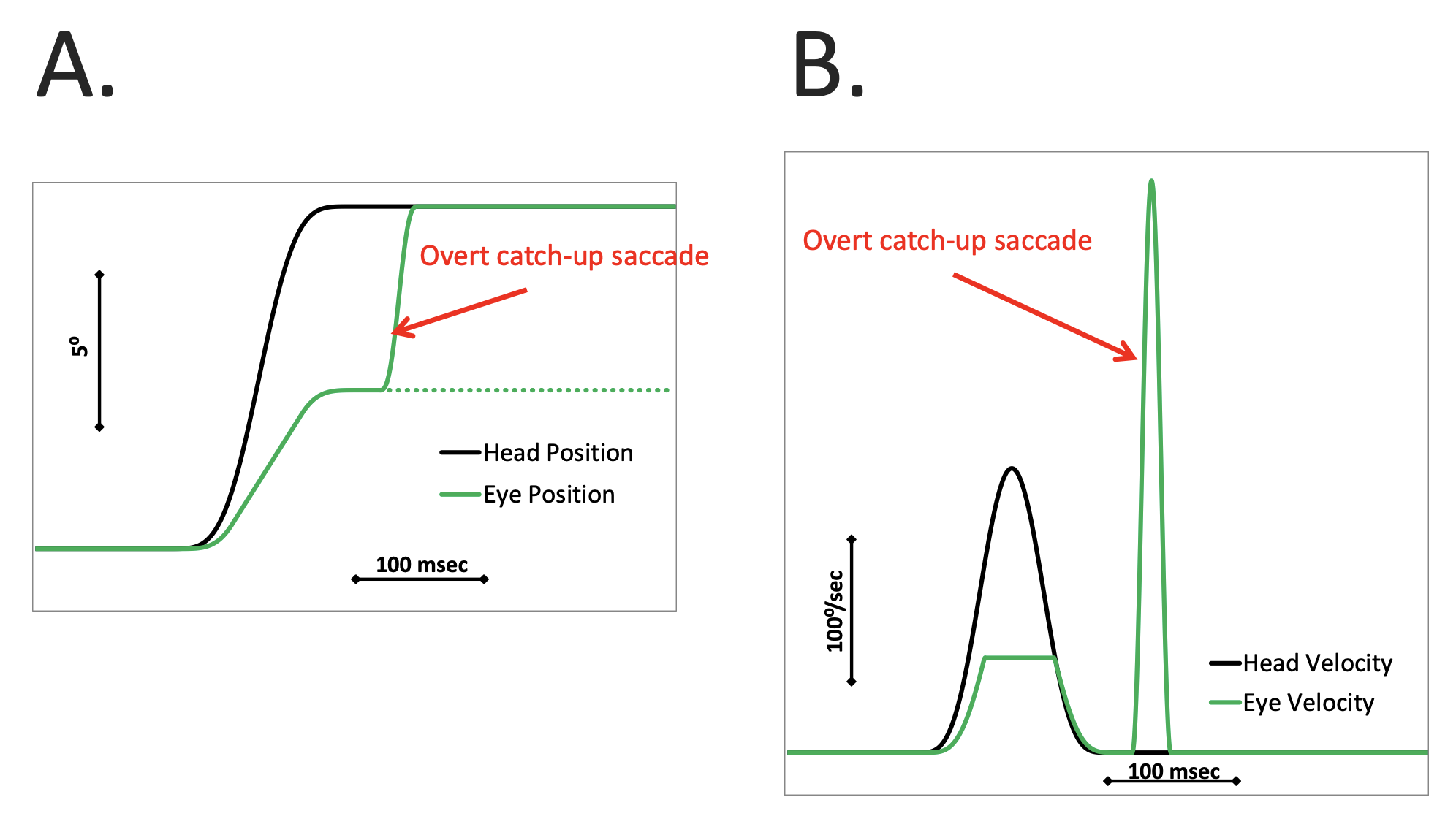
In this figure:
- Panel A shows head position and eye position. During the head impulse the magnitude of the eye rotation is much less than that of the head rotation, so the eyes get “dragged off target” by the head movement. When the head impulse is completed (i.e., head velocity returns to zero) the patient realizes that their eyes are off target, and in order to get the eyes back on target, they execute an overt corrective saccade.
- Panel B shows head velocity and eye velocity (and this is the way the results would be displayed in most formats of video head impulse testing). During the head impulse the absolute magnitude of the peak eye velocity is much smaller than that of the peak head velocity. After the head velocity returns to zero, the patient executes an overt corrective saccade, which on the tracing appears as a large spike in eye velocity.
In contrast, consider the Figure below, adapted from Hill (Hill 2017) and Dr. Kamran Barin, which shows an idealized example of covert saccade that is initiated during a rotational vestibulo-ocular response with low gain.
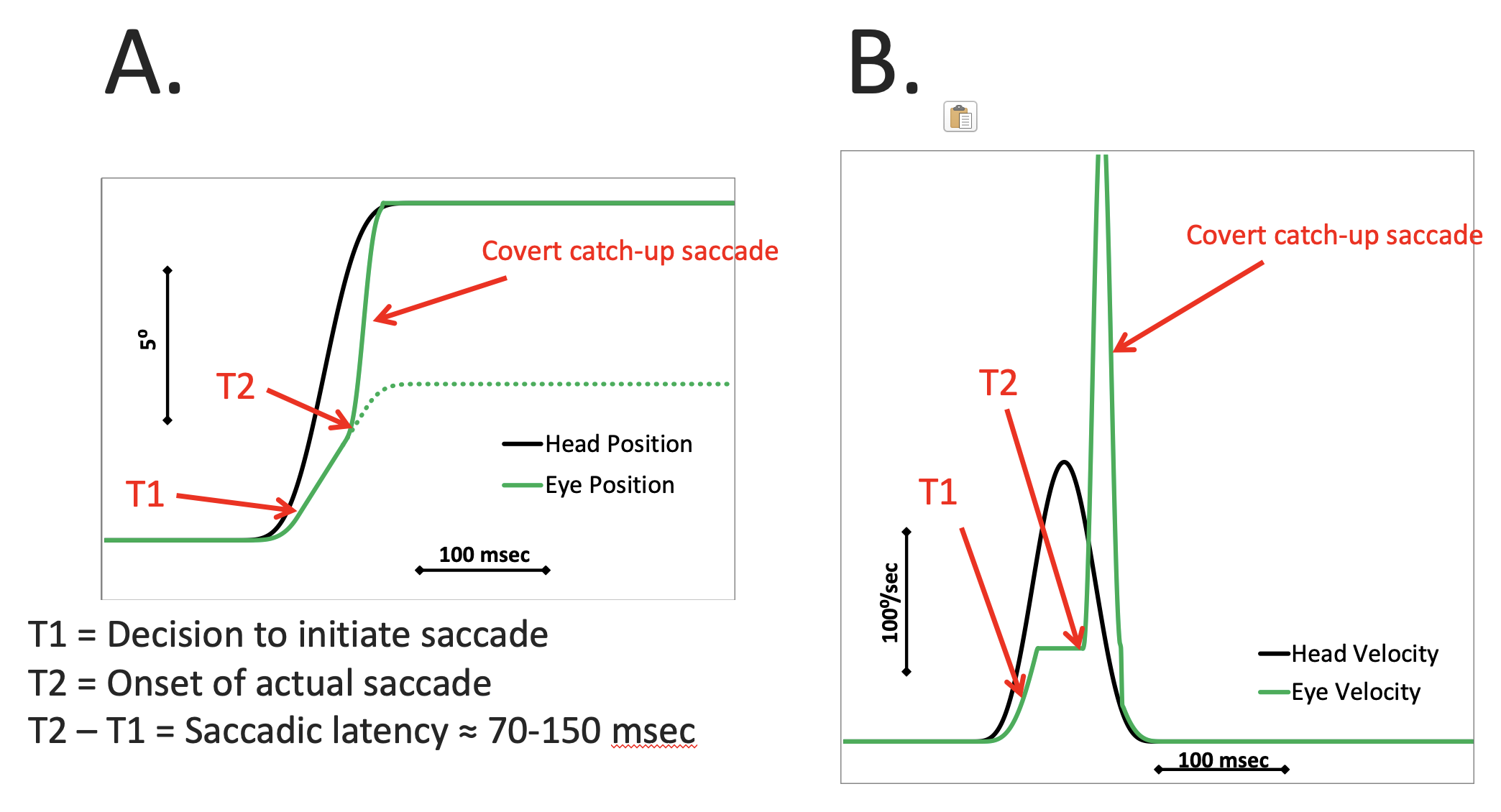
In this figure:
- Panel A shows head position and eye position. During the head impulse the magnitude of eye rotation is much less than that of the head rotation, so the eyes get “dragged off target” by the head movement. In response, a corrective saccade is initiated while the head is still in motion (thus it is a covert corrective saccade) bringing the eyes on to the target by the time the head velocity returns to zero.
- Panel B shows head velocity and eye velocity (and this is the way the results would be displayed in most formats of video head impulse testing). During the head impulse the absolute magnitude of the peak eye velocity is much smaller than that of the peak head velocity. In response, a corrective saccade is initiated while the head is still in motion (thus it is a covert corrective saccade), which manifests as a large spike in eye velocity.
It is unclear why some with vestibular hypofunction use overt corrective saccades while others use covert corrective saccades during video head impulse testing. In some cases, shortly after an acute vestibular insult (such as vestibular neuritis) a patient will exhibit overt corrective saccades, and over time (usually months) these transition into covert corrective saccades, which suggests that covert corrective saccades reflect central vestibular compensation — but this has not yet been proven.
Equipment needed
Several commercial video head impulse testing systems are available. These usually consist of a set of specialized lightweight goggles with a small infrared camera, all connected to a computer responsible for image processing and calculations.
How to perform the test
The patient is seated a prespecified distance from a screen such that their primary position of gaze is on a marked, earth-stationary target. The specialized lightweight goggles are placed on the patient’s head. Calibration is performed, which many systems execute by projecting a tiny laser (affixed to the nasal bridge of the goggles) at different points on the screen, and the patient must direct their gaze at those points while keeping the head still.
After calibration is completed, the examiner stands behind the patient, firmly grasps the patient’s head, and executes head impulses similar to what one would do with bedside head impulse testing (except that the examiner is standing behind the patient). In most systems an angular accelerometer is attached to the lightweight goggles and the examiner is given feedback about whether a given head impulse was executed with sufficient acceleration. The examiner executes the saccades randomly to the right or left.
It is possible to do video head impulse testing in planes other than that of the horizontal semicircular canals. Many practitioners find it considerably more challenging to perform oblique head impulses, and it requires experience to develop this skill.
What this test assesses
Video head impulse testing assesses the rotational vestibulo-ocular reflex, and can do so for each of the six semicircular canals.
How to interpret the test results
For head impulses of a given canal, if the responses exhibit low gain and there are compensatory saccades, then that suggests vestibular hypofunction (low gain) in that semicircular canal or its afferent pathways.
Other protocols of video head impulse testing
There are several other protocols that use the same lightweight goggles employed for video head impulse testing. These are less commonly performed in clinical practice, but we include them here for completeness.
One of these, called “suppression head impulse” testing (abbreviated “SHIMP”), is a protocol in which the forward-projecting laser attached to the nasal bridge of the goggles is left on, and the patient is instructed to keep their eyes on that target (rather than on an earth-stationary target).
Regular video head impulse testing, and the SHIMP protocol, use head movements that are very fast, and during which pursuit (mediated by the visual system) is temporarily suppressed; in this circumstance labyrinthine function (specifically the vestibulo-ocular reflex) predominates over visual function.
The second and third protocols are uncommonly performed, in part because they are more commonly done during rotatory chair testing. These protocols are “visually enhanced vestibulo-ocular reflex” testing (abbreviated “VVOR”) and “vestibulo-ocular reflex suppression” testing (abbreviated “VORS”), and are discussed in greater detail by Ramos and colleagues (Ramos et al. 2019).
In the Table below we compare all four protocols for which vHIT goggles can be used.
|
The task is to keep the eyes on a stationary target. In other words, the target is earth-fixed |
The task is to track a target that moves with the head. In other words, the target is head-fixed. |
|
|
Protocol notes: • High velocity (head impulse). Physiological notes: • Pursuit (visual system) is temporarily suppressed. • Labyrinthine function (VOR) predominates (over visual system). |
HIMP (head impulse paradigm), i.e., vHIT. In this protocol there are high velocity head movements (during which the visual pursuit system is suppressed), vision is available, and the patient is instructed to maintain gaze on an earth-fixed target. Unilateral loss: In brisk head movement towards the diseased side the eyes get partially dragged along with the head (i.e., towards the diseased side), away from the earth-fixed stationary target, after which there is a “compensatory saccade” (i.e., a corrective saccade in the direction opposite the head movement, towards the healthy side) to re-acquire the earth-fixed target. This is what MacDougall and colleagues (MacDougall et al. 2016) mean when they say, “with HIMP, compensatory saccades indicate vestibular loss.” In brisk head movement towards the healthy side the eyes get (appropriately) driven contraversive to the head movement (i.e., towards the diseased side) and thus remain on the earth-fixed stationary target, so no corrective saccade is needed. In some cases the compensatory saccade may “overcorrect,” because it is unopposed by vestibular input from the diseased ear. Bilateral loss: In head movement towards either (diseased) the eyes get partially dragged along with the head, away from the earth-fixed stationary target, after which there is a corrective saccade in the direction opposite the head movement to re-acquire the target. |
SHIMP (suppression head impulse paradigm). In this protocol there are high velocity head movements (during which the visual pursuit system is suppressed), vision is available, and the patient is instructed to maintain gaze on a head-fixed target. Unilateral loss: In brisk head movement towards the diseased side the eyes get dragged along with the head (i.e., towards the diseased side) and thus along with the target, so no corrective saccade is needed. In brisk head movement towards the healthy side the eyes get incompletely dragged along with the head (i.e., towards the healthy side), after which there is an “anti-compensatory saccade” (i.e., a corrective saccade in the same direction as the head, towards the healthy side) to re-acquire the head-fixed target. This is what MacDougall and colleagues (MacDougall et al. 2016) mean when they say, “with SHIMP, anti-compensatory saccades indicate vestibular function.” Bilateral loss: In head movement towards either diseased side there are few or no saccades (since there is no VOR to suppress). |
|
Protocol notes: • Low velocity head movements. Physiological notes: • Pursuit (visual system) is not suppressed. • Pursuit (visual system) predominates over the labyrinthine function (VOR). |
VVOR (visually enhanced vestibulo-ocular reflex). Here, “visually enhanced” means that an earth-fixed visual target is available in order to supplement the VOR during slow head movements. Unilateral loss: In slow head rotation towards the diseased side the eyes get dragged along with the head (i.e., towards the diseased side, away from the stationary target), so there are corrective saccades directed opposite the head movement (i.e., towards the healthy side) in order to re-acquire the earth-fixed target. In slow head rotation towards the healthy side the eyes get (appropriately) driven contraversive to the head movement (i.e., towards the diseased side) and thus remain on the earth-fixed stationary target, so no corrective saccades are needed. Bilateral loss: In slow head rotation towards either (diseased) side there are corrective saccades directed opposite to the head motion. |
VORS (vestibulo-ocular reflex suppression). This means that a head-fixed visual target is available to supplement suppression of the VOR during slow head movements. Unilateral loss: In slow head rotation towards the healthy side the eyes are incompletely dragged along with the head (because the intact VOR is driving the eyes contraversively), so there are larger corrective saccades in the same direction as the head movement (towards the healthy side) in order to keep up with the head-fixed target, compared with smaller corrective saccades when the head is turned towards the diseased side. Bilateral loss: In slow head rotation towards either (diseased) side there are no corrective saccades (since there is no VOR to suppress). |
Limitations
Many articles have been written that compare evaluation of horizontal semicircular canal function by caloric testing and by video head impulse testing (Awadie et al. 2018; Burston et al. 2018; Cordero-Yanza et al. 2017; Fukushima et al. 2018; Han et al. 2020; Jung et al. 2017; Mahringer and Rambold 2014; McCaslin et al. 2014; McGarvie et al. 2015; Park et al. 2005; Rambold 2015; Redondo-Martinez et al. 2016; Rohrmeier et al. 2013; Rubin et al. 2018; Schmid-Priscoveanu et al. 2001; Shugyo et al. 2020; Vallim et al. 2021; Wegmann-Vicuña et al. 2018; Yoo et al. 2016; Zellhuber et al. 2014). This literature often conveys perplexity over apparently discrepant results between the two tests.
Do not be misled by this apparent discrepancy, because it is not so much a discrepancy as a dissociation. Keep in mind that caloric testing assesses the low frequency of the vestibular tuning spectrum, while head impulse testing and video head impulse testing assess the high frequency of the vestibular tuning spectrum.
Although the original description of head impulse testing pertained to horizontal semicircular canal function (Halmagyi and Curthoys 1988), it was eventually realized that this technique applies to the vertical canals (posterior and anterior) as well (MacDougall et al. 2013a, b). However, literature regarding assessment of the vertical canals finds vHIT to be less reliable; for example, some authors note inconsistency in gain of the vertical canals and significant variability within and between examiners (Money-Nolan and Flagge 2023); other authors note weak replicability of compensatory saccades across testing sessions in one and the same patient (Singh et al. 2019).
Contraindications
Patients with significant limitations of cervical range of motion may simply be unable to undergo video head impulse testing.
Although there is no literature supporting the concern that video head impulse testing can cause or worsen cervical arterial dissection (Kaski and Bronstein 2012; Ullman and Edlow 2010), this theoretical possibility still makes many clinicians shy of this procedure. Our own practice is to refrain from performing video head impulse testing in patients with risk factors such as connective tissue diseases or a prior history of carotid or vertebral artery dissection.
Pitfalls
Like any test of ocular motor function, video head impulse testing needs to be calibrated. If calibration is incorrect, then the data will be corrupted, and in particular, assessment of the gain of the vestibulo-ocular reflex may have false positive or false negative results.
Some literature describes significant inter-operator variability in video head impulse testing results (Mutlu et al. 2020).
Target distance (from the subject’s eyes) and target size need to remain consistent (Judge et al. 2018). Changes in examiner hand position on the patient’s head can also introduce variability in the results (Fu et al. 2018). Some literature suggests that pupil size may have an effect on analysis, finds that smaller pupils give better readings, and therefore recommends conducting video head impulse testing in well-illuminated environments to promote miosis (Yoon et al. 2023).
There have been several attempts to arrive at normative ranges for video head impulse testing (Emekci et al. 2021; Hansson and Salzer 2020; Matino-Soler et al. 2015; Murnane et al. 2014). Some literature suggests that age-adjusted normative ranges (Hansson and Salzer 2020; Kim and Kim 2018; Matino-Soler et al. 2015; McGarvie et al. 2015b; Murnane et al. 2014) should probably be established, while other literature disputes this (Emekci et al. 2021; Yang et al. 2016). McGarvie and colleagues (McGarvie et al. 2015b) studied this question and concluded that age has minimal effect on VOR gain as measured by vHIT.
Some literature describes similar results from different video head impulse testing systems (Aalling et al. 2020). Other literature describes discrepant results (in one and the same patient) between different video head impulse testing devices (Janky et al. 2017; van Dooren et al. 2020), and it is not yet clear whether algorithmic uniformity can address these differences (Cleworth et al. 2017; Rey-Martinez et al. 2015).
Artifact can interfere with calculations of gain (Mantokoudis et al. 2016; Versino et al. 2014; Zamaro et al. 2020), and computational approaches have been proposed to address this (Trinidad-Ruiz et al. 2020).
Non-vestibular ocular motor abnormalities may interfere with results from video head impulse testing. For example, in unilateral internuclear ophthalmoplegia, during a head thrust towards the side of the lesion:
- The adducting eye’s response will exhibit hypometria and reduced velocity. If the vHIT’s camera happens to be viewing this adducting eye, the result may give the false impression of vestibular weakness on that side — though this impression is only partial because there will be no compensatory saccade.
- The abducting eye’s response, due to Herring’s law, will include abduction nystagmus (Zee et al. 1987), of which only the first beat might get recorded given the brief time frame tracked on vHIT. If the vHIT’s camera happens to be viewing this abducting eye, that first beat of nystagmus may appear to be a compensatory saccade, contributing to a false impression of vestibular weakness on that side — though this impression is only partial because the amplitude is eumetric (rather than hypometric).
Usually internuclear ophthalmoplegia will be detected on face-to-face ocular motor examination, but if a clinician is only provided with the tracings from video head impulse testing, the result (in isolation) may be misinterpreted as compatible with vestibular weakness.
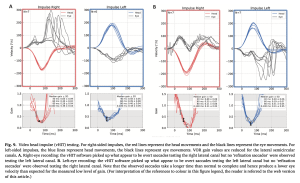
The Figure below, from Koohi and colleagues (Koohi et al. 2022), shows results of video head impulse testing in a patient with multiple sclerosis who has bilateral internuclear ophthalmoplegia. The two left panels show the results of head thrust with the camera on the patient’s right eye; rightward impulse shows low gain plus covert compensatory saccades, while leftward impulse only shows low gain. The two right panels show the results of head thrust with the camera on the patient’s left eye; rightward impulse shows low gain, while leftward impulse shows low gain and overt plus covert compensatory saccades. Interpreted in isolation, either pair of tracings could be misinterpreted as compatible with bilateral vestibular weakness.
When is the test indicated
It is reasonable to check video head impulse testing in patients complaining of disequilibrium in whom there are grounds to suspect vestibular weakness (unilateral or bilateral).
Diseases that may be diagnosed by this test
The bedside head impulse test of the horizontal rotational vestibulo-ocular reflex may help identify unilateral vestibular weakness or bilateral vestibular weakness, each of which in turn may have many etiologies.
Additional notes
Video head impulse testing is more sophisticated than bedside head impulse testing in several respects: (1) it can quantify gain of the vestibulo-ocular reflex; (2) it can more easily identify corrective saccades.
References
Aalling M, Skals RK, Abrahamsen ER, Hougaard DD (2020) Comparison of test results from two separate video head impulse test systems in a cohort of patients diagnosed with a unilateral vestibular schwannoma. Eur Arch Otorhinolaryngol 277: 3185-3193. doi: 10.1007/s00405-020-06116-2
Awadie A, Holdstein Y, Kaminer M, Shupak A (2018) The head impulse test as a predictor of videonystagmography caloric test lateralization according to the level of examiner experience: A prospective open-label study. Ear Nose Throat J 97: 16-23.
Burston A, Mossman S, Mossman B, Weatherall M (2018) Comparison of the video head impulse test with the caloric test in patients with sub-acute and chronic vestibular disorders. J Clin Neurosci 47: 294-298. doi: 10.1016/j.jocn.2017.10.040
Cleworth TW, Carpenter MG, Honegger F, Allum JHJ (2017) Differences in head impulse test results due to analysis techniques. J Vestib Res 27: 163-172. doi: 10.3233/VES-170614
Cordero-Yanza JA, Arrieta Vazquez EV, Hernaiz Leonardo JC, Mancera Sanchez J, Hernandez Palestina MS, Perez-Fernandez N (2017) Comparative study between the caloric vestibular and the video-head impulse tests in unilateral Meniere’s disease. Acta Otolaryngol 137: 1178-1182. doi: 10.1080/00016489.2017.1354395
Emekci T, Ugur KS, Cengiz DU, Men Kilinc F (2021) Normative values for semicircular canal function with the video head impulse test (vHIT) in healthy adolescents. Acta Otolaryngol 141: 141-146. doi: 10.1080/00016489.2020.1836396
Fu W, He F, Zhao R, Wei D, Bai Y, Wang X, Han J (2018) Effects of Hand Positions During Video Head-Impulse Test (vHIT) in Patients With Unilateral Vestibular Neuritis. Front Neurol 9: 531. doi: 10.3389/fneur.2018.00531
Fukushima M, Oya R, Nozaki K, Eguchi H, Akahani S, Inohara H, Takeda N (2018) Vertical head impulse and caloric are complementary but react opposite to Meniere’s disease hydrops. Laryngoscope. doi: 10.1002/lary.27580
Halmagyi GM, Curthoys IS (1988) A clinical sign of canal paresis. Arch Neurol 45: 737-9. doi: 10.1001/archneur.1988.00520310043015.
Han C, Paik SW, Yahg H, Park SY, Lee JH (2020) Comparison of Predictive Parameters between the Video Head Impulse Test and Caloric Test. Research in Vestibular Science 19: 55-61. doi: 10.21790/rvs.2020.19.2.55
Hansson A, Salzer J (2020) Normative video head impulse test data in subjects with and without vascular risk factors. European Archives of Oto-Rhino-Laryngology. doi: 10.1007/s00405-020-06332-w
Hill K (2017) Vestibular assessment using video head impulse testing (vHIT). https://slideplayer.com/slide/13290284/. Accessed May 2 2023
Hougaard D, Abrahamsen E (2019) Functional Testing of all Six Semicircular Canals with Video Head Impulse Test Systems. Journal of Visualized Experiments. doi: 10.3791/59012
Janky KL, Patterson JN, Shepard NT, Thomas MLA, Honaker JA (2017) Effects of Device on Video Head Impulse Test (vHIT) Gain. J Am Acad Audiol 28: 778-785. doi: 10.3766/jaaa.16138
Judge PD, Rodriguez AI, Barin K, Janky KL (2018) Impact of Target Distance, Target Size, and Visual Acuity on the Video Head Impulse Test. Otolaryngol Head Neck Surg: 194599818779908. doi: 10.1177/0194599818779908
Jung J, Suh MJ, Kim SH (2017) Discrepancies between video head impulse and caloric tests in patients with enlarged vestibular aqueduct. Laryngoscope 127: 921-926. doi: 10.1002/lary.26122
Kaski D, Bronstein AM (2012) Response to: Making a diagnosis in patients who present with vertigo. BMJ: e5809. doi: 10.1136/bmj.e5809
Kim TH, Kim MB (2018) Effect of aging and direction of impulse in video head impulse test. Laryngoscope 128: E228-E233. doi: 10.1002/lary.26864
Koohi N, Mendis S, Lennox A, Whelan D, Kaski D (2022) Video head impulse testing: Pitfalls in neurological patients. J Neurol Sci 442: 120417. doi: 10.1016/j.jns.2022.120417
MacDougall HG, McGarvie LA, Halmagyi GM, Curthoys IS, Weber KP (2013a) Application of the video head impulse test to detect vertical semicircular canal dysfunction. Otol Neurotol 34: 974-9. doi: 10.1097/MAO.0b013e31828d676d
Macdougall HG, McGarvie LA, Halmagyi GM, Curthoys IS, Weber KP (2013b) The video Head Impulse Test (vHIT) detects vertical semicircular canal dysfunction. PLoS One 8: e61488. doi: 10.1371/journal.pone.0061488
MacDougall HG, McGarvie LA, Halmagyi GM, Rogers SJ, Manzari L, Burgess AM, Curthoys IS, Weber KP (2016) A new saccadic indicator of peripheral vestibular function based on the video head impulse test. Neurology 87: 410-8. doi: 10.1212/WNL.0000000000002827
Mahringer A, Rambold HA (2014) Caloric test and video-head-impulse: a study of vertigo/dizziness patients in a community hospital. Eur Arch Otorhinolaryngol 271: 463-72. doi: 10.1007/s00405-013-2376-5
Mantokoudis G, Saber Tehrani AS, Wozniak A, Eibenberger K, Kattah JC, Guede CI, Zee DS, Newman-Toker DE (2016) Impact of artifacts on VOR gain measures by video-oculography in the acute vestibular syndrome. J Vestib Res 26: 375-385. doi: 10.3233/VES-160587
Matino-Soler E, Esteller-More E, Martin-Sanchez JC, Martinez-Sanchez JM, Perez-Fernandez N (2015) Normative data on angular vestibulo-ocular responses in the yaw axis measured using the video head impulse test. Otol Neurotol 36: 466-71. doi: 10.1097/MAO.0000000000000661
McCaslin DL, Jacobson GP, Bennett ML, Gruenwald JM, Green AP (2014) Predictive properties of the video head impulse test: measures of caloric symmetry and self-report dizziness handicap. Ear Hear 35: e185-91. doi: 10.1097/AUD.0000000000000047
McGarvie LA, Curthoys IS, MacDougall HG, Halmagyi GM (2015a) What does the dissociation between the results of video head impulse versus caloric testing reveal about the vestibular dysfunction in Meniere’s disease? Acta Otolaryngol 135: 859-65. doi: 10.3109/00016489.2015.1015606
McGarvie LA, MacDougall HG, Halmagyi GM, Burgess AM, Weber KP, Curthoys IS (2015b) The Video Head Impulse Test (vHIT) of Semicircular Canal Function – Age-Dependent Normative Values of VOR Gain in Healthy Subjects. Front Neurol 6: 154. doi: 10.3389/fneur.2015.00154
McGarvie LA, MacDougall HG, Halmagyi GM, Burgess AM, Weber KP, Curthoys IS (2015b) The Video Head Impulse Test (vHIT) of Semicircular Canal Function – Age-Dependent Normative Values of VOR Gain in Healthy Subjects. Front Neurol 6: 154. doi: 10.3389/fneur.2015.00154
Money-Nolan LE, Flagge AG (2023) Factors affecting variability in vestibulo-ocular reflex gain in the Video Head Impulse Test in individuals without vestibulopathy: A systematic review of literature. Front Neurol 14: 1125951. doi: 10.3389/fneur.2023.1125951
Murnane O, Mabrey H, Pearson A, Byrd S, Akin F (2014) Normative data and test-retest reliability of the SYNAPSYS video head impulse test. J Am Acad Audiol 25: 244-52. doi: 10.3766/jaaa.25.3.3
Mutlu B, Cesur S, Topcu MT, Gecici CR, Askin OE, Derinsu EU (2020) Evaluation of Interexaminer Variability in Video Head Impulse Test Results. J Am Acad Audiol 31: 613-619. doi: 10.1055/s-0040-1717124
Park HJ, Migliaccio AA, Della Santina CC, Minor LB, Carey JP (2005) Search-coil head-thrust and caloric tests in Meniere’s disease. Acta Otolaryngol 125: 852-7.
Rambold HA (2015) Economic management of vertigo/dizziness disease in a county hospital: video-head-impulse test vs. caloric irrigation. Eur Arch Otorhinolaryngol 272: 2621-8. doi: 10.1007/s00405-014-3205-1
Ramos BF, Cal R, Carmona S, Weber KP, Zuma EMF (2019) Corrective Saccades in Unilateral and Bilateral Vestibular Hypofunction During Slow Rotation Expressed by Visually Enhanced VOR and VOR Suppression: Role of the Cerebellum. Cerebellum. doi: 10.1007/s12311-019-01066-w
Redondo-Martinez J, Becares-Martinez C, Orts-Alborch M, Garcia-Callejo FJ, Perez-Carbonell T, Marco-Algarra J (2016) Relationship between video head impulse test (vHIT) and caloric test in patients with vestibular neuritis. Acta Otorrinolaringol Esp 67: 156-61. doi: 10.1016/j.otorri.2015.07.005
Rey-Martinez J, Batuecas-Caletrio A, Matino E, Perez Fernandez N (2015) HITCal: a software tool for analysis of video head impulse test responses. Acta Otolaryngol 135: 886-94. doi: 10.3109/00016489.2015.1035401
Rohrmeier C, Richter O, Schneider M, Wirsching K, Fiedler I, Haubner F, Strutz J, Kuhnel TS (2013) Triple test as predictive screen for unilateral weakness on caloric testing in routine practice. Otol Neurotol 34: 297-303.
Rubin F, Simon F, Verillaud B, Herman P, Kania R, Hautefort C (2018) Comparison of Video Head Impulse Test and Caloric Reflex Test in advanced unilateral definite Meniere’s disease. Eur Ann Otorhinolaryngol Head Neck Dis 135: 167-169. doi: 10.1016/j.anorl.2017.08.008
Schmid-Priscoveanu A, Bohmer A, Obzina H, Straumann D (2001) Caloric and search-coil head-impulse testing in patients after vestibular neuritis. J Assoc Res Otolaryngol 2: 72-8.
Shugyo M, Ito T, Shiozaki T, Nishikawa D, Ohyama H, Fujita H, Yamanaka T, Kitahara T (2020) Comparison of the video head impulse test results with caloric test in patients with Meniere’s disease and other vestibular disorders. Acta Otolaryngol 140: 728-735. doi: 10.1080/00016489.2020.1766700
Singh NK, Govindaswamy R, Jagadish N (2019) Test-Retest Reliability of Video Head Impulse Test in Healthy Individuals and Individuals with Dizziness. J Am Acad Audiol 30: 744-752. doi: 10.3766/jaaa.17080
Trinidad-Ruiz G, Rey-Martinez J, Matino-Soler E, Batuecas-Caletrio A, Martin-Sanz E, Perez-Fernandez N (2020) Relevance of Artifact Removal and Number of Stimuli for Video Head Impulse Test Examination. Ear Hear. doi: 10.1097/AUD.0000000000000849
Ullman E, Edlow JA (2010) Complete Heart Block Complicating the Head Impulse Test. Archives of Neurology 67: 1272-1274. doi: 10.1001/archneurol.2010.266
Vallim MGB, Gabriel GP, Mezzalira R, Stoler G, Chone CT (2021) Does the video head impulse test replace caloric testing in the assessment of patients with chronic dizziness? A systematic review and meta-analysis. Brazilian Journal of Otorhinolaryngology. doi: https://doi.org/10.1016/j.bjorl.2021.01.002
van Dooren TS, Starkov D, Lucieer FMP, Vermorken B, Janssen AML, Guinand N, Perez-Fornos A, Van Rompaey V, Kingma H, van de Berg R (2020) Comparison of three video head impulse test systems for the diagnosis of bilateral vestibulopathy. J Neurol 267: 256-264. doi: 10.1007/s00415-020-10060-w
Versino M, Colagiorgio P, Sacco S, Colnaghi S, Ramat S (2014) Artifact avoidance for head impulse testing. Clin Neurophysiol 125: 1071-3. doi: 10.1016/j.clinph.2013.09.024
Wegmann-Vicuña R, Garaycochea O, Domínguez-Echavarri P, Guajardo-Vergara C, García-Eulate R, Pérez-Fernández N (2018) Dissociated responses to caloric and head impulse stimulation in a case of isolated vestibule-lateral semicircular canal dysplasia. Acta Oto-Laryngologica Case Reports 3: 5-10. doi: 10.1080/23772484.2017.1416955
Yang CJ, Lee JY, Kang BC, Lee HS, Yoo MH, Park HJ (2016) Quantitative analysis of gains and catch-up saccades of video-head-impulse testing by age in normal subjects. Clin Otolaryngol 41: 532-8. doi: 10.1111/coa.12558
Yoo MH, Kim SH, Lee JY, Yang CJ, Lee HS, Park HJ (2016) Results of video head impulse and caloric tests in 36 patients with vestibular migraine and 23 patients with vestibular neuritis: a preliminary report. Clin Otolaryngol 41: 813-817. doi: 10.1111/coa.12556
Yoon HJ, Lee JH, Lee JH, Park E, Lee SU, Kim BJ, Kim JS (2023) Effects of pupil size in video head-impulse tests. J Neurol. doi: 10.1007/s00415-023-12026-0
Zamaro E, Saber Tehrani AS, Kattah JC, Eibenberger K, Guede CI, Armando L, Caversaccio MD, Newman-Toker DE, Mantokoudis G (2020) VOR gain calculation methods in video head impulse recordings. J Vestib Res 30: 225-234. doi: 10.3233/VES-200708
Zee DS, Hain TC, Carl JR (1987) Abduction nystagmus in internuclear ophthalmoplegia. Ann Neurol 21: 383-8. doi: 10.1002/ana.410210411
Zellhuber S, Mahringer A, Rambold HA (2014) Relation of video-head-impulse test and caloric irrigation: a study on the recovery in unilateral vestibular neuritis. Eur Arch Otorhinolaryngol 271: 2375-83. doi: 10.1007/s00405-013-2723-6
![]()