By Marcello Cherchi, MD PhD
For patients
Cogan syndrome (CS) is a rare autoimmune disease that usually causes problems with the eyes, hearing loss and dizziness. If your doctor suspects CS, then they may order a number of tests. CS is usually managed by a rheumatologist and ophthalmologist.
For clinicians
Overview
Cogan syndrome (CS) is a rare autoimmune vasculitis of uncertain etiology whose original description was a triad of interstitial keratitis, hearing loss and vestibular symptoms, though subsequent research documented symptoms involving multiple organ systems (musculoskeletal, abdominal, cardiac, genitourinary, cutaneous and other). The sensorineural hearing loss is usually (but not always) symmetrical, usually (though not always) involving the high greater than the low frequencies, and can be abrupt in onset or gradually progressive. Vestibular workup may show unilateral or bilateral caloric weakness, and an elevated SP/AP ratio on electrocochleography (ECoG). MRI may show T1 hyperintensity of the labyrinths. Management is usually multidisciplinary, often organized by a rheumatologist, and usually involving an ophthalmologist. Treatment usually begins with systemic corticosteroids which, if initiated early, may bring about some improvement in the hearing loss.
Introduction
David Glendenning Cogan (1908 – 1993) was an American ophthalmologist whose name became attached to numerous eponymously designated disorders and physical examination findings.

In 1934 R. F. Morgan and Dr. Conrad J. Baumgartner published a case of “Ménière’s disease” that seemed unusual insofar as the auditory and vestibular symptoms were additionally accompanied by interstitial keratitis (Morgan and Baumgartner 1934).
Beginning in 1945 Dr. Cogan published several articles describing a syndrome of interstitial keratitis, auditory and vestibular symptoms (Cogan 1945, 1949; Norton and Cogan 1959). This triad eventually became known as Cogan syndrome (CS).
Epidemiology
CS is rare. Writing in 2018, Shamriz and colleagues (Shamriz et al. 2018) stated that about 250 cases had been reported worldwide. Most reported cases are adults in their third decade; only a small proportion of cases are reported in children and the elderly. It is more common in Caucasians.
CS can occur in isolation, but it often is found in the context of other illnesses such as vasculitis (Azami et al. 2014; Shamriz et al. 2018).
Genetics
It is unknown whether there is any genetic predisposition to CS.
Pathophysiological mechanism of disease
CS is believed to be due to an autoimmune vasculitis (Greco et al. 2013), though the underlying etiology remains uncertain.
Clinical presentation
Typical Cogan syndrome presents with interstitial keratitis, sensorineural hearing loss and vestibular symptoms.
Atypical Cogan syndrome differs from typical Cogan syndrome in its ophthalmologic manifestations, which (instead of interstitial keratitis) can include scleritis, episcleritis, uveitis, retinal vasculitis and others (Grasland et al. 2004).
The hearing loss in CS generally begins bilaterally, though some cases present with unilateral loss and later become bilateral (Azami et al. 2014).
The cadence of hearing loss is variable (Baumann et al. 2005), ranging from slowly progressive (Cundiff et al. 2006) to abrupt in onset (Cote et al. 1993; Treviño González et al. 2018).
Greco and colleagues (Greco et al. 2013) state that:
“The clinical diagnosis is based on audiovestibular symptoms, ocular inflammation and nonreactive serologic tests for syphilis in the presence of histopathologically proven vasculitis. Due to the variable onset of symptoms and the lack of specific laboratory tests, the diagnosis of Cogan syndrome is a challenge and is often based upon a good response to corticosteroid treatment” (Greco et al. 2013).
Beyond the visual, auditory and vestibular symptoms, CS can involve other organ systems, causing “fever, fatigue, weight loss, lymphadenopathy, hepatitis, splenomegaly, pulmonary nodules, pericarditis, abdominal pain, arthralgia, arthritis, myalgia and urticaria” (Azami et al. 2014).
Vollertsen and colleagues (Vollertsen et al. 1986) provide a sense of the breadth of possible symptoms (beyond the visual, auditory and vestibular) in the Table below.
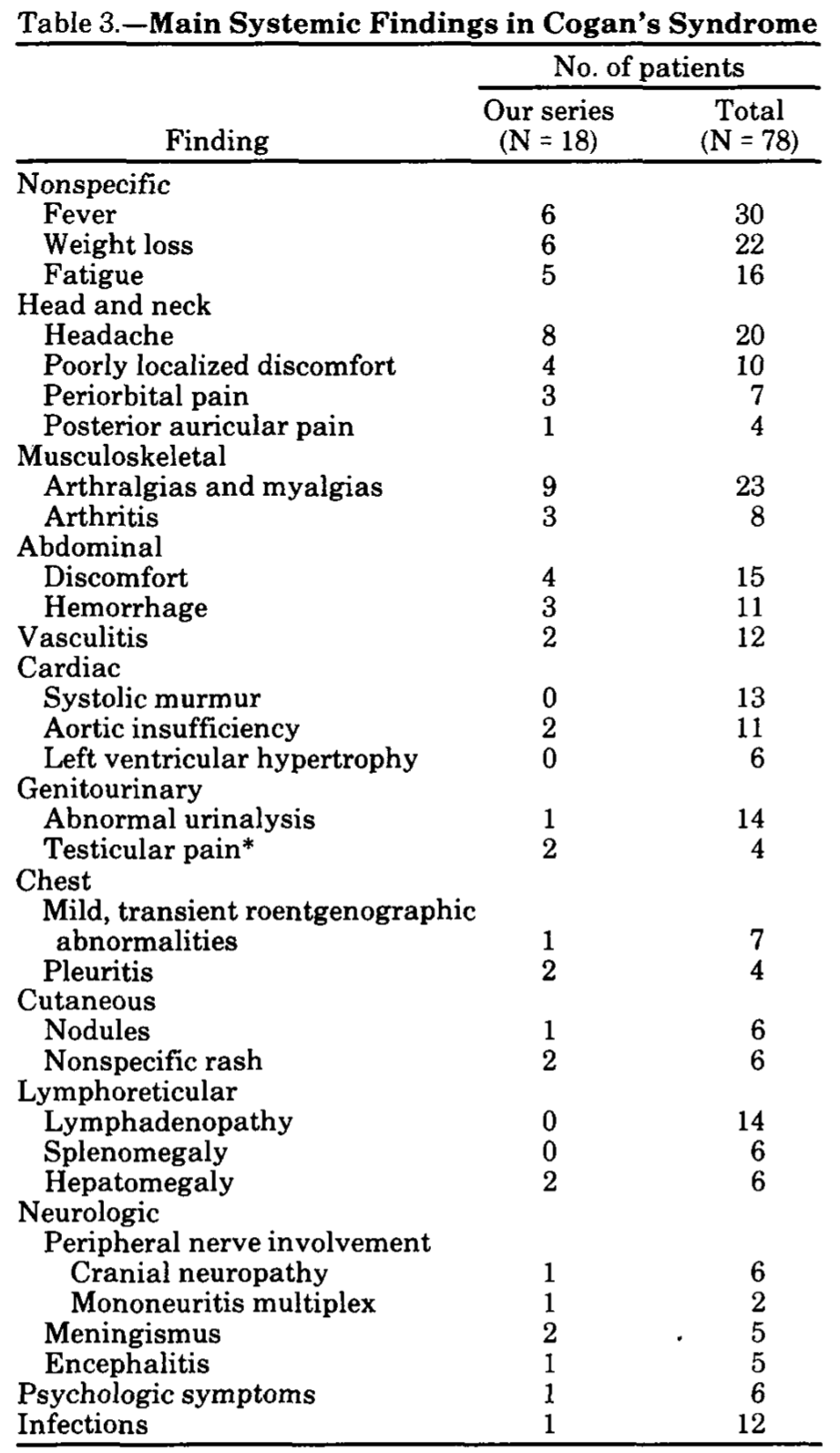
Physical examination
Since CS evolves over days to months, physical examination varies depending upon the organ systems involved at a particular time.
Ophthalmological examination
Some of the relevant ophthalmological findings in CS are shown to advantage by Vollertsen and colleagues (Vollertsen et al. 1986) in the Figure below.
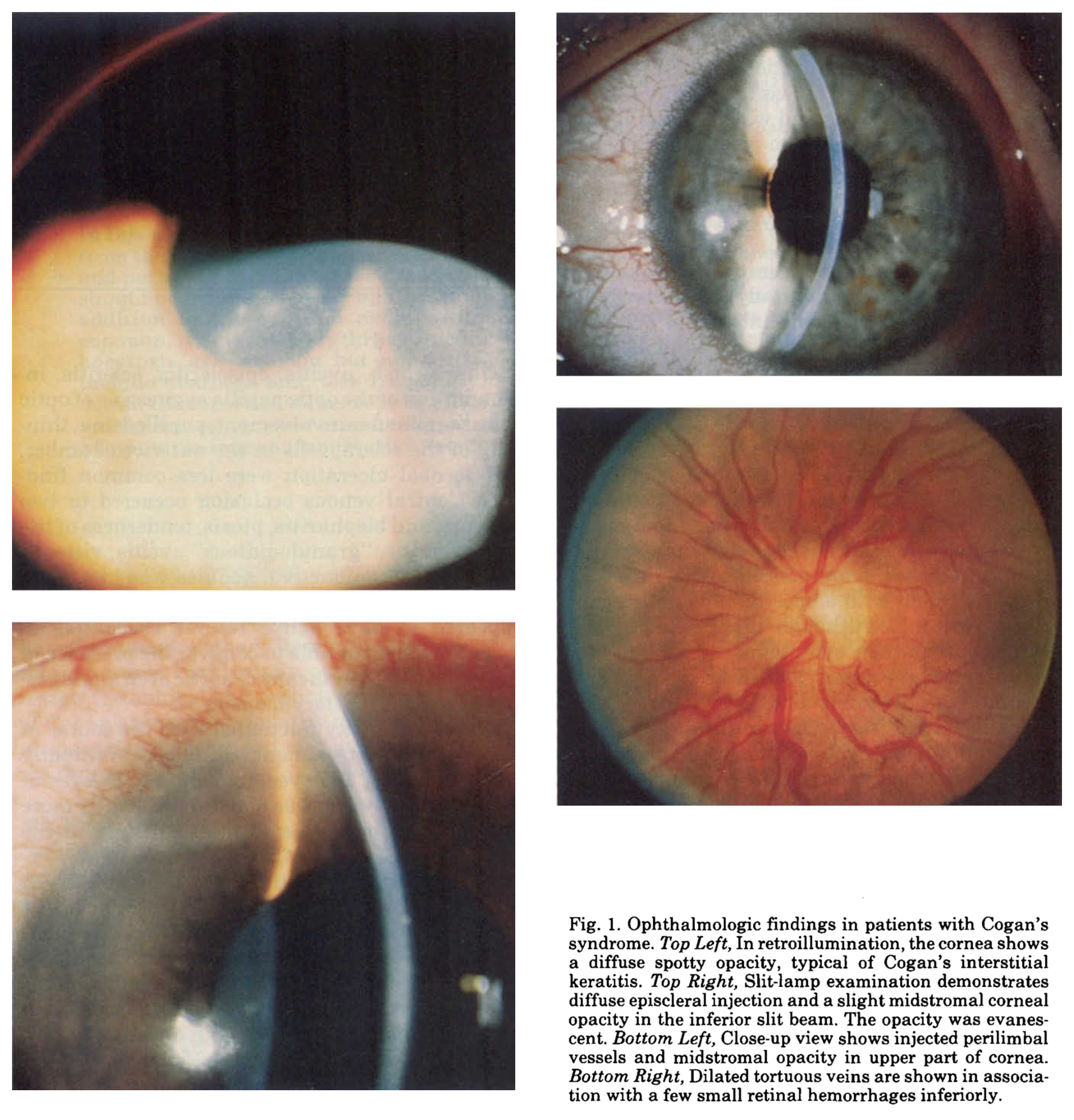
Testing: auditory
Audiometry usually shows approximately symmetrical sensorineural hearing loss. An example from Azami and colleagues (Azami et al. 2014) is displayed in the Figure below.
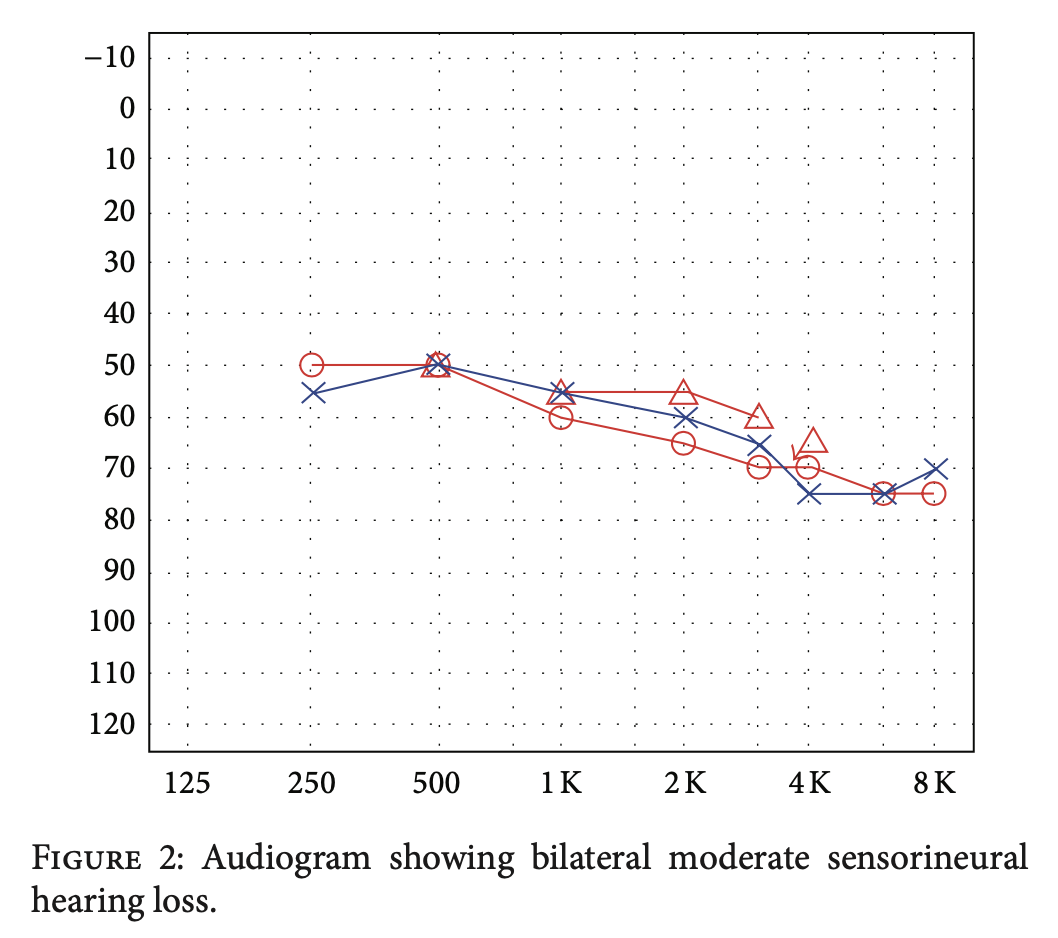
Less commonly, CS can cause low frequency sensorineural hearing loss that may fluctuate (resembling Ménière’s disease). An example of this is shown in the Figure below from Benitez and colleagues.
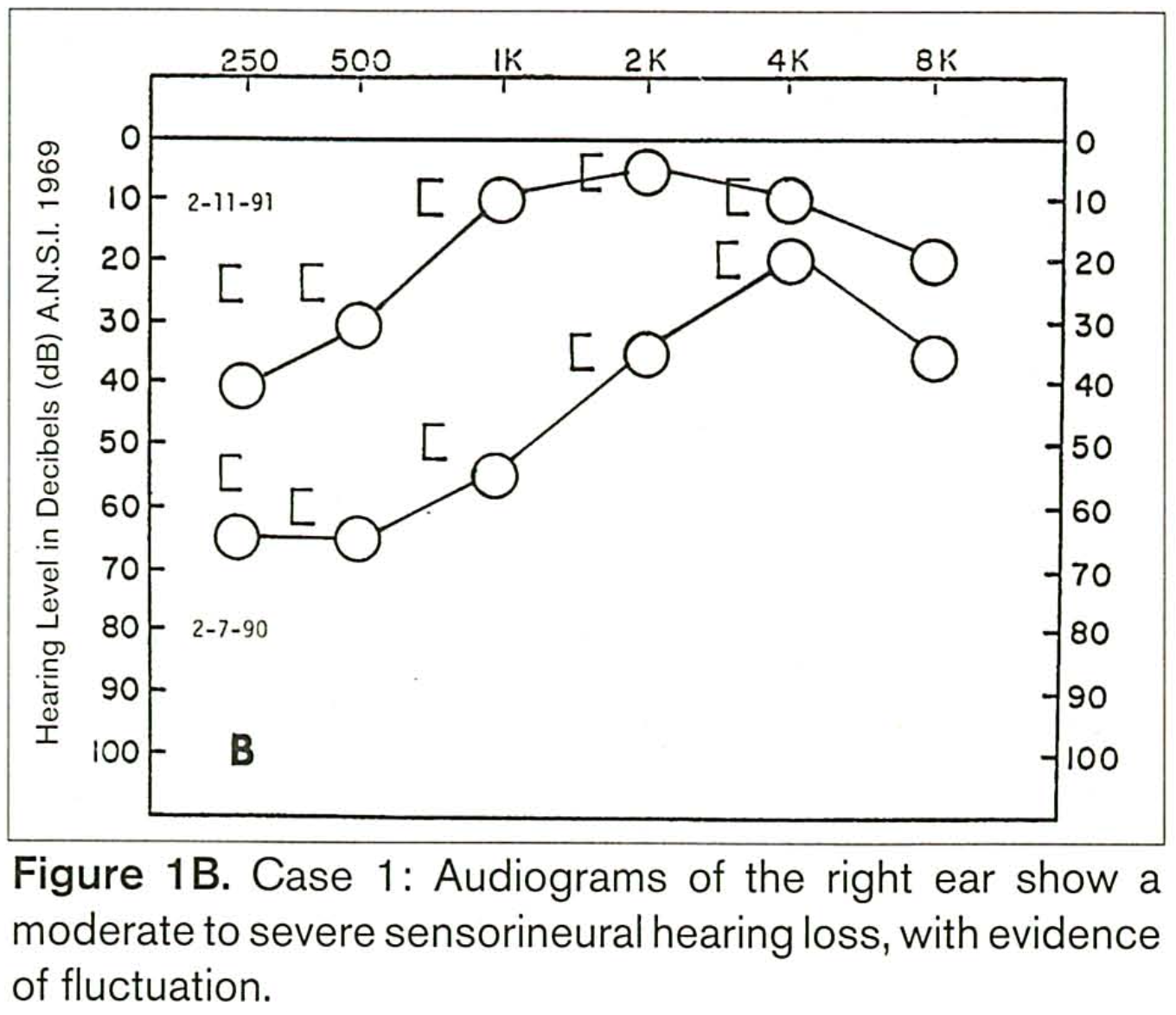
Testing: vestibular
Migliori and colleagues (Migliori et al. 2009) presented a case of a patient with CS. Workup after the initial vestibular symptoms revealed unilateral paresis on caloric testing. Several weeks later repeat caloric testing showed bilateral paresis.
Benitez and colleagues (Benitez et al. 1999) reported a case of typical Cogan syndrome in which the hearing loss on one side involved predominantly the low frequencies, and fluctuated over time (resembling Ménière’s disease). In this patient, electrocochleography (ECoG) showed an elevated SP/AP ratio, as displayed in the Figure below. Note that the tracing is “flipped” compared to the more conventional display of ECoG results.
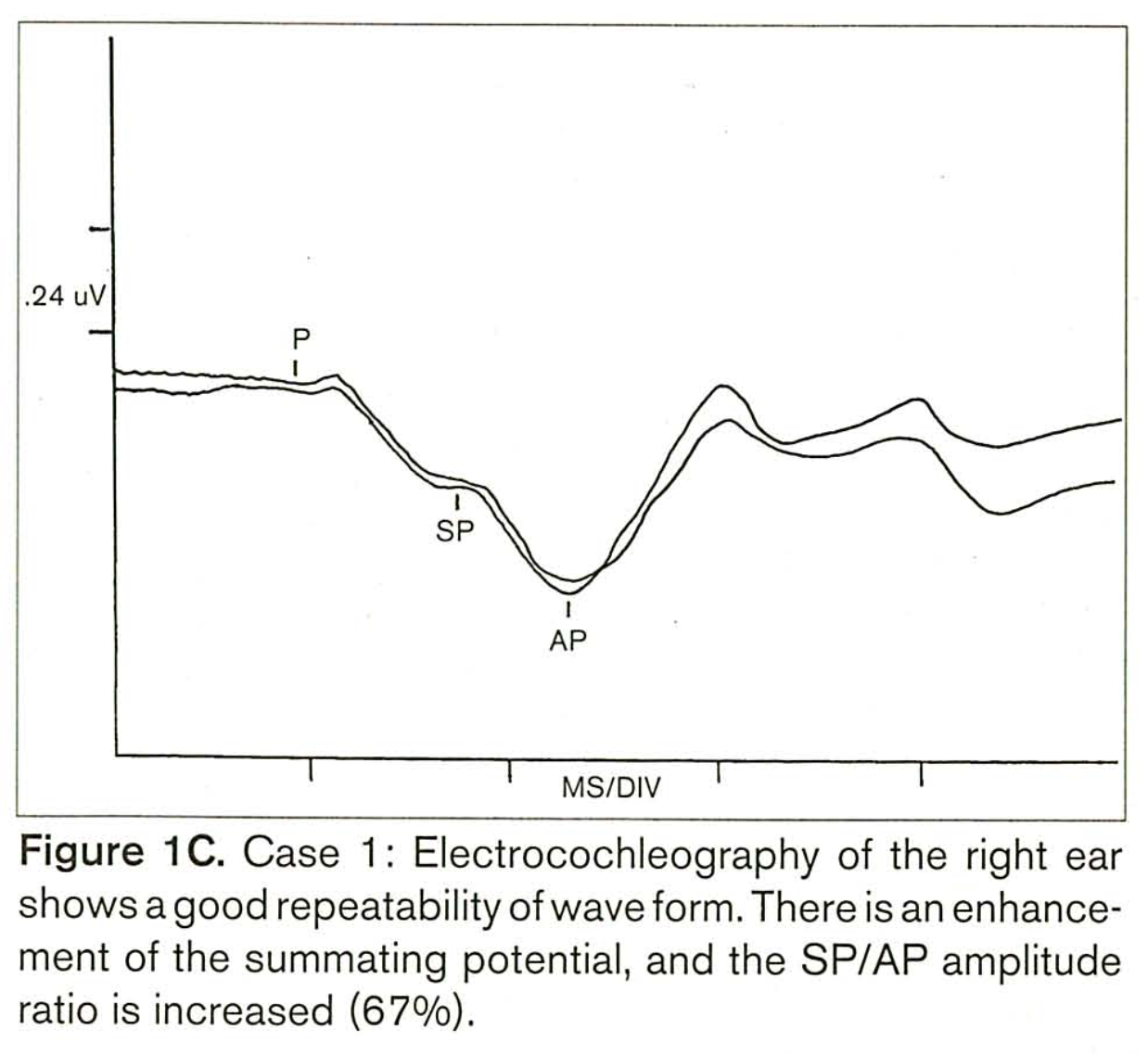
In the same article, Benitez and colleagues (Benitez et al. 1999) reported similar ECoG findings in a patient with atypical Cogan syndrome.
Imaging
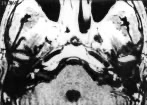
Brain MRI without contrast may reveal T1 hyperintensity in the labyrinths. This is shown in the Figure below from Garcia Berrocal and colleagues (García Berrocal et al. 1999).
Histopathology
Schuknecht and colleagues (Schuknecht and Nadol 1994) described the histopathological findings of both temporal bones in a patient who had had CS:



Differential diagnosis
The differential diagnosis of CS is broad. An example differential offered by Vollertsen and colleagues (Vollertsen et al. 1986) is:
- Nonulcerative, stromal, interstitial keratitis
- Congenital syphilis
- Tuberculosis
- Onchocerciasis
- Sarcoidosis
- Audiovestibular symptoms and eye involvement
- Congenital syphilis
- Congenital mumps
- Congenital rubella
- Viral labyrinthitis
- Systemic diseases associated with eye and audiovestibular involvement
- Connective tissue disorders
- Polyarteritis nodosa
- Wegener’s granulomatosis
- Giant cell arteritis
- Takayasu’s arteritis
- Bechet’s syndrome
- Relapsing polychondritis
- Sarcoidosis
- Bacterial, mycobacterial, and fungal infections
- Connective tissue disorders
- Miscellaneous manifestations
- Drug toxicity with infections or connective tissue disorders
- Vogt-Koyanagi-Harada syndrome
- Effects of a toxin
- Multiple concurrent disorders (for example, bilateral Meniere’s disease and unrelated eye disorders)
Other differential diagnostic suggestions from Azami and colleagues (Azami et al. 2014) include:
- Whipple’s disease
- KID (keratitis, ichthyosis, and deafness) syndrome
- Sjögren’s syndrome
- Rheumatoid arthritis
- Systemic lupus erythematosus
- Granulomatosis with polyangiitis (Wegener’s)
- Ulcerative colitis
- Crohn’s disease
- Central nervous system lymphoma/leukemia
- Anti-phospholipid antibody syndrome
- Chlamydial infection
- Viral infection
- Herpes simplex and varicella zoster infection
- Demyelinating disease (e.g., multiple sclerosis)
- Cerebellopontine angle tumor
Treatment
Management of CS is multidisciplinary, and may involve rheumatology, ophthalmology and audiology. Referral to ophthalmology (for slit lamp examination and possibly fundus photography) is required. If vestibular weakness is identified, then referral to vestibular rehabilitation therapy (VRT) is reasonable. An otolaryngologist may become involved if cochlear implantation is being considered (Minet et al. 1997; Vishwakarma and Shawn 2007).
Generally, “Systemic corticosteroids are always the most widely used and successful therapy in Cogan’s syndrome” (Azami et al. 2014).
Prognosis
The sensorineural hearing loss in CS may respond to steroids. The Figures below from Baumann and colleagues show pairs of pure tone audiograms from three patients before and after treatment with systemic corticosteroids.
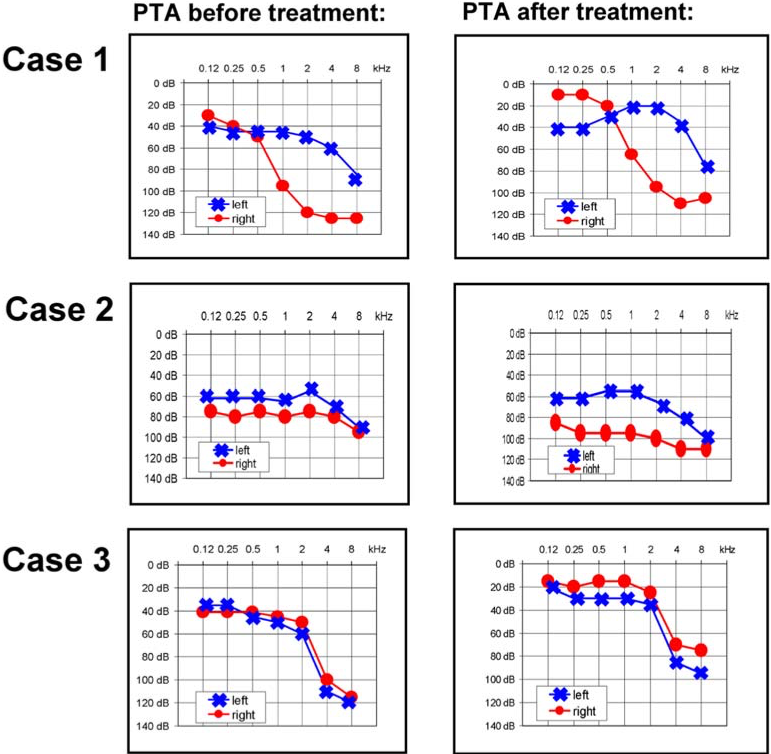
Similar improvement on serial audiometry is documented by Son and Uluap (Son and Ulualp 2009) and Vollertsen and colleagues (Vollertsen et al. 1986).
McDonald and colleagues, summarizing their experience managing 18 patients, state that, “If Cogan’s syndrome is diagnosed early and treatment with corticosteroids is initiated promptly, hearing can be stabilized; otherwise, the prognosis for auditory function is poor” (McDonald et al. 1985).
References
Azami A, Maleki N, Kalantar Hormozi M, Tavosi Z (2014) Interstitial Keratitis, Vertigo, and Vasculitis: Typical Cogan’s Syndrome. Case Rep Med 2014: 830831. doi: 10.1155/2014/830831
Baumann A, Helbling A, Oertle S, Häusler R, Vibert D (2005) Cogan’s syndrome: clinical evolution of deafness and vertigo in three patients. Eur Arch Otorhinolaryngol 262: 45-9. doi: 10.1007/s00405-004-0738-8
Benitez JT, Bojrab DI, Lubbers DE, Arsenault MD (1999) Investigation of endolymphatic hydrops by electrocochleography in patients with Cogan’s syndrome. Ear Nose Throat J 78: 929-33.
Cogan DG (1945) Syndrome of nonsyphilitic interstitial keratitis and vestibuloauditory symptoms. Archives of Ophthalmology 33: 144-149.
Cogan DG (1949) Nonsyphilitic interstitial keratitis with vestibuloauditory symptoms; report of four additional cases. Arch Ophthal 42: 42-9. doi: 10.1001/archopht.1949.00900050045004
Cote DN, Molony TB, Waxman J, Parsa D (1993) Cogan’s syndrome manifesting as sudden bilateral deafness: diagnosis and management. South Med J 86: 1056-60. doi: 10.1097/00007611-199309000-00018
Cundiff J, Kansal S, Kumar A, Goldstein DA, Tessler HH (2006) Cogan’s syndrome: a cause of progressive hearing deafness. Am J Otolaryngol 27: 68-70. doi: 10.1016/j.amjoto.2005.07.006
García Berrocal JR, Vargas JA, Vaquero M, Ramón y Cajal S, Ramírez-Camacho RA (1999) Cogan’s syndrome: an oculo-audiovestibular disease. Postgrad Med J 75: 262-4. doi: 10.1136/pgmj.75.883.262
Grasland A, Pouchot J, Hachulla E, Bletry O, Papo T, Vinceneux P, Study Group for Cogan’s S (2004) Typical and atypical Cogan’s syndrome: 32 cases and review of the literature. Rheumatology (Oxford) 43: 1007-15. doi: 10.1093/rheumatology/keh228
Greco A, Gallo A, Fusconi M, Magliulo G, Turchetta R, Marinelli C, Macri GF, De Virgilio A, de Vincentiis M (2013) Cogan’s syndrome: an autoimmune inner ear disease. Autoimmun Rev 12: 396-400. doi: 10.1016/j.autrev.2012.07.012
McDonald TJ, Vollertsen RS, Younge BR (1985) Cogan’s syndrome: audiovestibular involvement and prognosis in 18 patients. Laryngoscope 95: 650-4. doi: 10.1288/00005537-198506000-00003
Migliori G, Battisti E, Pari M, Vitelli N, Cingolani C (2009) A shifty diagnosis: Cogan’s syndrome. A case report and review of the literature. Acta Otorhinolaryngol Ital 29: 108-13.
Minet M, Deggouj N, Gersdorff M (1997) Cochlear implantation in patients with Cogan’s syndrome: a review of four cases. Eur Arch Otorhinolaryngol 254: 459-62. doi: 10.1007/bf02439981
Morgan RF, Baumgartner CJ (1934) Ménière’s disease complicated by recurrent interstitial keratitis: excellent result following cervical ganglionectomy. Western J Surgery 42: 628-631.
Norton EW, Cogan DG (1959) Syndrome of nonsyphilitic interstitial keratitis and vestibuloauditory symptoms; a long-term follow-up. AMA Arch Ophthalmol 61: 695-7. doi: 10.1001/archopht.1959.00940090697004
Schuknecht HF, Nadol JB, Jr. (1994) Temporal bone pathology in a case of Cogan’s syndrome. Laryngoscope 104: 1135-42. doi: 10.1288/00005537-199409000-00015
Sen M, Honavar SG (2022) David Glendenning Cogan: What’s in a name? Indian J Ophthalmol 70: 720-722. doi: 10.4103/ijo.IJO_377_22
Shamriz O, Tal Y, Gross M (2018) Autoimmune Inner Ear Disease: Immune Biomarkers, Audiovestibular Aspects, and Therapeutic Modalities of Cogan’s Syndrome. J Immunol Res 2018: 1498640. doi: 10.1155/2018/1498640
Son HJ, Ulualp SO (2009) Course of auditory impairment in Cogan’s syndrome. Am J Otolaryngol 30: 65-8. doi: 10.1016/j.amjoto.2008.02.003
Treviño González JL, Soto-Galindo GA, Moreno Sales R, Morales Del Ángel JA (2018) Sudden sensorineural hearing loss in atypical Cogan’s syndrome: A case report. Ann Med Surg (Lond) 30: 50-53. doi: 10.1016/j.amsu.2018.04.030
Vishwakarma R, Shawn TJ (2007) Cochlear implant in Cogan’s syndrome. Eur Arch Otorhinolaryngol 264: 1121-4. doi: 10.1007/s00405-007-0362-5
Vollertsen RS, McDonald TJ, Younge BR, Banks PM, Stanson AW, Ilstrup DM (1986) Cogan’s syndrome: 18 cases and a review of the literature. Mayo Clin Proc 61: 344-61. doi: 10.1016/s0025-6196(12)61951-x
![]()