By Marcello Cherchi, MD PhD
For patients
It is uncommon, but possible, to have a “stroke of the inner ear,” and this usually causes sudden hearing loss and disequilibrium, though often there are other symptoms as well. Management of inner ear strokes is similar to management of strokes in general.
For clinicians
Overview
Arterial blood supply to the inner ear relies entirely on the labyrinthine artery. Venous drainage is more diffuse, and there are intra-labyrinthine anastomoses; but, the single arterial supply renders the labyrinth fairly vulnerable to ischemia. That being said, the labyrinthine artery usually branches off the anterior inferior cerebellar artery (AICA), so usually embolic strokes will result in deficits in the territories of both the labyrinthine artery and the AICA. It is far less common for a stroke to be so focal as to affect only the inner ear, but when it does, the clinical presentation can mimic primary otologic diseases such as vestibular neuritis, labyrinthitis and sudden sensorineural hearing loss. Most inner ear strokes are ischemic; a minority are hemorrhagic. Management of inner ear strokes is essentially a combination of managing stroke, sensorineural hearing loss (if present) and vestibular weakness (if present). Prognosis is guarded, and tends to be poor if the inner ear stroke is hemorrhagic.
Introduction
Arterial blood supply to the inner ear relies entirely on the labyrinthine artery. Venous drainage is more diffuse, and there are intra-labyrinthine anastomoses; but, the single arterial supply renders the labyrinth fairly vulnerable to ischemia. However, the extent to which vascular disease accounts for otovestibular disorders is unclear.
Epidemiology
Having a discrete acute “stroke” (whether ischemic or hemorrhagic) isolated to the inner ear is uncommon because the territory is so focal. More commonly, inner ear “strokes” occur in association with a stroke in closely related vascular territories (typically the anterior inferior ischemic artery).
Whether gradually progressive age-related microvascular circulatory disturbances cause or contribute to age-related attrition of inner ear function is less clear (Johnsson and Hawkins 1972).
Pathophysiological mechanism of disease
The vascular anatomy of the inner ear is reasonably well-understood thanks to a number of meticulous temporal bone studies (Mazzoni 1990; Mei et al. 2018), including studies of individuals who in life had known vascular pathology (Belal 1979, 1980). There are several anastomotic complexes within the inner ear (Hansen 1971a, b). The inner ear vasculature probably also has some degree of autoregulation in addition to autonomic modulation (Lawrence 1980).
Animal studies suggest that temporary interruption of blood supply affects cochlear hair cells sooner than vestibular hair cells (Kimura 1986), though it is unknown whether this pattern also holds in humans.
The arterial supply of the inner ear is tenuous as it relies exclusively on the labyrinthine artery. Most reported cases of “inner ear strokes” are ischemic and presumed to be embolic in origin.
More rarely reported are cases of inner ear hemorrhage (Cherchi et al. 2006; Dumberger et al. 2021; Parikh and Frank 2017; Sabatini and Kutz 2009; Vivas et al. 2018). Some such cases appear to be spontaneous (Sabatini and Kutz 2009) or presumed secondary to hypertension (Vivas et al. 2018), but many occur in the context of malignancy with presumed hyperviscosity (Dumberger et al. 2021).
Clinical presentation
Clinically it is difficult to identify cases of inner ear ischemia confidently. Probably the most convincing cases are those in which the vertebral artery is compromised by dissection (confirmed by imaging), and in close temporal proximity the patient subsequently develops apoplectic unilateral audiologic and vestibular symptoms (Choi et al. 2006; Eliezer et al. 2019).
There are also cases of labyrinthine artery embolization even in the absence of vertebral artery dissection (Liqun et al. 2018; Nam et al. 2021). However, because the labyrinthine artery usually branches off the anterior inferior cerebellar artery (AICA), it is unusual for embolic events to be restricted to the territory of the labyrinthine artery. More commonly, embolic events leading to ischemia/infarction in the territory of the labyrinthine artery usually are accompanied by ischemia/infarction in territory of the AICA distal to the labyrinthine artery. Lee and colleagues (Lee et al. 2009) studied a series of 82 patients with AICA strokes and reported that 60% of cases included findings referable to infarction of the cochlea and labyrinth; however, only 5% of cases had isolated vestibular labyrinth infarction (without involvement of the cochlea or cerebellum), only 4% of cases had isolated cochlear involvement (without involvement of the vestibular labyrinth or cerebellum), and only 1% had “isolated audiovestibular loss without central symptoms or signs.”
Physical examination
The audiologic dysfunction of cochlear infarction will manifest as sensorineural hearing loss, and thus could mimic idiopathic sudden onset sensorineural hearing loss.
The vestibular dysfunction of infarction of the vestibular labyrinth will manifest similarly to vestibular neuritis.
If a patient exhibits any physical examination findings of cerebellar or brainstem dysfunction, then a diagnosis of infarction should always be high on the differential diagnosis (as opposed to primary inner ear disease).
Ocular motor examination
Ocular motor examination of a patient with an infarct limited to the vestibular labyrinth will appear similar to vestibular neuritis. For example, in the case reported by Nam and colleagues (Nam et al. 2021) of a patient with a left-sided vestibular labyrinthine infarction, the patient exhibited spontaneous right beat and right torsional nystagmus.
Testing: auditory
Cochlear infarction will manifest as sensorineural hearing loss.
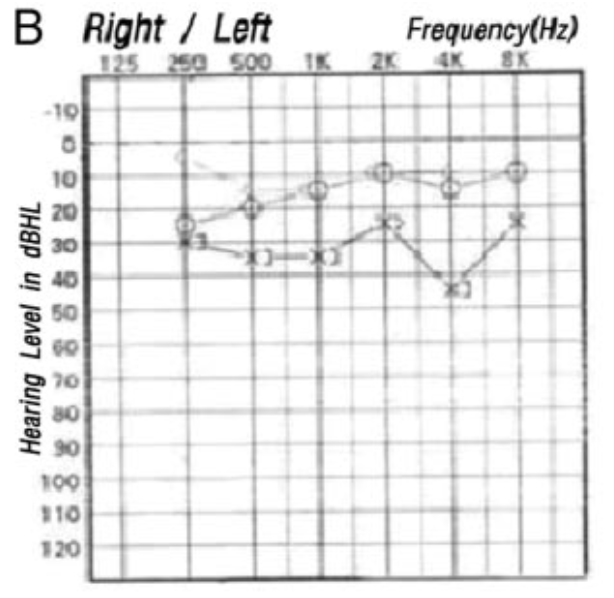
The Figure below, from Lee and colleagues (Lee et al. 2009), shows left-sided sensorineural hearing loss in a patient with a left AICA territory infarction.
Testing: vestibular
In a patient with labyrinthine vestibular function, otovestibular testing would be expected to show vestibular weakness.
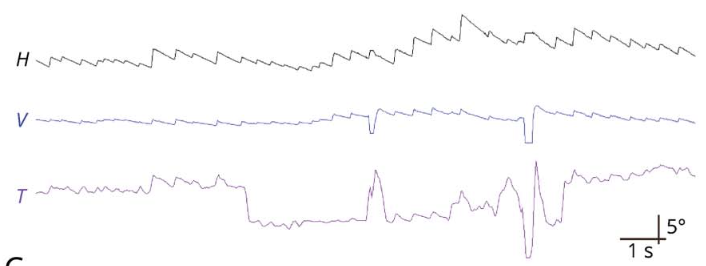
The Figure below, from Nam and colleagues (Nam et al. 2021), shows a video oculographic tracing from a patient with left-sided vestibular labyrinthine infarction. This shows spontaneous nystagmus that is predominantly right beat, with more modest components of right torsional and up beat.
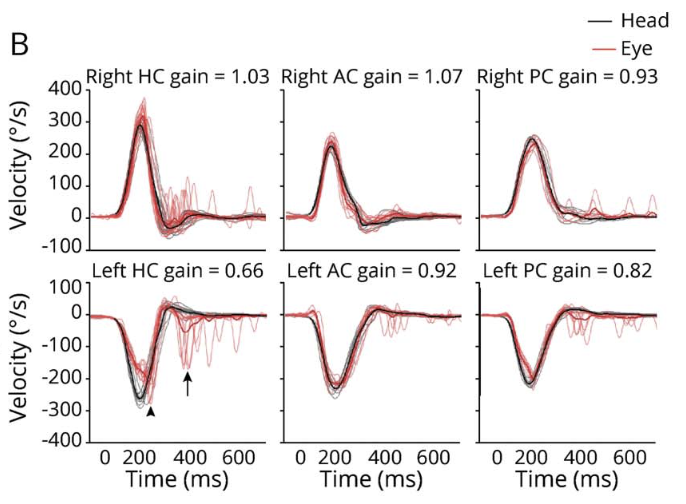
The Figure below, from Nam and colleagues (Nam et al. 2021), shows results of video head impulse testing (vHIT) from a patient with left-sided vestibular labyrinthine infarction. The lower left panel shows vHIT from the left horizontal canal, which exhibits low gain and overt compensatory saccades.
Testing: other
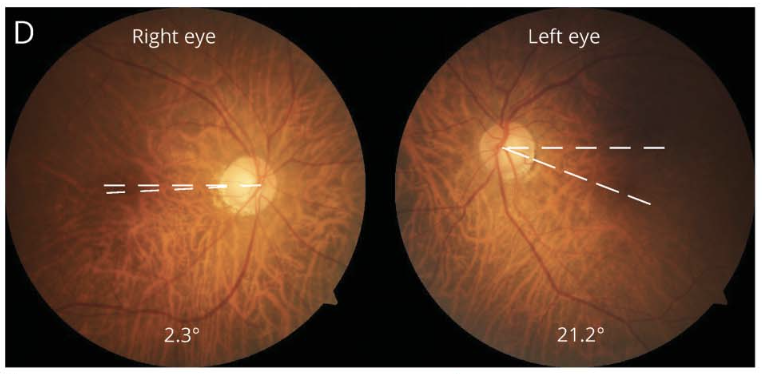
The Figure below, from Nam and colleagues (Nam et al. 2021), shows results of fundus photography from a patient with left-sided vestibular labyrinthine infarction. This shows leftward cyclodeviation of both eyes.
Imaging
Truly isolated arterial infarction of the labyrinth is rarely detectable on imaging. Venous infarction from hypercoagulable states, such as leukemia, may manifest with labyrinthine hemorrhage (Cherchi et al. 2006), which may be more readily detectable.
Histopathology
Temporal bone studies of patients who suffered labyrinthine artery occlusion generally show “severe degenerative changes and ossification of the cochlea and the entire vestibular labyrinth” (Belal 1979), or “hemorrhages followed by severe degenerative changes that ultimately produce total ossification” (Belal 1980).
Differential diagnosis
Practically, because of the vascular anatomy at issue, the majority of patients with audiologic and/or vestibular symptoms secondary to inner ear ischemia will also have signs and symptoms referrable to brainstem and cerebellar deficits in the territory of the anterior inferior cerebellar artery (Adams 1943), which should immediately alert the clinician that the case is not one of primary inner ear disease.
However, the rare cases of infarctions that are so focal as to involve just the inner ear are troubling because they can mimic other diseases; specifically, infarction of the vestibular structures of the labyrinth (in isolation) can mimic vestibular neuritis (Nam et al. 2021), infarction of the cochlea (in isolation) can mimic idiopathic sudden sensorineural hearing loss, and isolated infarction of the vestibular and cochlear labyrinth (in isolation) can mimic labyrinthitis. The physiologic deficit of infarction of the vestibular labyrinth will be functionally similar to vestibular neuritis, and thus would not be differentiated on HiNTS (Nam et al. 2021).
Despite the fact that such focal infarctions are possible, it is not practical to obtain advanced imaging in every patient whose presentation is compatible with vestibular neuritis, labyrinthitis, or sudden sensorineural hearing loss. However, the clinical index of suspicion for an inner ear stroke should be higher in patients with known risk factors such as cardiovascular disease, hyper coagulable states, malignancy, or a history suggestive of current or recent vertebral artery dissection.
Treatment
If auditory and vestibular symptoms are truly due to infarction in the territory of the labyrinthine artery, then the source of the infarct should be sought and treated, generally under the guidance of a vascular neurologist. The hearing loss would be treated similarly to other forms of sensorineural hearing loss. For the vestibular deficits, the patient should be offered a trial of vestibular rehabilitation therapy.
Whether the inner ear “component” of stroke merits different or additional management beyond what would normally be offered for stroke is unclear. Animal studies suggest that the rheologic agent pentoxifylline promotes cochlear blood flow (Coleman et al. 1990; LaRouere et al. 1992; Maass and Ludwig 1984; Quirk et al. 1988). Several studies in humans reach similar conclusions (Cesarone et al. 2002; Incandela et al. 2002), but it is unclear whether this has a clinically meaningful effect, so it is unclear whether pentoxifylline has any role in the treatment (or prevention) of inner ear ischemia.
Although aspirin is often used for secondary stroke prophylaxis in patients with atherosclerotic disease, aspirin is also ototoxic, so where medically feasible, it is preferable to use a non-ototoxic anti-platelet agent such as clopidogrel.
Prognosis
Infarcted neurons and labyrinthine structures do not regenerate, so if any recovery occurs it is likely due to compensatory mechanisms. The prognosis in cases of inner ear hemorrhage is generally poor (Dumberger et al. 2021).
References
Adams RD (1943) Occlusion of the anterior inferior cerebellar artery. Archives of Neurology & Psychiatry 49: 765-770. doi: 10.1001/archneurpsyc.1943.02290170135010
Belal A, Jr. (1979) The effects of vascular occlusion on the human inner ear. J Laryngol Otol 93: 955-68. doi: 10.1017/s0022215100087958
Belal A, Jr. (1980) Pathology of vascular sensorineural hearing impairment. Laryngoscope 90: 1831-9. doi: 10.1288/00005537-198011000-00011
Cesarone MR, Incandela L, Belcaro G, De Sanctis MT, Nicolaides AN, Griffin M, Geroulakos G, Ramaswami G (2002) Treatment of vascular inner ear disease in vascular patients with pentoxifylline: a controlled, randomized trial. Angiology 53 Suppl 1: S23-6.
Cherchi M, Huo EJ, Nelson NW, Frankfurt O, Russell EJ, Raizer JJ (2006) Gradual hearing loss with bilateral labyrinthine hemorrhage in chronic myelogenous leukemia. Neurology 67: 177-8. doi: 10.1212/01.wnl.0000223624.82210.21
Choi KD, Chun JU, Han MG, Park SH, Kim JS (2006) Embolic internal auditory artery infarction from vertebral artery dissection. J Neurol Sci 246: 169-72. doi: 10.1016/j.jns.2006.02.014
Coleman JK, Quirk WS, Dengerink HA, Wright JW (1990) Pentoxifylline increases cochlear blood flow while decreasing blood pressure in guinea pigs. Hear Res 47: 169-74. doi: 10.1016/0378-5955(90)90174-n
Dumberger LD, Hwa TP, Panara K, Husain S, Yver C, Bigelow DC (2021) Profound Sudden Sensorineural Hearing Loss in Hematologic Malignancy: A Case for Urgent Cochlear Implantation With Discussion and Systematic Review of the Literature. Otol Neurotol 42: e815-e824. doi: 10.1097/MAO.0000000000003131
Eliezer M, Verillaud B, Guichard JP, Kania R, Toupet M, Herman P, Houdart E, Hautefort C (2019) Labyrinthine infarction caused by vertebral artery dissection: consideration based on MRI. J Neurol. doi: 10.1007/s00415-019-09456-0
Hansen CC (1971a) Vascular anatomy of the human temporal bone. I. Anastomoses of the membranous labyrinth and its bony capsule. Arch Klin Exp Ohren Nasen Kehlkopfheilkd 200: 83-98. doi: 10.1007/BF00418192
Hansen CC (1971b) Vascular anatomy of the human temporal bone. II. Anastomoses inside the labyrinthine capsule. Arch Klin Exp Ohren Nasen Kehlkopfheilkd 200: 99-114. doi: 10.1007/BF00418193
Incandela L, Cesarone MR, Belcaro G, De Sanctis MT, Nicolaides AN, Griffin M, Geroulakos G, Ramaswami G (2002) Treatment of vascular inner ear disease with pentoxifylline: a 4-week, controlled, randomized trial. Angiology 53 Suppl 1: S19-22.
Johnsson LG, Hawkins JE, Jr. (1972) Vascular changes in the human inner ear associated with aging. Ann Otol Rhinol Laryngol 81: 364-76. doi: 10.1177/000348947208100307
Kimura RS (1986) Animal models of inner ear vascular disturbances. Am J Otolaryngol 7: 130-9. doi: 10.1016/s0196-0709(86)80042-4
LaRouere MJ, Sillman JS, Tsai MT, Nuttall AL (1992) The effect of pentoxifylline on cochlear blood flow. Otolaryngol Head Neck Surg 106: 87-91. doi: 10.1177/019459989210600133
Lawrence M (1980) Control mechanisms of inner ear microcirculation. Am J Otolaryngol 1: 324-33. doi: 10.1016/s0196-0709(80)80035-4
Lee H, Kim JS, Chung EJ, Yi HA, Chung IS, Lee SR, Shin JY (2009) Infarction in the territory of anterior inferior cerebellar artery: spectrum of audiovestibular loss. Stroke 40: 3745-51. doi: 10.1161/STROKEAHA.109.564682
Liqun Z, Park KH, Kim HJ, Lee SU, Choi JY, Kim JS (2018) Acute Unilateral Audiovestibulopathy due to Embolic Labyrinthine Infarction. Front Neurol 9: 311. doi: 10.3389/fneur.2018.00311
Maass B, Ludwig D (1984) Effect of pentoxifylline (‘Trental’) on cochlear blood flow as measured by hydrogen wash-out. Curr Med Res Opin 9: 52-5. doi: 10.1185/03007998409109559
Mazzoni A (1990) The vascular anatomy of the vestibular labyrinth in man. Acta Otolaryngol Suppl 472: 1-83. doi: 10.3109/00016489009121137
Mei X, Atturo F, Wadin K, Larsson S, Agrawal S, Ladak HM, Li H, Rask-Andersen H (2018) Human inner ear blood supply revisited: the Uppsala collection of temporal bone-an international resource of education and collaboration. Ups J Med Sci 123: 131-142. doi: 10.1080/03009734.2018.1492654
Nam HW, Yoo D, Lee SU, Choi JY, Yu S, Kim JS (2021) Pearls & Oy-sters: Labyrinthine Infarction Mimicking Vestibular Neuritis. Neurology 97: 787-790. doi: 10.1212/WNL.0000000000012297
Parikh SK, Frank SA (2017) Clinical Reasoning: Labyrinthine hemorrhage: An unusual etiology for peripheral vertigo. Neurology 88: e14-e18. doi: 10.1212/WNL.0000000000003481
Quirk WS, Dengerink HA, Bademian MJ, Hall KW, Wright JW (1988) The effects of pentoxifylline on cochlear blood flow in normotensive and spontaneously hypertensive rats. Hear Res 36: 175-80. doi: 10.1016/0378-5955(88)90059-7
Sabatini PR, Kutz JW, Jr. (2009) Radiology quiz case 1. Intralabyrinthine hemorrhage. Arch Otolaryngol Head Neck Surg 135: 612, 614. doi: 10.1001/archoto.2009.39-a
Vivas EX, Panella NJ, Baugnon KL (2018) Spontaneous Labyrinthine Hemorrhage: A Case Series. Otolaryngol Head Neck Surg 159: 908-913. doi: 10.1177/0194599818785900
![]()