By Marcello Cherchi, MD PhD
For patients
Mal de debarquement syndrome (MdDs) usually consists of a persistent sensation of rocking that occurs after traveling in some kind of vehicle for several hours or days. Oddly, the sensation improves when a patient gets into a moving vehicle. Patients with MdDs usually have normal physical examinations, and results of testing and brain imaging are normal. If symptoms are intrusive then some patients may consider treatment with medications or physical therapy.
For clinicians
Overview
Mal de debarquement syndrome (MdDs) is a disorder usually triggered by prolonged exposure to passive motion (typically vehicular travel for at least several hours) whose symptoms begin at some point after that exposure to motion and usually include rocking disequilibrium that, in the majority of cases, transiently improves when the patient gets back into a moving vehicle. The mechanism of disease is unclear. The overall duration of an episode can last weeks to years. The typical age at a first episode is in the 4th or 5th decade, and the majority of patients are women. Usually physical examination and workup (otovestibular testing, brain imaging) are normal. The main differential diagnosis is migraine associated vertigo. Some patients notice symptomatic improvement with a low dose benzodiazepine, SSRI or SNRI. Non-pharmacologic interventions include vestibular rehabilitation therapy and a strategy developed by Dr. Mingjia Dai.
Introduction
Mal de debarquement syndrome (MdDs) is a condition in which an individual is exposed to motion (usually vehicular travel of some sort) for a prolonged period, and shortly after returning to solid ground, the individual continues to feel as if in motion. A striking feature of this form of disequilibrium is that the sensation improves (often completely disappears) when the patient gets back into a moving vehicle. Almost no other vestibular disorder behaves in this fashion.
Nomenclature
The earlier definitions of MdDs required a trigger of prolonged exposure to motion.
However, subsequent literature recognized a subset of patients who did not have any such motion exposure, yet resembled MdDs in most other respects (such as symptomatic improvement when in a moving vehicle). Classifying such patients as a “sub-type of MdDs” seems peculiar since there is no “disembarkation” from any moving vehicle. However, in recognition of the other shared characteristics, this is sometimes called “non-motion-triggered MdDs,” although such terminology is rather internally contradictory.
History
Bronstein and colleagues (Bronstein et al. 2013), citing Pike (Pike 1986), state that Whittel published the first description of mal de debarquement in 1689:
“As we marched here upon good Ground, the Souldiers would stumble and sometimes fall because of a dissiness in their Heads after they had been so long toss’d at Sea, the very Ground seem’d to rowl up and down for some days, according to the manner of the Waves” (Whittel 1689).
Erasmus Darwin (1731 – 1802) offered another early description of MdDs in volume 1 of Zoonomia (Darwin et al. 1796):
“Those, who have been upon the water in a boat or ship so long, that they have acquired the necessary habits of motion upon that unstable element, at their return on land frequently think in their reveries, or between sleeping and waking, that they observe the room, they sit in, or some of its furniture, to librate like the motion of the vessel. This I have experienced myself, and have been told, that after long voyages, it is some time before these ideas entirely vanish. The same is observable in a less degree after having travelled some days in a stage coach, and particularly when we lie down in bed, and compose ourselves to sleep; in this case it is observable, that the rattling noise of the coach, as well as the undulatory motion, haunts us.”
Eighty-five years later Dr. Irwin wrote (Irwin and Cantab 1881):
“The ocean habit teaches the [semicircular] canals to adapt themselves to the new condition of things, and to pass over unheeded erroneous impressions which were noticed at first. In fact, the new habit may become so strong that a disturbance of it, by a return to the land, will be marked by similar phenomena; hence the unsteady gait sometimes observable in a not-drunken sailor during his first few hours on shore after a long and stormy voyage.”
Similar descriptions appear in various nautical literature.
Jack London (1876 – 1916) wrote in Sea-wolf (London 1904):
“The boat’s stern touched the hard shingle. I sprang out, extending my hand to Maud. The next moment she was beside me. As my fingers released hers, she clutched for my arm hastily. At the same moment I swayed, as about to fall to the sand. This was the startling effect of the cessation of motion. We had been so long upon the moving, rocking sea that the stable land was a shock to us. We expected the beach to lift up this way and that, and the rocky walls to swing back and forth like the sides of a ship; and when we braced ourselves, automatically, for these various expected movements, their non-occurrence quite overcame our equilibrium.”
And Cruise of the Snark (London 1911):
“So we went ashore with them across a level flashing sea to the wonderful green land. We landed on a tiny wharf, and the dream became more insistent; for know that for twenty-seven days we had been rocking across the ocean on the tiny Snark. Not once in all those twenty-seven days had we known a moment’s rest, a moment’s cessation from movement. This ceaseless movement had become ingrained. Body and brain we had rocked and rolled so long that when we climbed out on the tiny wharf kept on rocking and rolling. This, naturally, we attributed to the wharf. It was projected psychology. I spraddled along the wharf and nearly fell into the water. I glanced at Charmian, and the way she walked made me sad. The wharf had all the seeming of a ship’s deck. It lifted, tilted, heaved and sank; and since there were no handrails on it, it kept Charmian and me busy avoiding falling in. I never saw such a preposterous little wharf. Whenever I watched it closely, it refused to roll; but as soon as I took my attention off from it, away it went, just like the Snark. Once, I caught it in the act, just as it upended, and I looked down the length of it for two hundred feet, and for all the world it was like the deck of a ship ducking into a huge head-sea.”
Epidemiology
The majority of patients are women, but the reported sex ratio varies across studies. The population of 27 patients studied by Hain and colleagues (Hain, Hanna et al. 1999) consisted of 26 women (96%) and 1 man (4%). The population of 64 patients studied by Cha and colleagues (Cha, Brodsky et al. 2008) consisted of 48 women (75%) and 16 men (25%).
The peak age of onset varies among studies from the 4th to the 5th decades. Hain and colleagues (Hain, Hanna et al. 1999) report the age of onset as 49.3 ± 10.3 years. Cha and colleagues (Cha, Brodsky et al. 2008) report a peak age of onset of 39 ± 13 years.
Pathophysiological mechanism of disease
The underlying pathophysiology of MdDs is unknown. Proposed mechanisms have included:
- Vestibular dysfunction secondary to overstimulation (Murphy 1993).
- “Vestibular hallucination” due to inappropriate “release of stored vestibular information from the hippocampus” (Moeller and Lempert 2007).
- “Aberrant adaptation” (Lewis 2004). Lewis elaborated that, “During passive motion there may be a mismatch between the vestibular inputs that encode head motion and other sensory and motor cues that do not correlate with the labyrinthine signals. Central adaptation may minimize the symptoms provoked by this mismatch, but when the [passive] movement ends, the brain must readapt to the stationary environment. It has been suggested that patients with MDD [mal de debarquement syndrome] undergo a physioogic adaptative process during passive motion, but for unknown reasons they do not readily adapt back to the stable environment. This impaired return to baseline results in a perception of motion when the subject is stationary.”
- Dynamic multi-sensorimotor neural plasticity that adapts during exposure to motion but then maladaptively fails to revert when motion ceases (Gordon, Spitzer et al. 1995).
Further developing the last two suggestions, some investigators reasonably hypothesize that at some level MdDs must be an example of a mis-applied internal predictive model — specifically, a predictive model that had been developed and adopted during exposure to passive motion, which then is mistakenly maintained even after return to stable ground (Hain and Helminski 2007). Cha (Cha 2015) describes this as, “MdDs may be a disorder of oversynchronization of brain networks caused by entrainment to the background low-amplitude-oscillating environments typical of the triggers of MdDs.”
For the so-called “non-motion-triggered” type of MdDs, it is unclear how such entrainment would occur, or how the internal predictive modeling hypothesis would apply. Cha and colleagues (Cha, Brodsky et al. 2008) note that of 18 patients with so-called “mixed MdDs” (meaning subjects who experienced both motion-triggered and non-motion-triggered MdDs), the first-ever episode in all of them was a motion-triggered episode. This observation suggests that the initial motion-triggered MdDs episode did trigger development of an internal predictive model, and although that initial episode resolved, it later reappeared without any exposure to motion; in other words, a latent (previously learned) internal predictive model was “unleashed.” This hypothesis begs the question of what triggered the “unleashing” of the (previously) quiescent internal predictive model.
Clinical presentation
Cha and colleagues (Cha, Brodsky et al. 2008) retrospectively reviewed 64 patients with MdDs. The investigators distinguished patients with “pure” MdDs (those whose episodes were only triggered by motion) from those with “mixed” MdDs (patients in whom some episodes were triggered by motion and other episodes were spontaneous). Triggers were as shown in the Table below.
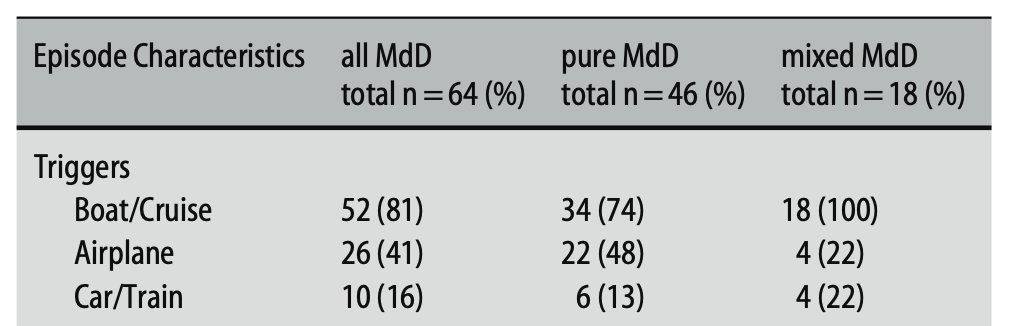
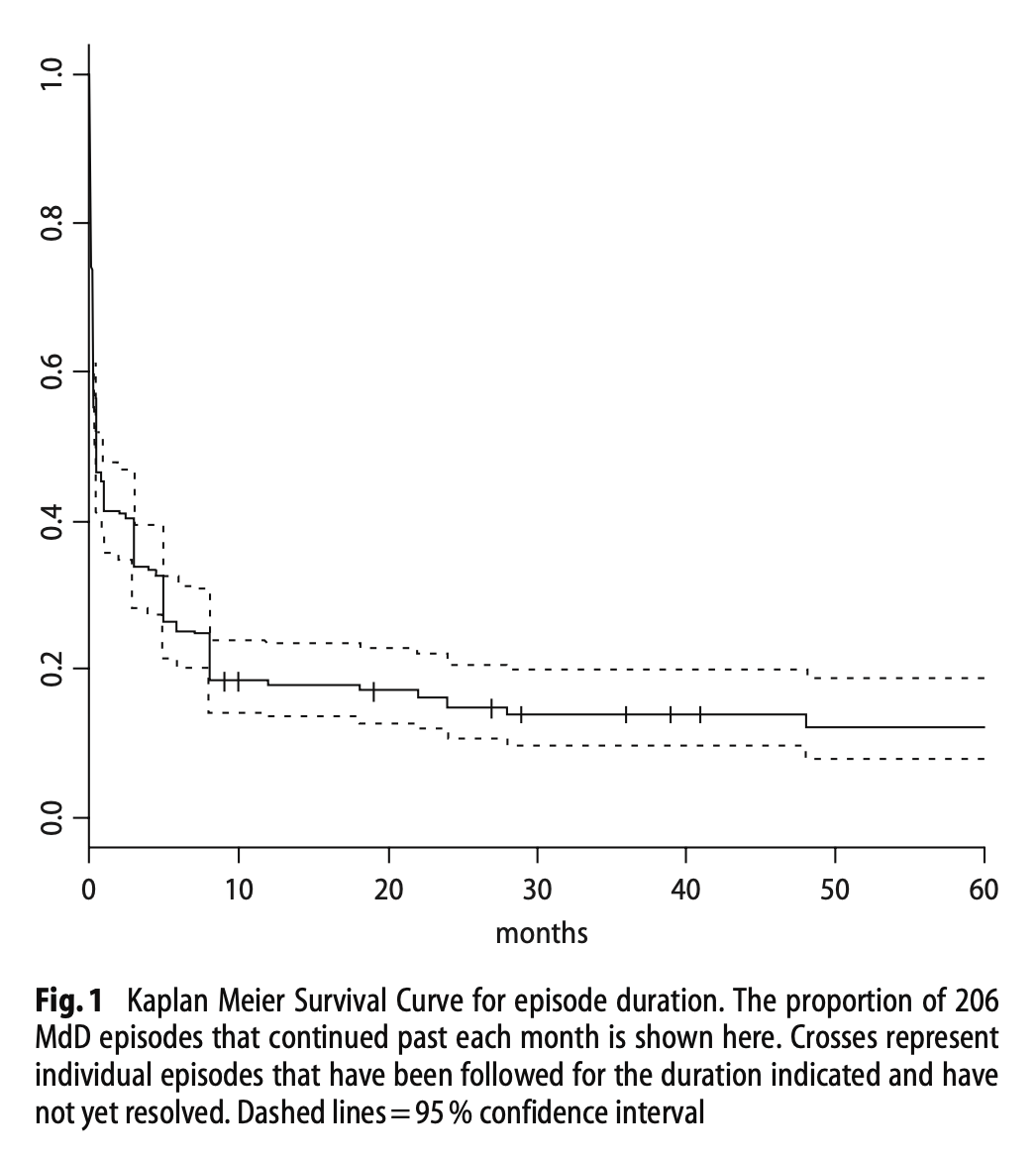
In the series studied by Cha and colleagues (Cha, Brodsky et al. 2008), the 64 patients experienced a total of 206 episodes. Of these:
- 51% lasted >1 month
- 18% lasted >1 year
- 15% lasted >2 years
- 12% lasted >4 years
- 11% lasted >5 years
The Figure below, from Cha and colleagues (Cha, Brodsky et al. 2008), shows a Kaplan-Meier curve for duration of 206 episodes of MdDs in 64 patients.
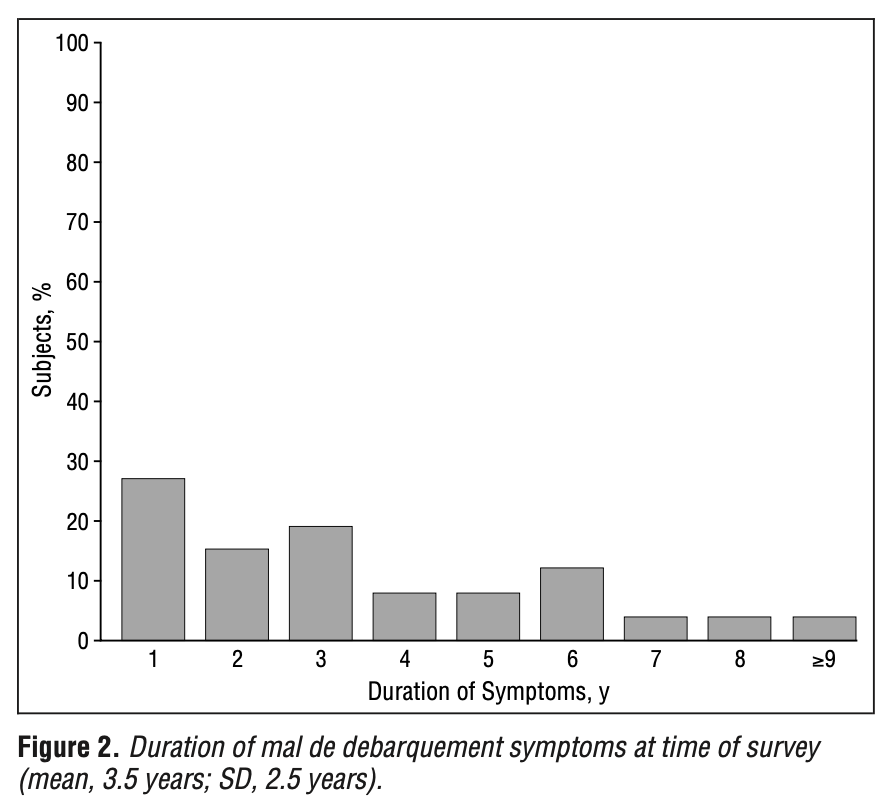
In the series studied by Hain and colleagues (Hain, Hanna et al. 1999) the reported mean duration of symptoms at the time of the survey was 3.5 ± 2.5 years. The Figure below shows a histogram of symptom duration of their patients.
During an “episode” of MdDs the symptoms are usually constant (except when relieved by getting into a vehicle). In the series studied by Hain and colleagues (Hain, Hanna et al. 1999), 85% of patients described symptoms as constant during an episode.
In the series of 64 patients studied by Cha and colleagues (Cha, Brodsky et al. 2008), 41 (64%) had more than one episode.
Characteristic of classical (motion-triggered) MdDs is that symptoms usually improve when the patient gets into a moving vehicle. The study by Cha and colleagues (Cha, Brodsky et al. 2008) reported that 17/22 (77%) of motion-triggered episodes improved with exposure to motion.
A smaller proportion of “non-motion-triggered” episodes of MdDs improve when a patient gets into a moving vehicle. The study by Cha and colleagues (Cha, Brodsky et al. 2008) reported that 4/10 (40%) of “mixed MdDs” episodes (meaning some that were motion-triggered and some that were not) improved with exposure to motion.
Cha and colleagues (Cha, Brodsky et al. 2008) commented that:
“The overall prevalence of migraine headache in our MdD patient group (38%) was higher than the population baseline (17.1% in women, 5.6% in men) but separating this group into the pur MdD group and the mixed MdD group showed that most of this risk came from the mixed MdD group (mixed = 73% vs pure = 22%).”
The sensation of disequilibrium, and other associated symptoms, are variable. The Tables below review symptoms reported by 27 MdDs patient studied by Hain and colleagues (Hain et al. 1999), and symptoms reported by 149 MdDs patient studied by Dai and colleagues (Dai et al. 2017).


In both series (Dai et al. 2017; Hain et al. 1999) “rocking” and “swaying” are the most common vestibular symptoms. The next most common symptoms in the series reported by Hain and colleagues (Hain et al. 1999) were “imbalance” (74%) and “tilting” (52%), while in the series reported by Dai and colleagues (Dai et al. 2017) were “bobbing” (46%) and “gravity pulling” (54%).
Hain and colleagues (Hain, Hanna et al. 1999) further noted that, “Although only 15 (56%) of the [27] subjects checked ear symptoms as part of their present illness, 24 (89%) described having experienced otologic symptoms at some time since the onset of MDD [mal de debarquement syndrome] when asked about specific ear problems. Otologic symptoms reported by 27 survey subjects” are shown in the Table below.

Physical examination
Patients with MdDs who are otherwise healthy will have normal physical examinations.
Ocular motor examination
Patients with MdDs who are otherwise healthy will usually have normal ocular motor examinations.
The study by Cha and colleagues (Cha, Brodsky et al. 2008) reported that 6/64 (9%) of all MdDs patients exhibited “positional torsional/downbeat nystagmus.”
Testing: audiologic and vestibular
The study by Cha and colleagues (Cha, Brodsky et al. 2008) reported that, “Peripheral vestibular function testing is normal” in patients with MdDs.
Lewis (Lewis 2004) studied an MdDs patient in detail with audiometry, vestibular evoked myogenic potentials, subjective visual vertical, videonystagmography (including caloric testing), rotatory chair testing and computerized dynamic posturography, and reported no abnormalities.
Nachum and colleagues (Nachum, Shupak et al. 2004) prospectively studied male sailors aged 18 – 22, including 17 patients who were “susceptible” do MdDs and 17 subjects who were not susceptible, testing them with computerized dynamic posturography before and after sailing. They found, “Abnormal SOT [sensory organization testing] results, defined as a composite and at least one equilibrium score below the normal values achieved by 95% of an age-matched, healthy population, were found in two and nine of the subjects in the SMD [susceptible to MdDs] group for the pre- and postsailig tests respectively,” whereas, “All SOT results for the NSMD [non-susceptible to MdDs] subjects were within normal limits.”
The main value of otovestibular testing in patients with MdDs is to exclude competing diagnoses. Our experience has been that some patients appropriately referred for MdDs sometimes have an additional, more common disorder (such as benign paroxysmal positional vertigo) which, if unrecognized, will interfere with identification and management of MdDs.
Imaging
The study by Cha and colleagues (Cha, Brodsky et al. 2008) reported that “there are no consistent structural brain imaging abnormalities in patients” with MdDs. Lewis (Lewis 2004) also reported no abnormalities on regular brain MRI.
Some studies of MRI voxel-based morphometry report distinct features, “in (a) visual-vestibular processing areas (IPL, SPL, V3, V5/MT), (b) default mode network structures (cerebellar IX, IPL, ACC), (c) salience network structures (ACC and IFG/AI) (d) somatosensory network structures (postcentral gyrus, MCC, anterior cerebellum, cerebellar lobule VIII), and (e) a structure within the central executive network (DLPFC)” (Cha and Chakrapani 2015).
Differential diagnosis
The Barany Society published diagnostic criteria for MdDs (Cha, Baloh et al. 2020).
Gordon and colleagues (Gordon, Spitzer et al. 1995) studied “116 members of seagoing vessels” and reported that 72% of subjects experienced “transient feeling of swinging, swaying, unsteadiness and disequilibrium” following the voyage; this resolved in ≤6 hours in 88% of subjects, and in ≤48 hours in all subjects. When this phenomenon resolves within 2 days following prolonged exposure to motion it is sometimes referred to as “land sickness,” and in such patients, the sensation is usually not relieved by getting into a moving vehicle, so this is probably distinct from MdDs.
Cha and colleagues (Cha, Brodsky et al. 2008) studied a series of patients with “pure” MdDs (whose episodes were triggered by motion) and “mixed” MdDs (in whom some episodes were triggered by motion, and others were not). Among patients with “mixed” episodes they observed that:
- Some episodes of “MdDs” in this group were not triggered by motion.
- The episodes of “MdDs” in this group were less likely to improve when the patient was re-exposed to motion.
- There was a significantly higher prevalence of migraine among this group.
Dai and colleagues (Dai, Cohen et al. 2017) report follow-up of the non-pharmacologic treatment protocol that they developed. The treatment group included patients with “classic” MdDs (episodes triggered by prolonged exposure to motion) and “spontaneous” MdDs (episodes not triggered by motion). They observed that in comparison to patients with “classic” MdDs, those patients with “spontaneous” MdDs overall were less responsive to treatment, treatment effect was less sustained, and fewer achieved remission.
In our view this constellation of characteristics raises the question of whether “non-motion-triggered MdDs” is truly mal de debarquement syndrome. The differential diagnosis should probably include migraine associated vertigo (MAV) (Cha and Cui 2013, Beh, Chiang et al. 2021).
Treatment
Before the diagnosis of MdDs is realized, patients often attempt self-treatment with (or are prescribed) over-the-counter motion sickness medications such as dimenhydrinate or meclizine, and these are always ineffective. Prescription motion-sickness medications, such as transdermal scopolamine, are also ineffective (Hain, Hanna et al. 1999).
Some patients notice short-term pharmacologic improvement with a low-dose benzodiazepine (Cha, Brodsky et al. 2008), but the use of such medications is limited by their significant addictive potential. In the patients studied by Hain and colleagues (Hain, Hanna et al. 1999), 3/6 (50%) reported benefit from clonazepam, 1/4 (25%) from alprazolam, and 2/10 (20%) from diazepam.
Some patients notice longer-term pharmacologic improvement from an SSRI or SNRI. Our experience with venlafaxine 25 – 50 mg once per day has been favorable.
Some practitioners advocate simply treating MdDs as if it were migraine associated vertigo. Ghavami and colleagues (Ghavami, Haidar et al. 2017) treated 15 MdDs patients with a “migraine protocol” that included some combination of nortriptyline, verapamil and topiramate, and reported that, “73% of patients with persisted MdDS had improvement in their QOL [quality of life] and symptom intensity when treated with migraine lifestyle changes and prophylactic therapy.”
Vestibular rehabilitation therapy is also commonly attempted for MdDs. Results are often difficult to interpret due to concomitant pharmacotherapy; for example, Hain and colleagues (Hain, Hanna et al. 1999) reported that, “Fifteen subjects had balance rehabilitation (9 on these took medication during the therapy), and 10 of them reported benefit.” Not all practitioners advocate a trial of vestibular rehabilitation therapy; Ghavami and colleagues (Ghavami, Haidar et al. 2017) report that, “In our experience, MdDS patients who were treated with physical therapy or vestibular rehabilitation did not have a significant improvement in their symptomatology or QOL [quality of life].” Nevertheless, since vestibular rehabilitation therapy incurs no medical risk, we are willing to offer it to patients who express an interest in vestibular therapy, or are averse to a trial of medication.
Some patients experience longer-term non-pharmacologic improvement with the treatment protocol devised by Dr. Mingjia Dai (Dai, Cohen et al. 2017, Cohen 2019) currently available at the Icahn School of Medicine at Mt. Sinai in New York (see https://labs.icahn.mssm.edu/dailab/, accessed 3/20/23). The efficacy of this protocol has been replicated in other laboratories (Mucci, Perkisas et al. 2018). Research is exploring whether a virtual reality version of this approach is effective (Yakushin, Zink et al. 2020).
Ongoing research is exploring the possible role of transcranial magnetic stimulation in the treatment of MdDs (Cha, Cui et al. 2013, Guofa, Han et al. 2014, Cha, Deblieck et al. 2016, Yuan, Shou et al. 2017). Transcranial alternating current stimulation has also been explored (Cha, Gleghorn et al. 2022).
Some research suggests that different facets (symptoms) of MdDs may respond differently to different interventions. For instance, Yakushin and colleagues (Yakushin et al. 2022) studied whether a treatment protocol involving optokinetic stimulation with vertically-moving horizontal stripes would be more helpful specifically for the “gravitational pulling sensation” (as opposed to optokinetic stimulation with horizontally-moving vertical stripes for the more common “rocking” sensation of MdDs).
As with other “invisible” diseases (illnesses for which patients “look normal” to an untrained outside observer), MdDs patients often appear stigmatized, leading to additional psychosocial problems beyond the disease’s original symptoms, including anxiety and depression (Mucci, Canceri et al. 2018). Seeking psychiatric help for this aspect of the disease is sensible (Arroll, Attree et al. 2016).
Prognosis
Prognosis is guarded because the overall duration of episodes is variable, and the response to treatments is variable.
In the majority of cases, an episode of MdDs will eventually end. The clinical question at that point then becomes: What is the risk of re-triggering an episode with another prolonged exposure to motion? There is not yet any proven algorithm to predict this, so some patients will simply practice avoidance of such exposure. Some patients will elect to travel again, and anecdotally report feeling “protected” by taking a benzodiazepine prophylactically, but this has never been studied systematically.
References
Arroll MA, Attree EA, Cha YH, Dancey CP (2016) The relationship between symptom severity, stigma, illness intrusiveness and depression in Mal de Debarquement Syndrome. J Health Psychol 21: 1339-50. doi: 10.1177/1359105314553046
Beh SC, Chiang HS, Sanderson C (2021) The Interconnections of Mal de Debarquement Syndrome and Vestibular Migraine. Laryngoscope 131: E1653-E1661. doi: 10.1002/lary.29214
Bronstein AM, Golding JF, Gresty MA (2013) Vertigo and dizziness from environmental motion: visual vertigo, motion sickness, and drivers’ disorientation. Semin Neurol 33: 219-30. doi: 10.1055/s-0033-1354602
Cha YH (2015) Mal de debarquement syndrome: new insights. Ann N Y Acad Sci 1343: 63-8. doi: 10.1111/nyas.12701
Cha YH, Baloh RW, Cho C, Magnusson M, Song JJ, Strupp M, Wuyts F, Staab JP (2020) Mal de debarquement syndrome: Diagnostic criteria consensus document of the classification committee of the Barany society. J Vestib Res. doi: 10.3233/VES-200714
Cha YH, Brodsky J, Ishiyama G, Sabatti C, Baloh RW (2008) Clinical features and associated syndromes of mal de debarquement. J Neurol 255: 1038-44. doi: 10.1007/s00415-008-0837-3
Cha YH, Chakrapani S (2015) Voxel Based Morphometry Alterations in Mal de Debarquement Syndrome. PLoS One 10: e0135021. doi: 10.1371/journal.pone.0135021
Cha YH, Cui Y (2013) Rocking dizziness and headache: a two-way street. Cephalalgia 33: 1160-9. doi: 10.1177/0333102413487999
Cha YH, Cui Y, Baloh RW (2013) Repetitive transcranial magnetic stimulation for mal de debarquement syndrome. Otol Neurotol 34: 175-9. doi: 10.1097/MAO.0b013e318278bf7c
Cha YH, Deblieck C, Wu AD (2016) Double-Blind Sham-Controlled Crossover Trial of Repetitive Transcranial Magnetic Stimulation for Mal de Debarquement Syndrome. Otol Neurotol 37: 805-12. doi: 10.1097/MAO.0000000000001045
Cha YH, Gleghorn D, Doudican BC (2022) Double-blind randomized N-of-1 trial of transcranial alternating current stimulation for mal de debarquement syndrome. PLoS One 17: e0263558. doi: 10.1371/journal.pone.0263558
Cohen B (2019) Dedication to Mingjia Dai, Ph.D. for Discovery of the First Successful Treatment of the Mal de Debarquement Syndrome. Front Neurol 10: 1196. doi: 10.3389/fneur.2019.01196
Dai M, Cohen B, Cho C, Shin S, Yakushin SB (2017) Treatment of the Mal de Debarquement Syndrome: A 1-Year Follow-up. Front Neurol 8: 175. doi: 10.3389/fneur.2017.00175
Darwin E, Mitchill SL, Joseph Meredith Toner Collection (Library of Congress) (1796) Zoonomia, or, The laws of organic life. Vol. I. Printed by T. & J. Swords, printers to the Faculty of Physic of Columbia College, No. 99 Pearl-Street, New-York
Ghavami Y, Haidar YM, Ziai KN, Moshtaghi O, Bhatt J, Lin HW, Djalilian HR (2017) Management of mal de debarquement syndrome as vestibular migraines. Laryngoscope 127: 1670-1675. doi: 10.1002/lary.26299
Gordon CR, Spitzer O, Doweck I, Melamed Y, Shupak A (1995) Clinical features of mal de debarquement: adaptation and habituation to sea conditions. J Vestib Res 5: 363-9.
Guofa S, Han Y, Urbano D, Yoon-Hee C, Lei D (2014) Changes of symptom and EEG in mal de debarquement syndrome patients after repetitive transcranial magnetic stimulation over bilateral prefrontal cortex: a pilot study. Conf Proc IEEE Eng Med Biol Soc 2014: 4294-7. doi: 10.1109/EMBC.2014.6944574
Hain TC, Hanna PA, Rheinberger MA (1999) Mal de debarquement. Arch Otolaryngol Head Neck Surg 125: 615-20.
Hain TC, Helminski JO (2007) Therapy for mal de debarquement syndrome. In: Herdman SJ (ed) Vestibular Rehabilitation, 3 edn. E.A. Davis Company, Philadelphia, pp 202-204
Irwin JA, Cantab MA (1881) The pathology of sea-sickness. The Lancet 118: 907-909. doi: https://doi.org/10.1016/S0140-6736(02)38129-7
Lewis RF (2004) Frequency-specific mal de debarquement. Neurology 63: 1983-4.
London J (1904) The Sea-wolf, New York and London
London J (1911) The cruise of the Snark. The Macmillan company, New York
Moeller L, Lempert T (2007) Mal de debarquement: pseudo-hallucinations from vestibular memory? J Neurol 254: 813-5. doi: 10.1007/s00415-006-0440-4
Mucci V, Canceri JM, Brown R, Dai M, Yakushin S, Watson S, Van Ombergen A, Topsakal V, Van de Heyning PH, Wuyts FL, Browne CJ (2018a) Mal de Debarquement Syndrome: a survey on subtypes, misdiagnoses, onset and associated psychological features. J Neurol 265: 486-499. doi: 10.1007/s00415-017-8725-3
Mucci V, Perkisas T, Jillings SD, Van Rompaey V, Van Ombergen A, Fransen E, Vereeck L, Wuyts FL, Van de Heyning PH, Browne CJ (2018b) Sham-Controlled Study of Optokinetic Stimuli as Treatment for Mal de Debarquement Syndrome. Front Neurol 9: 887. doi: 10.3389/fneur.2018.00887
Murphy TP (1993) Mal de debarquement syndrome: a forgotten entity? Otolaryngol Head Neck Surg 109: 10-3. doi: 10.1177/019459989310900103
Nachum Z, Shupak A, Letichevsky V, Ben-David J, Tal D, Tamir A, Talmon Y, Gordon CR, Luntz M (2004) Mal de debarquement and posture: reduced reliance on vestibular and visual cues. Laryngoscope 114: 581-6. doi: 10.1097/00005537-200403000-00036
Pike JR (1986) Tall ships in Torbay : a brief maritime history. Ex Libris, Bradford on Avon
Whittel J (1689) An exact diary of the late expedition of His Illustrious Highness, the Prince of Orange, (now King of Great Britain) from his palace at the Hague, to his landing at Torbay; and from thence to his arrival at White-Hall. Giving a particular account of all that happened, and every day’s march. R. Baldwin, London
Yakushin SB, Raphan T, Cho C (2022) Treatment of Gravitational Pulling Sensation in Patients With Mal de Debarquement Syndrome (MdDS): A Model-Based Approach. Front Integr Neurosci 16: 801817. doi: 10.3389/fnint.2022.801817
Yakushin SB, Zink R, Clark BC, Liu C (2020) Readaptation Treatment of Mal de Debarquement Syndrome With a Virtual Reality App: A Pilot Study. Front Neurol 11: 814. doi: 10.3389/fneur.2020.00814
Yuan H, Shou G, Gleghorn D, Ding L, Cha YH (2017) Resting State Functional Connectivity Signature of Treatment Effects of Repetitive Transcranial Magnetic Stimulation in Mal de Debarquement Syndrome. Brain Connect 7: 617-626. doi: 10.1089/brain.2017.0514
![]()