By Marcello Cherchi, MD PhD
For patients
Susac syndrome is a disease that affects blood vessels of the ear, eye and brain, so it causes problems with vision, hearing and thinking. It usually starts around age 30. If your doctor suspects Susac syndrome, then they may check tests of vision, hearing and balance, and may check a brain MRI. Treatment is sometimes tried with drugs that affect the immune system.
For clinicians
Overview
Susac syndrome (retinocochleocerebral vasculopathy) is believed to be an autoimmune disorder (perhaps mediated by CD8+ T-cells) preferentially affecting the precapillary arterioles of the brain, retina and labyrinth, manifesting with some combination of visual deficits, auditory symptoms (hearing loss, tinnitus), encephalopathy and headache. The disease is rare, affects women more than men, with a peak age of onset of around 30 years (range 14 – 63 years). The hearing loss often affects low and middle more than high frequencies, and can be unilateral or bilateral, and fluctuating. Vestibular workup may reveal unilateral weakness on vestibular evoked myogenic potentials (which may recover), caloric testing, or video head impulse testing. Fluorescein angiography shows branch retinal artery occlusions. Brain MRI shows T2/FLAIR lesions in the corpus callosum, periventricular, thalamic and internal capsular regions. There are no randomized treatment trials, but treatment is sometimes attempted with corticosteroids and other immunosuppressants or immunomodulatory agents.
Introduction
In 1979 neuro-ophthalmologist John O. Susac (1940 – 2012), pathologist John O. Hardman (1933 – 2006), and neurologist John B. Selhorst reported two cases of young women with neurological symptoms (headache, behavioral disturbances) and visual deficits who were found to have microangiopathy of the brain and retina (Susac et al. 1979). Subsequent reports also noted hearing loss.
This clinical triad (visual abnormalities, sensorineural hearing loss and cognitive dysfunction) resulting from retinocochleocerebral vasculopathy became known as Susac syndrome. This is currently believed to be an immune-mediated vascular disorder preferentially affecting blood vessels of the eye, ear and brain.

Epidemiology
Susac syndrome is rare. Bose and colleagues state that from the original description in 1979 until 2021 there were only 450 cases reported in the medical literature (Bose et al. 2023).
Bose and colleagues (Bose et al. 2023) studied a series of 16 patients and reported a female-to-male ratio of 2.2 to 1, with a mean onset of 30.5 years (±9.6 years).
Hardy and colleagues (Hardy et al. 2020) studied a series of 30 patients and reported a female-to-male ratio of 17 to 13 (1.3 to 1) with a mean age of onset of 36.8 (range 14 – 63).
Genetics
There has been little study of the genetics of Susac syndrome. David and colleagues (David et al. 2021) performed whole exome sequencing on 40 patients with Susac syndrome and identified a de novo missense variant in the ZNF76 (zinc finger protein 76) at chromosome 6p21.31 (OMIM 194549).
Pathophysiological mechanism of disease
Susac syndrome is a “microangiopathy affecting the precapillary arterioles of the brain, retina and inner ear (cochlea and semicircular canals)” manifesting with the “triad of encephalopathy, branch retinal artery occlusions… and sensorineural hearing loss” (Bose et al. 2023). Immunological studies suggest that the vasculopathy results from malfunctioning CD8+ T-cells (Hardy et al. 2020).
Clinical presentation
Of the three cardinal clinical features of Susac syndrome (visual deficits, hearing loss, encephalopathy), usually only a subset is present initially. It has been reported that of patients eventually diagnosed with Susac syndrome, only 13% present with all three symptoms at onset (Bose et al. 2023).
Hardy and colleagues (Hardy et al. 2020) described a series of 30 Susac patients, and reported:
“Neuro-otological involvement is relatively uncommon at the onset of SuS [Susac syndrome] but 93% of patients reported experiencing hearing loss, 54% tinnitus, and 67% vertigo or disequilibrium at some point during their disease course over an average of 4.5 years of follow-up. Moreover, 97% of patients experienced at least one of these symptoms during their disease course and 87% were left with residual neuro-otological symptoms” (Hardy et al. 2020).
Mean time to diagnosis has been reported to range from 3 weeks to 3 months (Bose et al. 2023).
Bose and colleagues reported a series of 16 Susac patients (Bose et al. 2023). The Table below shows presenting complaints and results of preliminary workup.
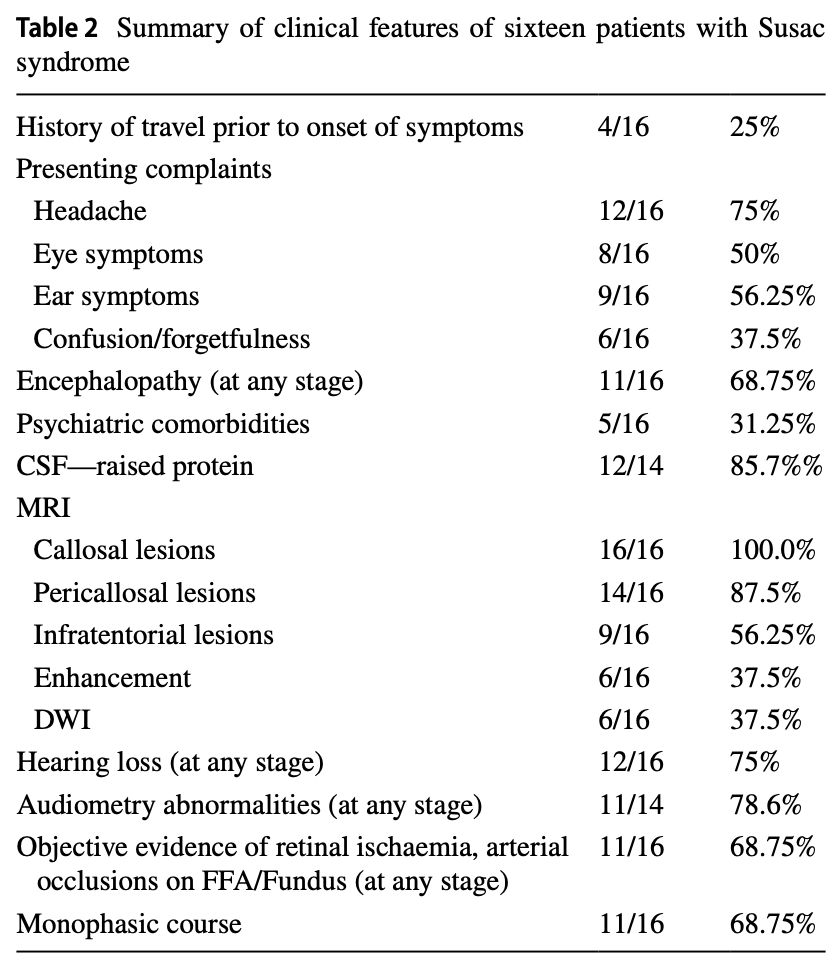
Notably, headache (75%) is the most common presenting feature. Encephalopathy occurs at some stage of the disease in 69% of cases.
Hardy and colleagues reported a series of 30 Susac patients (Hardy et al. 2020). The Table below shows the distribution of symptoms.
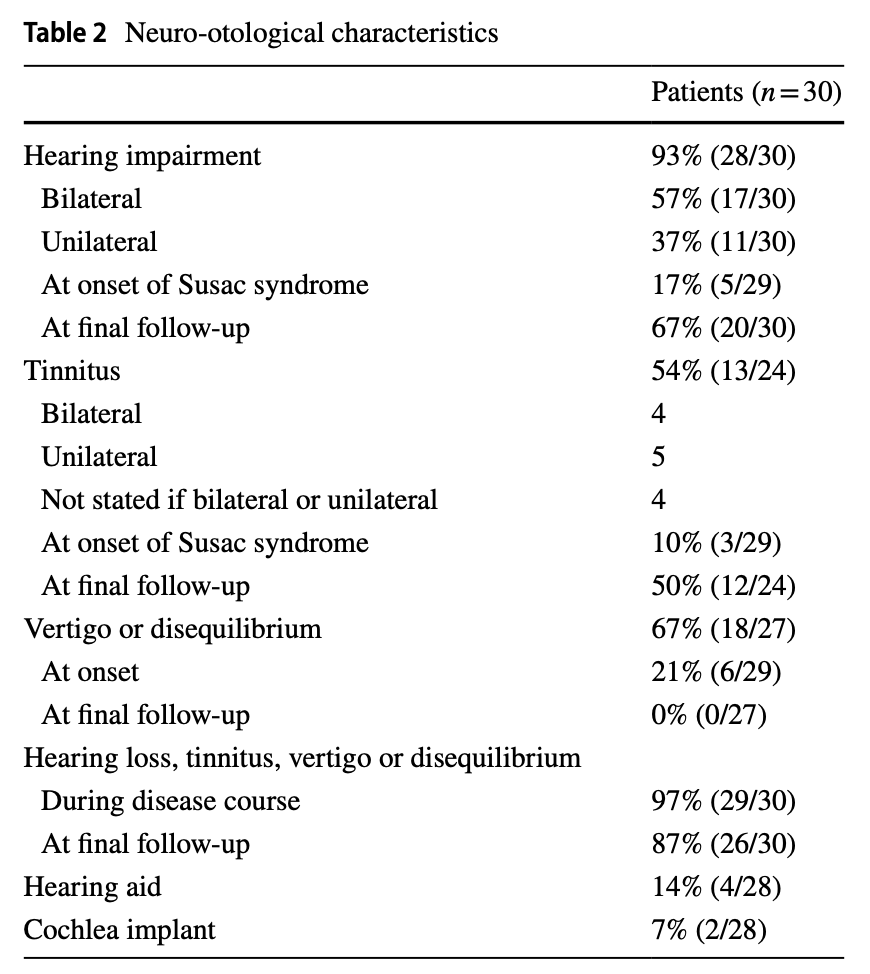
The encephalopathic features can range “from minor cognitive symptoms to frank confusion” (Hardy et al. 2020).
The visual symptoms may manifest abruptly (with visual loss, visual field changes, scintillating scotoma and other features), but visual loss may also develop insidiously and thus not be volunteered by the patient (Bose et al. 2023).
The hearing loss may be abrupt or insidious in onset (Bose et al. 2023), can progress in a step-wise fashion, and can be unilateral or bilateral (Hardy et al. 2020).
In the series reported by Hardy and colleagues (Hardy et al. 2020), disequilibrium was reported by 18 (67%) of 27 patients, was the initial symptom (or one of several presenting symptoms) in 6 (21%) of 29 patients, and was the only otoneurological symptom in 2 (7%) of patients. It is unusual for vestibular symptoms to be the exclusive initial presentation of Susac syndrome (Magliulo et al. 2008).
Testing (auditory): audiometry and otoacoustic emissions
When Susac syndrome involves hearing loss, pure tone audiometry “often shows low to mid-frequency sensorineural hearing loss in the early stages, but the pantonal loss can also occur in severe cases” (Hardy et al. 2020). The initial involvement of low and middle frequencies, particularly if unilateral, may resemble Ménière’s disease.
The Figure below, from Hardy and colleagues (Hardy et al. 2020), shows serial audiometry in a patient, demonstrating fluctuating sensorineural hearing loss.
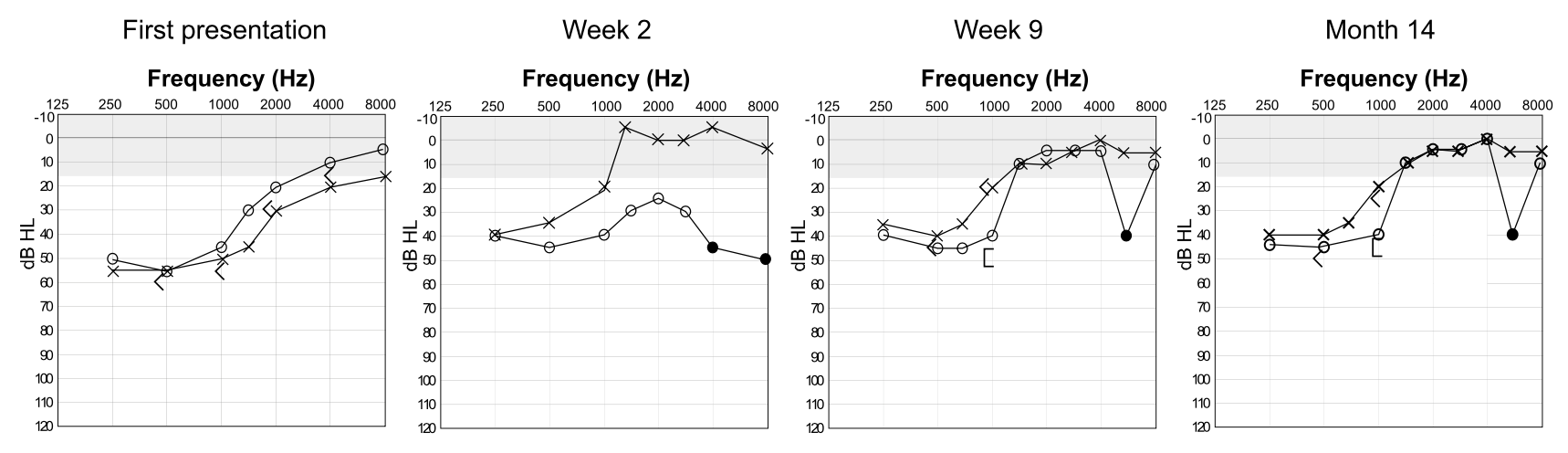
In the series of Susac patients studied by Hardy and colleagues (Hardy et al. 2020), some patients had the paradoxical finding of hearing loss on pure tone audiometry yet preserved otoacoustic emissions, suggesting dysfunction of only the type 1 (inner) cochlear hair cells, or retrocochlear pathways (such as an auditory neuropathy). The remaining patients with hearing loss on pure tone audiometry had commensurate loss on otoacoustic emissions.
Testing (vestibular): caloric testing
In the series of Susac patients studied by Hardy and colleagues (Hardy et al. 2020), 10 had caloric testing, of which one showed bilateral weakness, and two showed asymmetrical weakness. The case studied by River and colleagues (River et al. 2015) reported a 56% right unilateral caloric weakness.
Testing (vestibular): vestibular evoked myogenic potentials
Hardy and colleagues (Hardy et al. 2020) reported that patients initially found to have unilateral weakness on ocular vestibular evoked myogenic potentials or cervical vestibular evoked myogenic potentials subsequently appeared to recover and exhibit normal responses on these tests.
Oron and colleagues (Oron et al. 2020) studied several patients with Susac syndrome and reported one to have a unilaterally (left) absent cervical vestibular evoked myogenic potentials response, as shown in the Figure below.
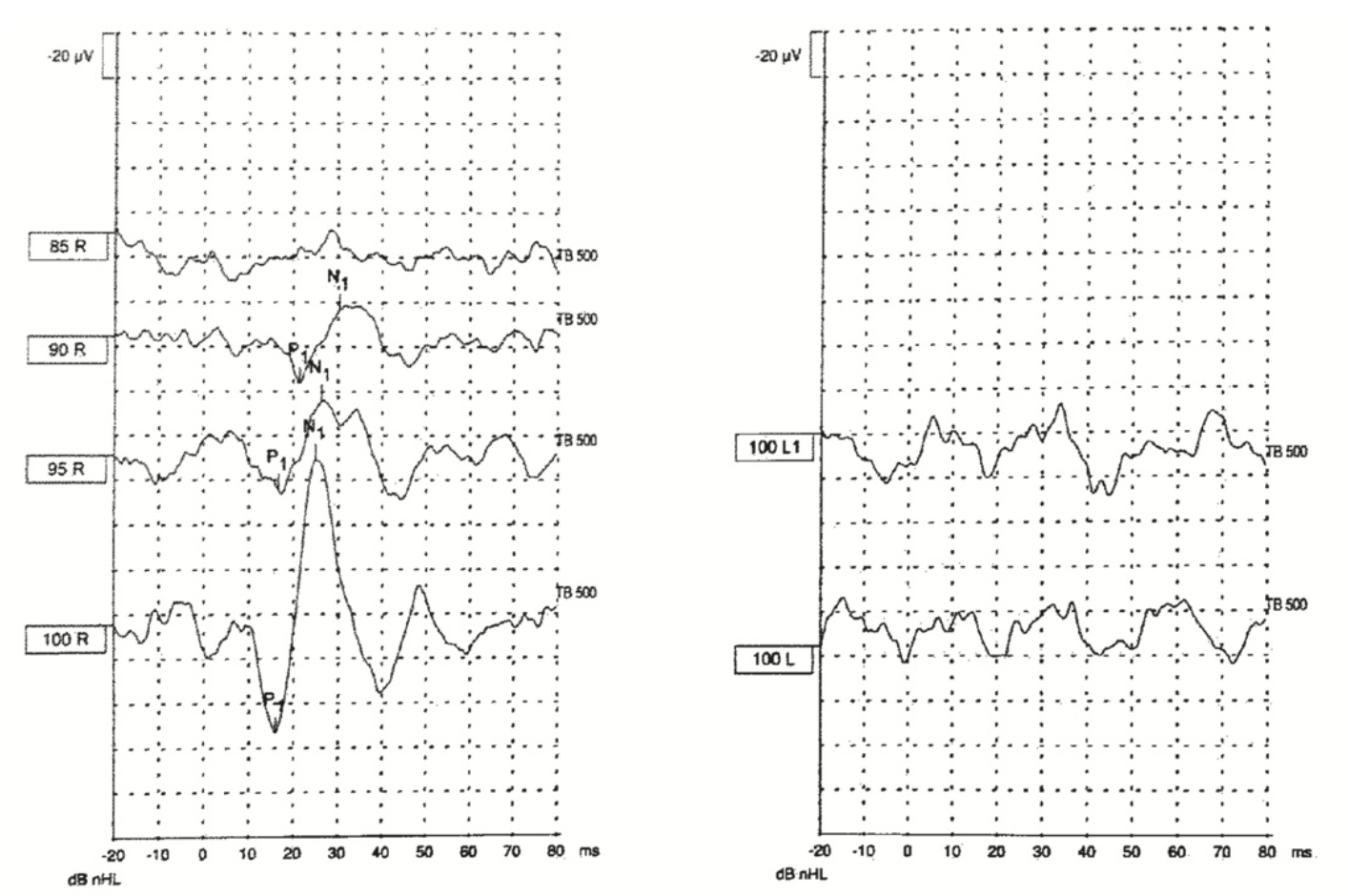
Suman and colleagues (Suman et al. 2022) reported a case of Susac syndrome in which serial cervical and ocular vestibular evoked myogenic potentials were performed. Cervical vestibular evoked myogenic potentials were unilaterally absent (left side) on the initial study but present on the second and third studies. Ocular vestibular evoked myogenic potentials were normal in all 3 studies.
Testing (vestibular): video head impulse testing
Marrodan and colleagues (Marrodan et al. 2019) reported the case of a 19-year-old lady diagnosed with Susac syndrome who underwent video head impulse testing showing that the right posterior semicircular canal exhibited low gain and over compensatory saccades. Of note, the patient had no vestibular complaints.
The Figure below, from Marrodan and colleagues (Marrodan et al. 2019), shows the focal deficit involving the right posterior canal.
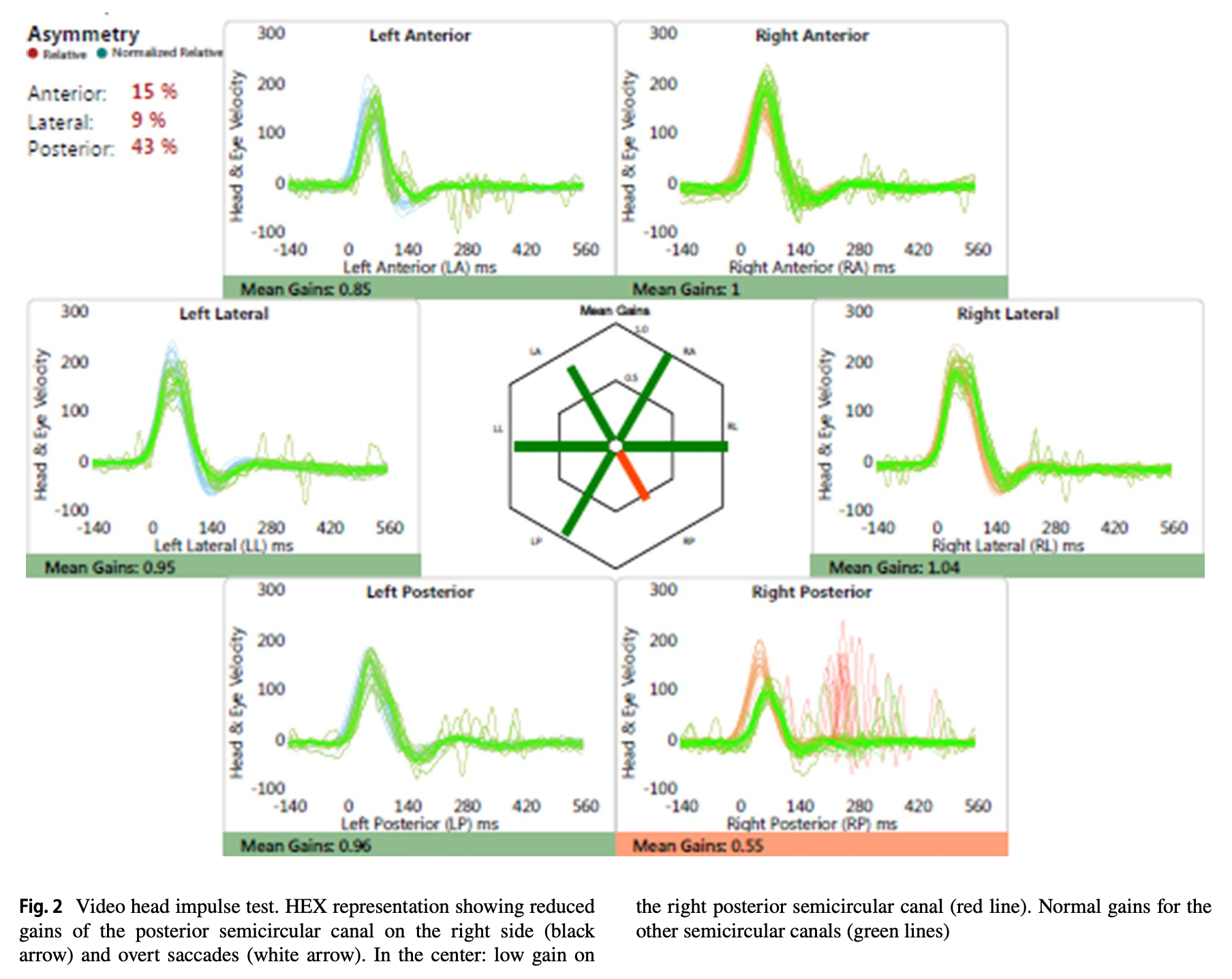
Testing: other
Fluorescein angiography shows branch retinal artery occlusions.
Imaging
Brain MRI usually shows lesions on FLAIR sequences involving the corpus callosum, periventricular and thalamic regions. The MRI lesions have been described with colorful terms such as “snowballs” (referring to the round lesions in the corpus callosum), “spokes and icicles” (for linear lesions in the corpus callosum) and “string of beads” pattern in the internal capsule (Marrodan et al. 2019), and may also involve the periventricular and thalamic regions (Bose et al. 2023).
The Figure below, from Bose and colleagues (Bose et al. 2023), shows typical imaging findings of Susac syndrome.
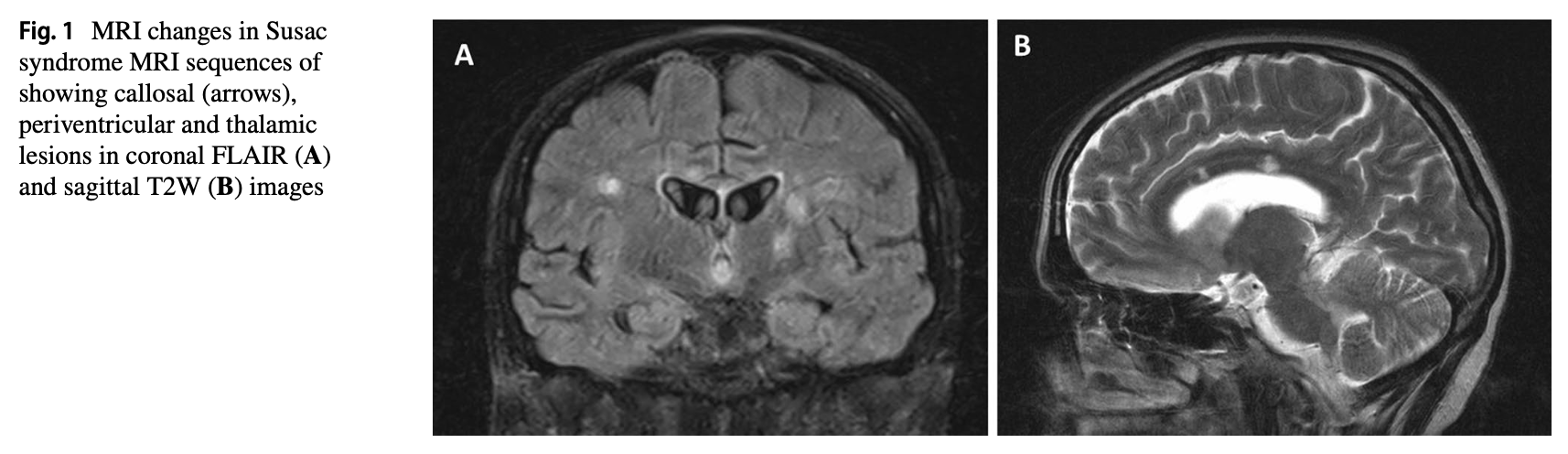
Histopathology
Bose and colleagues (Bose et al. 2023) reported on the brain biopsy of one patient, and the autopsy of another patient, which together showed “multiple microinfarcts involving grey/white matter, deep grey nuclei, brain stem and corpus callosum with endothelial cell necrosis and perivascular lymphocytic infiltration.”
Francis and colleagues studied the temporal bone of a 51-year-old woman with Susac syndrome and reported, “atrophy and degeneration involving the apical halves of the cochlear duct without inflammation or infection. The findings were consistent with capillary occlusion as being responsible for the atrophy” (Francis et al. 2011).
Differential diagnosis
The cognitive and visual symptoms of Susac syndrome may resemble demyelinating disease (such as multiple sclerosis, neuromyelitis optica, acute demyelinating encephalomyelitis), but imaging usually helps differentiate these.
The headache and encephalopathy of Susac syndrome may resemble CADASIL, but the visual and auditory symptoms may differentiate these.
Cases of Susac syndrome manifesting with abrupt onset cochlear symptoms and found to have ipsilateral vestibular deficits (River et al. 2015) may resemble labyrinthitis, but the cognitive symptoms will usually differentiate these.
Susac syndrome can also be mistaken for a CNS vasculitis.
Treatment and prognosis
There are no prospective, randomized, double-blinded, placebo-controlled treatment trials for Susac syndrome, so treatment decisions usually draw from case reports, case series and expert opinion. Attempted treatments have included corticosteroids, immunosuppressants (rituximab, cyclophosphamide, mycophenolate mofetil, azathioprine), immunomodulatory therapies (intravenous immunoglobulin) (Moheb et al. 2025).
Hearing loss and tinnitus, once established, are usually permanent (Hardy et al. 2020).
References
Bose S, Papathanasiou A, Karkhanis S, Appleton JP, King D, Batra R, Mollan SP, Jacob S (2023) Susac syndrome: neurological update (clinical features, long-term observational follow-up and management of sixteen patients). J Neurol 270: 6193-6206. doi: 10.1007/s00415-023-11891-z
David C, Papo T, Ba I, Ollivier E, Boileau C, Dieude P, Keren B, Kannengiesser C, Sacre K (2021) Hunting for the genetic basis of Susac syndrome. Eur J Neurol 28: e57-e59. doi: 10.1111/ene.14836
Egan R (2012) John O. Susac, MD (1940–2012). Journal of Neuro-Ophthalmology 32.
Francis HW, Makary C, Halpin C, Crane BT, Merchant SN (2011) Temporal bone findings in a case of Susac’s syndrome. Otol Neurotol 32: 1198-204. doi: 10.1097/MAO.0b013e31822e9665
Hardy TA, Taylor RL, Qiu J, O’Brien B, Gopinath S, Trewin B, Spring PJ, Shaffi M, Bolitho SJ, Garsia RJ, Roxburgh R, Mason DF, Ip J, Chan F, Chen L, Wilson I, Beadnall HN, Barnett MH, Parratt JDE, Watson JDG, Welgampola MS, Reddel SW, Tsang B, Halmagyi GM (2020) The neuro-otology of Susac syndrome. J Neurol 267: 3711-3722. doi: 10.1007/s00415-020-10086-0
Magliulo G, Al-Ansi W, Parrotto D, Gagliardi S, Stasolla A (2008) Susac syndrome and vestibular-evoked myogenic potentials. Otolaryngol Head Neck Surg 138: 542-3. doi: 10.1016/j.otohns.2007.10.033
Marrodan M, Laffue A, Fiol MP, Correale J, Gualtieri F (2019) Video head impulse test contributes to Susac syndrome diagnosis. J Neurol. doi: 10.1007/s00415-019-09357-2
Moheb N, Tajfirouz DA, Chodnicki KD, McKeon A, Rattanathamsakul N, Salman AR, Rettenmaier LA, Varma-Doyle A, Chwalisz BK, Chen JJ (2025) Outcomes of Maintenance Immunotherapy in a Cohort of Patients With Susac Syndrome: A 2-Center Large Case Series. Neurol Clin Pract 15: e200544. doi: 10.1212/CPJ.0000000000200544
Oron Y, Handzel O, Habot-Wilner Z, Regev K, Karni A, Zur D, Baraquet D, Goldstein M, Elkayam O, Ungar OJ (2020) Vestibular function assessment of Susac syndrome patients by the video head impulse test and cervical vestibular-evoked myogenic potentials. J Vestib Res 30: 393-399. doi: 10.3233/ves-200007
River Y, Shupak A, Tiosano B, Danilov V, Braverman I (2015) Susac’s Syndrome: A Case with Unusual Cardiac Vestibular and Imaging Manifestations. Case Rep Neurol Med 2015: 419408. doi: 10.1155/2015/419408
Suman NS, Poovaiah PP, Rangarajan A, Tiwari R, Nashi S, Yuvraj P, Rajasekaran AK (2022) Cervical and Ocular Vestibular Evoked Myogenic Potential Recovery in Susac Syndrome: A Case Report. Am J Audiol 31: 1059-1066. doi: 10.1044/2022_aja-22-00012
Susac JO, Hardman JM, Selhorst JB (1979) Microangiopathy of the brain and retina. Neurology 29: 313-6. doi: 10.1212/wnl.29.3.313
![]()