By Marcello Cherchi, MD PhD
For patients
A vestibular schwannoma, sometimes called an acoustic neuroma, is an uncommon, non-malignant tumor involving the vestibulocochlear nerve (the nerve between the ear and the brain that transmits hearing and balance signals), or less commonly the inner ear itself. It usually presents with gradually progressive hearing loss on one side, and sometimes with imbalance. The diagnosis may be suspected based on results of hearing and balance testing, and confirmed with an MRI of the inner ear without and with contrast. Vestibular schwannomas sometimes do not grow at all, or only grow very slowly, so monitoring symptoms (and sometimes repeating imaging) is appropriate in some cases. In other cases intervention may be required, and this can be in the form of radiation therapy (to shrink the tumor) or open surgery (to remove or de-bulk the tumor). Symptoms of imbalance may transiently worsen after surgery, and some patients benefit from vestibular rehabilitation therapy.
For practitioners
Overview
A vestibular schwannoma is an uncommon and (usually) non-malignant tumor involving the 8th cranial nerve that, depending on its size and location can cause auditory, vestibular symptoms, or symptoms secondary to brainstem compression. The symptoms may be due to a combination of (1) abnormal Schwann cells; (2) mechanical distortion of neural structures; (3) interference with signal propagation; (4) vascular compromise secondary to mass effect; and (5) change in composition of endolymph. Audiometry usually shows gradually progressive, predominantly high frequency sensorineural hearing loss, and otovestibular testing may show ipsilateral vestibular weakness; but the definitive diagnostic tool is MRI of the internal auditory canals without and with contrast. Depending on the cadence of symptom evolution, management strategies include (1) monitoring; (2) radiosurgery; and (3) open surgery. The radiosurgical and open surgical interventions often result in vestibular weakness (new, or greater than pre-surgical), so vestibular rehabilitation therapy is often needed post-surgically.
Terminology
A vestibular schwannoma is usually a non-malignant tumor involving the 8th cranial nerve (vestibulocochlear nerve), which contains both acoustic and vestibular afferent fibers.
Older literature referred to this tumor as an acoustic neuroma in the mistaken belief that it arose in the auditory (“acoustic”) portion of the nerve, and from actual nerve cells (“neuroma”).
Subsequent studies revealed that the tumor more commonly arises in the “vestibular” portion of the nerve (Komatsuzaki & Tsunoda, 2001), and from Schwann cells (“Schwannoma”) that ensheathe the nerve (Murray, Stout, & Bradley, 1940).
Thus, from the anatomical and histological perspective the term “vestibular schwannoma” is more correct. Nevertheless, many practitioners and some literature continue to use the term “acoustic neuroma.”
Epidemiology
Vestibular schwannomas are relatively uncommon, though there is controversy about the exact numbers. Longitudinal studies typically cite an annual incidence from as low as 5.4 to as high as 10 per million persons, and a lifetime incidence of 1 per thousand persons (Stangerup & Caye-Thomasen, 2012). Some studies describe a rising incidence of this disease (Evans et al., 2005), while others claim that this apparent increase is more likely due to increased recognition, as well as the incidental discovery of asymptomatic vestibular schwannomas on imaging (for example, in persons who underwent a brain MRI for some unrelated problem) (Hoa, Drazin, Hanna, Schwartz, & Lekovic, 2012; Jeyakumar, Seth, Brickman, & Dutcher, 2007; Lin, Hegarty, Fischbein, & Jackler, 2005; Puls, 2015; Schmidt et al., 2012; Selesnick, Deora, Drotman, & Heier, 1999).
Vestibular schwannoma is largely a disease of adulthood, with studies describing a mean age at diagnosis of 49 to 58 years (Stangerup & Caye-Thomasen, 2012). In children one study found only 39 cases of non-syndromic vestibular schwannomas (i.e., those not associated with tumor-predisposing genetic abnormalities such as neurofibromatosis type 2) published in world medical literature (Pothula, Lesser, Mallucci, May, & Foy, 2001).
Presentation
Vestibular schwannomas are generally very slowly growing tumors, so symptoms correspondingly present subtly and are very slowly progressive. Because of this insidious presentation, the median time between symptom onset and diagnosis is 5 years, though the range is very broad (from a few weeks to 40 years) (Van Abel et al., 2013).
Given the anatomical structures involved (vestibulocochlear nerve), the disease correspondingly presents with auditory symptoms (hearing loss, sound distortion, tinnitus) and vestibular symptoms.
Other symptoms are also possible, depending largely on whether the tumor is impinging on other structures. For example, facial weakness can occur because the facial nerve runs through part of the internal auditory canal and adjacent to the vestibulocochlear nerve for some of its course, so a vestibular schwannoma can compress the facial nerve as well; clumsiness or incoordination can result if the vestibular schwannoma is extracanalicular, in the cerebellopontine angle, impinging on the cerebellum; facial pain can occur if a vestibular schwannoma in the cerebellopontine angle impinges on the trigeminal nerve. A typical single-institution retrospective study (Tos, Charabi, & Thomsen, 1998) of this patient population (703 individuals) reported the following percentages of symptom presentation:
- 57%: Gradually progressive sensorineural hearing loss
- 14%: Vertigo
- 12%: Tinnitus
- 7%: Sudden deafness
- 4.1%: Trigeminal symptoms, such as facial pain
- 2.8%: Cerebellar symptoms
- 1.3%: Headache
- 1.1%: Facial nerve paresis
- 0.1%: Epilepsy
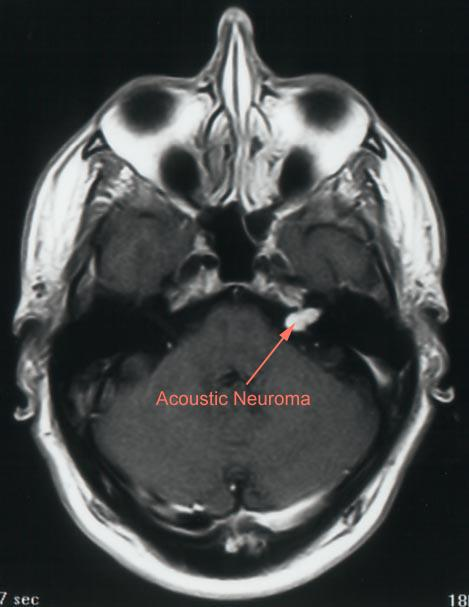
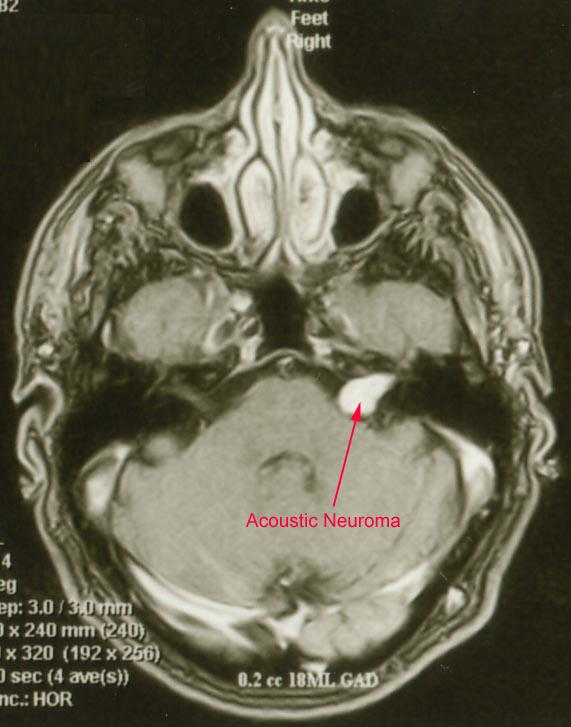
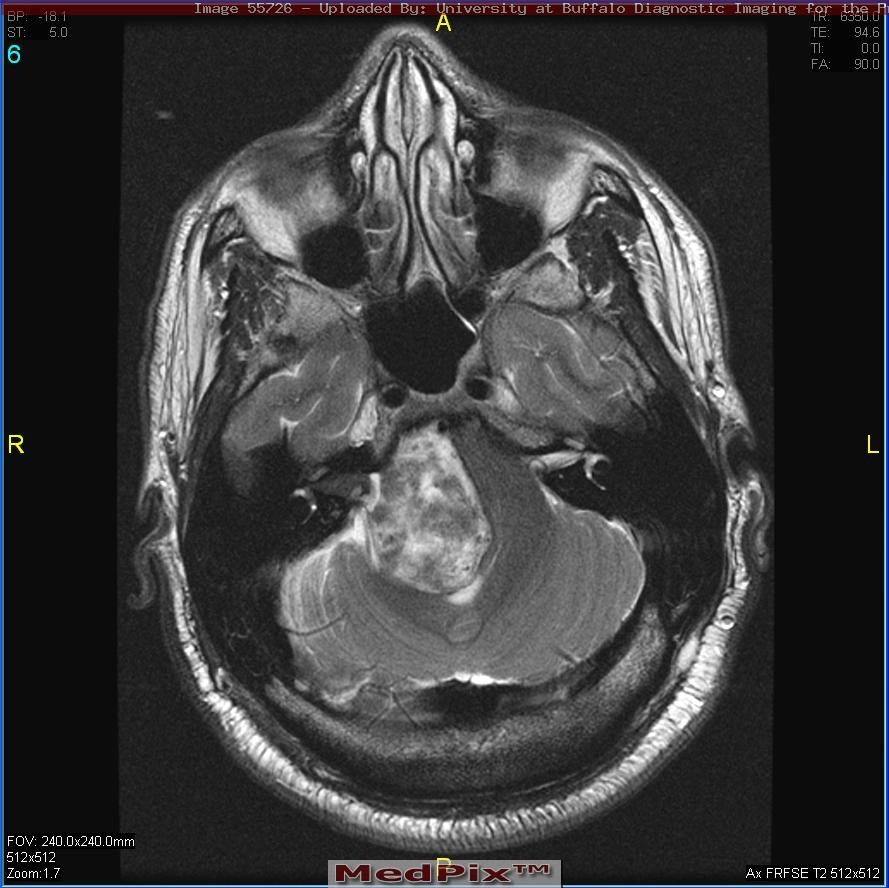
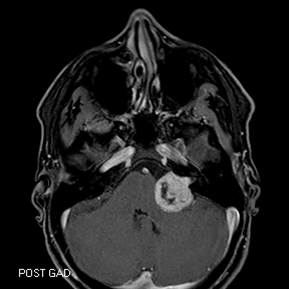
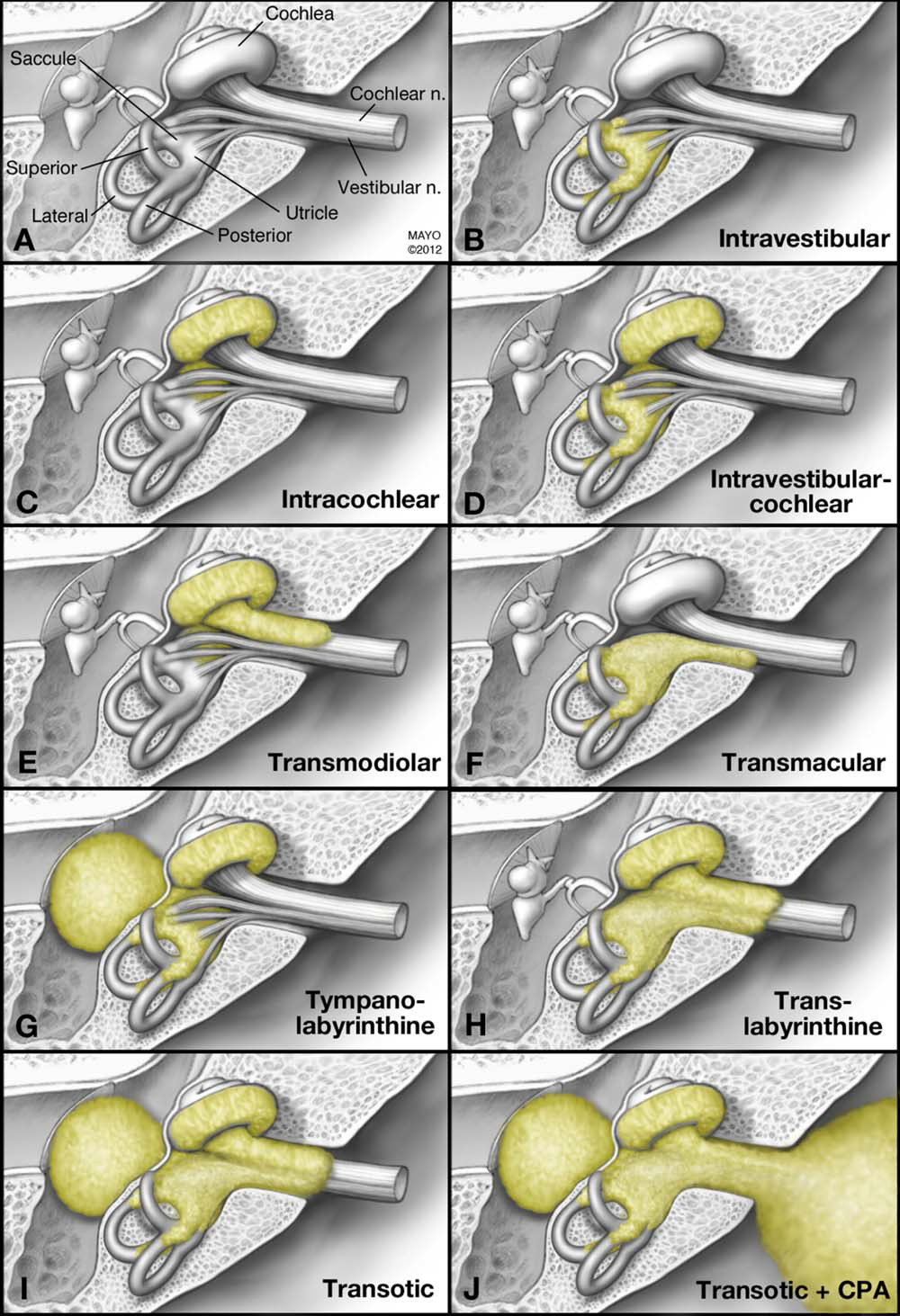
Anatomical location of vestibular schwannomas
The vestibulocochlear nerve is a relatively long cranial nerve. Vestibular schwannomas can arise anywhere along this structure, including:
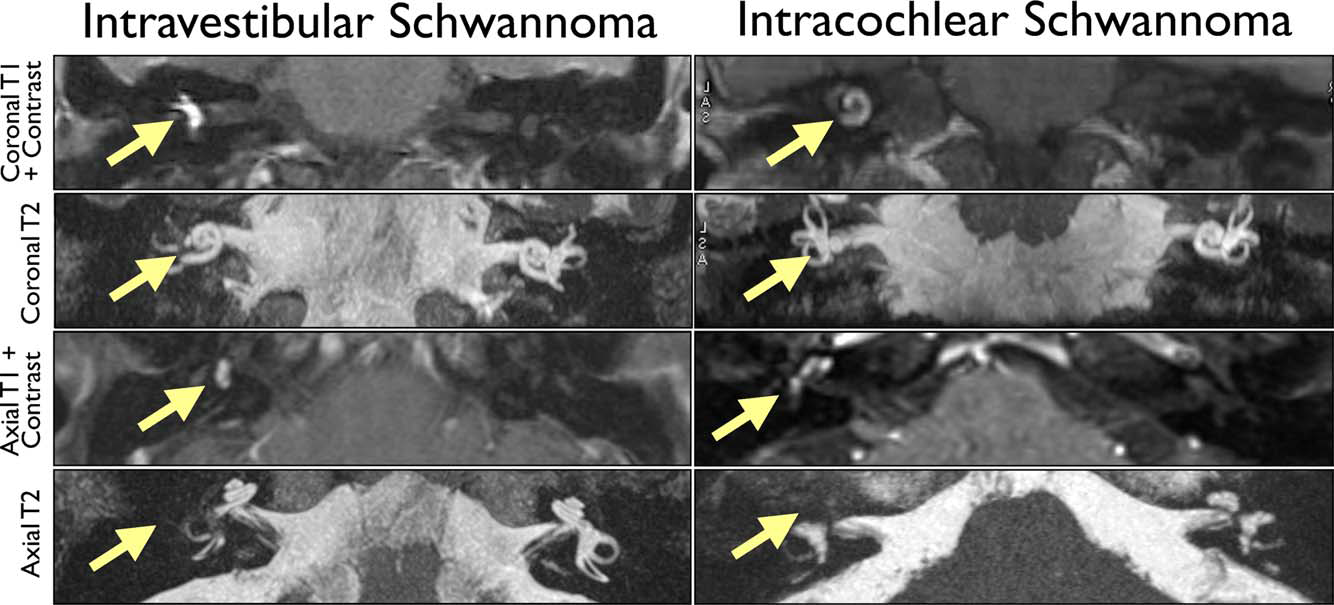
- Intralabyrinthine, meaning that the tumor lies within the labyrinth itself (Van Abel et al., 2013).
- Intracanalicular (intrameatal), meaning that the tumor lies entirely within the internal auditory canal.
- Intra- and extra-canalicular (intra- and extra-meatal), meaning that the tumor lies within the internal auditory canal, crosses the internal acoustic meatus, and extends into the cerebellopontine angle.
 |
 |
 |
 |
 |
 |
Mechanism of symptoms
There are probably multiple mechanisms by which vestibular schwannomas can cause auditory and vestibular symptoms, including:
First, vestibular schwannomas may have direct effects on the vestibulocochlear nerve, such as:
-
- The tumorous Schwann cells (i.e., the Schwann cells that are growing inappropriately) are not normal, and their usual function (namely, facilitating nerve conduction) is disturbed.
- Mechanical distortion of the vestibular nerve, damaging neuronal axons and healthy (non-tumorous) Schwann cells. This can disrupt both afferent signals (coming in from type 1 hair cells) and efferent signals (outgoing to type 2 hair cells).
- These result in conduction deficits, including failure to propagate signal; slow propagation; aberrant firing; ephaptic transmission.
Second, vestibular schwannomas may Interfere with circulation:
- If intracanalicular: compression of the labyrinthine artery, thereby compromising blood flow to the inner ear.
- Possible change of protein composition in labyrinth’s endolymphatic fluid (Asthagiri et al., 2012).
Finally, vestibular schwannomas that are extra-canalicular may exert mass effect on the brainstem or cerebellum.
Diagnosis: imaging
The cornerstone of diagnosis is MRI of the internal auditory canals without and with contrast, and this is currently considered the criterion standard. However, this is an expensive study, so when the history is suggestive of a vestibular schwannoma, practitioners may consider first checking other (less expensive) tests for screening, including audiometry, brainstem auditory evoked responses and certain vestibular tests. If those screening tests are all normal, then the likelihood of vestibular schwannoma is very low, and imaging may be avoided. In contrast, if those screening tests are abnormal, then seeking confirmation with imaging is appropriate.
Diagnosis: hearing tests
Audiometry. Since the most common presenting symptom is hearing loss, it is logical to assess hearing in greater detail. Although almost any pattern of hearing loss is possible (Thomsen, Terkildsen, & Tos, 1983), the most common is high frequency hearing loss.
Auditory brainstem evoked responses (ABRs), also called brainstem auditory evoked responses (BAERs). The sensitivity/specificity of this test for vestibular schwannoma remains controversial, but some clinicians still regard this as a useful screening test (Koors, Thacker, & Coelho, 2013).
Diagnosis: vestibular tests
Videonystagmography (VNG): Caloric weakness. Although symptoms of disequilibrium comprise the initial presentation in only a minority of cases, subclinical (i.e., asymptomatic) vestibular deficits can be detected in a larger percentage of patients. For example, a recent single-institution review of 703 patients with vestibular schwannoma (Tos et al., 1998) found that only 14% of individuals reported dizziness/unsteadiness as the presenting symptom, yet when assessed with videonystagmography, 400 (57%) had an absent caloric response on the affected side, 243 (35%) had a reduced caloric response on the affected side, and only 60 (8%) had a normal caloric response on the affected side; in other words, fully 92% of individuals had an abnormal (reduced or absent) caloric response on the side of the vestibular schwannoma. Other studies report slightly lower numbers, in the range of 77% – 87% (Hulshof, Hilders, & Baarsma, 1989).
Videonystagmography: Hyperventilation-induced nystagmus (HVIN). Although not in wide use, the finding of hyperventilation-induced nystagmus has been well-studied and is quite sensitive for vestibular schwannomas (Bance, O’Driscoll, Patel, & Ramsden, 1998; Bradley, Hullar, Neely, & Goebel, 2011; Choi, Cho, Koo, Park, & Kim, 2005; Mandala, Giannuzzi, Astore, Trabalzini, & Nuti, 2013; Minor, Haslwanter, Straumann, & Zee, 1999). HVIN is believed to arise from the fact that hyperventilation changes serum acid-base balance, and perhaps also extracellular free calcium concentration, thereby “unmasking” the abnormal neural transmission of the vestibular nerve (Burke, 1993).
Cervical vestibular evoked myogenic potentials (cVEMPs). Cervical VEMPs are often diminished in amplitude ipsilateral to a vestibular schwannoma (Matsuzaki, Murofushi, & Mizuno, 1999; Murofushi, Matsuzaki, & Mizuno, 1998; Suzuki et al., 2008; Takeichi, Sakamoto, Fukuda, & Inuyama, 2001; Ushio et al., 2009). The deficits detected on cVEMPs do not always correlate with those identified on caloric testing (C. H. Huang, Wang, & Young, 2012), though this is unsurprising since caloric testing assesses afferents from the horizontal semicircular canal (which run through the superior division of the vestibular nerve) whereas cVEMPs test afferents from the saccule (which run through the inferior division of the vestibular nerve).
Ocular vestibular evoked myogenic potentials (oVEMPs). Ocular VEMPs are often diminished in amplitude ipsilateral to a vestibular schwannoma, and this deficit frequently correlates with a caloric weakness (C. H. Huang et al., 2012), which makes sense since caloric testing assesses afferents from the horizontal semicircular canal (which run through the superior division of the vestibular nerve) and oVEMPs assess afferents from the utricle (which also run through the superior division of the vestibular nerve).
Video head impulse testing (vHIT) often shows abnormalities ipsilateral to a vestibular schwannoma (Aalling, Skals, Abrahamsen, & Hougaard, 2020; Blodow et al., 2013). One study (Blodow et al., 2013) reported caloric testing to be abnormal more frequently than video head impulse testing. This discrepancy is likely due to the fact that caloric testing assesses the low end of the vestibular frequency spectrum, while video head impulse testing assesses the high end of the vestibular frequency spectrum, and it may be that these fibers are differentially affected by the lesion.
Galvanic stimulation of the vestibular nerve often elicits asymmetrical ocular motor responses in patients with vestibular schwannomas (Mackenzie et al., 2018; Welgampola, Ramsay, Gleeson, & Day, 2013), but this test is generally restricted to research settings.
Practically, since the various parts of the vestibular nerve can be affected in nearly any pattern, our practice has been to check videonystagmography (with caloric testing, hyperventilation and neck vibration), cervical and ocular vestibular evoked myogenic potentials, and video head impulse testing, as these are readily available in most clinical laboratories.
Diagnosis: differential
Other tumors can manifest with symptoms that, at least initially, resemble those of a vestibular schwannoma. Considerations include meningiomas (Nakamura et al., 2004), malignant peripheral nerve sheath tumors (Gupta, Mammis, & Maniker, 2008; Kuzmik et al., 2013; Wei et al., 2012), and tumors metastatic from elsewhere, such as melanoma (Lopci, Monti, Balzarini, & Chiti, 2013).
Management: monitoring
If a vestibular schwannoma is suspected by history, if the suspicion is further raised by screening tests and then confirmed on imaging, then the question becomes one of management.
If a vestibular schwannoma is not posing any immediate danger (such as brainstem compression), and if the symptoms for which it is believed to be responsible are manageable, then many practitioners will take a conservative course of watchful waiting, which may include serial imaging.
The rationale behind serial imaging is that the tumor’s behavior may be quite variable. Some tumors appear never to change size after they are discovered. Other tumors are found to grow but then plateau and stop growing. Still others continue to grow. Surprisingly, some vestibular schwannomas appear spontaneously to shrink (X. Huang, Caye-Thomasen, & Stangerup, 2013a); one study reported that of 1261 patients with vestibular schwannoma, 48 (3.81%) exhibited spontaneous shrinkage of an average of 52.1% during the period of observation (X. Huang, Caye-Thomasen, & Stangerup, 2013b).
The frequency and interval of serial imaging has remained a matter of debate. For patients who are asymptomatic, or whose symptoms are manageable and not changing, one study (Stangerup & Caye-Thomasen, 2012) suggested that an MRI of the internal auditory canals without and with gadolinium should be performed:
- Every year for years 1 – 5 after diagnosis.
- Every other year for years 6 – 10 after diagnosis.
- Once at 15 years after diagnosis.
- At that point if there has been no growth, then no further imaging is recommended.
In parallel, a patient’s symptoms should be monitored clinically. Some clinicians also elect to perform annual audiometry. Interpretation of serial audiometry is made more complicated by the fact that expected age-related hearing loss may be superimposed on whatever component of hearing loss is attributable to the vestibular schwannoma.
Management: intervention
If symptomatic or radiographic progression warrants action, then an otolaryngologist may consider intervention. The otolaryngologist may take into account several factors, including the tumor’s location, size, what (if any) structures are being compressed, and whether any salvageable hearing remains.
Management: medical
Medical therapy, in the sense of oral medications, do not usually play a significant role in the treatment of vestibular schwannomas. However, there may be some rationale for considering anti-epileptic medications (particularly oxcarbazepine), on the physiologic basis that this approach is thought to stabilize cell membranes and may diminish abnormal signal transmission through the vestibular nerve (Ahle, Visser, Juergens, & Schlegel, 2008).
Management: radiosurgery
Radiosurgical modalities include gamma knife and stereotactic radiosurgeries. These approaches have both similarities and differences (Guadix et al., 2021).
Gamma knife surgery directs targeted beams of ionizing radiation such that they hit as much of the tumor as possible, and as little normal tissue as possible (Agarwal, Natanasabapathi, Bisht, Malhotra, & Kale, 2022). The intent is for the radiation to damage the tumorous Schwann cells so as to retard their growth, or even cause them to shrink (Guadix et al., 2021; Hafez, Morgan, Fahmy, & Hassan, 2020; Hwang et al., 2022; Kawashima et al., 2020; Kieu, Vuong, Mai, Pham, & Le, 2021; Killeen et al., 2022; Livingston et al., 2021; Wage, Mignano, & Wu, 2021; Yeole et al., 2022). Appropriate targeting of the radiation beams is a non-trivial task and requires considerable planning, usually by a team consisting of some combination of an otolaryngologist, neurosurgeon, radiation oncologist and medical physicist. In some cases the tumor may seem to grow, usually at 3 – 6 months following gamma knife surgery, but this appears to be due to swelling from tumor necrosis, and thus is sometimes termed “pseudo-progression” (Nasi, Zunarelli, Puzzolante, Moriconi, & Pavesi, 2020; Yang et al., 2021).
Other types of radiosurgery, including fractionated and unfractionated LINAC (linear particle accelerator) have been explored, though overall have been less studied (Anderson et al., 2014; Friedman, 2008; Friedman & Foote, 2003; Guadix et al., 2021; Matsuo et al., 2015; Okunaga et al., 2005; Spiegelmann et al., 2001) than gamma knife.
Management: open surgery
Vestibular schwannomas are located in the skull base, which is not easily accessible. There are several surgical approaches, each with its advantages and disadvantages. The otolaryngologist will decide which approach makes the most sense based on clinical history and tumor location.
Retrosigmoid approach. In this approach a neurosurgeon performs a suboccipital craniotomy and retracts the cerebellar hemisphere, providing access to the cerebellopontine angle and medial aspect of the temporal bone, from where it is possible to drill and expose the internal auditory canal. Advantages of this approach include that it can be used for tumors of any size, and it offers some possibility of hearing preservation. Disadvantages include that it is quote invasive, and there is a likelihood of damage to the cerebellum during retraction.
Middle fossa approach. In this approach a neurosurgeon performs a temporal craniotomy and retracts the temporal lobe, giving the otolaryngologist has access to the temporal bone and thus to the vestibular schwannoma. Advantages include that it is sometimes possible to preserve hearing. Disadvantages include that it is limited to small intracanalicular tumors.
Translabyrinthine approach. In this approach the otolaryngologists enters directly through the temporal bone. Advantages include that it is less invasive than the other approaches (not requiring neurosurgical assist). Disadvantages include that there is complete loss of auditory and vestibular function, as the labyrinth is destroyed.
Management: transtympanic gentamicin
For patients whose symptoms are predominantly vestibular, who have no salvageable hearing, and who are failing medical management, some practitioners may consider chemical ablation of vestibular function with a transtympanic gentamicin injection (a technique also used in Ménière’s disease) (Giannuzzi, Merkus, & Falcioni, 2013). This approach is controversial because the procedure ablates function of the inner ear rather than function of the vestibular nerve (where the true abnormality lies).
Outcomes after surgical intervention
Some studies report that patients in whom pre-operative vestibular testing found a residual caloric response in the ear with the vestibular schwannoma did worse (in terms of dizziness) post-operatively than those patients with absent vestibular function pre-operatively (Uehara et al., 2011). At some level this makes sense, because if a patient has no vestibular function pre-operatively, then there is no further vestibular function to lose, so a destructive procedure would cause no new deficit.
With open surgical approaches, post-procedural dizziness and changes in balance are usually fairly immediate.
In contrast, outcomes following gamma knife surgery are different in that some effects (including impact on equilibrium) may be delayed by several months (Wackym, Hannley, Runge-Samuelson, Jensen, & Zhu, 2008).
It is reasonable to ask why there should be new or worsening of symptoms following these interventions. Prior to surgery or radiosurgery the vestibulocochlear nerve was probably already behaving abnormally (due to adverse effects of the vestibular schwannoma on nerve conduction, such as impaired signal propagation, slowed conduction velocities, aberrant firing and ephaptic transmission). Although surgery and radiosurgery may debulk or shrink the tumor, this does not mean that the nerve is restored to its normal function; rather it becomes abnormal in a different way, and may therefore continue to behave abnormally. This may be due to mechanisms similar to what happens with demyelinating disease of the central nervous system (Rae-Grant, 2013; Smith & McDonald, 1999).
Post-surgical rehabilitation
Vestibular rehabilitation is the mainstay of therapy after surgical intervention, though the exact type of therapy, and the timing of therapy, is a matter of debate. One meta-analysis of randomized treatment trials suggested that adaptation exercises are superior to habituation exercises (Passier, Doherty, Smith, & McPhail, 2012), but more research is needed. Therapy involving visual feedback is emerging (Cakrt et al., 2010), but also requires further study.
In some cases, despite appropriately-targeted vestibular rehabilitation therapy, outcomes are poor even several years after surgery. One study found that approximately 10% of patients reported post-operative dizziness as “disabling” (Tufarelli et al., 2007).
It is difficult to predict which patients will have good outcomes, and which will have poor outcomes. Early studies reported correlations such as, “Worse imbalance pre-operatively correlates with poorer balance post-operatively,” or “Better results on vestibular testing pre-operatively correlates with poorer balance post-operatively,” but more recent review found insufficient evidence to support such correlations (Saman, Bamiou, & Gleeson, 2009).
Practically, it is probably always better to try some sort of formal vestibular rehabilitation therapy rather than no rehabilitation at all. The rehabilitative program should be supervised by a physical therapist with specific expertise in balance disorders. If post-operative disequilibrium is moderate to severe, then a home safety assessment may be warranted, or an occupational therapist should become involved.
Note on neurofibromatosis type 2
Neurofibromatosis type 2 (NF2) results from abnormalities in the NF2 gene on chromosome 22 that encodes the synthesis of the protein merlin. These genetic abnormalities can arise from autosomal dominant inheritance (accounting for about half of all cases of NF2), or from de novo mutations (accounting for the other half of all cases of NF2). These mutations result in abnormal production/proliferation of Schwann cells, manifesting with non-malignant neuroectodermal tumors in multiple areas of the body (central nervous system, peripheral nervous system, skin), including schwannomas, meningiomas, gliomas and neurofibromas.
The relevance to the present discussion is that NF2 classically causes bilateral vestibular schwannomas. Of all people with vestibular schwannomas, approximately 5% of cases are attributable to NF2.
The management of bilateral vestibular schwannomas in the context of NF2 is generally undertaken by a neuro-oncologist.
References
Aalling, M., Skals, R. K., Abrahamsen, E. R., & Hougaard, D. D. (2020). Comparison of test results from two separate video head impulse test systems in a cohort of patients diagnosed with a unilateral vestibular schwannoma. Eur Arch Otorhinolaryngol, 277(11), 3185-3193. doi:10.1007/s00405-020-06116-2
Agarwal, P., Natanasabapathi, G., Bisht, R. K., Malhotra, R. K., & Kale, S. S. (2022). Investigation of optimal planning strategy in gamma knife perfexion for vestibular schwannoma tumor using hybrid plan technique. Biomed Phys Eng Express, 8(6). doi:10.1088/2057-1976/ac9abb
Ahle, G., Visser, W., Juergens, A., & Schlegel, U. (2008). Successful treatment of hyperventilation-induced nystagmus in vestibular schwannoma with oxcarbacepine. J Neurol, 255(7), 1093. doi:10.1007/s00415-008-0842-6
Anderson, B. M., Khuntia, D., Bentzen, S. M., Geye, H. M., Hayes, L. L., Kuo, J. S., . . . Mehta, M. P. (2014). Single institution experience treating 104 vestibular schwannomas with fractionated stereotactic radiation therapy or stereotactic radiosurgery. J Neurooncol, 116(1), 187-193. doi:10.1007/s11060-013-1282-4
Asthagiri, A. R., Vasquez, R. A., Butman, J. A., Wu, T., Morgan, K., Brewer, C. C., . . . Lonser, R. R. (2012). Mechanisms of hearing loss in neurofibromatosis type 2. PLoS One, 7(9), e46132. doi:10.1371/journal.pone.0046132
Bance, M. L., O’Driscoll, M., Patel, N., & Ramsden, R. T. (1998). Vestibular disease unmasked by hyperventilation. Laryngoscope, 108(4, part 1), 610-614. doi:PMID: 9546279
Blodow, A., Helbig, R., Wichmann, N., Wenzel, A., Walther, L. E., & Bloching, M. B. (2013). [Video head impulse test or caloric irrigation?. Contemporary diagnostic tests for vestibular schwannoma]. HNO, 61(9), 781-785. doi:10.1007/s00106-013-2752-x
Bradley, J. P., Hullar, T. E., Neely, J. G., & Goebel, J. A. (2011). Hyperventilation-induced nystagmus and vertigo after stereotactic radiotherapy for vestibular schwannoma. Otol Neurotol, 32(8), 1336-1338. doi:10.1097/MAO.0b013e31822e8666
Burke, D. (1993). Microneurography, impulse conduction, and paresthesias. Muscle Nerve, 16(10), 1025-1032. doi:10.1002/mus.880161005
Cakrt, O., Chovanec, M., Funda, T., Kalitova, P., Betka, J., Zverina, E., . . . Jerabek, J. (2010). Exercise with visual feedback improves postural stability after vestibular schwannoma surgery. Eur Arch Otorhinolaryngol, 267(9), 1355-1360. doi:10.1007/s00405-010-1227-x
Choi, K. D., Cho, H. J., Koo, J. W., Park, S. H., & Kim, J. S. (2005). Hyperventilation-induced nystagmus in vestibular schwannoma. Neurology, 64(12), 2062. Retrieved from http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=15985572
Evans, D. G., Moran, A., King, A., Saeed, S., Gurusinghe, N., & Ramsden, R. (2005). Incidence of vestibular schwannoma and neurofibromatosis 2 in the North West of England over a 10-year period: higher incidence than previously thought. Otol Neurotol, 26(1), 93-97. doi:10.1097/00129492-200501000-00016
Friedman, W. A. (2008). Linear accelerator radiosurgery for vestibular schwannomas. Prog Neurol Surg, 21, 228-237. doi:10.1159/000157171
Friedman, W. A., & Foote, K. D. (2003). Linear accelerator-based radiosurgery for vestibular schwannoma. Neurosurg Focus, 14(5), e2. doi:10.3171/foc.2003.14.5.3
Giannuzzi, A. L., Merkus, P., & Falcioni, M. (2013). The use of intratympanic gentamicin in patients with vestibular schwannoma and disabling vertigo. Otol Neurotol, 34(6), 1096-1098. doi:10.1097/MAO.0b013e3182804c41
Guadix, S. W., Tao, A. J., An, A., Demetres, M., Tosi, U., Chidambaram, S., . . . Pannullo, S. C. (2021). Assessing the long-term safety and efficacy of gamma knife and linear accelerator radiosurgery for vestibular schwannoma: A systematic review and meta-analysis. Neurooncol Pract, 8(6), 639-651. doi:10.1093/nop/npab052
Gupta, G., Mammis, A., & Maniker, A. (2008). Malignant peripheral nerve sheath tumors. Neurosurg Clin N Am, 19(4), 533-543, v. doi:10.1016/j.nec.2008.07.004
Hafez, R. F. A., Morgan, M. S., Fahmy, O. M., & Hassan, H. T. (2020). Outcomes of Gamma Knife Surgery retreatment for growing vestibular schwannoma and review of the literature. Clin Neurol Neurosurg, 198, 106171. doi:10.1016/j.clineuro.2020.106171
Hoa, M., Drazin, D., Hanna, G., Schwartz, M. S., & Lekovic, G. P. (2012). The approach to the patient with incidentally diagnosed vestibular schwannoma. Neurosurg Focus, 33(3), E2. doi:10.3171/2012.6.FOCUS12209
Huang, C. H., Wang, S. J., & Young, Y. H. (2012). Correlation between caloric and ocular vestibular evoked myogenic potential test results. Acta Otolaryngol, 132(2), 160-166. doi:10.3109/00016489.2011.624120
Huang, X., Caye-Thomasen, P., & Stangerup, S. E. (2013a). Distinct spontaneous shrinkage of a sporadic vestibular schwannoma. Auris Nasus Larynx, 40(2), 243-246. doi:10.1016/j.anl.2012.01.011
Huang, X., Caye-Thomasen, P., & Stangerup, S. E. (2013b). Spontaneous tumour shrinkage in 1261 observed patients with sporadic vestibular schwannoma. J Laryngol Otol, 127(8), 739-743. doi:10.1017/S0022215113001266
Hulshof, J. H., Hilders, C. G., & Baarsma, E. A. (1989). Vestibular investigations in acoustic neuroma. Acta Otolaryngol, 108(1-2), 38-44. doi:10.3109/00016488909107390
Hwang, I., Choi, S. H., Kim, J. W., Yeon, E. K., Lee, J. Y., Yoo, R. E., . . . Sohn, C. H. (2022). Response prediction of vestibular schwannoma after gamma-knife radiosurgery using pretreatment dynamic contrast-enhanced MRI: a prospective study. Eur Radiol, 32(6), 3734-3743. doi:10.1007/s00330-021-08517-1
Jeyakumar, A., Seth, R., Brickman, T. M., & Dutcher, P. (2007). The prevalence and clinical course of patients with ‘incidental’ acoustic neuromas. Acta Otolaryngol, 127(10), 1051-1057. doi:10.1080/00016480701200210
Kawashima, M., Hasegawa, H., Shin, M., Takahashi, W., Shinya, Y., Iwasaki, S., . . . Saito, N. (2020). Long-term Outcomes of Gamma Knife Radiosurgery for Treating Vestibular Schwannoma With a Lower Prescription Dose of 12 Gy Compared With Higher Dose Treatment. Otol Neurotol, 41(10), e1314-e1320. doi:10.1097/MAO.0000000000002885
Kieu, H. D., Vuong, D. N., Mai, K. T., Pham, P. C., & Le, T. D. (2021). Long-term outcomes of rotating gamma knife for vestibular schwannoma: A 4-year prospective longitudinal study of 89 consecutive patients in Vietnam. Surg Neurol Int, 12, 585. doi:10.25259/SNI_687_2021
Killeen, D. E., Tolisano, A. M., Isaacson, B., Kutz, J. W., Barnett, S., Wardak, Z., & Hunter, J. B. (2022). Vestibular Schwannoma Tumor Size and Growth Rate Predict Response with Gamma Knife Stereotactic Radiosurgery. J Neurol Surg B Skull Base, 83(1), 11-18. doi:10.1055/s-0040-1716677
Komatsuzaki, A., & Tsunoda, A. (2001). Nerve origin of the acoustic neuroma. J Laryngol Otol, 115(5), 376-379. doi:10.1258/0022215011907910
Koors, P. D., Thacker, L. R., & Coelho, D. H. (2013). ABR in the diagnosis of vestibular schwannomas: a meta-analysis. Am J Otolaryngol, 34(3), 195-204. doi:10.1016/j.amjoto.2012.11.011
Kuzmik, G. A., Michaelides, E. M., Chiang, V. L., Nonaka, Y., Fukushima, T., Vortmeyer, A. O., & Bulsara, K. R. (2013). Rapidly progressive epithelioid malignant peripheral nerve sheath tumor of the vestibular nerve. Otol Neurotol, 34(9), 1739-1742. doi:10.1097/MAO.0b013e31829ab8be
Lin, D., Hegarty, J. L., Fischbein, N. J., & Jackler, R. K. (2005). The prevalence of “incidental” acoustic neuroma. Arch Otolaryngol Head Neck Surg, 131(3), 241-244. doi:10.1001/archotol.131.3.241
Livingston, A. J., Espahbodi, M., Harvey, S. A., Albano, K., Quinet, S. A., Bovi, J. A., & Friedland, D. R. (2021). Gamma Knife Treatment of Vestibular Schwannoma Planned With Computed Tomography Cisternography. Adv Radiat Oncol, 6(3), 100631. doi:10.1016/j.adro.2020.100631
Lopci, E., Monti, L., Balzarini, L., & Chiti, A. (2013). Cardiac and acoustic metastases in relapsing melanoma. Clin Nucl Med, 38(2), e85-88. doi:10.1097/RLU.0b013e31826c0cc0
Mackenzie, S. W., Irving, R., Monksfield, P., Kumar, R., Dezso, A., & Reynolds, R. F. (2018). Ocular torsion responses to electrical vestibular stimulation in vestibular schwannoma. Clin Neurophysiol, 129(11), 2350-2360. doi:10.1016/j.clinph.2018.08.023
Mandala, M., Giannuzzi, A., Astore, S., Trabalzini, F., & Nuti, D. (2013). Hyperventilation-induced nystagmus in vestibular schwannoma and unilateral sensorineural hearing loss. Eur Arch Otorhinolaryngol, 270(7), 2007-2011. doi:10.1007/s00405-012-2236-8
Matsuo, T., Okunaga, T., Kamada, K., Izumo, T., Hayashi, N., & Nagata, I. (2015). Long-term follow-up results of linear accelerator-based radiosurgery for vestibular schwannoma using serial three-dimensional spoiled gradient-echo MRI. J Clin Neurosci, 22(2), 320-325. doi:10.1016/j.jocn.2014.06.100
Matsuzaki, M., Murofushi, T., & Mizuno, M. (1999). Vestibular evoked myogenic potentials in acoustic tumor patients with normal auditory brainstem responses. Eur Arch Otorhinolaryngol, 256(1), 1-4. doi:10.1007/s004050050112
Minor, L. B., Haslwanter, T., Straumann, D., & Zee, D. S. (1999). Hyperventilation-induced nystagmus in patients with vestibular schwannoma. Neurology, 53(9), 2158-2168. doi:PMID: 10599798
Murofushi, T., Matsuzaki, M., & Mizuno, M. (1998). Vestibular evoked myogenic potentials in patients with acoustic neuromas. Arch Otolaryngol Head Neck Surg, 124(5), 509-512. doi:PMID: 9604975
Murray, M. R., Stout, A. P., & Bradley, C. F. (1940). Schwann cell versus fibroblast as the origin of the specific nerve sheath tumor: Observations upon normal nerve sheaths and neurilemomas in vitro. Am J Pathol, 16(1), 41-60. Retrieved from https://pubmed.ncbi.nlm.nih.gov/19970485/
Nakamura, M., Roser, F., Mirzai, S., Matthies, C., Vorkapic, P., & Samii, M. (2004). Meningiomas of the internal auditory canal. Neurosurgery, 55(1), 119-127; discussion 127-118. doi:10.1227/01.neu.0000126887.55995.e7
Nasi, D., Zunarelli, E., Puzzolante, A., Moriconi, E., & Pavesi, G. (2020). Early life-threating enlargement of a vestibular schwannoma after gamma knife radiosurgery. Acta Neurochir (Wien), 162(8), 1977-1982. doi:10.1007/s00701-020-04434-2
Okunaga, T., Matsuo, T., Hayashi, N., Hayashi, Y., Shabani, H. K., Kaminogo, M., . . . Nagata, I. (2005). Linear accelerator radiosurgery for vestibular schwannoma: measuring tumor volume changes on serial three-dimensional spoiled gradient-echo magnetic resonance images. J Neurosurg, 103(1), 53-58. doi:10.3171/jns.2005.103.1.0053
Passier, L., Doherty, D., Smith, J., & McPhail, S. (2012). Vestibular rehabilitation following removal of acoustic neuroma: a systematic review of randomised trials. Head Neck Oncol, 4(2), 59. Retrieved from https://www.oapublishinglondon.com/article/233#
Pothula, V. B., Lesser, T., Mallucci, C., May, P., & Foy, P. (2001). Vestibular schwannomas in children. Otol Neurotol, 22(6), 903-907. doi:10.1097/00129492-200111000-00032
Puls, T. (2015). Incidental findings of a Vestibular Schwannoma on Brain MRI. B-ENT, 11(4), 309-312. Retrieved from https://www.ncbi.nlm.nih.gov/pubmed/26891545
Rae-Grant, A. D. (2013). Unusual symptoms and syndromes in multiple sclerosis. Continuum (Minneap Minn), 19(4 Multiple Sclerosis), 992-1006. doi:10.1212/01.CON.0000433287.30715.07
Saman, Y., Bamiou, D. E., & Gleeson, M. (2009). A contemporary review of balance dysfunction following vestibular schwannoma surgery. Laryngoscope, 119(11), 2085-2093. doi:10.1002/lary.20648
Schmidt, R. F., Boghani, Z., Choudhry, O. J., Eloy, J. A., Jyung, R. W., & Liu, J. K. (2012). Incidental vestibular schwannomas: a review of prevalence, growth rate, and management challenges. Neurosurg Focus, 33(3), E4. doi:10.3171/2012.7.FOCUS12186
Selesnick, S. H., Deora, M., Drotman, M. B., & Heier, L. A. (1999). Incidental discovery of acoustic neuromas. Otolaryngol Head Neck Surg, 120(6), 815-818. doi:10.1016/S0194-5998(99)70319-0
Smith, K. J., & McDonald, W. I. (1999). The pathophysiology of multiple sclerosis: the mechanisms underlying the production of symptoms and the natural history of the disease. Philos Trans R Soc Lond B Biol Sci, 354(1390), 1649-1673. doi:10.1098/rstb.1999.0510
Spiegelmann, R., Lidar, Z., Gofman, J., Alezra, D., Hadani, M., & Pfeffer, R. (2001). Linear accelerator radiosurgery for vestibular schwannoma. J Neurosurg, 94(1), 7-13. doi:10.3171/jns.2001.94.1.0007
Stangerup, S. E., & Caye-Thomasen, P. (2012). Epidemiology and natural history of vestibular schwannomas. Otolaryngol Clin North Am, 45(2), 257-268, vii. doi:10.1016/j.otc.2011.12.008
Suzuki, M., Yamada, C., Inoue, R., Kashio, A., Saito, Y., & Nakanishi, W. (2008). Analysis of vestibular testing in patients with vestibular schwannoma based on the nerve of origin, the localization, and the size of the tumor. Otol Neurotol, 29(7), 1029-1033. doi:10.1097/MAO.0b013e3181845854
Takeichi, N., Sakamoto, T., Fukuda, S., & Inuyama, Y. (2001). Vestibular evoked myogenic potential (VEMP) in patients with acoustic neuromas. Auris Nasus Larynx, 28(Suppplement), S39-S41. doi:PMID: 11683341
Thomsen, J., Terkildsen, K., & Tos, M. (1983). Acoustic neuromas. Progression of hearing impairment and function of the eighth cranial nerve. Am J Otol, 5(1), 20-33. Retrieved from https://www.ncbi.nlm.nih.gov/pubmed/6881306
Tos, M., Charabi, S., & Thomsen, J. (1998). Clinical experience with vestibular schwannomas: epidemiology, symptomatology, diagnosis, and surgical results. Eur Arch Otorhinolaryngol, 255(1), 1-6. doi:10.1007/s004050050012
Tufarelli, D., Meli, A., Labini, F. S., Badaracco, C., De Angelis, E., Alesii, A., . . . Sanna, M. (2007). Balance impairment after acoustic neuroma surgery. Otol Neurotol, 28(6), 814-821. doi:10.1097/mao.0b013e31811f40ad
Uehara, N., Tanimoto, H., Nishikawa, T., Doi, K., Katsunuma, S., Kimura, H., . . . Nibu, K. (2011). Vestibular dysfunction and compensation after removal of acoustic neuroma. J Vestib Res, 21(5), 289-295. doi:10.3233/VES-2011-0429
Ushio, M., Iwasaki, S., Murofushi, T., Sugasawa, K., Chihara, Y., Fujimoto, C., . . . Yamasoba, T. (2009). The diagnostic value of vestibular-evoked myogenic potential in patients with vestibular schwannoma. Clin Neurophysiol, 120(6), 1149-1153. doi:10.1016/j.clinph.2009.01.017
Van Abel, K. M., Carlson, M. L., Link, M. J., Neff, B. A., Beatty, C. W., Lohse, C. M., . . . Driscoll, C. L. (2013). Primary inner ear schwannomas: a case series and systematic review of the literature. Laryngoscope, 123(8), 1957-1966. doi:10.1002/lary.23928
Wackym, P. A., Hannley, M. T., Runge-Samuelson, C. L., Jensen, J., & Zhu, Y. R. (2008). Gamma Knife surgery of vestibular schwannomas: longitudinal changes in vestibular function and measurement of the Dizziness Handicap Inventory. J Neurosurg, 109 Suppl, 137-143. doi:10.3171/JNS/2008/109/12/S21
Wage, J., Mignano, J., & Wu, J. (2021). Tufts Medical Center Experience With Long-Term Follow-Up of Vestibular Schwannoma Treated With Gamma Knife Stereotactic Radiosurgery: Novel Finding of Delayed Pseudoprogression. Adv Radiat Oncol, 6(4), 100687. doi:10.1016/j.adro.2021.100687
Wei, C., Heman-Ackah, S. E., Newman, K., Zagzag, D., Golfinos, J. G., & Roland, J. T., Jr. (2012). Temporal bone histopathology case of the month: Malignant peripheral nerve sheath tumor arising within vestibular schwannoma. Otol Neurotol, 33(9), e83-84. doi:10.1097/MAO.0b013e3182659b44
Welgampola, M. S., Ramsay, E., Gleeson, M. J., & Day, B. L. (2013). Asymmetry of balance responses to monaural galvanic vestibular stimulation in subjects with vestibular schwannoma. Clin Neurophysiol, 124(9), 1835-1839. doi:10.1016/j.clinph.2013.03.015
Yang, H. C., Wu, C. C., Lee, C. C., Huang, H. E., Lee, W. K., Chung, W. Y., . . . Lu, C. F. (2021). Prediction of pseudoprogression and long-term outcome of vestibular schwannoma after Gamma Knife radiosurgery based on preradiosurgical MR radiomics. Radiother Oncol, 155, 123-130. doi:10.1016/j.radonc.2020.10.041
Yeole, U., Prabhuraj, A. R., Arivazhagan, A., Narasingarao, K. V. L., Vazhayil, V., Bhat, D., . . . Sampath, S. (2022). Gamma Knife Radiosurgery for Large Vestibular Schwannoma More Than 10 cm (3) : A Single-Center Indian Study. J Neurol Surg B Skull Base, 83(Suppl 2), e343-e352. doi:10.1055/s-0041-1729977
![]()