By Marcello Cherchi, MD PhD
For patients
Behcet disease (BD) is an autoimmune disease that usually begins in adulthood with ulcers involving the mouth, genitals and skin. It can affect other organs as well. Less commonly it can cause hearing problems or disequilibrium. If you have been diagnosed with Behcet disease and are experiencing hearing problems or disequilibrium, your doctor may want to check some tests of hearing and balance to figure out whether those symptoms are due to a more common cause, or whether they are due to Behcet disease itself.
For clinicians
Overview
Behcet disease (BD) is an immunologically mediated vasculopathy (venulitis more than arteritis) of unknown etiology, thought to occur after an environmental exposure in an otherwise genetically predisposed individual. It has a higher prevalence among people in or from the area stretching from Eastern Asia to the Mediterranean basin. Onset of symptoms is in the 4th or 5th decade; it is uncommon for it to begin in childhood or in people over the age of 50 years. The cardinal features include recurrent oral and genital ulcers, skin lesions and uveitis. BD can affect the central nervous system (sometimes called “neuro-Behcet disease”) in 4% – 49% of cases. About 47% of BD patients complain of some combination of audiologic symptoms (hearing loss, tinnitus, aural fullness) and vestibular symptoms, and these can be synchronous or metachronous with non-audiovestibular BD symptoms. Audiometry typically shows high frequency sensorineural hearing loss. Ocular motor abnormalities can include spontaneous nystagmus, dysmetric saccades, abnormal smooth pursuit or aperiodic alternating nystagmus. Caloric testing may or may not show vestibular paresis. Vestibular evoked myogenic potentials (VEMP) may show delayed latencies, diminished amplitudes or absent responses. Video head impulse testing (vHIT) may show low gain and compensatory saccades. None of these audiovestibular findings (or their combnation) is sensitive or specific for Behcet disease. Management of BD usually requires a multidisciplinary approach organized by a rheumatologist.
Introduction
The clinical syndrome that eventually became known as Behcet disease (BD) was probably documented as early as the 5th century BCE by Hippocrates (Feigenbaum 1956). However, a more complete description was given by Hulusi Behcet (1889 – 1948), a Turkish dermatologist who cultivated a particular interest in infectious diseases (Shakeri 2018; Tan and Poole 2016). In 1937 Behcet published a description in German of several patients with recurrent aphthous ulcers of the mouth, eyes and genitalia (Behçet 1937) which has been translated into English (Behcet and Matteson 2010). Behcet attributed these findings to a viral infection (Behcet 1938).

The malady now eponymously designated Behcet disease (BD) is currently believed to be an immunologically-mediated systemic vasculopathy (venulitis more than arteritis) (Ertugrul et al. 2019) typically manifesting with recurrent oral and genital ulcers, skin lesions and uveitis (Lavalle et al. 2024), though other organ systems can be affected.
Epidemiology
BD occurs worldwide, but “is more prevalent in countries along the ancient Silk Road, extending from Eastern Asia to the Mediterranean basin” (Lavalle et al. 2024; Verity et al. 1999), as shown in the Figure below from Cho and colleagues (Cho et al. 2012).
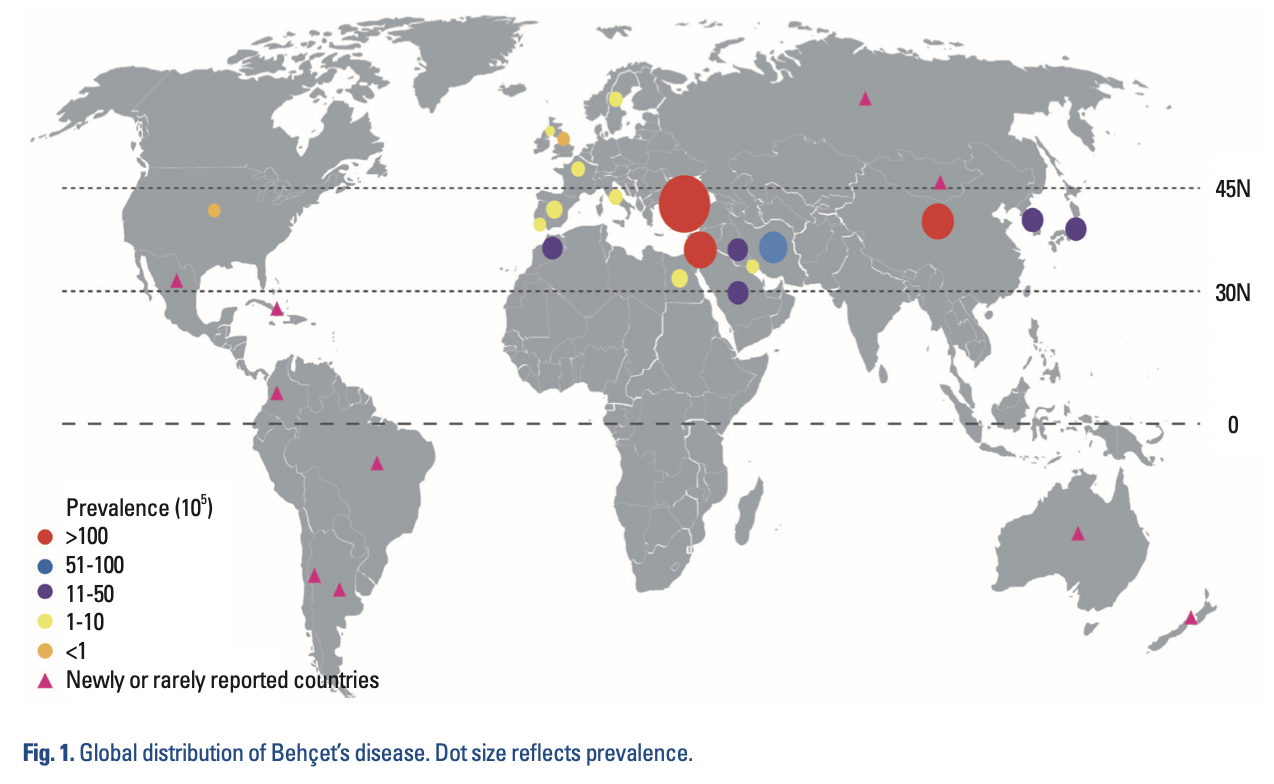
In these endemic areas the prevalence per 100,000 people is as shown in the Table below from Cho and colleagues (Cho et al. 2012).

Genetics
The HLA-B51 allele is found in up to 60% of patients with BD but only 10% – 20% of healthy controls (Lavalle et al. 2024), and thus is believed to confer increased risk for manifesting the disorder. Genetic markers less strongly associated with BD include variations in the genes for IL-10, IL-23R and ERAP1 (Lavalle et al. 2024).
Pathophysiological mechanism of disease
The underlying pathophysiologic mechanism of BD is unknown, though most investigators believe it “to result from an abnormal immune response triggered by an environmental agent in a genetically susceptible individual” (Lavalle et al. 2024).
The Figure below, from Cho and colleagues (Cho et al. 2012), diagrams potential mechanisms of BD.
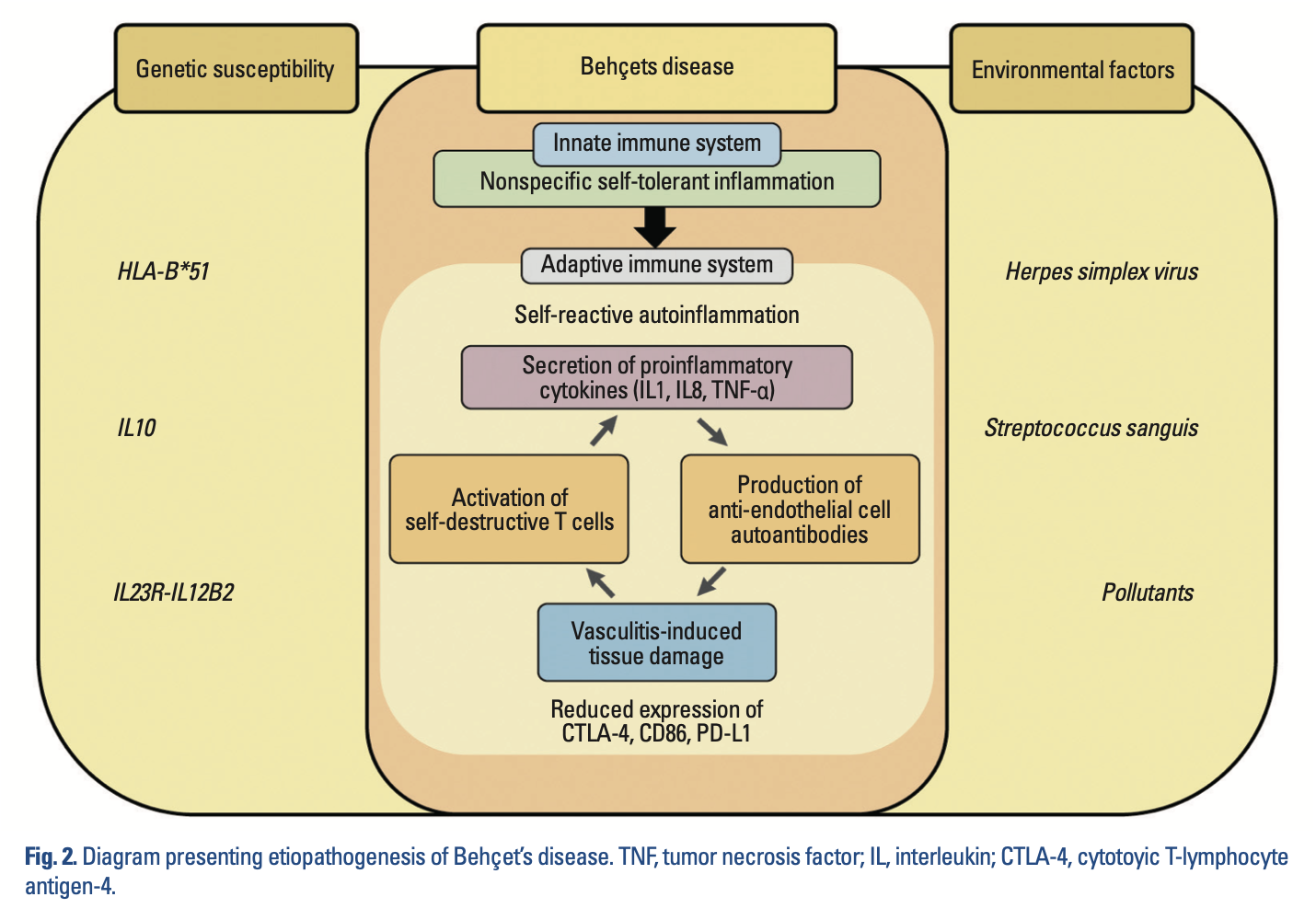
These inflammatory responses cause a vasculopathy. Ertugrul and colleagues state that in BD, “The inflammation of the venous vessels causes inflammatory thrombi which were thought to lead to the lumen being occluded. With regards to the arterial vessels, focal aneurysmal dilatation was mostly suspected and perivascular lymphocytic infiltration with corruption of medial elastic fibers was thought to lead to this pathology” (Ertugrul et al. 2019).
Clinical presentation
The onset of BD is usually in the third to fourth decade (Cho et al. 2012). Because it is a multisystem disorder, its clinical presentation is often variable (Lavalle et al. 2024) and can resemble other diseases. Over 95% of patients present with painful recurrent oral ulcers (Lavalle et al. 2024). Dermatologic manifestations include “erythema nodosum-like lesions” and “acneiform nodules;” ocular involvement includes anterior or posterior uveitis, which can cause blindness (Lavalle et al. 2024).
The International Study Group for Behcet Disease published diagnostic criteria in 1990 which are still used (International Study Group for Behcet’s Disease 1990). The criteria are:
- Recurrent oral ulceration, defined as “Minor aphthous, major aphthous, or herpetiform ulceration observed by physician or patient, which recurred at least 3 times in one 12-month period.”
- Plus any 2 of the following:
- Recurrent genital ulceration, defined as “Aphthous ulceration or scarring, observed by physician or patient.”
- Eye lesions, defined as “Anterior uveitis, posterior uveitis, or cells in vitreous on slit lamp examination; or retinal vasculitis observed by ophthalmologist.”
- Skin lesions, defined as “Erythema nodosum observed by physician or patient, pseudofolliculitis, or papulopustular lesions; or acneiform nodules observed by physician in post-adolescent patients not on corticosteroid treatment.”
- Positive pathergy test, “read by physician at 24 – 48 hours.”
Neurological involvement, sometimes designated “neuro-Behcet disease,” is less common, thought to affect from 4% – 49% of all Behcet patients (Park et al. 2020).
Central nervous system involvement in BD can manifest with auditory and vestibular symptoms, though to be clear, there is no pattern of audiovestibular deficits that is sensitive or specific for BD.
In the series of 17 BD with audiologic or vestibular symptoms studied by Choung and colleagues (Choung et al. 2006), 7 (41%) complained of auditory symptoms (hearing loss, tinnitus, aural fullness) and 14 (82%) complained of vestibular symptoms.
In a larger series of 62 BD patients, Kulahli and colleagues (Kulahli et al. 2005) reported the distribution of audiovestibular symptoms shown in the Table below.
|
Audiovestibular symptom |
Number of patients |
Percentage of patients |
|
Tinnitus |
7 |
11% |
|
Hearing loss |
6 |
10% |
|
Vertigo |
5 |
8% |
|
Hearing loss + tinnitus + vertigo |
5 |
8% |
|
Hearing loss + tinnitus |
4 |
7% |
|
Tinnitus + vertigo |
2 |
3% |
|
No auditory or vestibular symptoms |
33 |
53% |
|
Total |
62 |
100% |
Table : Audiovestibular symptoms in patients with Behcet disease, as reported by Kulahli et al. (Kulahli et al. 2005).
The timing of audiovestibular symptoms (compared to other symptoms) in BD, is variable, as they can constitute the initial presentation (Marsili et al. 2016) or follow other symptoms by a decade (Brama and Fainaru 1980).
Testing: auditory
Bayram and colleagues (Bayram et al. 2015) studied 30 BD patients and reported that 77% (23/30) exhibited high frequency sensorineural hearing loss, compared with only 30% of control subjects.
Erbek and colleagues (Erbek et al. 2008) studied 26 BD patients and reported that 42% (11/26) exhibited high frequency sensorineural hearing loss, and 54% (14/26) exhibited sensorineural hearing loss at some frequencies, as compared to control subjects of whom only 20% exhibited sensorineural hearing loss.
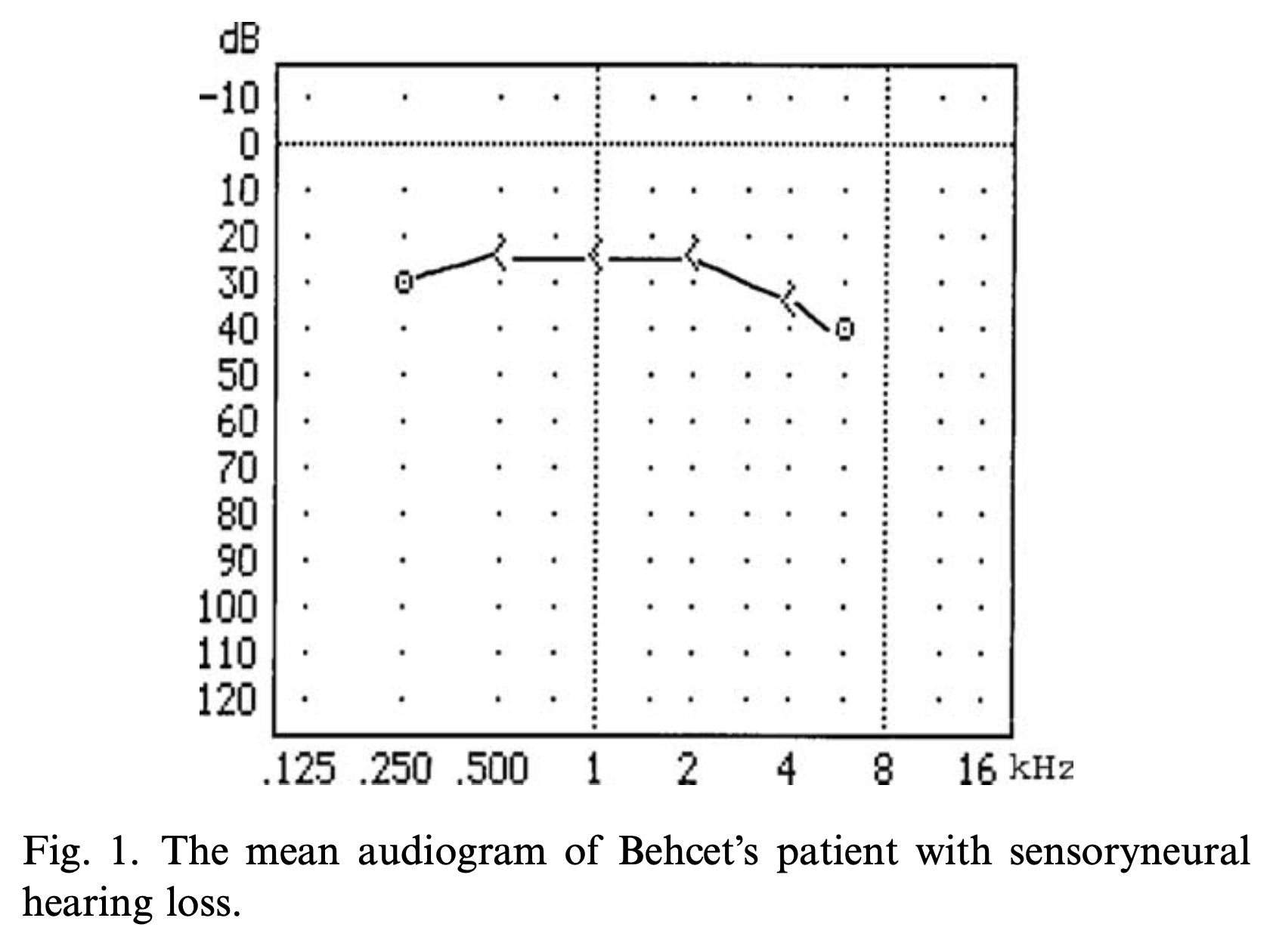
The most common pattern of sensorineural hearing loss in BD involves the high frequencies. The Figure below displays a composite audiogram of 62 BD patients studied by Kulahli and colleagues (Kulahli et al. 2005).
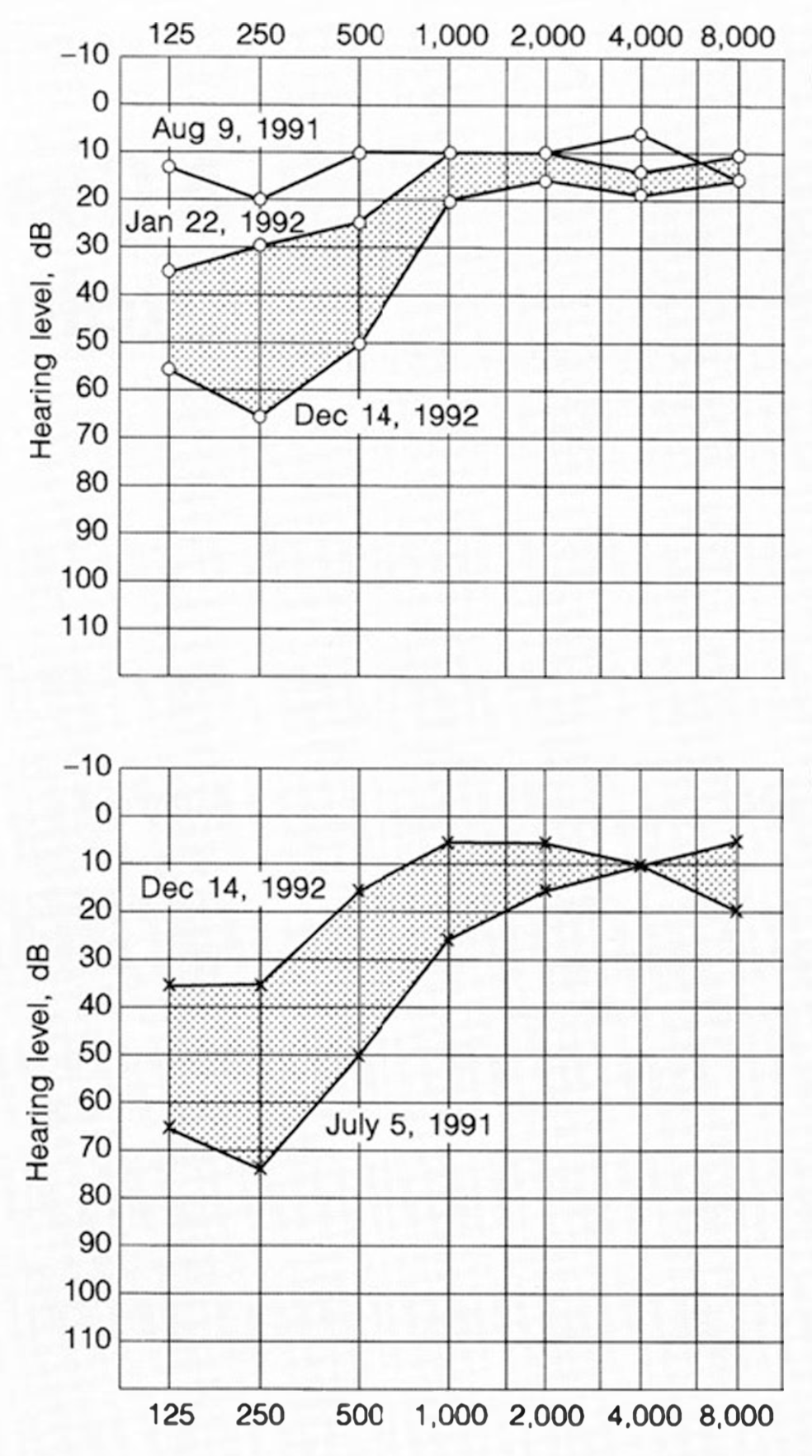
Less commonly, audiometry can show progressive or fluctuating sensorineural hearing loss in the low frequencies, resembling Ménière’s disease. An example of this is shown in the Figure below from Igarashi and colleagues (Igarashi et al. 1994).
Testing: ocular motor testing
Park and colleagues (Park et al. 2020), citing Gemigniani and colleagues (Gemignani et al. 1991), state that about 25% of neuro-Behcet disease patients exhibit abnormal ocular motor findings.
Choung and colleagues (Choung et al. 2006) studied instrumented ocular motor testing in 17 BD patients and reported that:
- 12% (2/17) exhibited spontaneous nystagmus.
- 6% (1/17) exhibited abnormal saccades.
- 59% (10/17) exhibited abnormalities on rotatory chair testing (not further specified).
Kulahli and colleagues studied electronystagmography (ENG) in 62 BD patients (Kulahli et al. 2005) and reported that:
- 34% (21/62) “had hypometric or hypermetric saccades.”
- 6% (4/62) “had pathological changes” in “slow pursuit” (not further specified).
Park and colleagues (Park et al. 2020) reported the case of a 46-year-old woman (eventually diagnosed with BD) who developed a headache, and two days later developed an “isolated vestibular syndrome” who, under video oculography (VOG), exhibited aperiodic alternating nystagmus, which they defined as, “a horizontal nystagmus that reverses direction without periodicity of the cycles, thereby presenting relatively irregular and short cycles of alternating nystagmus.”
Testing: caloric testing
Results of caloric testing in BD patients are variable.
- Choung and colleagues (Choung et al. 2006) reported that 29% (5/17) of patients exhibited “caloric abnormalities” (without further specification).
- Erbek and colleagues (Erbek et al. 2008) reported that 15% (4/26) of patients exhibited “canal paresis” (without further description).
- Kulahli and colleagues (Kulahli et al. 2005) and reported that 0% (0/62) of patients exhibited caloric abnormalities.
Testing: vestibular evoked myogenic potentials (VEMP)
Erbek and colleagues (Erbek et al. 2008) studied cervical vestibular evoked myogenic potentials (cVEMP) in 26 BD patients and reported that 27% (7/26) exhibited delayed p13 latencies.
Bayram and colleagues (Bayram et al. 2015) studied 30 BD patients with vestibular evoked myogenic potentials (VEMP) and reported that:
- For cervical vestibular evoked myogenic potentials (cVEMP):
- 17% (5/30) had delayed latencies.
- 7% (2/30) had diminished amplitudes.
- 10% (3/30) had absent responses.
- For ocular vestibular evoked myogenic potentials (oVEMP):
- 10% (3/30) had delayed latencies.
- 3% (1/30) had diminished amplitudes.
- 10% (3/30) had absent responses.
Testing: video head impulse testing (vHIT)
Some patients with BD exhibit vestibular weakness, evident on video head impulse testing (vHIT) with low gain and compensatory saccades. An example of this is shown in the Figure below from Ertugrul and colleagues (Ertugrul et al. 2019).
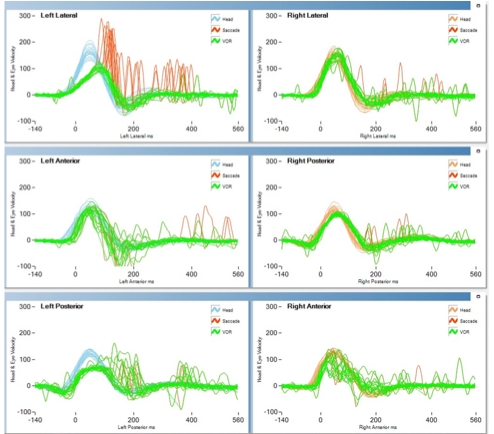
Testing: electrocochleography (ECoG)
Electrocochleography (ECoG) may show an elevated SP/AP ratio, as shown in the Figure below from Igarashi and colleagues (Igarashi et al. 1994).
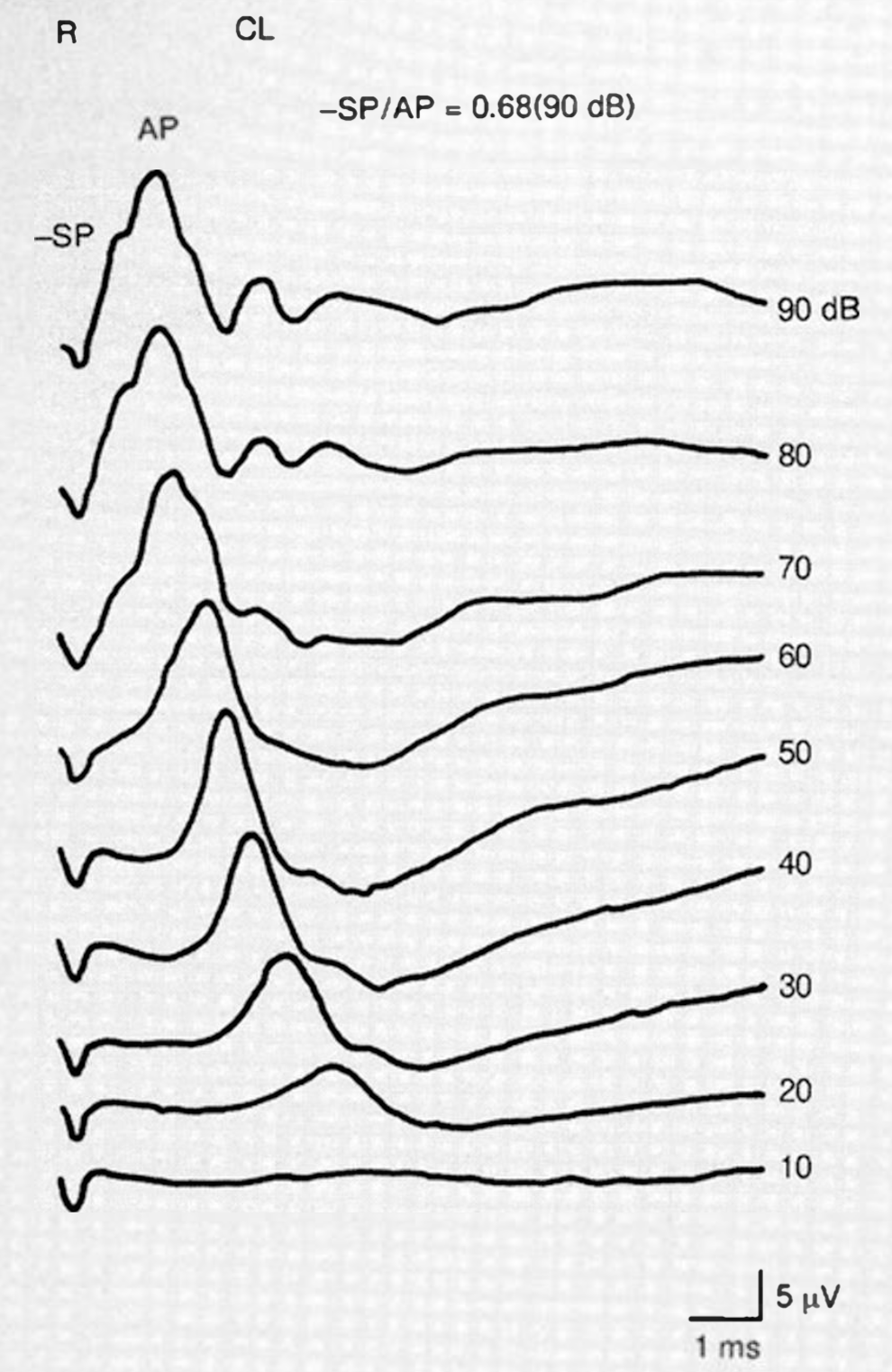
Testing: other
The HLA-B51 allele is found in up to 60% of patients with BD but only 10% – 20% of healthy controls (Lavalle et al. 2024), and thus is believed to confer increased risk for manifesting the disorder. Genetic markers less strongly associated with BD include variations in the genes for IL-10, IL-23R and ERAP1 (Lavalle et al. 2024).
Imaging
Rarely, BD can involve the inferior olivary nuclei and thus cause palatal tremor or oculopalatal tremor. The mechanism is presumably ischemic. An example of this is shown in the Figure below from Morgan and colleagues (Morgan et al. 2015).
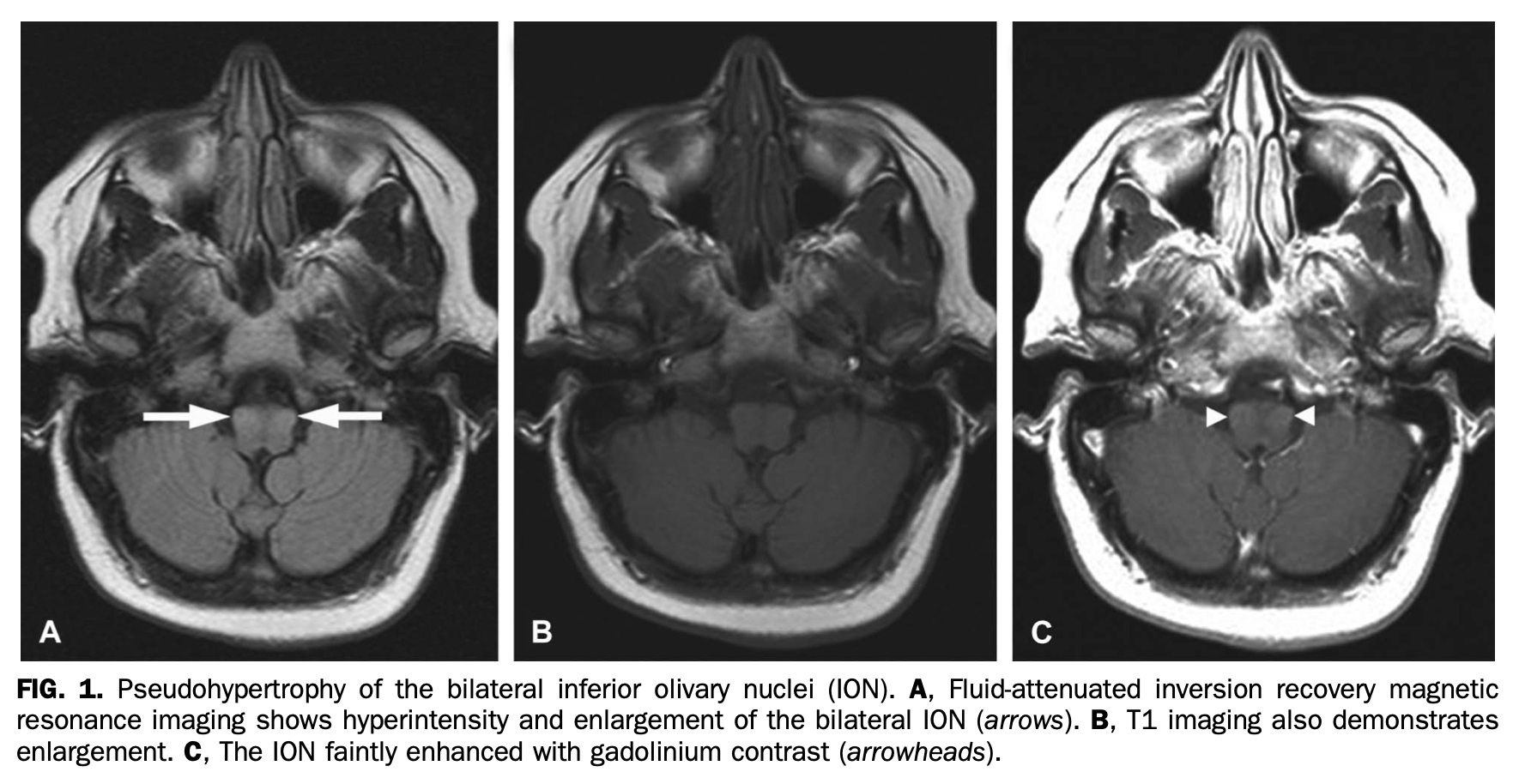
Histopathology
The histopathological manifestations of BD differ depending on the affected tissue.
Differential diagnosis
White and colleagues (White et al. 2014) reported the case of a man who developed “an acute vestibular syndrome with profound unilateral hearing loss.” The differential diagnosis includes labyrinthitis.
Since some patients with Behcet disease can exhibit fluctuating low frequency sensorineural hearing loss and have abnormal SP/AP ratios on electrocochleography (ECoG), the differential diagnosis includes Ménière’s disease.
Marsili and colleagues (Marsili et al. 2016) reported the case of a 15-year-old boy (ultimately diagnosed with Behcet disease) whose initial presentation consisted of “recurrent episodes of fever, vomiting, dizziness, headache, tinnitus and ataxia,” for which the initial differential diagnosis could have included migraine with migraine associated vertigo (MAV). A diagnosis of Behcet disease became clear when the boy subsequently developed oral ulcers and was found to have anterior uveitis and retinitis.
Treatment
Management of BD usually requires a multidisciplinary approach (Lavalle et al. 2024) organized by a rheumatologist.
It would be extraordinarily unusual for an undiagnosed BD patient to present initially to a vestibular clinic. The more common scenario would be one in which a patient already diagnosed with BD complains of audiologic or vestibular symptoms or both, and the clinical query for an otoneurologist or neuro-otologist is whether those symptoms are from BD itself or due to some other independently occurring pathology. In such cases it is reasonable to seek more common causes of such symptoms (such as evaluating for benign paroxysmal positional vertigo). If workup identifies hearing loss, then referral to audiology is appropriate. If workup identifies vestibular weakness then it is medically reasonable to refer to vestibular rehabilitation therapy (VRT).
Prognosis
The prognosis of BD is extremely variable. Cho and colleagues state that BD, “is rarely seen in children or patients above the age of 50. The clinical courses of childhood-onset Behcet’s disease and late-onset Behcet’s disease are relatively benign” (Cho et al. 2012).
References
Bayram A, Doğan M, Koç A, Kalkan M, Akçadağ A, Özcan İ (2015) Cervical and ocular vestibular evoked myogenic potentials in Behcet’s disease. Am J Otolaryngol 36: 503-8. doi: 10.1016/j.amjoto.2015.03.004
Behcet H (1938) Considerations sur les lesions aphteuses de la bouche et des parties genitals, ainsi que sur les manifestations oculaires d’origine probablement virutique et observations concernant leur foyer d’infection [Considerations on aphthous lesions of the mouth and genitals, as well as on ocular manifestations of probable viral origin and observations concerning their focus of infection]. Bull Soc Fr Dermatol Syphiligr 45: 420-433.
Behçet H (1937) Über rezidivierende, aphthöse, durch ein Virus verursachte Geschwüre am Mund, am Auge und an den Genitalien [Regarding recurrent, aphthous ulcers of the mouth, eyes and genitals caused by a virus]. Dermatologische Wochenschrift 105: 1152-1163.
Behcet H, Matteson EL (2010) On relapsing, aphthous ulcers of the mouth, eye and genitalia caused by a virus. 1937. Clin Exp Rheumatol 28: S2-5.
Brama I, Fainaru M (1980) Inner ear involvement in Behçet’s disease. Arch Otolaryngol 106: 215-7. doi: 10.1001/archotol.1980.00790280023004
Cho SB, Cho S, Bang D (2012) New insights in the clinical understanding of Behcet’s disease. Yonsei Med J 53: 35-42. doi: 10.3349/ymj.2012.53.1.35
Choung YH, Cho MJ, Park K, Choi SJ, Shin YR, Lee ES (2006) Audio-vestibular disturbance in patients with Behçet’s disease. Laryngoscope 116: 1987-90. doi: 10.1097/01.mlg.0000237442.80711.65
Erbek S, Erbek SS, Yilmaz S, Yucel E, Ozluoglu LN (2008) Vestibular evoked myogenic potentials in Behcet’s disease. Eur Arch Otorhinolaryngol 265: 1315-20. doi: 10.1007/s00405-008-0650-8
Ertugrul O, Mutlu A, Zindanci I, Cam OH, Ozluoglu L (2019) Audiological and vestibular measurements in Behçet’s disease. Eur Arch Otorhinolaryngol 276: 1625-1632. doi: 10.1007/s00405-019-05403-x
Feigenbaum A (1956) Description of Behcet’s syndrome in the Hippocratic third book of endemic diseases. Br J Ophthalmol 40: 355-7. doi: 10.1136/bjo.40.6.355
Gemignani G, Berrettini S, Bruschini P, Sellari-Franceschini S, Fusari P, Piragine F, Pasero G, Olivieri I (1991) Hearing and vestibular disturbances in Behçet’s syndrome. Ann Otol Rhinol Laryngol 100: 459-63. doi: 10.1177/000348949110000606
Igarashi Y, Watanabe Y, Aso S (1994) A case of Behçet’s disease with otologic symptoms. ORL J Otorhinolaryngol Relat Spec 56: 295-8. doi: 10.1159/000276678
International Study Group for Behcet’s Disease (1990) Criteria for diagnosis of Behcet’s disease. International Study Group for Behcet’s Disease. Lancet 335: 1078-80.
Kulahli I, Balci K, Koseoglu E, Yuce I, Cagli S, Senturk M (2005) Audio-vestibular disturbances in Behcet’s patients: report of 62 cases. Hear Res 203: 28-31. doi: 10.1016/j.heares.2004.11.020
Lavalle S, Caruso S, Foti R, Gagliano C, Cocuzza S, La Via L, Parisi FM, Calvo-Henriquez C, Maniaci A (2024) Behcet’s Disease, Pathogenesis, Clinical Features, and Treatment Approaches: A Comprehensive Review. Medicina (Kaunas) 60. doi: 10.3390/medicina60040562
Marsili M, Marzetti V, Lucantoni M, Lapergola G, Gattorno M, Chiarelli F, Breda L (2016) Autoimmune sensorineural hearing loss as presenting manifestation of paediatric Behçet disease responding to adalimumab: a case report. Ital J Pediatr 42: 81. doi: 10.1186/s13052-016-0291-2
Morgan ML, Espino Barros Palau A, Lee AG, Foroozan R (2015) Neuro-Behçet disease presenting with oculopalatal tremor. J Neuroophthalmol 35: 51-3. doi: 10.1097/wno.0000000000000159
Park E, Lee SU, Kim HJ, Choi JY, Im GJ, Yu S, Kim JS (2020) Neuro-Behçet’s Disease Presenting as Isolated Vestibular Syndrome. J Clin Neurol 16: 499-501. doi: 10.3988/jcn.2020.16.3.499
Shakeri A (2018) Hulusi Behcet-A Life of Passion and Endeavor in Dermatology. JAMA Dermatol 154: 440. doi: 10.1001/jamadermatol.2017.6364
Tan SY, Poole PS (2016) Hulusi Behcet (1889-1948): Passion for dermatology. Singapore Med J 57: 408-9. doi: 10.11622/smedj.2016123
Verity DH, Marr JE, Ohno S, Wallace GR, Stanford MR (1999) Behcet’s disease, the Silk Road and HLA-B51: historical and geographical perspectives. Tissue Antigens 54: 213-20. doi: 10.1034/j.1399-0039.1999.540301.x
White AS, Taylor RL, McNeill C, Garsia R, Welgampola MS (2014) Behçet’s disease presenting as a peripheral vestibulopathy. J Clin Neurosci 21: 1060-3. doi: 10.1016/j.jocn.2013.08.021
![]()