By Marcello Cherchi, MD PhD
For patients
An autoimmune disease is a disorder in which the body’s immune system mistakenly attacks part of the body. There are many autoimmune disorders. Occasionally, an autoimmune disease may cause problems with hearing or balance. If your doctor suspects that an autoimmune disease is the cause of hearing or balance problems, then they may consider checking tests of hearing, balance, blood tests, imaging studies, and others.
For clinicians
Overview
As a group, autoimmune disorders can affect any organ system. Specific autoimmune diseases may have a predilection for one or another body system, and manifest in a particular fashion. Most autoimmune diseases do not involve audio-vestibular system, but some can.
Introduction
Numerous articles have been written on the general relationships between autoimmunity and the audiovestibular system (Agrup 2008; Barna and Hughes 1988; Bovo et al. 2010; Girasoli et al. 2018; Russo et al. 2018). Our intention in this section is not to provide an exhaustive review of this subject, but rather to give an overview of several autoimmune diseases with overlapping presentations that may be referred to an otoneurologist or neuro-otologist.
Pathophysiological mechanism of disease
The underlying immunological mechanisms of diseases that can affect the otovestibular system are generally poorly understood. There does not seem to be any one-to-one relationship between, say, a specific gene, a specific HLA subtype, or a specific autoantibody, and a particular diagnosis. This is one reason why most investigators view these diseases as the result of several interacting factors, such as genetic predisposition, immune system profile, and environmental exposure. This viewpoint is illustrated in the Figure below from Bovo and colleagues.
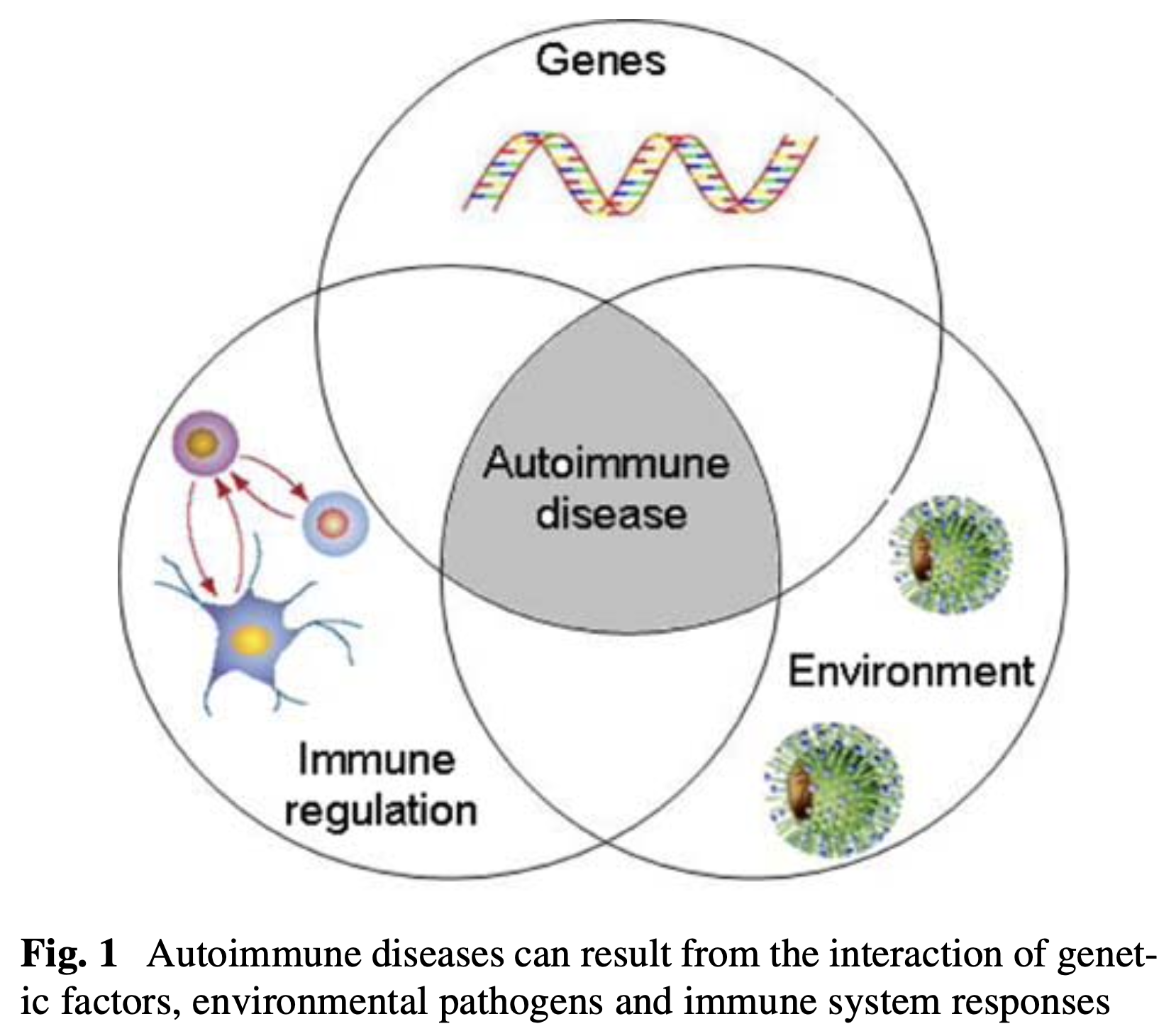
In the Table below we juxtapose several representative diseases. Many others have been omitted, such as rheumatoid arthritis, various autoimmune vasculidites, Alport syndrome, thyroid autoimmune disease, multiple sclerosis, paraneoplastic syndromes, hypertrophic cranial pachymeningitis, antiphospholipid antibody syndrome, anti-cardiolipin antibody syndrome, Ig4-related disease, relapsing polychondritis, ulcerative colitis and others. We have also omitted diseases for which there is uncertainty whether an autoimmune mechanism is involved at all, such as Ménière’s disease, sudden-onset sensorineural hearing loss, delayed endolymphatic hydrops, and others.
|
Diagnosis |
Audio-vestibular features |
Ophthalmological features |
Other features |
|
Auditory: Usually bilateral sensorineural hearing loss that is rapidly progressive (weeks to months). Vestibular: Vestibular symptoms are inconsistently present. Can cause vestibular weakness. |
None. |
Clinically defined by steroid-responsive hearing loss. |
|
|
Auditory: Sensorineural hearing loss that can be unilateral or bilateral, often affects low and middle more than high frequencies, and can fluctuate. Tinnitus often present. Vestibular: Workup may show unilateral weakness. |
Vision loss. Fluorescein angiography shows branch retinal artery occlusion. |
Mechanism: CD-8 T-cell mediated retinocochleocerebral vasculopathy, preferentially affecting precapillary arterioles. Neurological symptoms: headache, encephalopathy. Demographics: women>men, peak onset around 30 years (range 14 – 63 years). Imaging: MRI shows T2/FLAIR lesions in the corpus callosum, periventricular, thalamic and internal capsular regions. |
|
|
Auditory: Hearing loss, tinnitus, aural symptoms. Audiometry usually shows high frequency sensorineural hearing loss. Less commonly can show low frequency hearing loss. Hearing loss may fluctuate. Vestibular: Disequilibrium, which may be synchronous or metachronous with the auditory symptoms. Workup may or may not show evidence of vestibular weakness. |
Uveitis. |
Other clinical features: Recurrent oral and genital ulcers, and other dermatologic changes. Mechanism: immunologically mediated vasculopathy (venulitis > arteritis). Demographics: More common in peoples of the geographical stretch from East Asia to the Mediterranean basin. Onset usually in 4th or 5thdecades. |
|
|
Auditory: Hearing loss, may be abrupt in onset or gradually progressive. Audiometry usually shows high > low frequency sensorineural hearing loss. Less commonly there is primarily low frequency hearing loss. The hearing loss may fluctuate. ECoG may show elevated SP/AP ratio. Vestibular: Disequilibrium. Vestibular workup may show unilateral or bilateral vestibular weakness. |
Typical Cogan syndrome: interstitial keratitis. Atypical Cogan syndrome: scleritis, episcleritis, uveitis, retinal vasculitis. |
Other clinical features: Multiple organ systems may be involved, including musculoskeletal, abdominal, cardiac, genitourinary, cutaneous. Mechanism: Vasculitis. Imaging: MRI may show T1 hyperintensity of the labyrinths. |
|
|
Auditory: Hearing loss, tinnitus. Vestibular: Disequilibrium. Workup may show unilateral or bilateral vestibular weakness. |
Panuveitis. Optical coherence tomography eventually shows decreased retinal and choroidal vessel density. |
Other clinical features: Poliosis (whitening of the eyelashes), vitiligo. Mechanism: T-cell mediated attack on melanocytes in multiple tissues (uvea, meninges, skin, labyrinth). Imaging: May show abnormalities in the eyes, meninges, labyrinth, brain, spinal cord. |
|
|
Auditory: Hearing loss. Audiometry usually shows high frequency sensorineural hearing loss. Vestibular: Disequilibrium. Workup may show vestibular weakness. |
Xerophthalmia (dry eyes) from keratoconjunctivitis sicca. |
Other clinical features: Xerostomia (dry mouth), peripheral somatosensory neuropathy, autonomic neuropathy. Mechanism: Autoimmune-mediated exocrinopathy. Demographics: Very common (0.1% – 4.8% depending on geographic region). Significantly more common in women. |
|
|
Auditory: Hearing loss, variably present. Audiometry usually shows high frequency sensorineural hearing loss; this can be unilateral or bilateral (symmetric or asymmetric). Vestibular: Disequilibrium, variably present. Workup may show vestibular weakness. Some patients additionally develop a cerebellar ataxia. |
Xerophthalmia (dry eyes) from kerato-conjunctivitis sicca; anterior uveitis; episcleritis; posterior scleritis; papillary edema; inflammatory optic neuritis (Kedia et al. 2022). |
Other clinical features: Any organ system can be involved, so clinical presentation is variable. Mechanism: Uncertain. Affectation of the labyrinth may involve deposition of immune complexes. |
|
|
Auditory: Hearing loss (which may be abrupt in onset, or gradually progressive), tinnitus. Audiometry may show unilateral or bilateral (usually asymmetric) sensorineural hearing loss. Vestibular: Disequilibrium. May be abrupt in onset, or gradually progressive. Workup may show vestibular weakness (unilateral or bilateral). |
Uveitis, optic neuropathy (Seve et al. 2020) and optic neuritis (Yates et al. 2022). |
Mechanism: Caseating granulomas can develop in any organ system. |
|
|
Auditory: Hearing loss is variably present; it is usually (though not always) bilateral, can occur in any pattern (sensorineural, conductive or mixed), and any tempo (abrupt onset or progressive). Some patients also develop tinnitus. Vestibular: Disequilibrium is uncommon. |
Conjunctivitis, corneal ulceration, scleritis, episcleritis, uveitis, retinal vasculitis, optic neuropathy. |
Mechanism: Vasculitic damage and formation of granulomas. |
Table 1: Comparison of several autoimmune diseases that can affect the auditory and vestibular systems.
Testing: auditory and vestibular
No profile of audiologic findings is sensitive or specific for any autoimmune-mediated audio-vestibular disorder.
These autoimmune diseases are relatively uncommon. Having an autoimmune disease (including those listed here) does not prevent a person from developing more common audio-vestibular disorders. Therefore, even when a patient confidently diagnosed with one of these disorders is referred to a vestibular medicine clinic, it is medically reasonable to undertake a screening audio-vestibular workup to evaluate for more common causes of these symptoms (e.g., labyrinthitis, vestibular neuritis, benign paroxysmal positional vertigo, etc.).
Testing: other
Workup of these diseases is usually overseen by a rheumatologist. Initial laboratory tests suggested by Agrup (Agrup 2008) include:
- Erythrocyte sedimentation rate and c-reactive protein
- Serum immunoglobulins (IgM, IgG, IgA, IgE)
- Anti-thyroid antibodies (anti-thyroglobulin, anti-thyrotropin)
- Anti-nuclear antibodies (ANA)
- Anti-neutrophil cytoplasmic antibodies (ANCA)
- Anti-endothelial cell antibodies (AECA)
- Anti-phospholipid/anti-cardiolipin antibodies
Imaging
Imaging, such as MRI of the brain and internal auditory canals without and with contrast, may provide evidence to support or refute autoimmune involvement of otovestibular and neurologic structures.
Management
Management of these autoimmune disorders is undertaken by a rheumatologist, sometimes working with subspecialists in the various organ systems affected (e.g., ophthalmology, otolaryngology, neurology, dermatology).
Prognosis
Prognosis depends on the specific autoimmune disorder.
References
Agrup C (2008) Immune-mediated audiovestibular disorders in the paediatric population: a review. Int J Audiol 47: 560-5. doi: 10.1080/14992020802282268
Barna BP, Hughes GB (1988) Autoimmunity and otologic disease: clinical and experimental aspects. Clin Lab Med 8: 385-98.
Bovo R, Ciorba A, Martini A (2010) Vertigo and autoimmunity. Eur Arch Otorhinolaryngol 267: 13-9. doi: 10.1007/s00405-009-1122-5
Girasoli L, Cazzador D, Padoan R, Nardello E, Felicetti M, Zanoletti E, Schiavon F, Bovo R (2018) Update on Vertigo in Autoimmune Disorders, from Diagnosis to Treatment. J Immunol Res 2018: 5072582. doi: 10.1155/2018/5072582
Kedia N, Theillac V, Paez-Escamilla M, Indermill C, Gallagher DS, Adam R, Qu-Knafo AL, Amari F, Bottin C, Chotard G, Caillaux V, Streho M, Sedira N, Heron E, Becherel PA, Bodaghi B, Mrejen-Uretski S, Sahel AJ, Saadoun D, Errera MH (2022) The full range of ophthalmological clinical manifestations in systemic lupus erythematosus. Front Ophthalmol (Lausanne) 2: 1055766. doi: 10.3389/fopht.2022.1055766
Russo FY, Ralli M, De Seta D, Mancini P, Lambiase A, Artico M, de Vincentiis M, Greco A (2018) Autoimmune vertigo: an update on vestibular disorders associated with autoimmune mechanisms. Immunol Res. doi: 10.1007/s12026-018-9023-6
Seve P, Jamilloux Y, Tilikete C, Gerfaud-Valentin M, Kodjikian L, El Jammal T (2020) Ocular Sarcoidosis. Semin Respir Crit Care Med 41: 673-688. doi: 10.1055/s-0040-1710536
Yates WB, McCluskey PJ, Fraser CL (2022) Neuro-ophthalmological manifestations of sarcoidosis. J Neuroimmunol 367: 577851. doi: 10.1016/j.jneuroim.2022.577851
![]()