By Marcello Cherchi, MD PhD
For patients
The phrase “internuclear ophthalmoplegia” (abbreviated “INO”) refers to a specific problem with eye movements that happens when certain diseases affect a particular part of the brainstem. Many diseases can cause an INO.
For clinicians
Overview
Internuclear ophthalmoplegia (INO) refers to the ocular motor disorder resulting from a lesion in the medial longitudinal fasciculus (interfering with excitatory projections to the ipsilateral oculomotor nucleus from the contralateral abducens nucleus) in which, for saccades directed contraversive to that lesion, the eye ipsilateral to the lesion fails to adduct fully, whereas the eye contralateral to the lesion abducts fully but then may exhibit abducting nystagmus. (This deficit does not affect saccades directed ipsiversive to the lesion, and does not affect vergence movements.) This dysconjugate eye movement can manifest clinically with intermittent diplopia, and some patients experience this visual deficit as interfering with their equilibrium. INO can be unilateral or bilateral. The most common causes of INO in younger patients include demyelination, and in older patients include infarction. MRI is the preferred imaging modality for identifying lesions of the medial longitudinal fasciculus.
Introduction
Internuclear ophthalmoplegia (INO) refers to the ocular motor disorder resulting from a lesion in the medial longitudinal fasciculus in which, for saccades directed contraversive to that lesion, the eye ipsilateral to the lesion fails to adduct fully, whereas the eye contralateral to the lesion abducts fully but then may exhibit abducting nystagmus.
Pathophysiological mechanism of disease
In the phrase “internuclear ophthalmoplegia,” the term “internuclear” refers to the fact that one of the pathways in the medial longitudinal fasciculus lies between two nuclei relevant to ocular motor function; specifically, the pathway begins in the abducens (CN VI) nucleus, decussates and ascends to the oculomotor (CN III) nucleus.
The Figure below, from Frohman and colleagues (Frohman et al. 2008), depicts the relevant neuroanatomy.
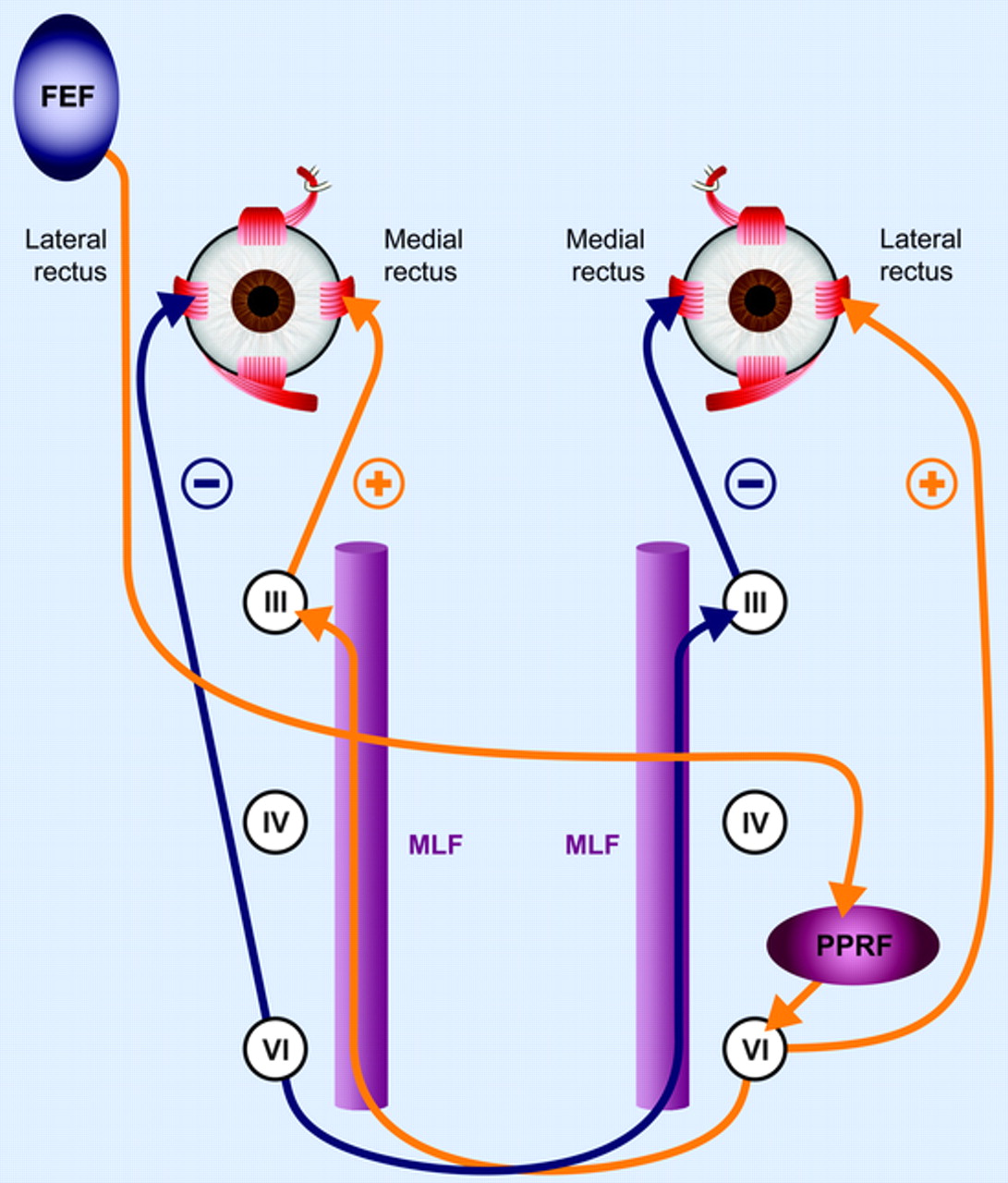
Figure : Neuroanatomical substrate for a leftward-directed saccade (Frohman et al. 2008). Excitatory pathways are shown in orange and the reciprocal inhibitory pathways are shown in blue. The particular pathway shown emanates from the frontal eye field (FEF), which projects through the anterior limb of the internal capsule, decussates to the opposite side at the midbrain-pontine junction, and then innervates the paramedian pontine reticular formation (PPRF). From there, projections directly innervate the lateral rectus (ipsilateral to the PPRF). A second decussation, back to the side of origin of FEF activation, via the MLF, innervates the medial rectus subnucleus of cranial nerve III and then neurons here project to innervate the medial rectus muscle. The right FEF command to trigger a saccade culminates in conjugate eye movements to the left. According to Herring’s law, the horizontal yoke pair, the medial and lateral recti, are activated in synchrony.
When executing a voluntary saccade directed towards the patient’s left, the normal physiology is as follows:
- The right frontal eye field projects to the left abducens nucleus.
- The left abducens nucleus sends an excitatory projection to the left lateral rectus muscle, resulting in abduction of the left eye. (There is a separate inhibitory pathway for the left medial rectus.)
- The left abducens nucleus also sends a projection that decussates, ascends through the right medial longitudinal fasciculus, and synapses in right the oculomotor nucleus.
- The right oculomotor nucleus projects to the right medial rectus, resulting in adduction of the right eye. (There is a separate inhibitory pathway for the right lateral rectus.)
(This anatomy is reversed for saccades directed towards the patient’s right.)
In a unilateral right-sided INO, the following will occur with attempted leftward saccades:
- A lesion in the right medial longitudinal fasciculus (step 3) will result in failure of step 4, and the right eye will not fully adduct (move leftward).
- The brain keeps attempting to complete the leftward saccade by sending additional excitation from the left abducens nucleus. By Hering’s law of equal innervation (Hering 1868, 1879, 1942), these additional excitatory signals are projected from the left abducens nucleus to the right oculomotor nucleus (but fail to reach that destination because the pathway through the right medial longitudinal fasciculus is lesioned) and also project from the left abducens nucleus to the left lateral rectus — which is successful, but results in abduction nystagmus of the left eye (Zee et al. 1987).
In a unilateral right-sided INO, rightwardly-directed saccades are unaffected.
(In a unilateral left-sided INO, the above descriptions would be reversed.)
INO usually leaves convergence movements (which are mediated by different pathways) intact (Zee et al. 1987).
Clinical presentation
While internuclear ophthalmoplegia might not be considered a “vestibular” disorder, its ocular motor effects interfere with vision, resulting in intermittent diplopia, and can be experienced by some patients as a form of disequilibrium.
Ocular motor examination
Unilateral or bilateral INO can often be identified on face-to-face ocular motor examination, though may evade detection if subtle.
Testing: instrumented ocular motor testing
The Figure below, from Omary and colleagues (Omary et al. 2023), shows ocular motor plots of eye position and velocity, and an MRI demonstrating a lesion involving the right medial longitudinal fasciculus.
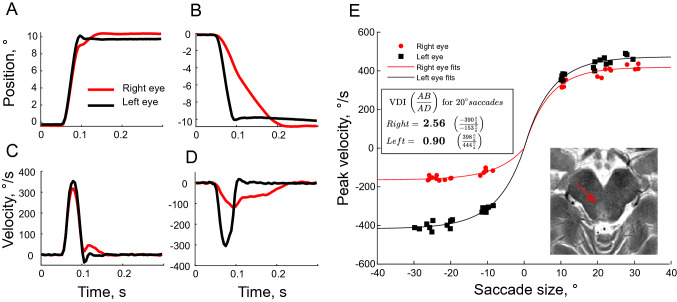
Figure : Ocular motor plots of eye position and velocity, and an MRI demonstrating a lesion involving the right medial longitudinal fasciculus (Omary et al. 2023). Saccadic eye movements from a patient with right INO. Saccadic eye movements and summary results of a patient with right INO. Panel (A) 10° rightward saccade, with peak velocities around 350°/s. Panel (C): Both eyes show a similar velocity profile for this saccade. Panel (B): 10° leftward saccade, with velocity shown in Panel (D). The left abducting eye (black) moves faster and reaches the target earlier than the right adducting eye (red), indicating a right INO. Panel (E): Peak eye velocity is plotted against saccade size for all trials. For rightward (positively directed) saccades, both eyes have approximately the same peak velocity, but for leftward saccades, the right adducting eye (red) moves with a peak velocity of less than half that of the left abducting eye (black). We defined the versional disconjugacy index (VDI) as the ratio of the peak velocity of abducting to adducting eyes, for 20° saccades based on exponential fits, resulting in an increased VDI of 2.56 for saccades to the left (indicating a right INO) and a normal VDI of 0.9 to the right. The inset shows the corresponding MR image from this patient, where all three of our raters diagnosed a lesion of the right MLF in the brainstem.
The Video below shows the ocular motor video and associated tracings for a 56-year-old lady with bilateral INO from multiple sclerosis.
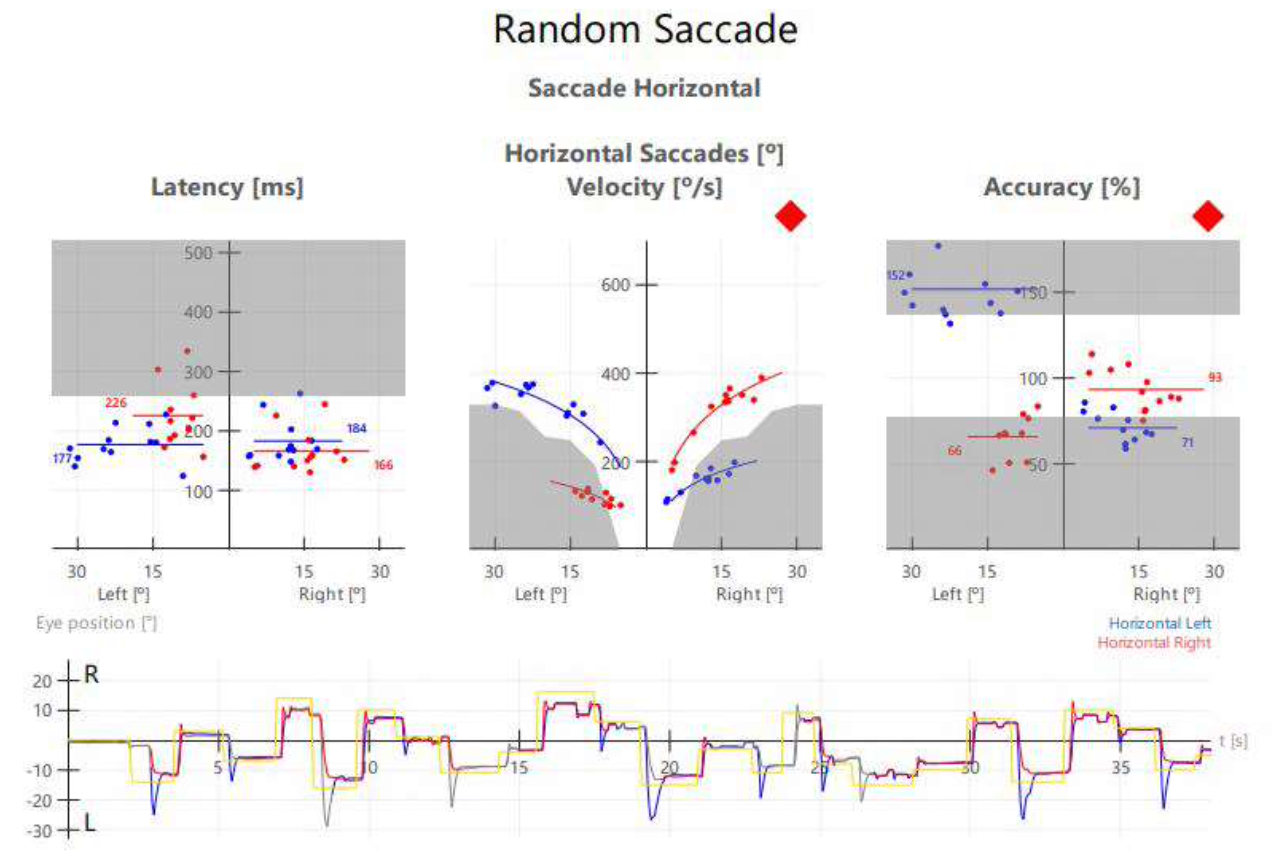
The Figure below shows binocular oculography of horizontal saccades in the patient with bilateral INO.
- The leftmost panel shows that latencies for horizontal saccades are normal in both eyes.
- The middle panels shows plots of horizontal saccade velocity; on rightward saccades the velocities for the abducting right eye (red) are normal, while the velocities for the adducting left eye (blue) are diminished; on leftward saccades the velocities for the abducting left eye are normal, while the velocities for the adducting right eye are diminished.
- The rightmost panel shows plots of horizontal saccadic accuracy; on rightward saccades the abducting right eye (red) is eumetric, while the adducting left eye (blue) is hypometric; on leftward saccades the abducting left eye is hypermetric and the adducting right eye is hypometric.
- The bottom panel shows a binocular tracing. On rightward saccades (represented by an upward deflection) the right eye (red tracing) is hypermetric; on leftward saccades (represented by a downward deflection) the left eye (blue tracing) is hypermetric.
Testing: vestibular
The ocular motor consequences of INO can interfere with vestibular tests that rely on ocular motor function, such as video head impulse testing.
INO can also interfere with results of ocular vestibular evoked myogenic potentials (Rosengren and Colebatch 2011).
Imaging
MRI is the imaging modality of choice to detect lesions involving the medial longitudinal fasciculus from etiologies such as infarction, demyelination, hemorrhage and neoplasm.
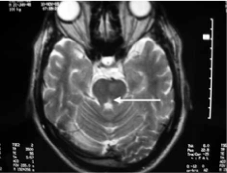
The Figure below, from Deleau and colleagues (Deleu et al. 2005), shows a focal area of high T2 signal in the left medial longitudinal fasciculus, attributed to ischemic infarction.
Differential diagnosis
The differential diagnosis of INO includes any disease process that can lesion the medial longitudinal fasciculus. In younger patients the most common etiology is demyelination, typically from multiple sclerosis. In older patients the most common etiology is infarction (Kim 2004). Less common etiologies include other autoimmune diseases (such as sarcoidosis, systemic lupus erythematosus), infections, neoplasm, head trauma, and others. It can also occur in the context of Wernicke encephalopathy.
Treatment
Treatment of the underlying etiology of INO is appropriate. For example, if the underlying etiology is an ischemic infarct, then secondary stroke prophylaxis is appropriate; if the underlying etiology is from multiple sclerosis, then treatment for that diagnosis may be advanced.
In addition, if (despite such treatments) the visual deficit persists, then consultation with neuro-ophthalmology or neuro-optometry may be appropriate.
Prognosis
The prognosis of INO depends on the underlying etiology.
Eggenberger and colleagues (Eggenberger et al. 2002) state that, “most ischemic-based INO become asymptomatic in primary position over 2 to 3 months.”
Kanhai and colleagues report a prospective, randomized, placebo-controlled trial of patients with internuclear ophthalmoplegia from multiple sclerosis, and reported improvement in certain INO parameters (such peak velocity) in the treatment group (Kanhai et al. 2019).
References
Deleu D, Sokrab T, Salim K, El Siddig A, Hamad AA (2005) Pure isolated unilateral internuclear ophthalmoplegia from ischemic origin: report of a case and literature review. Acta Neurol Belg 105: 214-7.
Eggenberger E, Golnik K, Lee A, Santos R, Suntay A, Satana B, Vaphlades M, Stevens C, Kaufman D, Wall M, Kardon R (2002) Prognosis of ischemic internuclear ophthalmoplegia. Ophthalmology 109: 1676-8. doi: 10.1016/s0161-6420(02)01118-1
Frohman TC, Galetta S, Fox R, Solomon D, Straumann D, Filippi M, Zee D, Frohman EM (2008) Pearls & Oy-sters: The medial longitudinal fasciculus in ocular motor physiology. Neurology 70: e57-67. doi: 10.1212/01.wnl.0000310640.37810.b3
Hering KEK (1868) Die lehre vom binocularen sehen. Engelmann, Leipzig
Hering KEK (1879) Der Raumsinn und die Bewegung des Auges. In: Hermann L (ed) Handbuch der Physiologie, vol III, part 1, Leipzig, pp 343-601
Hering KEK (1942) Spatial Sense and Movements of the Eye. American Academy of Optometry, Baltimore
Kanhai KMS, Nij Bijvank JA, Wagenaar YL, Klaassen ES, Lim K, Bergheanu SC, Petzold A, Verma A, Hesterman J, Wattjes MP, Uitdehaag BMJ, van Rijn LJ, Groeneveld GJ (2019) Treatment of internuclear ophthalmoparesis in multiple sclerosis with fampridine: A randomized double-blind, placebo-controlled cross-over trial. CNS Neurosci Ther 25: 697-703. doi: 10.1111/cns.13096
Kim JS (2004) Internuclear ophthalmoplegia as an isolated or predominant symptom of brainstem infarction. Neurology 62: 1491-6. doi: 10.1212/01.wnl.0000123093.37069.6d
Omary R, Bockisch CJ, De Vere-Tyndall A, Pazahr S, Barath K, Weber KP (2023) Lesion follows function: video-oculography compared with MRI to diagnose internuclear ophthalmoplegia in patients with multiple sclerosis. J Neurol 270: 917-924. doi: 10.1007/s00415-022-11428-w
Rosengren SM, Colebatch JG (2011) Ocular vestibular evoked myogenic potentials are abnormal in internuclear ophthalmoplegia. Clin Neurophysiol 122: 1264-7. doi: 10.1016/j.clinph.2010.10.040
Zee DS, Hain TC, Carl JR (1987) Abduction nystagmus in internuclear ophthalmoplegia. Ann Neurol 21: 383-8. doi: 10.1002/ana.410210411
![]()