By Marcello Cherchi, MD PhD
For patients
In superficial siderosis, a very slow bleed into the brain results in accumulation of iron on the surface of the brain, cranial nerves and spinal cord. This can cause symptoms of hearing loss and imbalance that usually start between 40 – 70 years of age and progress very slowly. In order to distinguish this disease from others that it may closely resemble, your doctor may consider checking several tests of balance function and a brain MRI. If a source of bleeding is identified then this should be repaired, but often no source of bleeding is found.
For clinicians
Overview
In superficial siderosis (SS) chronic slow bleeding into cerebrospinal fluid leads to iron deposition along the surfaces of the brain, brainstem and cranial nerves. This results in neuronal damage to which the vestibulocochlear nerve and cerebellum are selectively vulnerable. The peak age of symptom onset in SS is the fifth through seventh decades, and men are affected more than women by a ratio of 3:1. Common presenting symptoms include bilateral hearing loss and impaired gait. Less common symptoms include cognitive deficits, anosmia, extraocular muscle palsies, sensory abnormalities in the lower extremities and urinary incontinence. Physical examination shows cerebellar findings of appendicular and gait ataxia. Ocular motor examination may show positive bedside head impulse test bilaterally. Audiologic examination shows bilateral sensorineural hearing loss, usually more pronounced in the high frequencies, which may be asymmetrical. On instrumented vestibular testing, videonystagmography shows caloric weakness (which may be asymmetrical); video head impulse testing shows low gain with compensatory saccades; rotatory chair testing may show poor visual suppression of the vestibulo-ocular reflex, the presence of saccadic breakdown of smooth pursuit, and poor optokinetic responses. Cerebrospinal fluid may show xanthochromia, red blood cells and elevated protein. Brain MRI is the imaging modality of choice; T2 imaging shows dark outlines along the surfaces of neural structures such as the vestibulocochlear nerve, brainstem and cerebellar folia. If a source of bleeding can be identified it is medically reasonable to repair, but it is unknown whether this changes the symptom trajectory of SS.
Introduction
Superficial siderosis, sometimes also called superficial hemosiderosis, is a condition in which chronic, slow bleeding into the cerebrospinal fluid of the subarachnoid space results in deposition of iron in the leptomeninges and subjacent gray matter, with a predilection for the cerebellum (particularly the vermis) and certain cranial nerves (cochleovestibular and olfactory).
Dr. Ralph Hamill (1877 – 1961) was the first to describe the gross pathological pigmentary characteristics of what eventually became known as superficial siderosis (Hamill 1908). The German pathologist, Dr. H. Hoetzel (Noetzel 1940) was the first to connect these pigmentary changes to subarachnoid hemorrhage (Fearnley et al. 1995).
Epidemiology
Symptomatic superficial siderosis is very uncommon (Fearnley et al. 1995), though the precise incidence and prevalence are unknown. Incidental discovery of superficial siderosis (by imaging or at autopsy) in patients without corresponding symptoms suggests that superficial siderosis itself may not be rare.
The Figure below, from Fearnley and colleagues (Fearnley et al. 1995), shows a histogram of the age distribution of symptom onset in patients with superficial siderosis. Symptom onset peaks in the 5th – 7th decades.
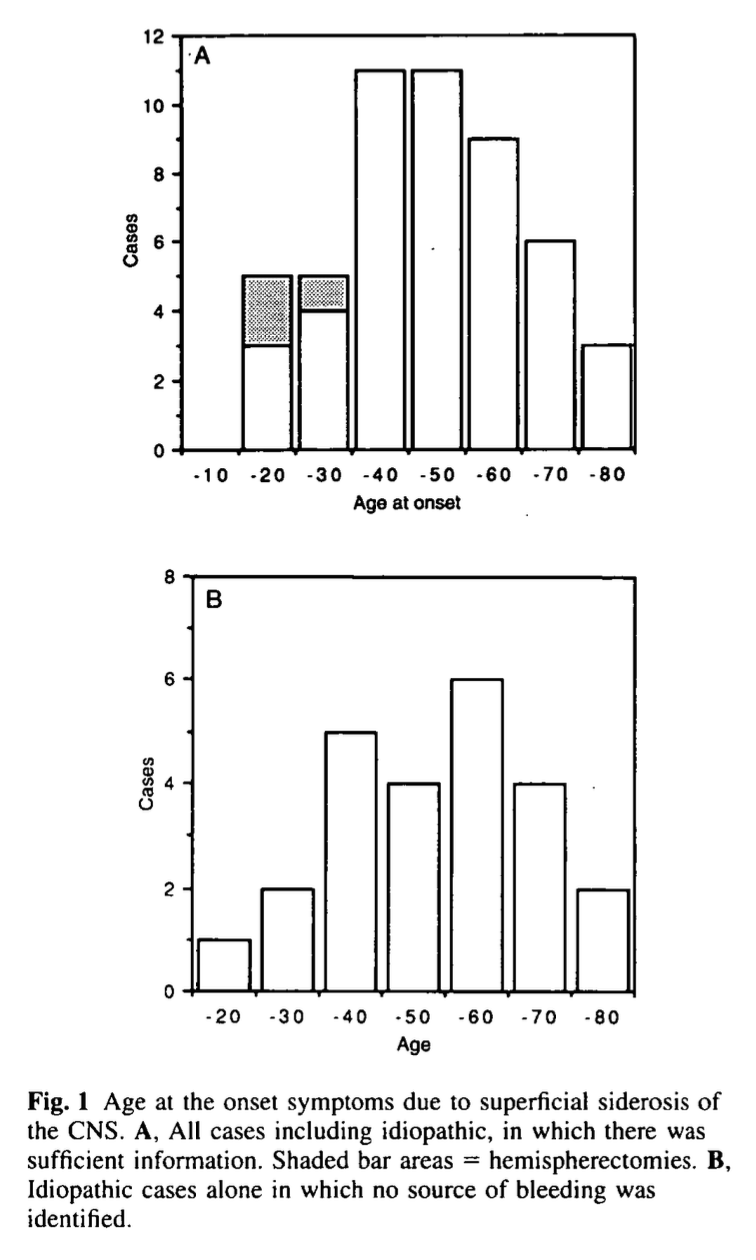
Fearnley and colleagues (Fearnley et al. 1995) cite a male-to-female predominance at a ratio of 3:1.
Pathophysiological mechanism of disease
When extravasated blood enters the subarachnoid space, its deoxyhemoglobin gets metabolized to iron (which the brain cannot conjugate with ferritin), and thus it gets deposited as insoluble hemosiderin in tissues with which it comes into contact (Aran Yoo and Kattah 2017). The vulnerable tissues are those in close proximity to cerebrospinal fluid, which includes those in the subpial and subependymal spaces (Fearnley et al. 1995). This unbound iron induces free radical formation and lipid peroxidation, resulting in tissue damage (Aran Yoo and Kattah 2017).
Fearnley and colleagues (Fearnley et al. 1995) comment that:
“There have been several cases in which superficial siderosis was found at autopsy in the absence of symptoms during life and it seems likely that there is a pre-symptomatic phase to the illness during which superficial siderosis is present, but not sufficient to cause symptoms” (Fearnley et al. 1995).
How long does this pre-symptomatic phase last? Fearnley and colleagues add:
“An attempt to calculate the duration of this phase in symptomatic cases can be made by taking the interval from the onset of bleeding to the onset of symptoms referable to superficial siderosis… The pre-symptomatic phase in these cases ranged from 4 months to 30 years with a mean of 15 years” (Fearnley et al. 1995)
Koeppen and colleagues state that, “Only CNS [central nervous system] tissues will convert heme in CSF to hemosiderin, and some tissues, such as the cerebellar cortex and the eight cranial nerves, are especially vulnerable” (Koeppen et al. 2008). Why this selective vulnerability?
With regard to the apparent selective vulnerability of the vestibulocochlear nerve in superficial siderosis, Feanley and colleagues state:
“The fact that the VIIIth nerve is particularly vulnerable is due to its having a long glial segment. This results in greater length of haemosiderin deposition and, thereby, a greater chance of axonal damage. Another important factor is that the VIIIth nerve runs through the pontine cistern, which not only contains a large pool of CSF [cerebrospinal fluid] but also has a greater flow of CSF. This potentially delivers a larger amount of iron and haem causing a correspondingly greater degree of neuronal damage” (Fearnley et al. 1995).
With regard to the apparent selective vulnerability of the cerebellum, Koeppen and colleagues state that, “The susceptibility of the cerebellar cortex is likely due to Bergmann glia that serve as conduits for heme; and the abundance of microglia” (Koeppen et al. 2008). This is apparently based on, “Animal studies [that] suggest that the ferritin repressor protein-immunoreactive Bergmann glia and ferritin-containing microglia in the molecular layer of the cerebellum are involved in conversion of heme to ferritin and finally hemosiderin” (Kumar et al. 2006).
Clinical presentation
Superficial siderosis affects multiple areas of the central nervous system, corresponding with multiple dysfunctions and symptoms.
In the 87 cases reviewed by Fearnley and colleagues (Fearnley et al. 1995) they reported that:
- Bilateral sensorineural hearing loss is the most common feature, occurring in (95%) of cases (Fearnley et al. 1995). The hearing loss begins insidiously and is gradually progressive.
- Cerebellar symptoms are almost as common as sensorineural hearing loss, occurring in 88% of cases (Fearnley et al. 1995).
- Other cranial nerves are less commonly affected, but the next most common is the olfactory nerve, manifesting with anosmia in as many as 17% of patients (Fearnley et al. 1995). Extraocular muscle palsies are relatively infrequent, occurring in 5% – 10% of cases (Fearnley et al. 1995)
- Cognitive impairment occurred in 24% of patients in one study (Fearnley et al. 1995), and in 40% of patients in another series (Kumar et al. 2006).
- Bilateral lower extremity paresthesias (Revesz et al. 1988) or somatosensory deficits may occur in 13% of patients (Fearnley et al. 1995).
- Urinary incontinence is less common, occurring in 7 (8%) of patients (Fearnley et al. 1995).
Physical examination
Most cases (88%) exhibit cerebellar findings of appendicular ataxia and gait ataxia (Fearnley et al. 1995).
A smaller proportion of cases (76%) exhibit pyramidal tract findings, and if present, these predominantly involve the lower extremities (Fearnley et al. 1995).
Ocular motor examination
Ocular motor testing occasionally shows nystagmus. This may consist of horizontal gaze-evoked nystagmus or spontaneous down beat nystagmus. Even if present, in some cases the nystagmus may only be detectable on instrumented oculography (Fearnley et al. 1995).
Bedside head impulse testing (Choi et al. 2014) may show a deficient vestibulo-ocular reflex gain, and the presence of compensatory saccades.
Testing: auditory
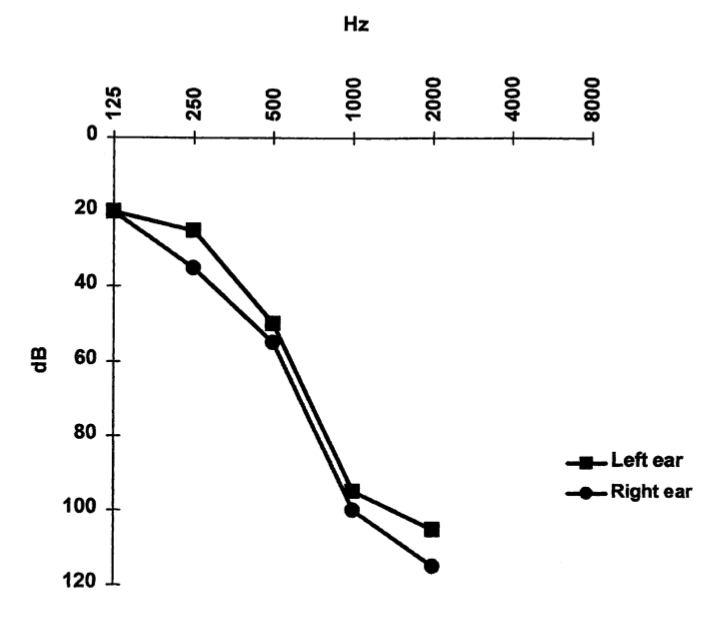
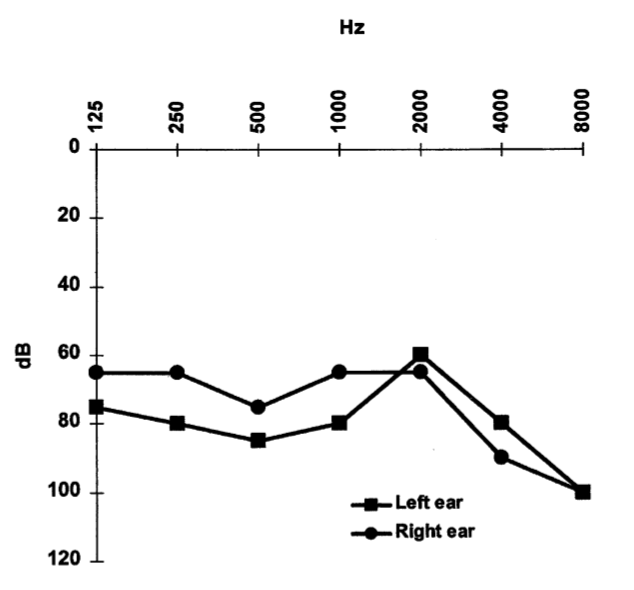
When Fearnley and colleagues reviewed the literature of 87 published cases of superficial siderosis they commented that, “In all cases with sufficient clinical details there was a sensori-neural [hearing] loss with the high tones being more severely affected” (Fearnley et al. 1995).
The sensorineural hearing loss on audiometry can be predominantly high frequency, or “across the board,” and may be modestly asymmetrical, as shown in the Figures below from Vibert and colleagues (Vibert et al. 2004).
 |
 |
 |
Testing: vestibular
When Fearnley and colleagues reviewed the literature of 87 published cases of superficial siderosis they lamented that, “In only four papers are vestibular tests reported” (Fearnley et al. 1995). This deficiency has unfortunately persisted in subsequent literature, with a few exceptions.
Vestibular studies in patients with superficial siderosis show evidence of bilateral vestibular weakness.
Several studies of videonystagmography in patients with superficial siderosis report caloric responses that are reduced or nearly absent (Choi et al. 2014; Revesz et al. 1988). In some cases the vestibular weakness may be asymmetrical (Vibert et al. 2004).
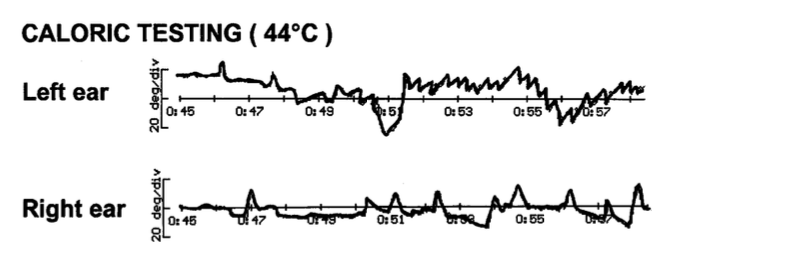
The Figure below, from Choi and colleagues (Choi et al. 2014), shows essentially absent bilateral caloric responses in a patient with superficial siderosis.
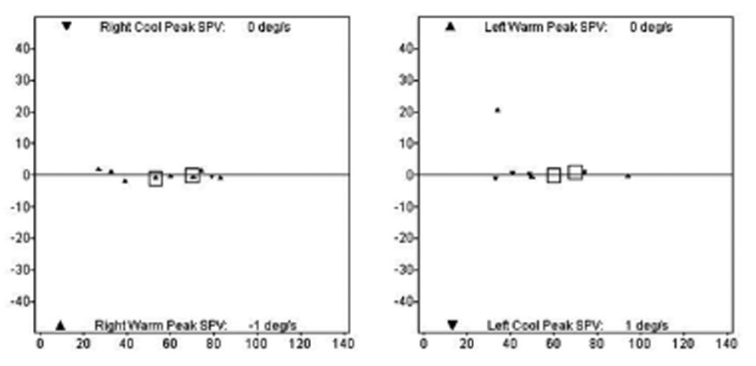
In some cases, the vestibular weakness may be asymmetrical. The Figure below, from Vibert and colleagues, demonstrates asymmetrical caloric responses, with a weaker response on the right.
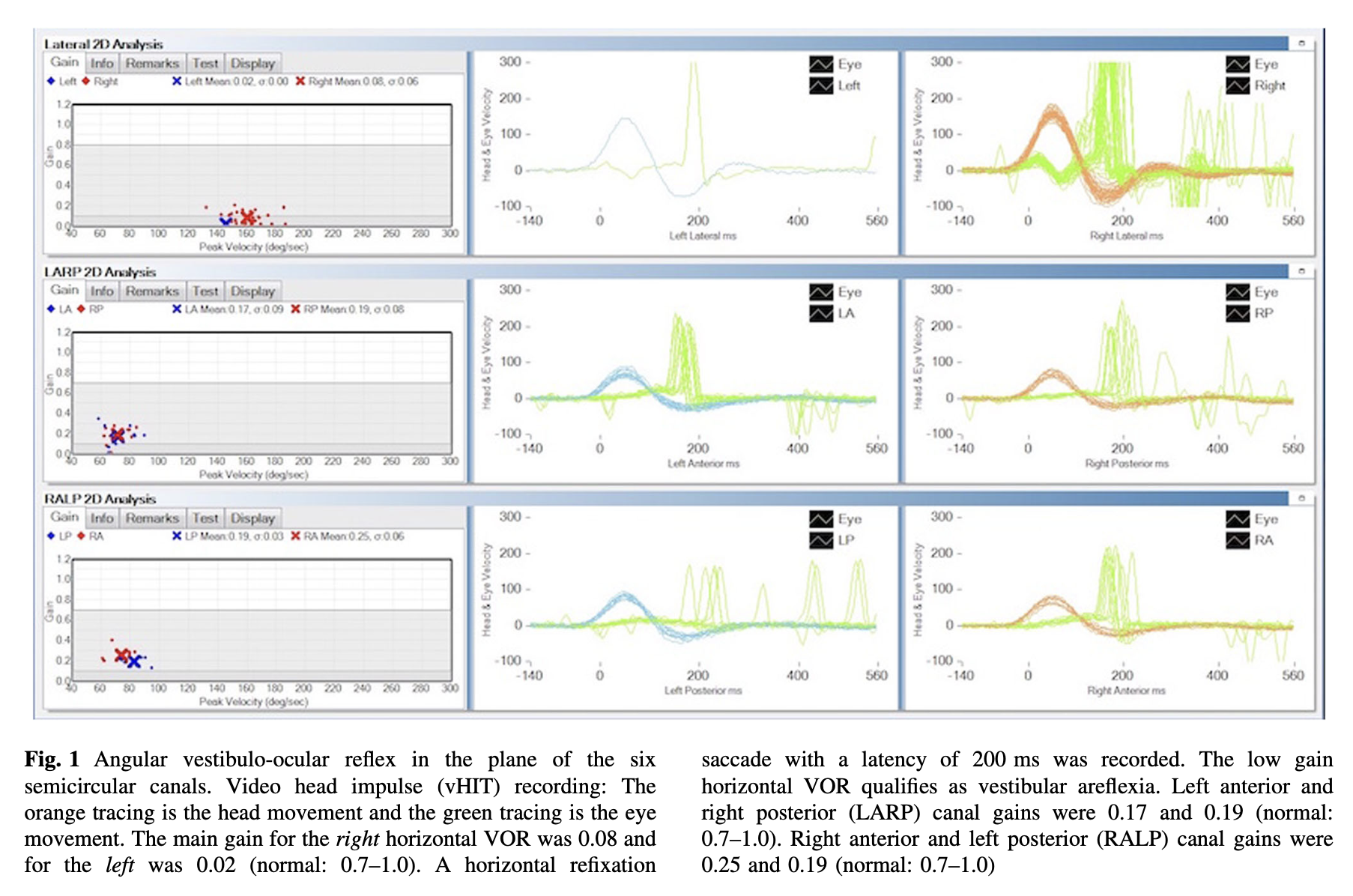
Several published studies of patients with superficial siderosis have documented bilateral vestibular weakness on video head impulse testing (Aran Yoo and Kattah 2017; Choi et al. 2014; Kang et al. 2015).
The Figure below, from Yoo and Kattah (Aran Yoo and Kattah 2017), shows dramatically reduced gain of the vestibulo-ocular reflex, and overt compensatory saccades, in all semicircular canals.
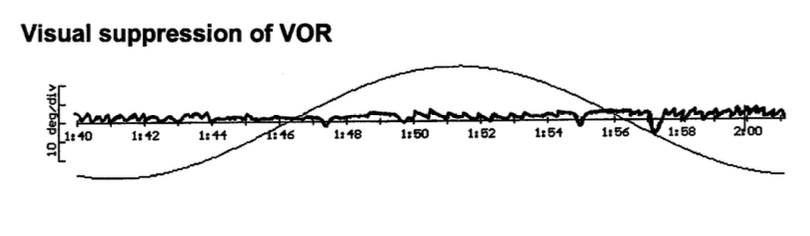
Instrumented oculography may also reveal abnormalities suggesting localization to the cerebellum, such as poor visual suppression of the vestibulo-ocular reflex, saccadic smooth pursuit and poor optokinetic responses.
The Figure below, from Vibert and colleagues (Vibert et al. 2004), shows incomplete visual suppression of the vestibulo-ocular reflex on rotatory chair testing, which suggests cerebellar dysfunction.
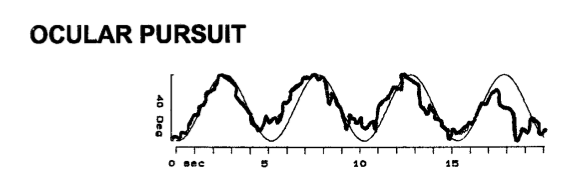
The Figure below, from Vibert and colleagues (Vibert et al. 2004), shows saccadic pursuit, which suggests cerebellar dysfunction.
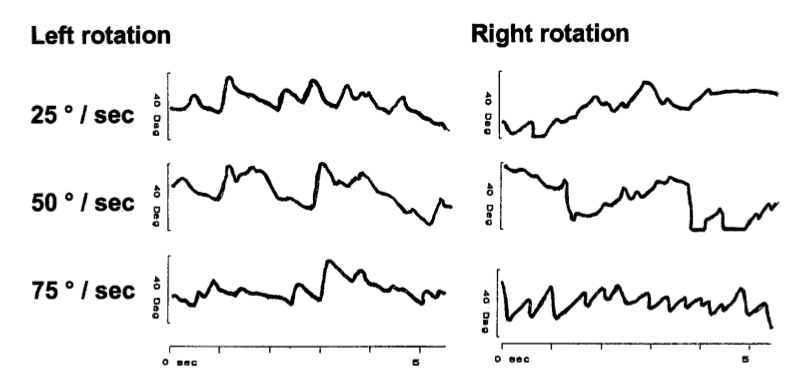
The Figure below, from Vibert and colleagues (Vibert et al. 2004), shows poor optokinetic responses (except to the rightward 75 deg/sec rightward stimulus), suggesting cerebellar dysfunction.
Testing: other
Cerebrospinal fluid shows several abnormalities in patients with superficial siderosis (Kumar et al. 2006). Feanley and colleagues summarize these as follows:
“Irrespective of headache, the cardinal CSF signs in superficial siderosis are haemorrhage and xanthochromia. Other features include the presence of erythrophages and siderophages and raised iron and ferritin levels. The protein may be elevated as a result of arachnoiditis and very occasionally a monoclonal or oligoclonal band may be present” (Fearnley et al. 1995).
Practically, given the high sensitivity of brain MRI for superficial siderosis (see below), lumbar puncture no longer plays a significant role in diagnosing this disease.
Imaging
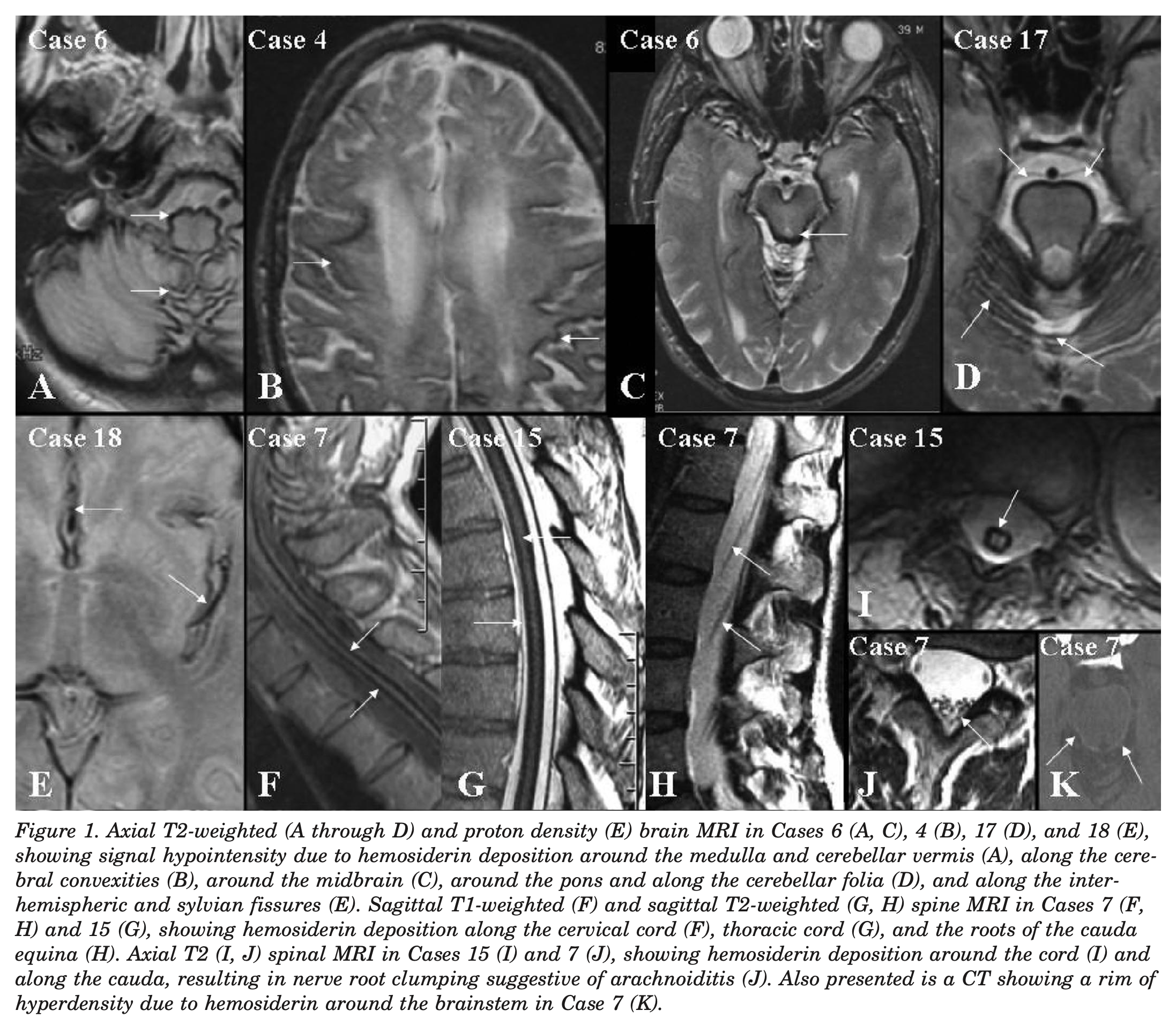
Brain MRI is the diagnostic imaging modality of choice (Fearnley et al. 1995) because of its very high sensitivity to hemosiderin (Kumar et al. 2006). “MRI reveals a rim of hypointensity on T2-weighted images, enveloping the surface of the brainstem, cerebellum, and sometimes the cortical fissures.” However, “Even with MRI, the diagnosis may still be missed because the imaging abnormalities follow the contours of the brain and can be overlooked” (Kumar et al. 2006).
The Figure below, from Kumar and colleagues (Kumar et al. 2006), shows such abnormalities involving the cerebrum, brainstem and spinal cord.
The Figure below, from Vibert and colleagues (Vibert et al. 2004), demonstrates to advantage involvement of the cerebellum by superficial siderosis.

Gross pathology and histopathology
Fearnley and colleagues summarize the gross pathology of superficial siderosis as follows:
“The pathology of superficial siderosis is characterized by marginal siderosis, granular ependymitis and in some cases an obstructive hydrocephalus. Macroscopically, there is a brownish discoloration of the leptomeninges, usually sparing the dura mater, and of the adjacent CNS parenchyma up to a depth of 3 mm (Rosenthal, 1958). Particularly pigmented regions were the cerebellum, especially the superior vermis and crests of the folia, basal frontal lobe, olfactory bulbs, temporal cortex, brainstem and cranial nerves (I, II, V, X and especially VIII), spinal cord and spinal nerve roots” (Fearnley et al. 1995).
With regard to histopathology they state, “Microscopically, there is haemosiderin deposition, neuronal loss, reactive gliosis, intra-cellular ovoid bodies and demyelination” (Fearnley et al. 1995). Koeppen and colleagues add, “In addition to perivascular collections of hemosiderin-laden macrophages, affected tissues displayed iron-positive anuclear foamy structures in the neuropil that resembled axonal spheroids. They were especially abundant in eighth cranial nerves and spinal cord” (Koeppen et al. 2008)
Differential diagnosis
Choi and colleagues state that, “When hearing impairment was associated, superficial hemosiderosis was the most common disorder in chronic bilateral combined vestibulopathy” (Choi et al. 2014).
Nevertheless, since each of the manifestations of superficial siderosis can occur in other diseases as well, the differential diagnosis includes:
- The associated hearing loss differentiates superficial siderosis from, for example, CANVAS and normal pressure hydrocephalus.
- The slowly progressive nature differentiates superficial siderosis from, for example, ototoxicity and autoimmune inner ear disease.
- The bilaterality of the hearing loss differentiates superficial siderosis from, for example, vestibular schwannoma or labyrinthitis.
Treatment
If a source of bleeding can be identified, this should be treated, though the long-term outcome of this is unknown. It is noteworthy that in a substantial minority of cases no bleeding source can be found; Koeppen and colleagues (Koeppen et al. 2008) review several studies documenting that in 30% – 46% of cases of superficial siderosis no source of bleeding remains undetermined.
Chelation therapy has been attempted in several cases, without success (Fearnley et al. 1995).
Practically, it is medically reasonable to refer patients to audiology to be evaluated for hearing amplification, and to vestibular rehabilitation therapy to be treated for bilateral vestibular weakness.
Prognosis
By the time a patient has developed symptoms, neuronal death has already occurred, and there are currently no therapies that can reverse this process.
Logically one would think that stopping any further bleeding into the subarachnoid space should retard progression, but this remains to be demonstrated, and would require longitudinal studies. Kumar and colleagues comment that, “It is unclear that elimination of the source of chronic bleeding slows or halts the clinical progression of this disorder… Judging clinical outcomes is difficult, given the slow progression of SS [superficial siderosis] and the possible episodic nature of the bleeding. It is unclear if the process continues despite eradication of the bleeding” (Kumar et al. 2006).
References
Aran Yoo BS, Kattah JC (2017) Superficial siderosis syndrome with progressive hearing loss and bilateral vestibular failure, 51 years after a neurosurgical procedure: diagnostic value of combined MRI and video head impulse test. J Neurol 264: 391-393. doi: 10.1007/s00415-016-8358-y
Choi SY, Kee HJ, Park JH, Kim HJ, Kim JS (2014) Combined peripheral and central vestibulopathy. J Vestib Res 24: 443-51. doi: 10.3233/VES-140524
Fearnley JM, Stevens JM, Rudge P (1995) Superficial siderosis of the central nervous system. Brain 118 ( Pt 4): 1051-66. doi: 10.1093/brain/118.4.1051
Hamill RC (1908) Report of a case of melanosis of the brain, cord and meninges. The Journal of Nervous and Mental Disease 35. doi: 10.1097/00005053-190809000-00027
Kang KW, Lee C, Kim SH, Cho HH, Lee SH (2015) Bilateral Vestibulopathy Documented by Video Head Impulse Tests in Superficial Siderosis. Otol Neurotol 36: 1683-6. doi: 10.1097/MAO.0000000000000865
Koeppen AH, Michael SC, Li D, Chen Z, Cusack MJ, Gibson WM, Petrocine SV, Qian J (2008) The pathology of superficial siderosis of the central nervous system. Acta Neuropathol 116: 371-82. doi: 10.1007/s00401-008-0421-z
Kumar N, Cohen-Gadol AA, Wright RA, Miller GM, Piepgras DG, Ahlskog JE (2006) Superficial siderosis. Neurology 66: 1144-52. doi: 10.1212/01.wnl.0000208510.76323.5b
Noetzel H (1940) Diffusion von Blutfarbstoff in der inneren Randzone und äußeren Oberfläche des Zentralnervensystems bei subarachnoidaler Blutung. Archiv für Psychiatrie und Nervenkrankheiten 111: 129-138. doi: 10.1007/BF01814021
Revesz T, Earl CJ, Barnard RO (1988) Superficial siderosis of the central nervous system presenting with longstanding deafness. J R Soc Med 81: 479-81.
Vibert D, Hausler R, Lovblad KO, Schroth G (2004) Hearing loss and vertigo in superficial siderosis of the central nervous system. Am J Otolaryngol 25: 142-9.
![]()