By Marcello Cherchi, MD PhD
For patients
Here are answers to some of the most common questions about rotatory chair testing.
|
What is this test? |
Rotatory chair testing assesses part of the balance system. |
|
What is this test looking for? |
This test examines eye movements during rotational movements of the body. |
|
Is this test experimental or investigational? |
This test is approved by the Food and Drug Administration. It is neither experimental nor investigational. |
|
What happens during this test? |
During this test you are seated in a secure chair that keeps the head and body still. The chair can rotate in different patterns. During the test you are wearing special goggles that monitor your eye movements. |
|
Is this test uncomfortable? |
Some patients experience dizziness during parts of this test. |
|
How long does this test take? |
The test takes about 60 minutes. |
|
Do I have to prepare for this test? |
You should avoid wearing eye makeup to this test. If possible, you should avoid taking meclizine (Bonine®) and dimenhydrinate (Dramamine®) for 24 hours prior to this test. There are no other specific preparations for this test. |
|
Are there any special instructions for what to do after the test? |
There are no special instructions for what to do after this test. |
For clinicians
Overview
Rotatory chair testing (RCT) is primarily useful as a detailed evaluation of the low- to middle- range of the vestibular tuning spectrum for the rotational vestibulo-ocular reflex (rVOR). It is not widely available because it requires expensive equipment and an experienced audiologist or ototechnician. A typical protocol for rotatory chair testing includes step velocity testing and slow harmonic acceleration (which can assess for vestibular weakness), visual fixation suppression (which can assess for certain cerebellar lesions), and optokinetic testing.
Introduction
Rotatory chair testing (RCT) is primarily useful as a detailed evaluation of the low- to middle- range of the vestibular tuning spectrum for the rotational vestibulo-ocular reflex (rVOR), and is considered the gold standard for evaluation of bilateral vestibular weakness. Less commonly used parts of the test can evaluate for certain cerebellar disorders.
Technique
The rotational position of the chair is driven and monitored by a very accurate servo-controlled torque motor. The patient’s body and head are secured (strapped) into the chair, so that head position can be inferred from chair position. The head is tilted about 30˚ forward (in order to align the plane of the horizontal semicircular canals in the earth-horizontal plane), and VNG goggles (or ENG electrodes) are placed on the patient. Information about head/body position and eye position are acquired as the chair rotates in various protocols. In this arrangement:
-
- The position of the chair (and therefore head/body) is the independent variable (“input” or “stimulus”).
- The position of the eye is the dependent variable (“output” or “response”).
For many sub-parts of rotatory chair testing, it is essential that the patient be in complete darkness, because even the tiniest light source (called a “light leak”) can allow the patient to fixate, which will distort test results. This is most easily achieved by using special light-occluding goggles.
Although the raw acquired data pertain to eye position, it is the calculated slow phase eye velocity (SPV) that is of interest.
The Figure below, from Fatima and colleagues (Fatima et al. 2000), shows the slow phases and quick phases of nystagmus.
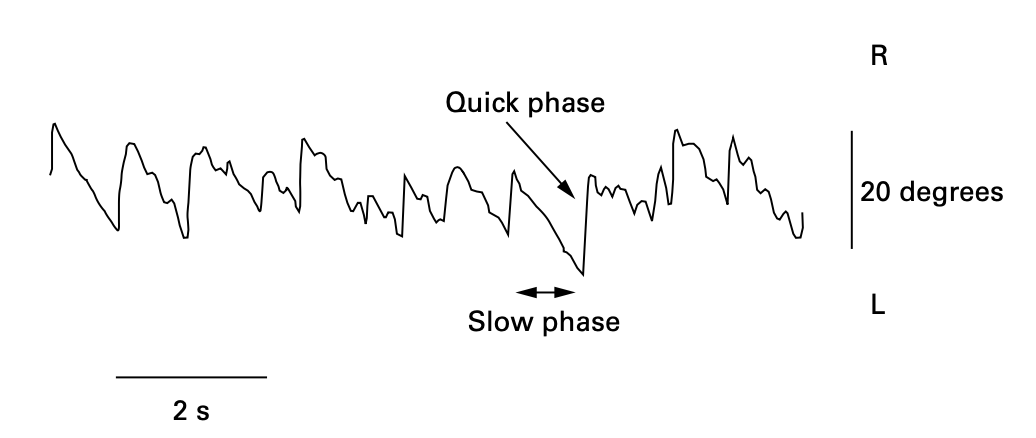
In RCT the objective is to compare the stimulus (chair velocity, from which we infer head velocity) with the response (slow phase eye velocity), and figure out the relationship between the two. In engineering terms this “systems identification” problem is one in which we are trying to determine the “transfer function” between input (stimulus) and output (response).
Physiology and neuroanatomy
We review the general concepts of the vestibulo-ocular reflex (rVOR) elsewhere. Here we discuss the physiology and neuroanatomy pertinent to specific sections of RCT.
Step velocity testing
Step velocity testing consists of an abrupt, brief, constant acceleration in a given direction, after which the achieved velocity is maintained for 30 seconds, then the chair is decelerated in the reverse direction to stop its movement. Most laboratories use an acceleration of 100˚/sec2 for 1 second. Each direction is tested separately.
The Video below, from https://www.youtube.com/watch?v=wPBgXqiSJig (accessed 5/3/23), shows step velocity testing.
The Figure below, from Stockwell and Bojrab (Stockwell and Bojrab 1997) diagrams step velocity testing with a leftward stimulus.
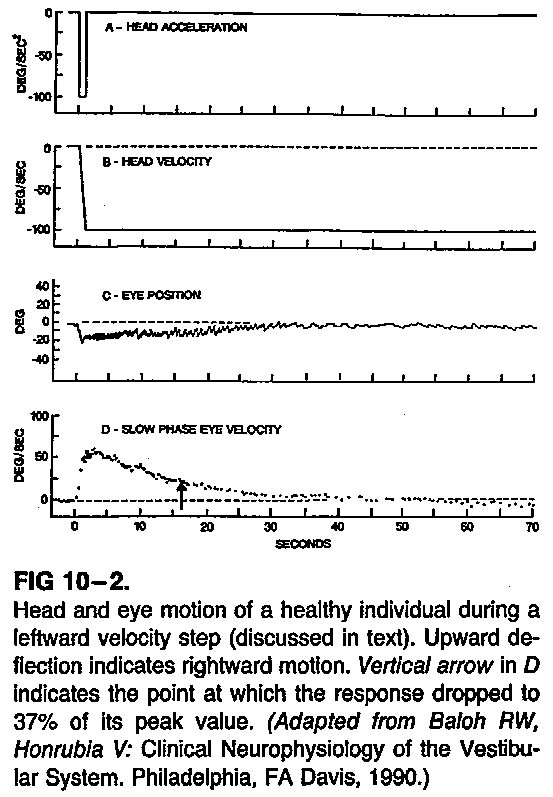
This figure shows abrupt leftward angular acceleration of 100 deg/sec/sec, after which head angular velocity is constant at 100 deg/sec to the left. There is left beat nystagmus, with rightward slow phases. This nystagmus and its slow phase decay over time.
The Figure below, from http://archimede.bibl.ulaval.ca/archimede/fichiers/23976/ch09.html (accessed 2012), shows composite plots of step velocity testing across a series of velocities.
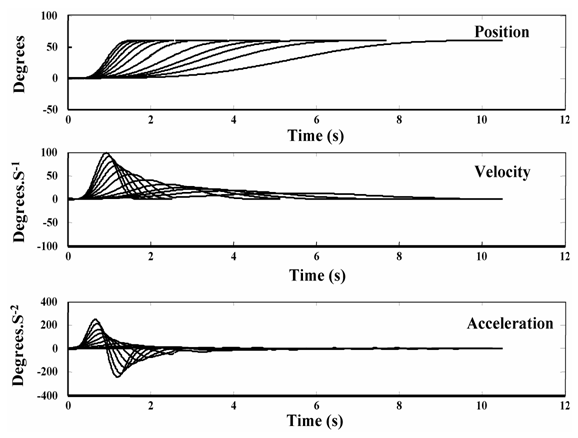
There are several points to note about this figure.
- By about 4 seconds, the traces of eye acceleration are converging.
- This reflects the fact that, as the change in the stimulus (head acceleration) decreases, change in the response (eye acceleration) disproportionately decreases, i.e., becomes more sluggish.
- In other words, the vestibular system is good at detecting large acceleration, and poor at detecting small acceleration.
As Stockwell and Bojrab describe it:
“The system performs well only for relatively fast head motions. The oscillation frequencies used in the slow harmonic acceleration test are at the lower end of its operating range, and therefore, as oscillation frequency decreases, the system becomes less sensitive to the head motion and fails to integrate the head acceleration signal completely, yielding lower gains and greater phase leads” (Stockwell and Bojrab 1997).
The pattern of eye movements induced by step velocity testing is complex. In order to explain it, vestibular physiologists have adapted modeling techniques from engineering. The model comprises three components:
- The mechanical response / direct pathway
- The velocity storage mechanism / indirect pathway
- Adaptation
To understand these components, consider the analogy of a coffee cup. The cup is the bony and membranous labyrinth. The coffee is the endolymph. Picture this sequence of events:
- The coffee cup is rapidly accelerated counterclockwise (looking from above) and then maintains a steady angular velocity in that direction.
- Initially, because of inertia, the coffee remains still (from the perspective of an outside observer).
- Thus, a tiny velocimeter at the edge of the cup (measuring the outermost rim of coffee) would initially measure the angular velocity of the coffee to be equal in amplitude but opposite in direction of the angular velocity of the cup.
- The movement of the coffee lags behind.
- But eventually (because of viscosity) the coffee slowly “catches up” with the motion of the cup. In other words, the coffee reaches the same angular velocity as the cup.
- At this point, the coffee is remaining still relative to the cup. Thus, the tiny velocimeter at the edge of the cup (measuring the outermost rim of coffee) would now measure the angular velocity of the coffee to be zero.
- This is the point at which the angular velocity of the coffee (relative to the cup) has “decayed” to zero.
Everything described so far pertains to the mechanical response of the deflection of the cupula by the endolymph in the lateral semicircular canal. This mechanical response (Stockwell and Bojrab 1997) is sometimes also referred to as the “direct pathway” (Baloh and Honrubia 2001), since its contribution to eye movements is directly proportional to afferents from the cupula. The direct response is mediated by a chain of three neurons, whose cell bodies reside in (1) the vestibular ganglion, (2) the vestibular nuclei, and (3) the oculomotor nuclei.
After the angular acceleration ceases (i.e., the cup is rotating at a constant angular velocity), the endolymph will continue moving for a little while, but shearing forces will soon decelerate it to an angular velocity of zero, and the rate of firing from the cupula will return to baseline — these fluid dynamics and electrophysiology have been experimentally confirmed. It turns out that, even after the angular velocity of the endolymph is zero (and the rate of firing from the cupula has returned to baseline tonic rate), there is still nystagmus for a little while. How can this be?
In order to explain this persistence of nystagmus, vestibular physiologists have postulated a second factor at play, called the velocity storage mechanism (Baloh and Honrubia 2001; Stockwell and Bojrab 1997). This is a positive feedback loop in the central vestibular system that performs the equivalent of mathematical integration of the labyrinthine inputs and yields a response with a time constant (more on this below) longer than that generated by the afferents (from the cupula’s mechanical response). The neural substrate for this is believed to reside in the interneurons mediating the commissural connections between the vestibular nuclei (Katz et al. 1991; Yakushin et al. 2017), though this is controversial. There are other reasons to postulate the existence of this mechanism (that come from optokinetic testing, discussed below).
The term “velocity storage” is intended to reflect the fact that this mechanism holds, or “stores,” activity from the vestibular periphery and discharges it over a more prolonged time span (i.e., more prolonged than the afferents themselves). For this reason, some refer to it as an “indirect pathway” (Baloh and Honrubia 2001). As discussed above, the vestibular system is generally better at detecting larger accelerations than smaller ones, so from a teleological perspective it is thought that the velocity storage mechanism “improves the ability of the rotational vestibulo-ocular reflexes to transduce the low-frequency components of head rotation” (Leigh and Zee 2015). Since the nystagmus induced by step velocity testing eventually ceases, it is clear that the velocity storage mechanism does not perpetuate the velocity signal forever. Something causes it to decay.
In order to explain this decay, vestibular physiologists postulated a third factor at play, adaptation. Stockwell and Bojrab (Stockwell and Bojrab 1997) describes adaptation as, “a zero velocity reference level within the central vestibular system that gradually shifts during sustained stimulation in the direction of the ongoing response. Adaptation progressively reduces the strength of a unidirectional response and, after the response finally subsides, induces a response reversal.” The neural substrate of adaptation is unknown.
The three components of step velocity testing are illustrated in the Figure below, from Stockwell and Bojrab (Stockwell and Bojrab 1997).
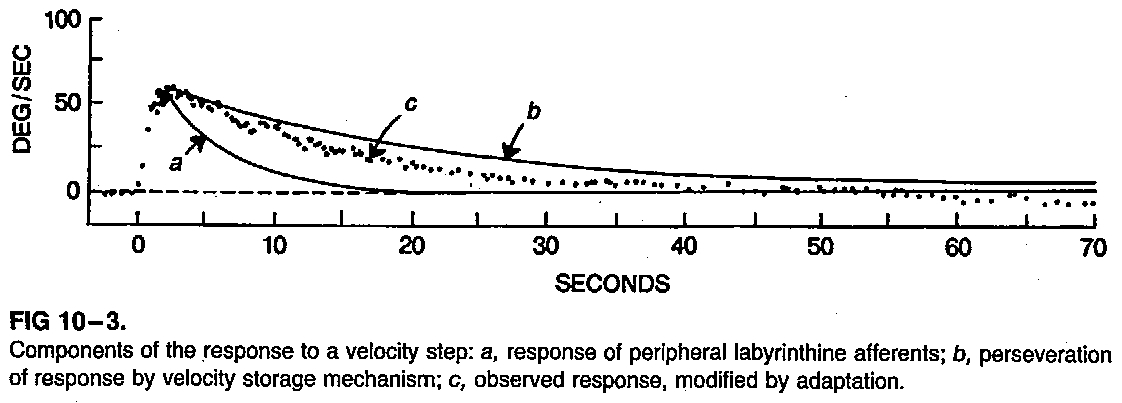
In this figure, the output (plotted dots) response is the net result of:
- The mechanical response (direct pathway).
- The velocity storage mechanism (indirect pathway).
- Adaptation.
From the output of step velocity testing two parameters can be calculated:
- Gain. This is the ratio of response (peak slow phase eye velocity) to stimulus (peak head velocity). In a perfect system (in which gain=1), peak slow phase eye velocity would be exactly equal and opposite to peak head velocity. This assesses the contribution from (1) the mechanical response/direct pathway.
- Time constant. This is the time, in seconds, for the response (peak slow phase eye velocity) to decay to 37% (1/e) of the stimulus (peak head velocity). This assesses the contribution from (2) the velocity storage mechanism/indirect pathway and (3) adaptation.
Step velocity testing has the advantage of relatively rapid assessment, since only a single velocity “step” (usually 100˚/sec) is tested. However, since it is based on a more limited set of data, its characterization of vestibular function is also limited. For this reason, rotatory chair testing usually employs the complementary technique of slow harmonic acceleration.
Slow harmonic acceleration
The slow harmonic acceleration (SHA) portion of rotatory chair testing consists of alternating rightward (clockwise) and leftward (counterclockwise) rotation in a smooth, sinusoidal pattern, at several frequencies (typically 0.01, 0.02, 0.04, 0.08, 0.16, 0.32 and 0.64 Hz), with peak angular velocities of 50˚/sec at each frequency. Baloh and Honrubia explain that, “With sinusoidal rotational testing, the gain of the canalith-ocular reflex can be measured at multiple discrete frequencies after the subject has attained a steady-state response. It usually provides a more accurate assessment of gain than is obtained with the step test” (Baloh and Honrubia 2001).
The Video below, from https://www.youtube.com/watch?v=0TIreo1oRzs (accessed 5/3/23), shows slow harmonic acceleration at 0.08 Hz.
The Video below, from https://www.youtube.com/watch?v=d-JldPU-qig (accessed 5/3/23), shows slow harmonic acceleration at 0.64 Hz.
The Video below from https://www.youtube.com/watch?v=XaI5s5IRWJo (accessed 2012), is a cartoon depiction of slow harmonic acceleration with corresponding nystagmus and oculographic tracing.
From the output of SHA two parameters can be calculated (Stockwell and Bojrab 1997):
- Gain. Similar to step velocity testing, this is the ratio of response (peak slow phase eye velocity) to stimulus (peak head velocity). In a perfect system (in which gain=1), peak slow phase eye velocity would be exactly equal in magnitude to peak head velocity.
- Phase angle. This is a measure of the timing relationship between eye slow phase velocity and head velocity. In a perfect system the phase angle=180˚, which means that peak slow phase eye velocity is exactly opposite in direction to peak head velocity.
The Figure below, from Leigh and Zee (Leigh and Zee 2015), shows gain and phase in an ideal system in which gain=1 and phase angle=180 deg.
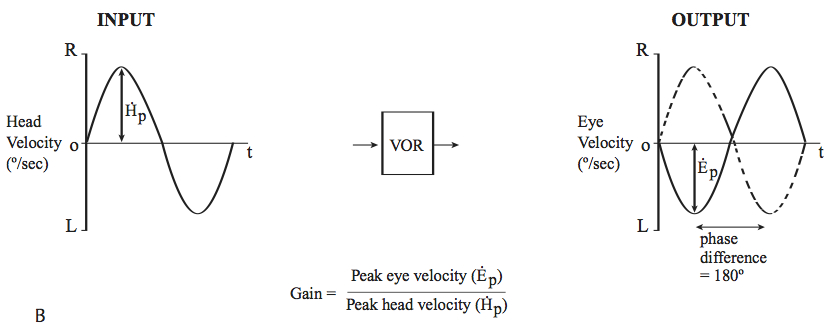
The figure above depicts the behavior of an ideal system in which gain=1 and phase angle=180 degrees. As mentioned earlier, the vestibular system is more effective at detecting larger acceleration than smaller acceleration, and this is reflected by the series of frequencies assessed in slow harmonic acceleration.
This is illustrated in the Figure below, adapted from Stockwell and Bojrab (Stockwell and Bojrab 1997).
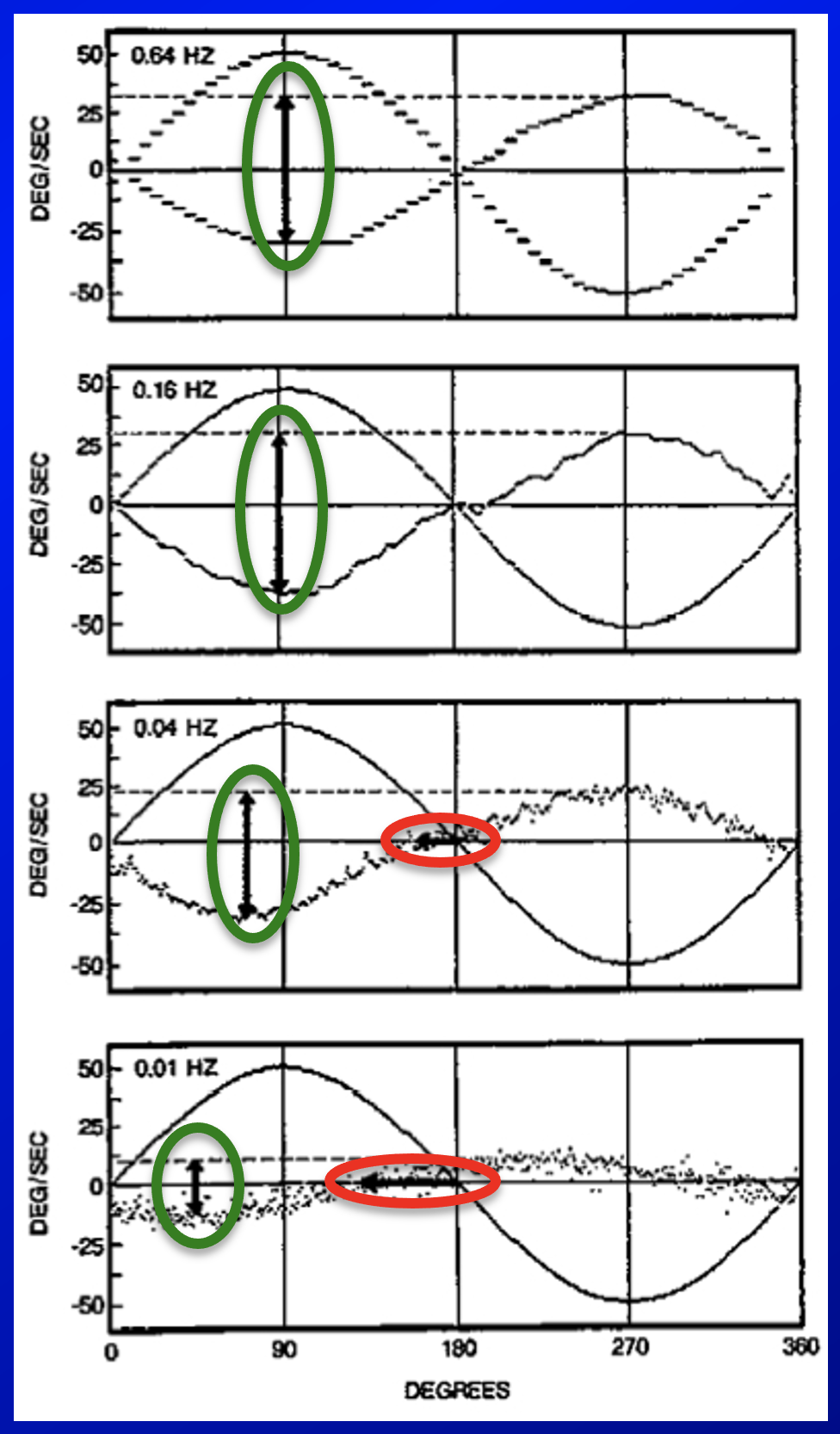
As they describe it:
“Head and slow-phase velocities of a normal individual during oscillation at four different frequencies. Upward deflection denotes rightward velocities. Response gains (vertical arrows) are 0.61 at 0.64 Hz, 0.67 at 0.16 Hz, 0.52 at 0.04 Hz and 0.25 at 0.01 Hz. Phase leads (horizontal arrows) are 0˚ at 0.64 and 0.16 Hz, 15˚ at 0.04 Hz, and 45˚ at 0.01 Hz” (Stockwell and Bojrab 1997).
The Figure below, from Ahmed (Ahmed 2014), shows the normal change in gain and phase seen in the series of frequencies tested in slow harmonic acceleration.
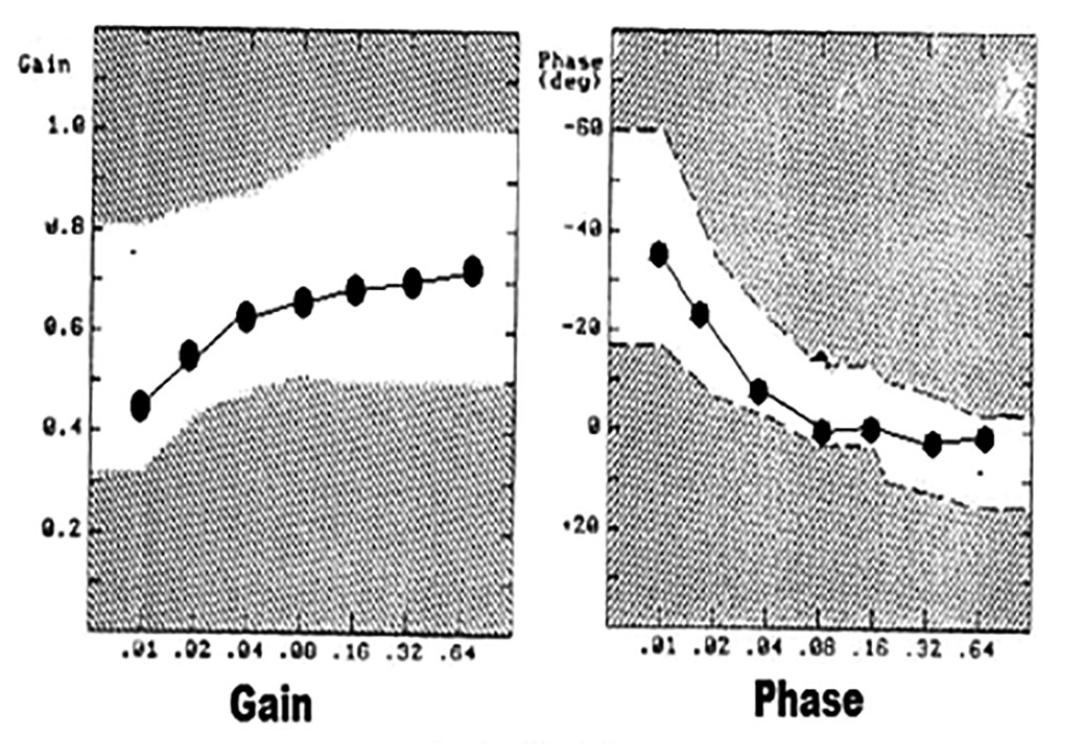
Since the system functions more poorly at lower frequencies of slow harmonic acceleration, it is at these lower frequencies that certain pathologies are more likely to be detected.
The Figure below, from Hain and colleagues (Hain et al. 2013), shows the results of slow harmonic acceleration in a patient with bilateral vestibular weakness.
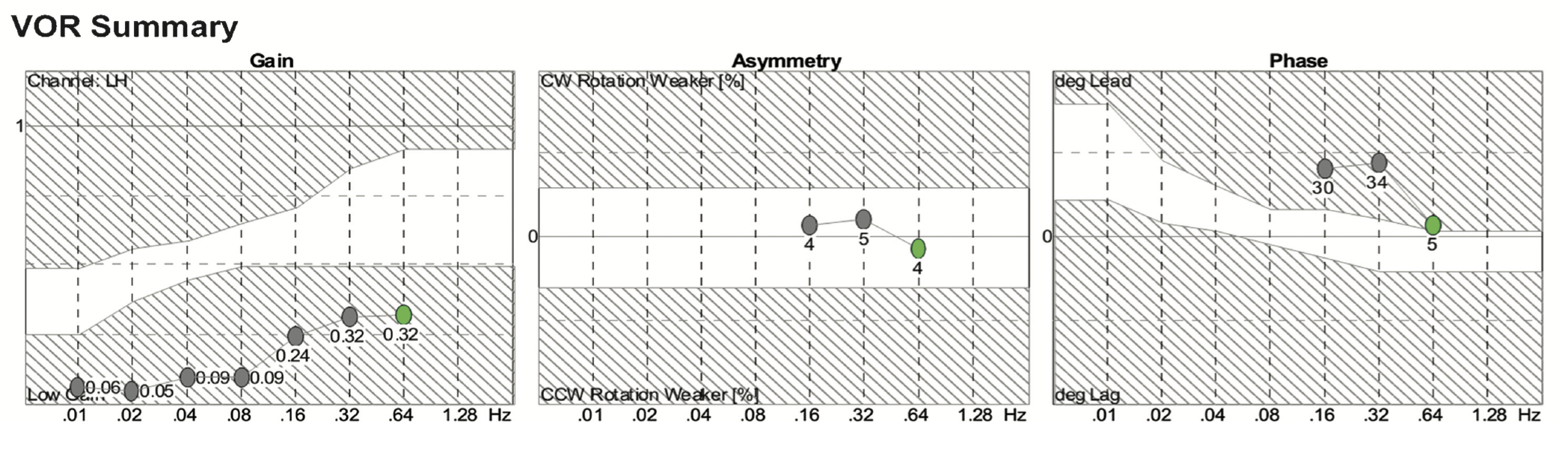
In this figure, gain (left panel) shows reduced gain across all frequencies. Phase (right panel) is elevated at 0.16 – 0.64 Hz, and the phase angle at lower frequencies was so great that it could not be displayed on this plot.
In comparison to step velocity testing, slow harmonic acceleration has the advantage of characterizing the function of the rotational vestibulo-ocular reflex more accurately because of its assessment over a series of frequencies. Slow harmonic acceleration also has disadvantages.
“The main disadvantage [of slow harmonic acceleration] is the lengthy procedure required to test a broad frequency range. Also, unlike the step [velocity] test, sinusoidal testing measures only a single [i.e., bilateral] time constant (the low-frequency phase lead reflects the average time constant in both directions)” (Baloh and Honrubia 2001).
In other words, slow harmonic acceleration cannot characterize a unilateral vestibular deficit, since its results at a given frequency necessarily reflect the composite function of both lateral semicircular canals.
Optokinetic testing
Optokinetic testing consists of assessing two types of responses: optokinetic nystagmus (OKN), and optokinetic afternystagmus (OKAN).
Optokinetic testing: optokinetic nystagmus
The eye movements that constitute optokinetic nystagmus (OKN) belong to a phylogenetically older system since they are present in afoveate animals (such as rabbits), whereas only animals that have evolved foveae exhibit true smooth pursuit movements (Baloh and Honrubia 2001). Baloh and Honrubia state that, “The optokinetic system is generally considered to be a primitive form of smooth pursuit involving the whole retina instead of the fovea alone” (Baloh and Honrubia 2001).
Optokinetic eye movements are not truly smooth pursuit because:
“Optokinetic nystagmus (OKN) is the eye movement elicited by tracking of a field [rather than tracking of a point target in foveal vision]. This movement is produced in response to a moving full-field visual surrounding… The purpose of OKN is to stabilize an entire visual field, unlike pursuit, which has to do with maintaining the position of a single target upon the fovea” (Hain 1997a).
The optokinetic system accomplishes this by combining several functions:
“OKN is the sum of two tracking mechanisms, namely, the smooth pursuit system, which uses foveal vision, and a separate tracking system, which uses both foveal and extrafoveal vision” (Hain 1997b).
Given these multiple mechanisms, it appears that optokinetic responses are mediated by both cortical and subcortical structures (Baloh and Honrubia 2001).
Of these several mechanisms, different ones are active at different points in the optokinetic responses:
- “The initial eye movement response to motion of the entire visual environment has characteristics slightly different from either smooth pursuit or OKN, and is called ocular following” (Carl 1997).
- “At the onset of the stimulus, the smooth pursuit system is most important and causes eye velocity to reach its maximum within a second or two” (Leigh and Zee 2015).
As in other parts of rotatory chair testing, one of the parameters calculated from optokinetic nystagmus is the gain which, in the case of OKN, is the ratio of peak slow phase eye velocity to visual field stimulus velocity. In normal subjects exposed to a full field stimulus moving at 60 deg/sec, gain is approximately 0.5 (Hain 1997a).
Optokinetic testing: optokinetic after-nystagmus
Optokinetic afternystagmus (OKAN) is the nystagmus that continues even after the visual stimulus has disappeared. The OKAN response is weak and variable between human subjects, and even varies substantially in the same individual from trial to trial (Hain 1997b). For technical reasons, the initial velocity is measured (rather than gain). The time constant is also assessed. Regarding the underlying neuroanatomical mechanisms, “Unlike OKN, OKAN is not produced by cortical neuronal structures analogous to pursuit, but rather is generated in the brain stem” (Hain 1997a).
Optokinetic afternystagmus is very “fragile” in that its function can be impaired by a variety of peripheral and central disorders.
Additional comments on optokinetic testing
An interesting point about optokinetic testing is that the responses exhibit their own time constants, even though there has been no peripheral vestibular stimulation at all (since the patient has remained stationary).
This is because purely visual input (i.e., in isolation from other changing vestibular or proprioceptive stimuli) also appears to “feed into” the velocity storage mechanism (Leigh and Zee 2015). Experiments in monkeys have shown that optokinetic stimulation does cause firing in the vestibular nuclei (Leigh and Zee 2015).
It is also interesting that optokinetic afternystagmus is abolished in bilateral vestibular loss (Cohen et al. 1973). It appears that some baseline level of tonic peripheral vestibular input is necessary to maintain the velocity storage mechanism.
Visual fixation suppression
In visual fixation suppression (VFX) the patient is asked to try to maintain gaze on a target (“visual fixation”) whose position is “locked” with respect to head position, and to carry out this task while ignoring (“suppressing”) peripheral vestibular input (such as the head oscillating). The arrangement is one in which a laser that is secured to the top of the head and aimed directly in front of the subject. The point of light projected by that laser will move in synchrony with the head, and the patient is instructed to stay focused on that point even when the body (and head) is rotating.
Normally functioning individuals are generally fairly good (though not perfect) at visual fixation suppression – essentially, they successfully almost completely ignore (suppress) peripheral vestibular input.
In disease where there is no peripheral vestibular input (such as complete bilateral vestibular loss), this task becomes very easy (because there is no input to suppress), so visual fixation suppression is perfect.
In contrast, there are some disease states in which VFX is impaired. This usually occurs with central lesions, such as those involving the cerebellum (particularly the flocculus) (Cohen et al. 1973; Takemori and Cohen 1974).
Visually enhanced vestibulo-ocular reflex
In visually enhanced vestibulo-ocular reflex (VVOR) testing the patient is instructed to try to maintain gaze on an earth-stationary target while the chair (and thus the patient’s head) rotates. In the dark, rotation of the chair (and thus of the stabilized head) would provoke contraversive horizontal ocular rotation of nearly the same magnitude, thanks to the vestibulo-ocular reflex; but by additionally providing an earth-stationary visual target, the VOR is “enhanced,” and the gain should be very close to 1.0 in a healthy individual.
The VVOR is reported to have abnormally low gain in CANVAS (Migliaccio et al. 2004).
The VVOR has been reported to have abnormally elevated gain in migraine associated vertigo (Arriaga et al. 2006; Kim et al. 2023), and in patients with hypersensitivity to motion or sound (Kim et al. 2023).
Equipment needed
A rotatory chair is one of the most expensive pieces of equipment in a well-outfitted otoneurology or neuro-otology clinic. Good products include fully-enclosed RCTs, such as that of the illustration shown in the Figure below, from Hain (Hain 1997b).
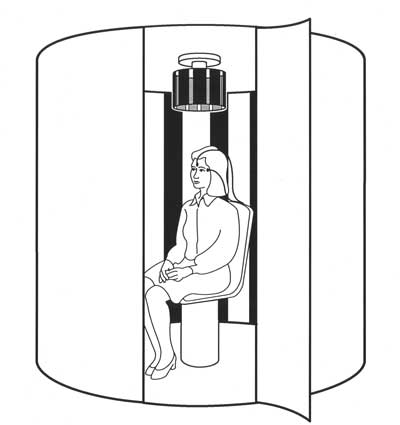
Many offices have only a partially enclosed RCT.
How to perform the test
RCT can be performed in various ways depending on the clinical needs. A common protocol would include:
- Calibration
- Step velocity testing
- Slow harmonic acceleration
- Optokinetic testing
- Visual fixation suppression
Calibration
Calibration should be performed for every patient, and consists of two parts:
- Oculomotor calibration. The patient keeps the head still and is instructed to glance from a target straight ahead (0˚) to another target at 30˚. Calibration checks whether the degree of eye movement matches the degree of target movement.
- Eye-chair calibration. The patient foveates a target straight ahead. Then the chair is rotated by 30˚ (the head is fixed with respect to the body and chair), and the patient is instructed to keep the eyes on the earth-fixed target. The eyes should rotate by an amount that is equal in magnitude but opposite in direction to the chair (and thus head) movement.
If either of these two aspects of calibration is off, then the results of the test will be difficult to interpret, or will be non-diagnostic. Unfortunately, many audiology reports do not describe calibration. This is problematic because in some circumstances it may be difficult to ascertain whether a given abnormality reflects true pathology or is an artifact of poor calibration. If the original tracings are provided, and if they include horizontal saccade tests or smooth pursuit tests, then one can infer correct calibration if the target position and eye position are equal.
Step velocity testing
This portion of the test consists of an abrupt, brief, constant acceleration in a given direction, after which the achieved velocity is maintained for 30 seconds, then the chair is decelerated in the reverse direction to stop its movement. Most laboratories use an acceleration of 100˚/sec2 for 1 second. Each direction is tested separately.
Slow harmonic acceleration
The slow harmonic acceleration (SHA) portion of rotatory chair testing consists of alternating rightward (clockwise) and leftward (counterclockwise) rotation in a smooth, sinusoidal pattern, at several frequencies (typically 0.01, 0.02, 0.04, 0.08, 0.16, 0.32 and 0.64 Hz), with peak angular velocities of 50˚/sec at each frequency.
Optokinetic testing
Generation of a true full-field visual stimulus usually requires special equipment, such as an enclosed rotatory chair chamber with a specialized light source that generates vertical bars of light that move laterally and uniformly across the horizon.
The Figure below, from Davies (Davies 2004), shows such an arrangement.

Most clinical laboratories that claim to offer rotatory chair testing do not have the proper facilities for true optokinetic testing. Instead, they tend to use “partial field” devices (Hain 1997a). Since that stimulus only occupies part of the visual field, this technique actually ends up eliciting either only smooth pursuit, or a combination of smooth pursuit and OKN. Most rotatory chair test reports do not specify what type of stimulus was employed, which can make it difficult to interpret apparent abnormalities.
For optokinetic testing it is important that the patient be in complete darkness (except for the intended stimulus). Any “light leak” will distort the results. The most common protocol involves:
- Abruptly the full field optokinetic stimulus is presented, consisting of light bars moving horizontally and uniformly across the visual field at a constant velocity (typically anywhere from 30˚ to 60˚/sec). “Optokinetic nystagmus” (OKN) is then measured for 30 seconds.
- After that the stimulus is abruptly turned off, returning the patient to complete darkness. At this point, “optokinetic afternystagmus” (OKAN) is measured for 30 seconds.
- The protocol is performed once in each direction.
Visual fixation suppression
In visual fixation suppression (VFX) the patient is asked to try to maintain gaze on a target (“visual fixation”) whose position is “locked” with respect to head position, and to carry out this task while ignoring (“suppressing”) peripheral vestibular input (such as the head oscillating). The arrangement is one in which a laser that is strapped to the top of the head and aimed directly in front of the subject. The point of light projected by that laser will move in synchrony with the head, and the patient is instructed to stay focused on that point even when the body (and head) are rotating.
Visually enhanced vestibulo-ocular reflex
In visually enhanced vestibulo-ocular reflex (VVOR) testing the patient is instructed to try to maintain gaze on an earth-stationary target while the chair (and thus the patient’s head) rotates. In the dark, rotation of the chair (and thus of the stabilized head) would provoke contraversive horizontal ocular rotation of nearly the same magnitude, thanks to the vestibulo-ocular reflex; but by additionally providing an earth-stationary visual target, the VOR is “enhanced.”
What this test assesses
In summary:
- Step velocity testing reveals the parameters of gain and phase in each direction.
- Slow harmonic acceleration reveals the gain and phase angle over a series of frequencies.
- Optokinetic nystagmus is characterized by gain in each direction.
- Optokinetic afternystagmus is characterized by the initial velocity and time constant in each direction.
- Visual fixation suppression is characterized by gain in each direction.
How to interpret the test results
Very few resources have been written encompassing the range of rotatory chair sub-tests and their interpretation. We cannot here replicate the detailed discussions from works such as that of Zalewski’s textbook (Zalewksi 2018), but we offer some summary points:
- If slow harmonic acceleration shows reduced gain and phase lead, this suggests bilateral vestibular weakness.
- If step velocity testing shows reduced gain and reduced time constant in one or both directions, then this suggests unilateral or bilateral vestibular weakness.
- Optokinetic nystagmus and optokinetic afternystagmus can be abnormal in a variety of central lesions, and can also be abolished by bilateral vestibular loss.
- Visual fixation suppression has abnormally high gain in bilateral vestibular weakness (because there is no peripheral vestibular input to suppress), and has abnormally low gain in cerebellar flocculus lesions (because the cerebellum fails to enable visual fixation to override normal peripheral vestibular input).
Limitations
Centrally acting medications, particularly those with vestibular suppressant affects (intentional or unintentional), can introduce errors into the results of rotatory chair testing.
Contraindications
There are no firm contraindications to rotatory chair testing.
Pitfalls
There are multiple potential sources of error in rotatory chair testing, including:
- Poor technique.
- Improper calibration.
- Light leaks.
- Use of a partial field stimulus for optokinetic testing.
- Inadequate instruction of patient.
- Poor patient cooperation.
- Eye-blink artifact.
- Failure of technician to report:
- Patient’s current medications and recent alcohol use.
- What method was employed to elicit optokinetic nystagmus.
When is the test indicated
Rotatory chair testing is always sensible to check in cases of suspected vestibular weakness, particularly if bilateral.
Rotatory chair testing may also be helpful in characterizing some cerebellar dysfunction.
Diseases that may be diagnosed by this test
Rotatory chair testing remains the gold standard for assessing bilateral vestibular weakness, but it should be kept in mind that it only assesses the low- to middle-frequency range of the vestibular tuning spectrum of the horizontal canal rotational vestibulo-ocular reflex.
The diagnostic utility of optokinetic nystagmus (OKN) is limited.
-
- Symmetrically reduced OKN gain can occur in visual disorders that impair foveal vision (such as macular degeneration) and/or extrafoveal vision (such as pigmentary retinopathy).
- Symmetrically reduced OKN gain also occurs in “fast phase disorders,” such as progressive supranuclear palsy, in which there is “hang-up” at end trajectory, because there is a failure of saccades (“fast phase”) to return the eyes towards the center of the orbit after the pursuit (“slow phase”) follows the stimulus out to the periphery.
- Asymmetrical OKN occurs infrequently, “presumably… because it requires lesions in two tracking systems: foveal and extrafoveal” (Hain 1997b). Asymmetrical OKN appears briefly following unilateral parieto-occipital lesions.
- Reversed OKN occurs in patients with congenital nystagmus.
The diagnostic utility of optokinetic afternystagmus (OKAN) is also limited.
- Reduced OKAN:
- Symmetrically absent (or reduced) OKAN occurs in bilateral vestibular loss.
- Asymmetrically absent (or reduced) OKAN occurs in patients with unilateral vestibular loss.
- Increased OKAN:
- Symmetrically increased OKAN has been reported to occur in mal de debarquement syndrome.
Cerebellar deficits, particularly those involving the flocculus, may be detected by poor gain on visual fixation suppression.
The diagnostic utility of visually enhanced vestibulo-ocular reflex (VVOR) is limited.
- Abnormally low gain of the VVOR occurs in CANVAS (Migliaccio et al. 2004).
- Abnormally high gain of the VVOR has been reported in in migraine associated vertigo (Arriaga et al. 2006) and in patients with hypersensitivity to motion or sound (Kim et al. 2023).
Additional notes
There have been proposals to modify the rotatory chair test configuration so as to assess vertical canal function. An example of this is illustrated in the Figure below, from Morita and colleagues (Morita et al. 2003). This has never come into wide use.
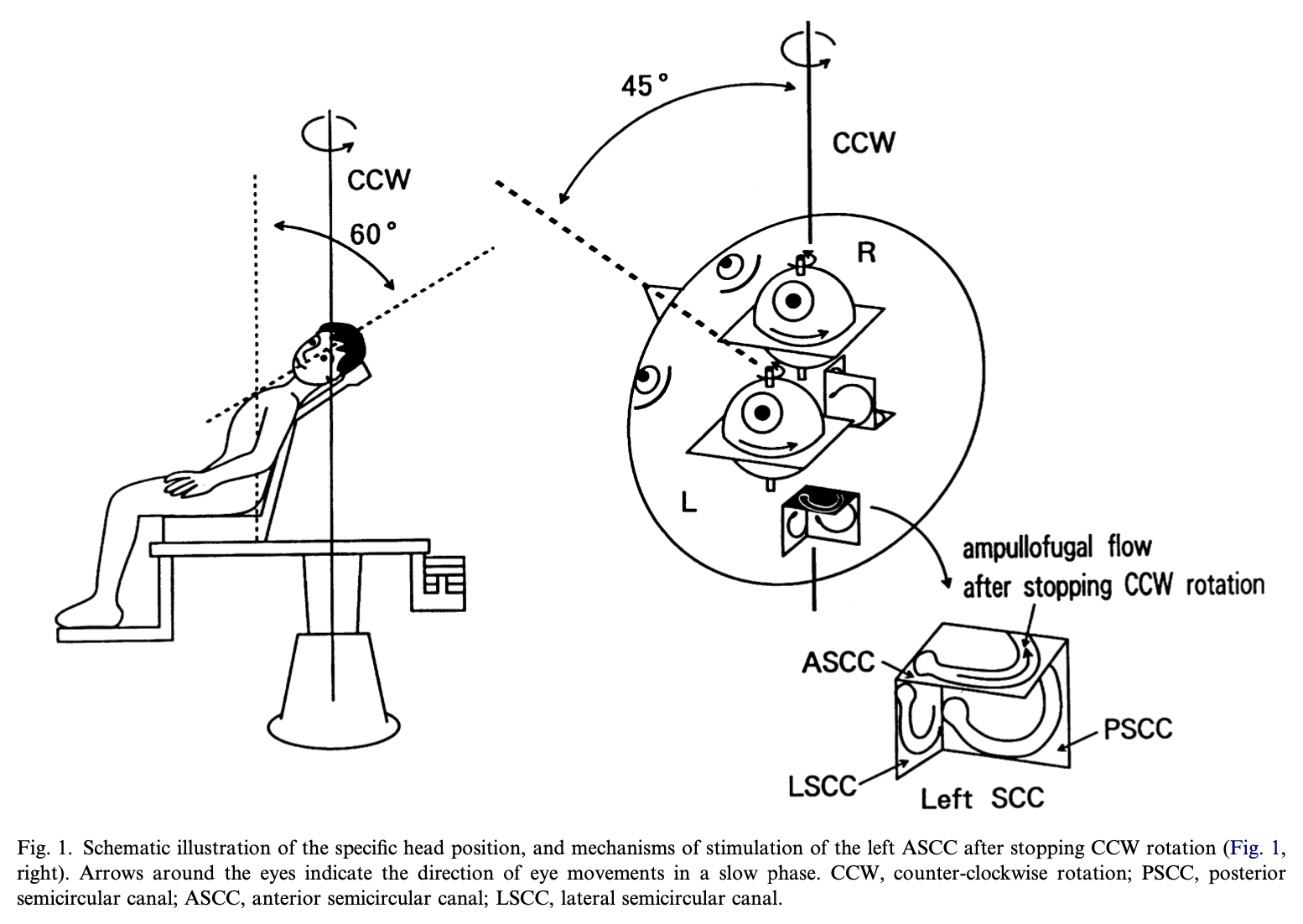
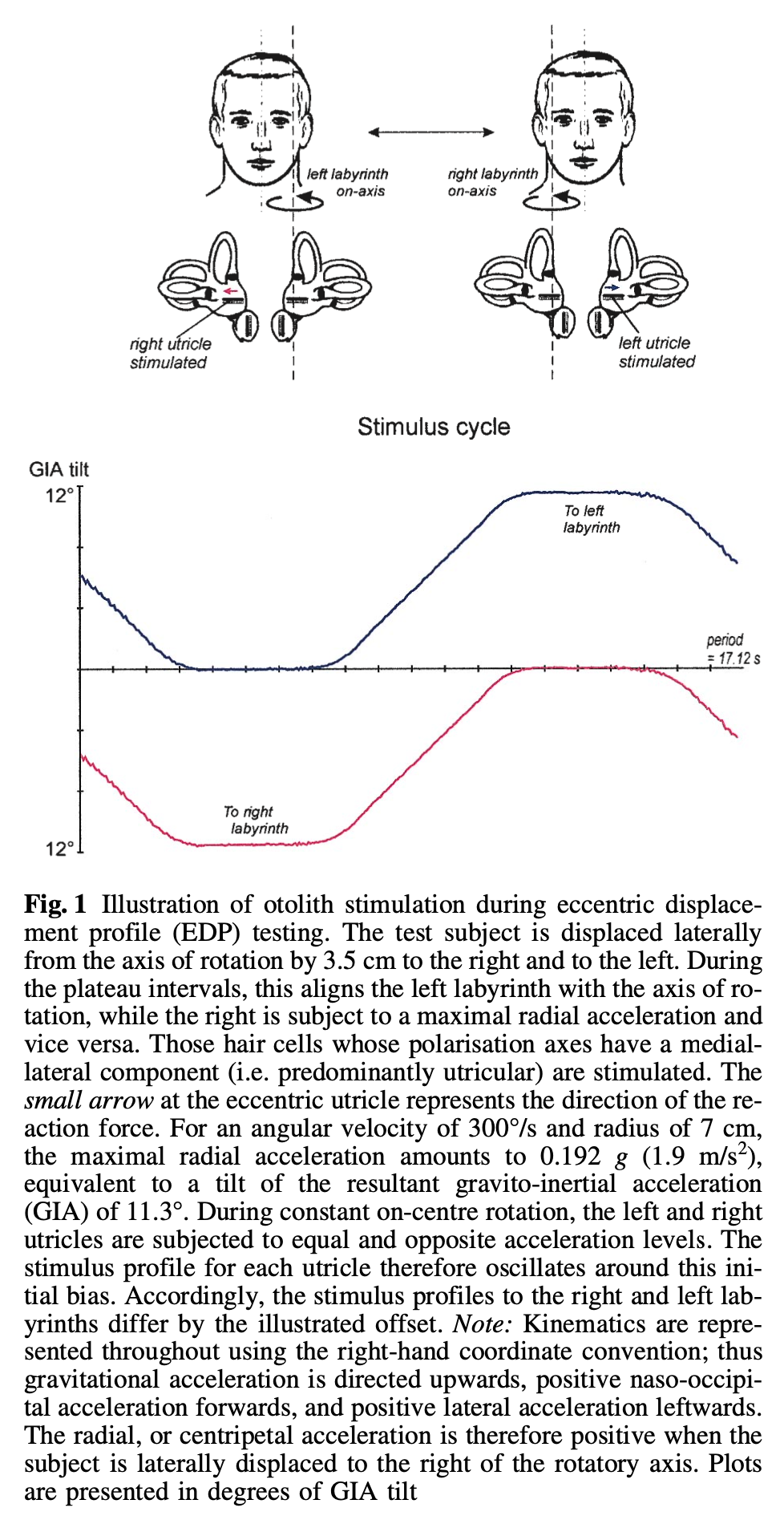
Eccentric rotation protocols have tried to use RCT to assess utricular function, as illustrated in the Figure below from Clarke and Engelhorn (Clarke and Engelhorn 1998), based on the idea that the utricle situated at the axis of rotation should not be stimulated. This has never come into wide use, as it requires a specially modified rotatory chair. Currently, in the clinical setting, utricular function is more easily assessed with ocular vestibular evoked myogenic potentials.
References
Ahmed MF (2014) Standardization of rotatory chair velocity step and sinusoidal harmonic acceleration tests in an adult population. Advanced Arab Academy of Audiovestibulogy 1: 80-86. doi: 10.4103/2314-8667.149016
Arriaga MA, Chen DA, Hillman TA, Kunschner L, Arriaga RY (2006) Visually enhanced vestibulo-ocular reflex: a diagnostic tool for migraine vestibulopathy. Laryngoscope 116: 1577-9. doi: 10.1097/01.mlg.0000231308.48145.f6
Baloh RW, Honrubia V (2001) Clinical Neurophysiology of the Vestibular System, 3rd edn. Oxford University Press, New York
Carl JR (1997) Practical anatomy and physiology of the ocular motor system. In: Jacobson GP, Newman CW, Kartush JM (eds) Handbook of Balance Function Testing. Delmar, New York, pp 53-68
Clarke AH, Engelhorn A (1998) Unilateral testing of utricular function. Exp Brain Res 121: 457-64. doi: 10.1007/s002210050481
Cohen B, Uemura T, Takemori S (1973) Effects of labyrinthectomy on optokinetic nystagmus (OKN) and optokinetic after-nystagmus (OKAN). International Journal of Equilibrium Research 3: 88-93.
Davies R (2004) Bedside neuro-otological examination and interpretation of commonly used investigations. J Neurol Neurosurg Psychiatry 75 Suppl 4: iv32-44. doi: 10.1136/jnnp.2004.054478
Fatima SS, Anthony K, Dorothy T, Isabelle R-E, David T, Christopher H (2000) Vertical or asymmetric nystagmus need not imply neurological disease. British Journal of Ophthalmology 84: 175. doi: 10.1136/bjo.84.2.175
Guillaud E, Gauthier G, Vercher JL, Blouin J (2006) Fusion of visuo-ocular and vestibular signals in arm motor control. J Neurophysiol 95: 1134-46. doi: 10.1152/jn.00453.2005
Hain TC (1997a) Background and technique of ocular motility testing. In: Jacobson GP, Newman CW, Kartush JM (eds) Handbook of Balance Function Testing. Delmar, New York, pp 83-100
Hain TC (1997b) Interpretation and usefulness of ocular motility testing. In: Jacobson GP, Newman CW, Kartush JM (eds) Handbook of Balance Function Testing. Delmar, New York, pp 101-122
Hain TC, Cherchi M, Yacovino DA (2013) Bilateral vestibular loss. Semin Neurol 33: 195-203. doi: 10.1055/s-0033-1354597
Katz E, Vianney de Jong JM, Buettner-Ennever J, Cohen B (1991) Effects of midline medullary lesions on velocity storage and the vestibulo-ocular reflex. Exp Brain Res 87: 505-20.
Kim EK, Sienko N, Gardi A, Krauter R, Pasquesi L, Sharon JD (2023) Visually enhanced vestibulo-ocular reflex gain in patients with vestibular disease. Laryngoscope Investig Otolaryngol 8: 1061-1067. doi: 10.1002/lio2.1106
Leigh RJ, Zee DS (2015) The neurology of eye movements, 5th edn. Oxford University Press, Oxford ; New York
Migliaccio AA, Halmagyi GM, McGarvie LA, Cremer PD (2004) Cerebellar ataxia with bilateral vestibulopathy: description of a syndrome and its characteristic clinical sign. Brain 127: 280-93. doi: 10.1093/brain/awh030
Morita M, Imai T, Kazunori S, Takeda N, Koizuka I, Uno A, Kitahara T, Kubo T (2003) A new rotational test for vertical semicircular canal function. Auris Nasus Larynx 30: 233-7. doi: 10.1016/s0385-8146(03)00098-1
Stockwell CW, Bojrab DI (1997) Background and technique of rotational testing. In: Jacobson GP, Newman CW, Kartush JM (eds) Handbook of Balance Function Testing. Delmar, New York, pp 237-248
Takemori S, Cohen B (1974) Loss of visual suppression of vestibular nystagmus after flocculus lesions. Brain Res 72: 213-24. doi: 10.1016/0006-8993(74)90860-9
Yakushin SB, Raphan T, Cohen B (2017) Coding of Velocity Storage in the Vestibular Nuclei. Front Neurol 8: 386. doi: 10.3389/fneur.2017.00386
Zalewksi CK (2018) Rotational vestibular assessment. Plural Publishing, San Diego, CA
![]()