By Marcello Cherchi, MD PhD
For patients
If the body’s immune system recognizes a cancer, then it may mount an immunological attack against that cancer. However, in so doing, it may mistakenly also attack some of the body’s healthy (non-cancerous) cells. This is called a paraneoplastic syndrome. When a paraneoplastic syndrome involves the brain, it often attacks a part called the cerebellum, which can result in unsteadiness, clumsiness and deficits in coordination. In order to arrive at this diagnosis, your doctor may order a variety of tests, which may include imaging studies (such as an MRI of the brain). If a cancer is discovered, then the treatment is removal of the cancer.
For clinicians
Overview
Paraneoplastic syndromes arise when the immune system’s recognition of a malignancy results in mis-directed immune-mediated damage of healthy tissues. Although any part of the body, and any part of the nervous system, can be affected, what usually comes to the attention of an otoneurologist is a paraneoplastic cerebellar disorder (which usually presents with ataxia), sometimes with a cochleovestibular component (which can present with vertigo, hearing loss and tinnitus). These symptoms can begin rapidly, or evolve gradually, and they may precede recognition of the cancer by months or even years. Paraneoplastic syndromes have been described with a broad variety of malignancies and antibodies. Physical examination usually shows signs of appendicular and/or midline cerebellar dysfunction; ocular motor abnormalities usually include spontaneous down beat nystagmus, but broad array of ocular motor abnormalities have been reported. These ocular motor abnormalities may be more easily detected and characterized with instrumented testing. Other vestibular tests will not confirm a paraneoplastic syndrome, but may help exclude alternative diagnoses. Brain MRI sometimes will show cerebellar atrophy, less commonly enhancement. The differential diagnosis includes other cerebellar diseases. In a patient with stigmata of malignancy (e.g., weight loss, blood in stool, hemoptysis) in whom no cancer has yet been identified, it is reasonable to consider a paraneoplastic etiology of symptoms. If an apparently paraneoplastic syndrome occurs, then a search for malignancy should be undertaken, usually with CT of the chest/abdomen/pelvis without and with oral and intravenous contrast. If that is unrevealing, then a full-body FDG‑PET scan is reasonable. The main treatment is removal of the cancer. If that fails, then there are no clear guidelines about what to try next, though a variety of immunosuppressive/immunomodulatory strategies have been attempted. Prognosis is unfavorable.
Introduction
When the immune system identifies “non-self” antigens, it may mount an immune response. “Non-self” antigens may be present on infectious agents (viruses, bacteria), and the immune system response serves the purpose of destroying the invading pathogen. Cancerous cells, which are the body’s own cells whose genes have mutated, may express surface antigens that to the immune system appear “non-self,” and the immune system response serves the purpose of destroying the cancer cells (sometimes referred to as “immune surveillance”).
In some cases the “non-self” antigens (in the infectious agent or on the cancer cell) are similar to antigens of the body’s own healthy cells — sometimes called “molecular mimicry.” This is problematic because the immune system response to the infectious agent or cancer cell will mistakenly also attack the body’s own healthy cells. When this mis-directed immune attack is triggered by an infectious agent it is called a “para-infectious” phenomenon. When this mis-directed immune attack is triggered by a cancer it is called a “paraneoplastic” phenomenon.
These immune-mediated phenomena can attack any tissue in the body, including the nervous system. They can attack any part of the peripheral or central nervous system (Dalmau and Rosenfeld 2008). Within the central nervous system, the cerebellum is often affected; when triggered by an infectious agent this is called “para-infectious cerebellitis;” when triggered by a malignancy this is called “paraneoplastic cerebellar disease.”
In this section we will focus on paraneoplastic syndromes primarily or exclusively involving the cerebellum and audio-vestibular system.
Epidemiology
The prevalence of paraneoplastic syndromes is unknown (Dalmau and Rosenfeld 2008), and estimates vary dramatically, from 1 per 100,000 cancer patients (0.01%) to 553 per 60,000 cancer patients (0.9%) — a difference of nearly two orders of magnitude. Paraneoplastic syndromes have been reported with many different types of cancers.
Pathophysiological mechanism of disease
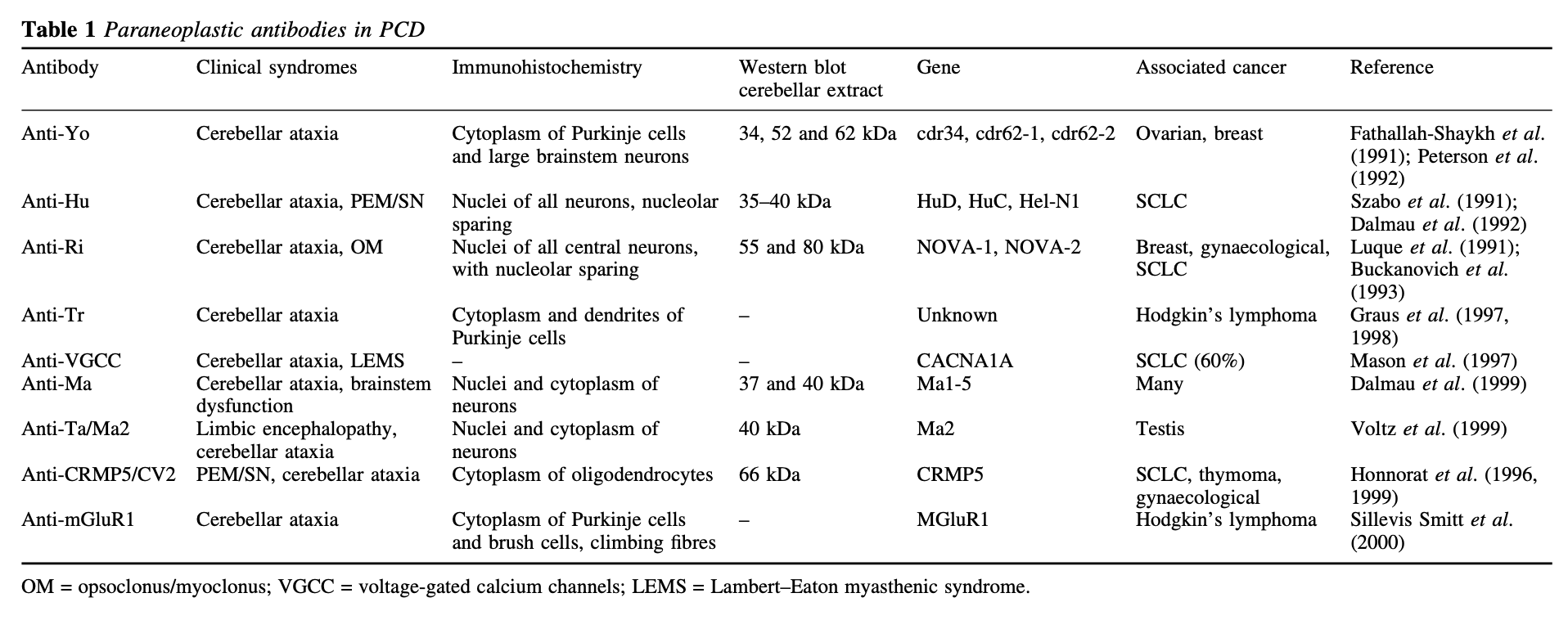
In a paraneoplastic syndrome, an immune response, whether humoral or cell-mediated (Dalmau and Rosenfeld 2008), erroneously attacks the body’s own healthy cells, presumably because of similarity between the epitopes of the cancer and the epitopes of healthy cells. A variety of antibodies has been identified.
Shams’ili and colleagues (Shams’ili, Grefkens et al. 2003) studied 50 patients with paraneoplastic cerebellar degeneration, and identified antibodies listed in the Table below.
Clinical presentation
Paraneoplastic syndromes have a wide range of tempo in symptom onset, ranging from hours to months.
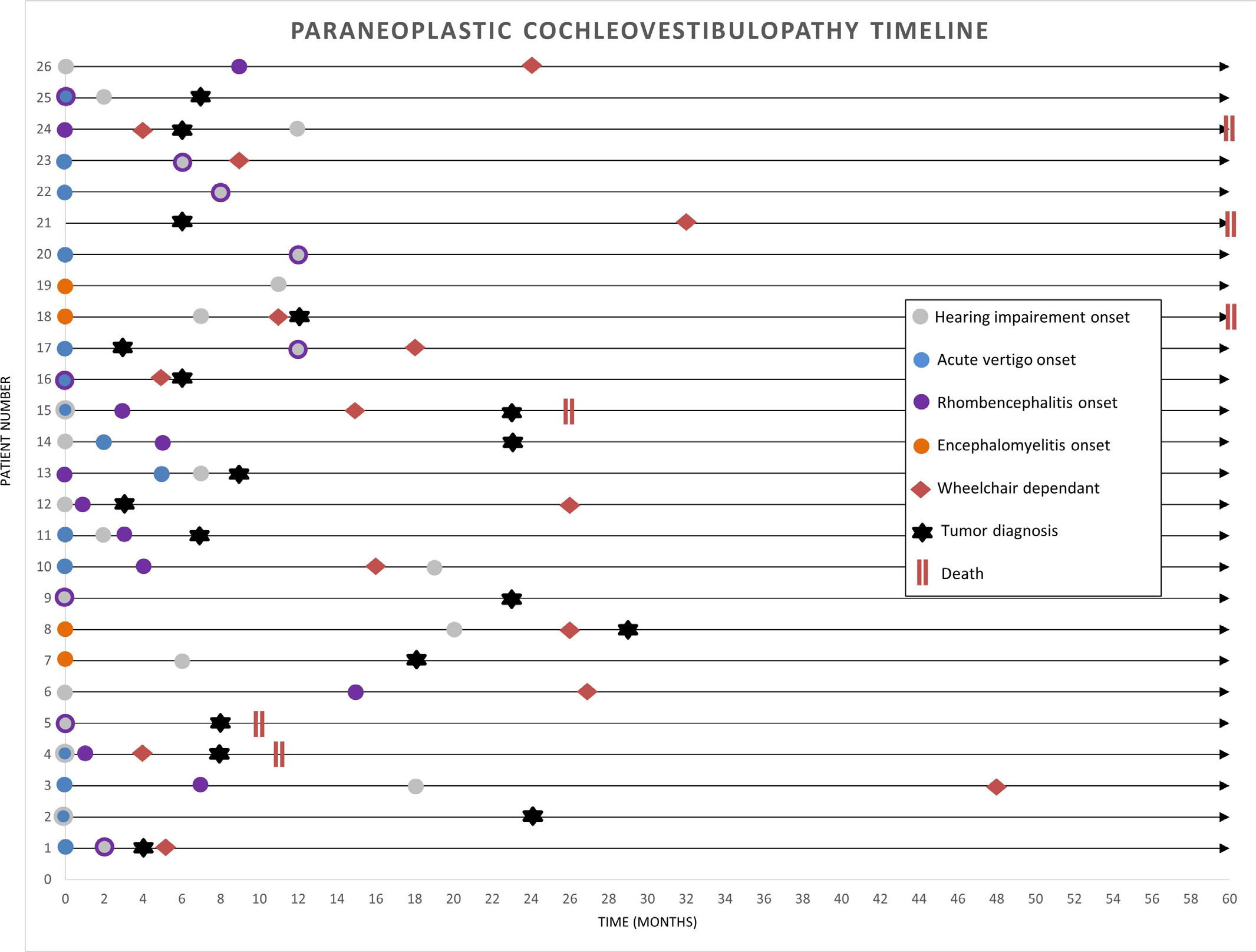
The Figure below, from Hammami and colleagues (Hammami, Eggers et al. 2021), shows the chronology of cochlear and vestibular symptom development in 26 patients with paraneoplastic syndromes.
Physical examination
The neurological symptoms of patients with paraneoplastic syndromes often manifests with cerebellar symptoms such as ataxia, dysarthric speech, and abnormal eye movements.
Audiologic evaluation
While not common, paraneoplastic syndromes can cause hearing loss, and on audiometry this can be unilateral, bilateral and symmetric, or bilateral and asymmetrical.
Hammami and colleagues (Hammami, Eggers et al. 2021) studied 26 patients with “cochleovestibular” paraneoplastic manifestations, and stated that 16/26 (62%) of these patients presented with both cochlear and vestibular symptoms, 8/26 (31%) presented only with hearing loss, and 2/26 (8%) presented only with tinnitus. Of the patients with hearing loss, 13/26 (50%) had asymmetrical progression.
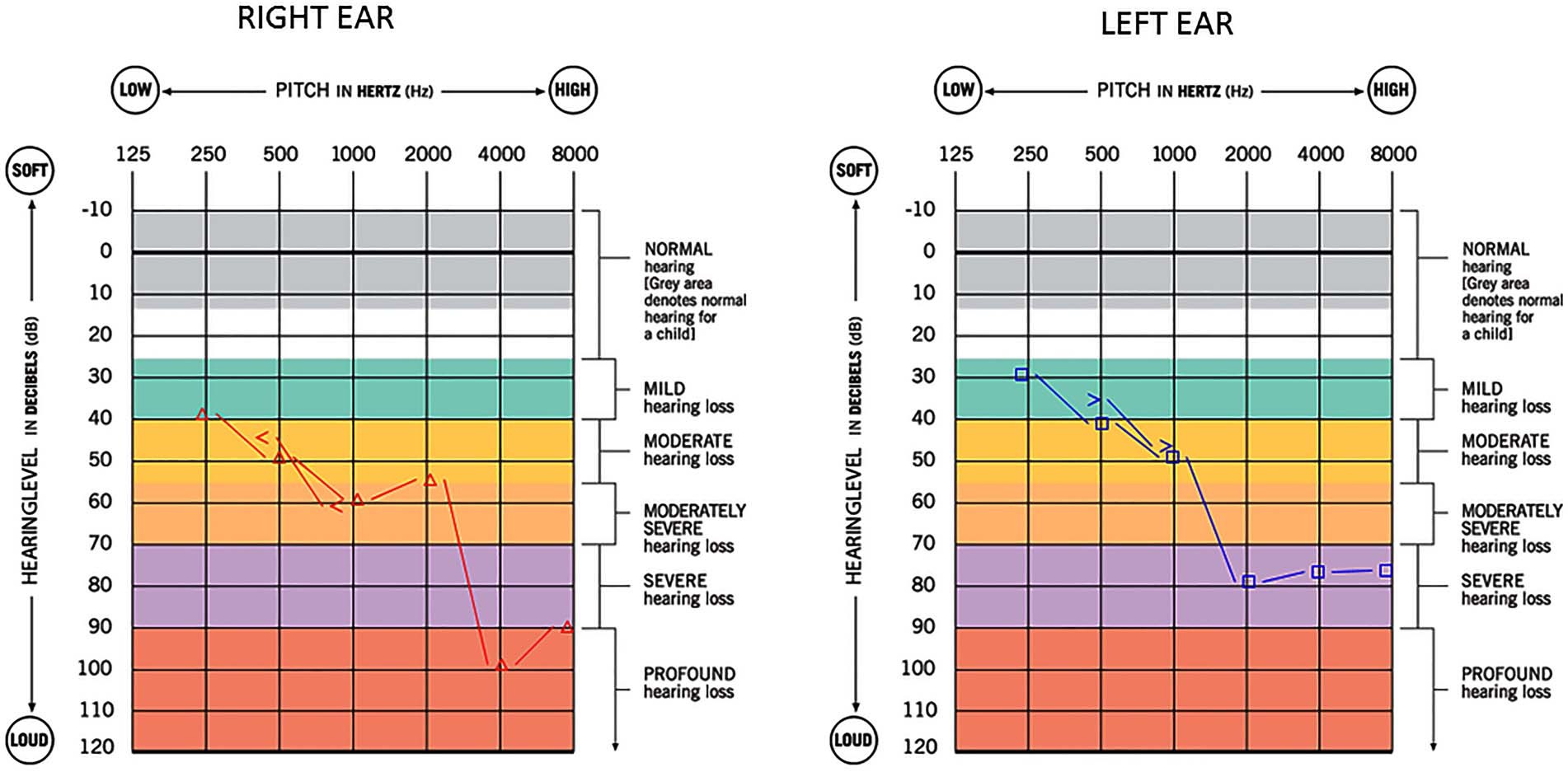
Kattah and colleagues (Kattah, Eggers et al. 2021) studied a 45-year-old man with a testicular seminoma whose paraneoplastic syndrome included bilateral, modestly asymmetrical, high greater than low frequency sensorineural hearing loss, as shown in the Figure below.
The hearing loss in paraneoplastic syndromes depends on the presence of malignancy, and thus can occur at any age. Since cancer tends to be more a disease of adults, it is unsurprising that a larger proportion of these cases occur in older age (Kearsley, Johnson et al. 1985), though it can occur in middle age (Kattah, Eggers et al. 2021) or even early childhood. For example, paraneoplastic hearing loss has been identified in a 24-month old child (by brainstem auditory evoked responses) with an anti‑Hu neuroblastoma (Fisher, Wechsler et al. 1994).
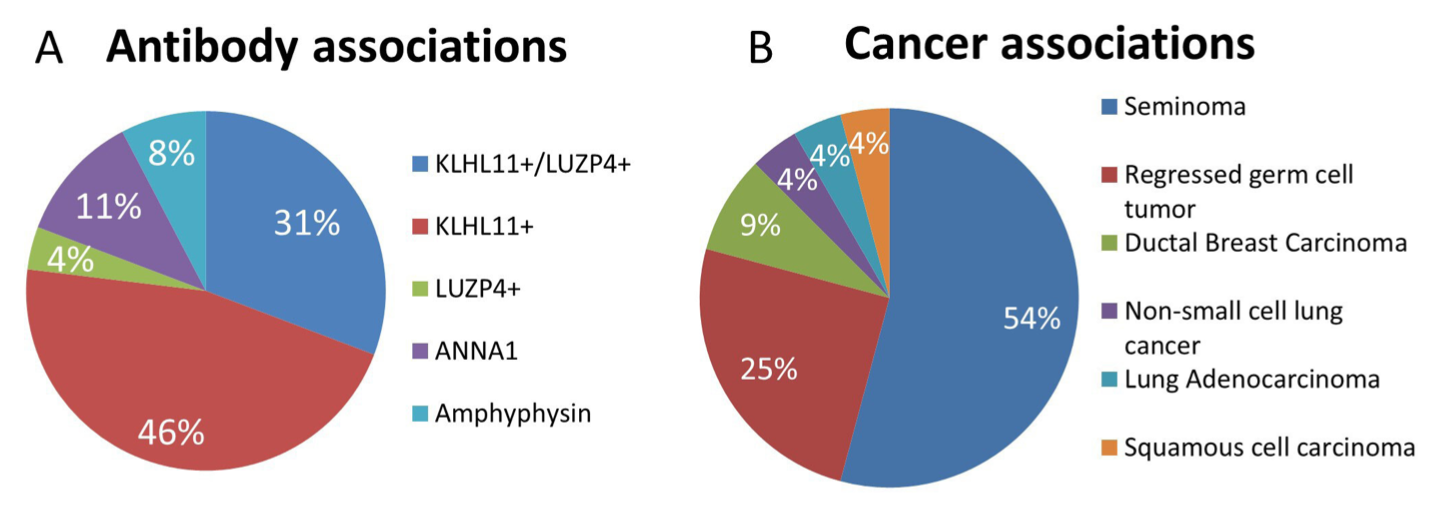
In paraneoplastic syndrome patients with cochlear and/or vestibular symptoms, the variety of antibodies and associated cancers is broad.
The Figure below, from Hammami and colleagues (Hammami, Eggers et al. 2021), shows the proportions of antibodies and associated cancers in 26 patients with paraneoplastic syndromes who had cochlear and/or vestibular symptoms.
Ocular motor examination
An impressive variety of ocular motor abnormalities has been reported in association with paraneoplastic syndromes, including:
- Centripetal nystagmus (Cherchi and Hac 2023; Hac et al. 2023)
- Spontaneous down beat nystagmus (Kearsley, Johnson et al. 1985, Guy and Schatz 1988, McLellan, Currie et al. 1988, Hammack, Kotanides et al. 1992, McCrystal, Anderson et al. 1995, Bussiere, Al-Khotani et al. 2008, Ogawa, Sakakibara et al. 2011, Choi, Park et al. 2014, Tsuyusaki, Sakakibara et al. 2014, Martin, Dillon et al. 2017, Kattah, Eggers et al. 2021, Kherallah, Samaha et al. 2022, Shivaram, Tallapalli et al. 2022, Khatib, Do et al. 2023)
- Elliptical nystagmus (Mistry, Lee et al. 2016)
- Abnormal visual fixation suppression (Kearsley, Johnson et al. 1985, Ogawa, Sakakibara et al. 2011, Kattah, Eggers et al. 2021, Kherallah, Samaha et al. 2022)
- Gaze evoked nystagmus, both horizontal and vertical (Taylor, Mason et al. 1999, Choi, Park et al. 2014)
- Ocular flutter (Kearsley, Johnson et al. 1985, Furman, Eidelman et al. 1988, Kruger, Yonekawa et al. 2014, Shivaram, Tallapalli et al. 2022)
- Opsoclonus, usually in the context of opsoclonus-myoclonus (Fisher, Wechsler et al. 1994, Desai and Mitchell 2012)
- Periodic alternating nystagmus (Ogawa, Sakakibara et al. 2011, Eggers, Pittock et al. 2012)
- Perverted nystagmus (Inui, Saito et al. 2020)
- Positional nystagmus (Kearsley, Johnson et al. 1985, Choi, Park et al. 2014)
- Abnormalities on rotatory chair testing, such as low gain and phase lead (Kattah, Eggers et al. 2021), suggestive of bilateral vestibular weakness
- Rebound nystagmus (Hac, Murphy et al. 2023)
- Saccadic dysmetria (Wray, Dalmau et al. 2011, Wray, Martinez-Hernandez et al. 2011)
- Saccadic intrusions (Furman, Eidelman et al. 1988, Wray, Martinez-Hernandez et al. 2011)
- See-saw nystagmus (Rizvi, Cameron et al. 2018)
- Skew deviation (McLellan, Currie et al. 1988, Taylor, Mason et al. 1999, Martin, Dillon et al. 2017)
- Smooth pursuit abnormalities, usually with saccadic intrusions (Kearsley, Johnson et al. 1985, Wray, Dalmau et al. 2011, Wray, Martinez-Hernandez et al. 2011, Martin, Dillon et al. 2017, Kattah, Eggers et al. 2021, Kherallah, Samaha et al. 2022)
- Spontaneous up beat nystagmus (Kearsley, Johnson et al. 1985, Wray, Dalmau et al. 2011, Wray, Martinez-Hernandez et al. 2011, Garcia-Reitboeck, Thompson et al. 2014, Cherchi 2015)
- Windmill nystagmus (Lee, Kim et al. 2018)
The majority of these ocular motor abnormalities localize to the cerebellum; some localize to the brainstem.
Testing: vestibular
Instrumented ocular motor testing (such as with videonystagmography) may characterize the eye movement abnormalities better than a face-to-face examination.
Computerized dynamic posturography may show increased postural sway (Wessel, Diener et al. 1988), but this is neither sensitive nor specific for paraneoplastic cerebellar degeneration.
Other tests (such as vestibular evoked myogenic potentials, video head impulse testing) have not been studied in paraneoplastic cerebellar degeneration. Their main value is to exclude competing diagnoses.
Imaging
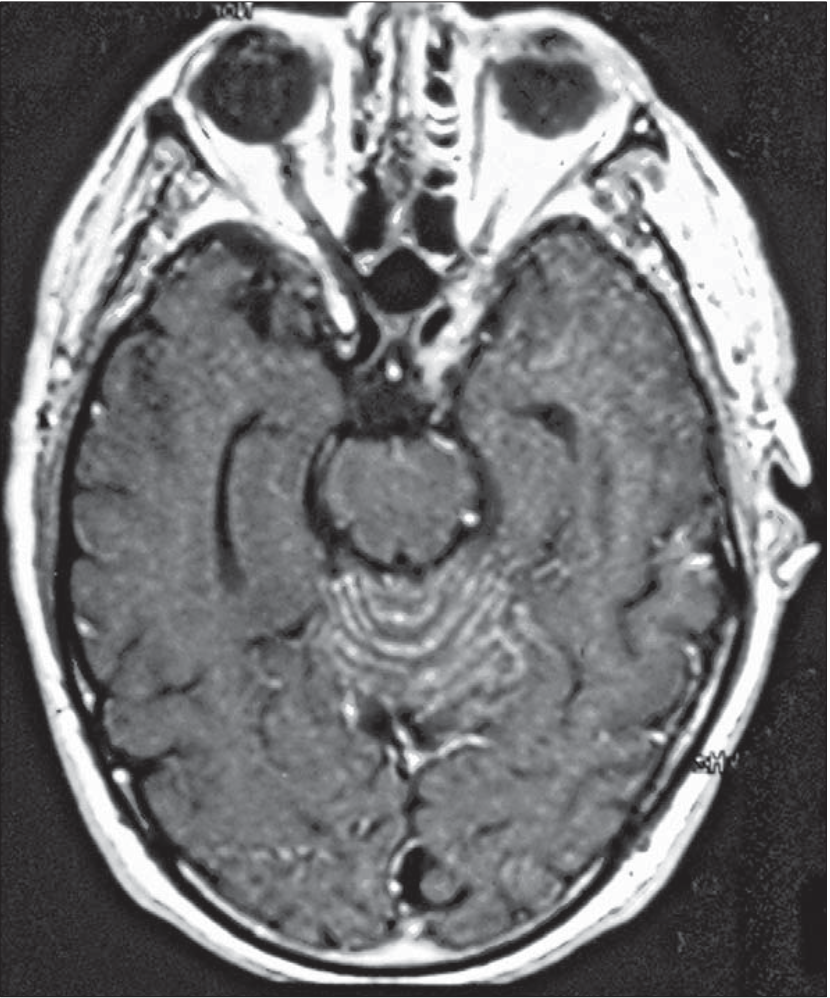
Brain MRI may show cerebellar atrophy in paraneoplastic cerebellar degeneration (Schlake, Husstedt et al. 1989, Akpinar, Berk et al. 1990, Emir, Kutluk et al. 2000, Scheid, Voltz et al. 2006, Fancellu, Corsini et al. 2014, Venkatraman and Opal 2016, Escudero-Fernandez, Garcia-Carpintero et al. 2020). Less commonly there may be enhancement, particularly of the cerebellar folia (Darnell and Posner 2003, Dalmau and Rosenfeld 2008).
The Figure below, from Dalmau and colleagues (Dalmau and Rosenfeld 2008) shows contrast enhancement of the sulci of the cerebellar vermis.
Histopathology
Paraneoplastic cerebellar degeneration characteristically shows extensive loss of Purkinje cells, as well as degeneration in the deep cerebellar nuclei and inferior olivary nuclei (Dalmau and Rosenfeld 2008).
Differential diagnosis
In a patient with neurological symptoms compatible with a paraneoplastic syndrome, in whom a cancer has already been identified, and paraneoplastic antibodies have already been detected, the diagnosis of a paraneoplastic syndrome is secure.
However, the clinical scenario is often not so straightforward; either the symptom presentation may not be “classic” for a paraneoplastic syndrome, or no malignancy has yet been identified, or no antibodies have been detected (Dalmau and Rosenfeld 2008). For this reason, diagnostic criteria have been suggested.
The Table below, from Graus and colleagues (Dalmau and Rosenfeld 2008), reviews the diagnostic criteria for definite and for possible paraneoplastic neurological syndromes.

The Figure below, from Graus and colleagues (Dalmau and Rosenfeld 2008), shows the diagnostic clinical criteria in a flow-chart format.

The differential diagnosis of a paraneoplastic cerebellar syndrome includes other cerebellar insults. Since some cases of paraneoplastic cerebellar syndromes evolve over many months, they may be mistaken for a primary neurodegenerative process, such as multiple systems atrophy or a late-onset cerebellar ataxia.
Treatment
The standard of care in paraneoplastic syndromes with an identified malignancy is to remove the cancer. After that, if symptoms persist or worsen, there are no clear guidelines on how to proceed (Darnell and Posner 2003, Dalmau and Rosenfeld 2008). A variety of immune system manipulation strategies have been attempted, including steroids such as methylprednisolone (Keime-Guibert, Graus et al. 2000), other immunosuppressants — cyclophosphamide (Keime-Guibert, Graus et al. 2000), tacrolimus (Albert, Austin et al. 2000), rituximab (Esposito, Penza et al. 2008) — plasmapheresis (Cocconi, Ceci et al. 1985), intravenous immunoglobulin (Uchuya, Graus et al. 1996, Keime-Guibert, Graus et al. 2000, Widdess-Walsh, Tavee et al. 2003, Phuphanich and Brock 2007) and others. While there are occasional reports of dramatic responses to such interventions (Croteau, Owainati et al. 2001), in most cases the outcome is not satisfactory.
For a patient with suspected paraneoplastic syndrome a search should be undertaken for malignancy. This often involves a CT of the chest, abdomen and pelvis, with and without intravenous and oral contrast. If that is unrevealing, then a full-body FDG‑PET scan is reasonable (Marchand, Graveleau et al. 2007). Some investigators advocate doing CT and FDG‑PET simultaneously (Linke, Schroeder et al. 2004, Frings, Antoch et al. 2005). If no malignancy is identified, then close follow-up is appropriate, with the idea that an occult malignancy will eventually declare itself.
Prognosis
Even when malignancy is treated and immunomodulatory therapies are attempted, prognosis is unfavorable.
References
Akpinar S, Berk O, Karaca L (1990) Paraneoplastic cerebellar degeneration associated with multiple myeloma–3.4 years follow up. J Neurooncol 9: 63-8. doi: 10.1007/BF00167070
Albert ML, Austin LM, Darnell RB (2000) Detection and treatment of activated T cells in the cerebrospinal fluid of patients with paraneoplastic cerebellar degeneration. Ann Neurol 47: 9-17.
Bussiere M, Al-Khotani A, Steckley JL, Nicolle M, Nicolle D (2008) Paraneoplastic downbeat nystagmus. Can J Ophthalmol 43: 243-5. doi: 10.3129/i08-026
Cherchi M (2015) Paraneoplastic upbeat nystagmus in renal cell carcinoma. Journal of Neurology and Neurophysiology 6. doi: 10.4172/2155-9562.1000287
Cherchi M, Hac NEF (2023) Paraneoplastic Gaze-Evoked Centripetal Nystagmus in Ovarian Teratoma. Journal of Neuro-Ophthalmology. doi: 10.1097/WNO.0000000000002027
Choi SY, Park SH, Kim HJ, Kim JS (2014) Paraneoplastic downbeat nystagmus associated with cerebellar hypermetabolism especially in the nodulus. J Neurol Sci 343: 187-91. doi: 10.1016/j.jns.2014.05.030
Cocconi G, Ceci G, Juvarra G, Minopoli MR, Cocchi T, Fiaccadori F, Lechi A, Boni P (1985) Successful treatment of subacute cerebellar degeneration in ovarian carcinoma with plasmapheresis. A case report. Cancer 56: 2318-20. doi: 10.1002/1097-0142(19851101)56:9<2318::aid-cncr2820560930>3.0.co;2-9
Croteau D, Owainati A, Dalmau J, Rogers LR (2001) Response to cancer therapy in a patient with a paraneoplastic choreiform disorder. Neurology 57: 719-22. doi: 10.1212/wnl.57.4.719
Dalmau J, Rosenfeld MR (2008) Paraneoplastic syndromes of the CNS. Lancet Neurol 7: 327-40. doi: 10.1016/S1474-4422(08)70060-7
Darnell RB, Posner JB (2003) Paraneoplastic syndromes involving the nervous system. N Engl J Med 349: 1543-54. doi: 10.1056/NEJMra023009
Desai J, Mitchell WG (2012) Acute cerebellar ataxia, acute cerebellitis, and opsoclonus-myoclonus syndrome. J Child Neurol 27: 1482-8. doi: 10.1177/0883073812450318
Eggers SD, Pittock SJ, Shepard NT, Habermann TM, Neff BA, Klebig RR (2012) Positional periodic alternating vertical nystagmus with PCA-Tr antibodies in Hodgkin lymphoma. Neurology 78: 1800-2. doi: 10.1212/WNL.0b013e3182583085
Emir S, Kutluk MT, Gogus S, Buyukpamukcu M (2000) Paraneoplastic cerebellar degeneration and Horner syndrome: association of two uncommon findings in a child with Hodgkin disease. J Pediatr Hematol Oncol 22: 158-61. doi: 10.1097/00043426-200003000-00015
Escudero-Fernandez JM, Garcia-Carpintero AS, Delgado-Alvarez I, Castellote-Alonso A, Vazquez-Mendez EJ (2020) Brain paraneoplastic syndromes in a patient with mediastinal ganglioneuroma. Radiol Case Rep 15: 2059-2062. doi: 10.1016/j.radcr.2020.08.010
Esposito M, Penza P, Orefice G, Pagano A, Parente E, Abbadessa A, Bonavita V (2008) Successful treatment of paraneoplastic cerebellar degeneration with Rituximab. J Neurooncol 86: 363-4. doi: 10.1007/s11060-007-9479-z
Fancellu R, Corsini E, Bernardi G, Buzzo P, Ferrari ML, Lamantea E, Garaventa A, Truini M, Salvarani S (2014) Paraneoplastic cerebellar ataxia associated with anti-Hu antibodies and benign ganglioneuroma. Funct Neurol 29: 277-80.
Fisher PG, Wechsler DS, Singer HS (1994) Anti-Hu antibody in a neuroblastoma-associated paraneoplastic syndrome. Pediatr Neurol 10: 309-12. doi: 10.1016/0887-8994(94)90127-9
Frings M, Antoch G, Knorn P, Freudenberg L, Bier U, Timmann D, Maschke M (2005) Strategies in detection of the primary tumour in anti-Yo associated paraneoplastic cerebellar degeneration. J Neurol 252: 197-201. doi: 10.1007/s00415-005-0635-0
Furman JM, Eidelman BH, Fromm GH (1988) Spontaneous remission of paraneoplastic ocular flutter and saccadic intrusions. Neurology 38: 499-501. doi: 10.1212/wnl.38.3.499
Garcia-Reitboeck P, Thompson G, Johns P, Al Wahab Y, Omer S, Griffin C (2014) Upbeat nystagmus in anti-Ma2 encephalitis. Pract Neurol 14: 36-8. doi: 10.1136/practneurol-2013-000524
Guy JR, Schatz NJ (1988) Paraneoplastic downbeating nystagmus. A sign of occult malignancy. J Clin Neuroophthalmol 8: 269-72.
Hac NEF, Murphy OC, Butala AA, Newsome SD, Gold DR (2023) Centripetal Nystagmus, Slow Saccades, Cerebellar Ataxia, and Parkinsonism in a Patient With Anti-GAD65-Associated Stiff Person Syndrome Spectrum Disorder. Journal of Neuro-Ophthalmology: 1-5. doi: 10.1097/WNO.0000000000001774
Hammack J, Kotanides H, Rosenblum MK, Posner JB (1992) Paraneoplastic cerebellar degeneration. II. Clinical and immunologic findings in 21 patients with Hodgkin’s disease. Neurology 42: 1938-43. doi: 10.1212/wnl.42.10.1938
Hammami MB, Eggers SDZ, Madhavan A, Montalvo MJ, Pittock SJ, Dubey D (2021) Paraneoplastic cochleovestibulopathy: clinical presentations, oncological and serological associations. J Neurol Neurosurg Psychiatry 92: 1181-1185. doi: 10.1136/jnnp-2021-326775
Inui R, Saito K, Shimomura Y, Yamashita D, Kawamoto M, Ishikawa T (2020) Anti-Ma-associated paraneoplastic cerebellar degeneration in a patient with nodular lymphocyte-predominant Hodgkin lymphoma: a case report. BMC Neurol 20: 355. doi: 10.1186/s12883-020-01929-4
Kattah JC, Eggers SD, Bach SE, Dubey D, McKeon AB (2021) Paraneoplastic Progressive Downbeat Nystagmus, Ataxia and Sensorineural Hearing Loss due to the ANTI-Kelch-11 Protein Antibody. J Neuroophthalmol 41: 261-265. doi: 10.1097/WNO.0000000000001194
Kearsley JH, Johnson P, Halmagyi GM (1985) Paraneoplastic cerebellar disease. Remission with excision of the primary tumor. Arch Neurol 42: 1208-10. doi: 10.1001/archneur.1985.04060110090026
Keime-Guibert F, Graus F, Fleury A, Rene R, Honnorat J, Broet P, Delattre JY (2000) Treatment of paraneoplastic neurological syndromes with antineuronal antibodies (Anti-Hu, anti-Yo) with a combination of immunoglobulins, cyclophosphamide, and methylprednisolone. J Neurol Neurosurg Psychiatry 68: 479-82. doi: 10.1136/jnnp.68.4.479
Khatib L, Do LD, Benaiteau M, Villagran-Garcia M, Scharf M, Meyer P, Haidar LA, Demeret S, Honnorat J (2023) Autoimmune Cerebellar Ataxia Associated with Anti-Glutamate Receptor delta2 Antibodies: a Rare but Treatable Entity. Cerebellum. doi: 10.1007/s12311-023-01523-7
Kherallah B, Samaha E, Bach SE, Guede C, Kattah JC (2022) Case report: Acute vestibular syndrome and cerebellitis in anti-Yo paraneoplastic syndrome. Front Neurol 13: 960584. doi: 10.3389/fneur.2022.960584
Kruger JM, Yonekawa Y, Skidd P, Cestari DM (2014) Ocular flutter as the presenting sign of lung adenocarcinoma. Digit J Ophthalmol 20: 4-6. doi: 10.5693/djo.02.2013.10.002
Lee SU, Kim HJ, Oh SW, Song EY, Choi JY, Kim JS (2018) Pearls & Oy-sters: Windmill nystagmus in paraneoplastic cerebellar degeneration. Neurology 91: e1831-e1833. doi: 10.1212/WNL.0000000000006477
Linke R, Schroeder M, Helmberger T, Voltz R (2004) Antibody-positive paraneoplastic neurologic syndromes: value of CT and PET for tumor diagnosis. Neurology 63: 282-6. doi: 10.1212/01.wnl.0000129983.06983.4e
Marchand V, Graveleau J, Lanctin-Garcia C, Bourbouloux E, Bridji B, Resche I, Kraeber-Bodere F, Rousseau C (2007) A rare gynecological case of paraneoplastic cerebellar degeneration discovered by FDG-PET. Gynecol Oncol 105: 545-7. doi: 10.1016/j.ygyno.2007.01.047
Martin AN, Dillon PM, Jones DE, Brenin DR, Lapides DA (2017) Anti-Yo Mediated Paraneoplastic Cerebellar Degeneration Associated with Pseudobulbar Affect in a Patient with Breast Cancer. Case Rep Oncol Med 2017: 8120689. doi: 10.1155/2017/8120689
McCrystal M, Anderson NE, Jones RW, Evans BD (1995) Paraneoplastic cerebellar degeneration in a patient with chemotherapy-responsive ovarian cancer. Int J Gynecol Cancer 5: 396-399. doi: 10.1046/j.1525-1438.1995.05050396.x
McLellan R, Currie JL, Royal W, Rosenshein NB (1988) Ovarian carcinoma and paraneoplastic cerebellar degeneration. Obstet Gynecol 72: 922-5. doi: 10.1097/00006250-198812000-00023
Mistry EA, Lee AG, Lai EC (2016) Teaching Video NeuroImages: P/Q-type voltage-gated calcium channel-associated paraneoplastic elliptical nystagmus. Neurology 87: e109. doi: 10.1212/WNL.0000000000003068
Ogawa E, Sakakibara R, Kawashima K, Yoshida T, Kishi M, Tateno F, Kataoka M, Kawashima T, Yamamoto M (2011) VGCC antibody-positive paraneoplastic cerebellar degeneration presenting with positioning vertigo. Neurol Sci 32: 1209-12. doi: 10.1007/s10072-011-0648-7
Phuphanich S, Brock C (2007) Neurologic improvement after high-dose intravenous immunoglobulin therapy in patients with paraneoplastic cerebellar degeneration associated with anti-Purkinje cell antibody. J Neurooncol 81: 67-9. doi: 10.1007/s11060-006-9198-x
Rizvi MT, Cameron L, Kilbane C, Shaikh AG (2018) Paraneoplastic seesaw nystagmus and opsoclonus provides evidence for hyperexcitable reciprocally innervating mesencephalic network. J Neurol Sci 390: 239-245. doi: 10.1016/j.jns.2018.05.002
Scheid R, Voltz R, Briest S, Kluge R, von Cramon DY (2006) Clinical insights into paraneoplastic cerebellar degeneration. J Neurol Neurosurg Psychiatry 77: 529-30. doi: 10.1136/jnnp.2005.082206
Schlake HP, Husstedt IW, Grotemeyer KH, Potter R (1989) Paraneoplastic subacute cerebellar degeneration in Hodgkin’s disease. Report of three cases and review of the literature. Clin Neurol Neurosurg 91: 329-35. doi: 10.1016/0303-8467(89)90010-3
Shams’ili S, Grefkens J, de Leeuw B, van den Bent M, Hooijkaas H, van der Holt B, Vecht C, Sillevis Smitt P (2003) Paraneoplastic cerebellar degeneration associated with antineuronal antibodies: analysis of 50 patients. Brain 126: 1409-18. doi: 10.1093/brain/awg133
Shivaram S, Tallapalli AVR, Gupta M, Nashi S, Kulkarni GB, Alladi S (2022) Spontaneous Downbeat Nystagmus in Anti-GAD-Antibody-Associated Paraneoplastic Syndrome. J Neurosci Rural Pract 13: 546-549. doi: 10.1055/s-0042-1749404
Taylor RB, Mason W, Kong K, Wennberg R (1999) Reversible paraneoplastic encephalomyelitis associated with a benign ovarian teratoma. Can J Neurol Sci 26: 317-20. doi: 10.1017/s0317167100000469
Tsuyusaki Y, Sakakibara R, Kishi M, Tateno F, Yoshida T (2014) Downbeat nystagmus as the initial manifestation of anti-NMDAR encephalitis. Neurol Sci 35: 125-6. doi: 10.1007/s10072-013-1511-9
Uchuya M, Graus F, Vega F, Rene R, Delattre JY (1996) Intravenous immunoglobulin treatment in paraneoplastic neurological syndromes with antineuronal autoantibodies. J Neurol Neurosurg Psychiatry 60: 388-92. doi: 10.1136/jnnp.60.4.388
Venkatraman A, Opal P (2016) Paraneoplastic cerebellar degeneration with anti-Yo antibodies – a review. Ann Clin Transl Neurol 3: 655-63. doi: 10.1002/acn3.328
Wessel K, Diener HC, Dichgans J, Thron A (1988) Cerebellar dysfunction in patients with bronchogenic carcinoma: clinical and posturographic findings. J Neurol 235: 290-6. doi: 10.1007/BF00314176
Widdess-Walsh P, Tavee JO, Schuele S, Stevens GH (2003) Response to intravenous immunoglobulin in anti-Yo associated paraneoplastic cerebellar degeneration: case report and review of the literature. J Neurooncol 63: 187-90. doi: 10.1023/a:1023931501503
Wray SH, Dalmau J, Chen A, King S, Leigh RJ (2011a) Paraneoplastic disorders of eye movements. Ann N Y Acad Sci 1233: 279-84. doi: 10.1111/j.1749-6632.2011.06113.x
Wray SH, Martinez-Hernandez E, Dalmau J, Maheshwari A, Chen A, King S, Bishop Pitman M, Leigh RJ (2011b) Paraneoplastic upbeat nystagmus. Neurology 77: 691-3. doi: 10.1212/WNL.0b013e318229e6a5
![]()