By Marcello Cherchi, MD PhD
For patients
Semicircular canal dehiscence (SCD) is an abnormal connection (hole) between the inner ear and some other space. It can occur spontaneously or sometimes following trauma. Symptoms can include some combination of disequilibrium, hearing loss, overly sensitive hearing, and tinnitus. Your physician may order a hearing test and vestibular evoked myogenic potentials; depending on those results, imaging (usually a temporal bone CT) may be appropriate. If a diagnosis of SCD is confirmed, then management depends on how intrusive or limiting the symptoms are. If symptoms are tolerable, then no treatment is needed. If symptoms are intolerable, then surgical repair of SCD may be warranted.
For practitioners
Overview
In semicircular canal dehiscence (SCD) a bony dehiscence creates an abnormal connection between one of the semicircular canals with another space, and thus is classified as one of several so-called “third window phenomena.” Clinically this can manifest with auditory symptoms (hyperacusis, autophony, pulsatile tinnitus), or vestibular symptoms (Valsalva-induced disequilibrium), or both. The superior semicircular canal is the most commonly involved, though any of the semicircular canals can be affected. Overall SCD is rare. It can occur spontaneously, but has also been documented after head trauma. The diagnosis is suspected based on a combination of a compatible clinical history, results of audiometry (conductive hyperacusis on the affected side) and vestibular evoked myogenic potentials (abnormally large amplitude and abnormally low threshold on the affected side). The diagnosis can be radiographically confirmed on a high resolution temporal bone CT. Not all cases require treatment; in those that do, surgery can be considered; options include canal plugging and (in the case of the superior semicircular canal) canal re-roofing.
Introduction
Semicircular canal dehiscence (SCD) is one of several so-called “third window phenomena” (Ota, Sakagami et al. 2021). In semicircular canal dehiscence an abnormal hole connects one of the semicircular canals with another space, and this has the two consequences. First, less energy is required to stimulate inner ear hair cells; clinically this manifests with overly sensitive hearing (and conductive hyperacusis on audiometry). Second, since the hole is within the semicircular canals, fluid waves will propagate from the oval window to the semicircular canal and thereby stimulate vestibular hair cells; clinically this manifests with oscillopsia from Tullio’s phenomenon (loud sounds cause disequilibrium), and ocular motor examination may show corresponding nystagmus.
A “third window” can appear in any of the three semicircular canals. Superior semicircular canal dehiscence (SSCD) is by far the most common variant of this problem, whereas lateral semicircular canal dehiscence (LSCD) and posterior semicircular canal dehiscence (PSCD) are much rarer.
The first SCD described was that involving the superior canal. This was recognized and reported by Minor and colleagues (Minor, Solomon et al. 1998).
The Figure below shows post-mortem surgical pathology of a patient with bilateral superior SCD reported by Carey and colleagues (Carey, Minor et al. 2000).
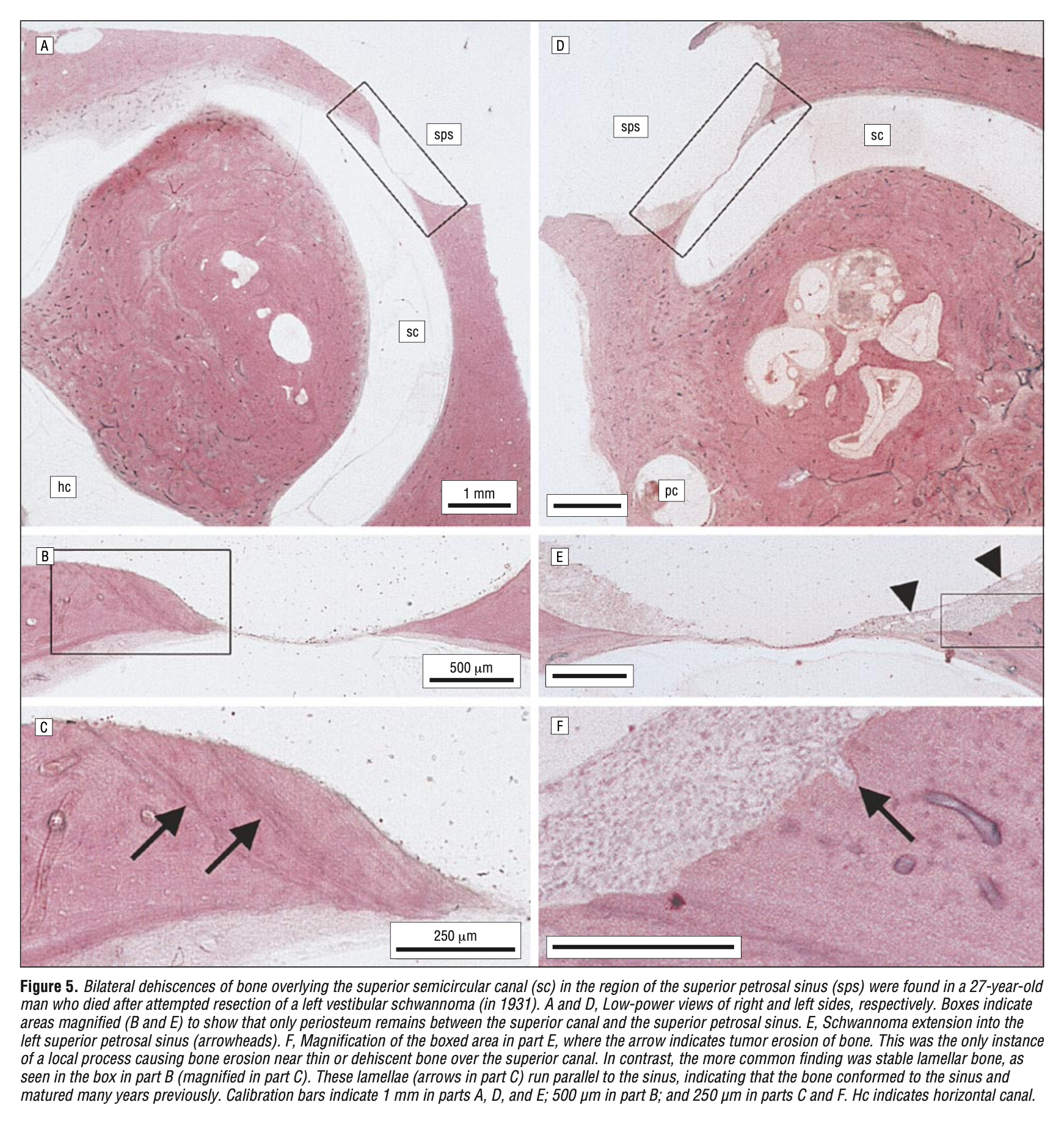
SCD is rare. It can occur spontaneously or after trauma. The clinical history includes some combination of vestibular symptoms, hearing loss, hyperacusis and tinnitus, with exacerbations often caused by Valsalva. On physical examination, Rinne’s test may show that bone conduction is superior to air conduction, and ocular motor examination may show nystagmus elicited by loud sounds. Audiometry often shows conductive hyperacusis in the affected ear. On vestibular evoked myogenic potentials (whether cervical or ocular) the affected ear may show an unusually large amplitude, and a low threshold. The imaging modality of choice is a temporal bone CT without contrast, with reconstructions in Stenver’s and Pöschl’s planes. If symptoms are tolerable than management can be expectant. If symptoms are intolerable, then treatment is surgical, though surgery is not always successful or durable.
Epidemiology
Semicircular canal dehiscence is rare, though the exact incidence and prevalence of SCD is unknown. Part of what makes this difficult to study is the incomplete overlap between the anatomical abnormality and the symptoms.
The phrase “semicircular canal dehiscence” is merely a description of anatomical pathology and is neutral with respect to any clinical manifestations. In contrast, the phrase “semicircular canal dehiscence syndrome” refers to a circumstance in which the anatomical abnormality is present, and it is believed to account for a specific set of symptoms.
This nomenclatural distinction draws attention to the fact that there is incomplete overlap between (1) the group of individuals with radiographic SCD, and (2) the group of individuals with symptoms compatible with SCD.
Clinical history
Semicircular canal dehiscence can occur after head trauma (McCrary, Babajanian et al. 2021), though it can also occur spontaneously. Some “spontaneously occurring” cases are sometimes suspected to be due to strong Valsalva events, such as coughing or sneezing. The symptoms of SCD are variable.
The Chart below from Naert and colleagues (Naert, Van de Berg et al. 2018) shows the frequency of symptoms in a meta-analysis that included 431 patients from 66 articles about superior SCD.
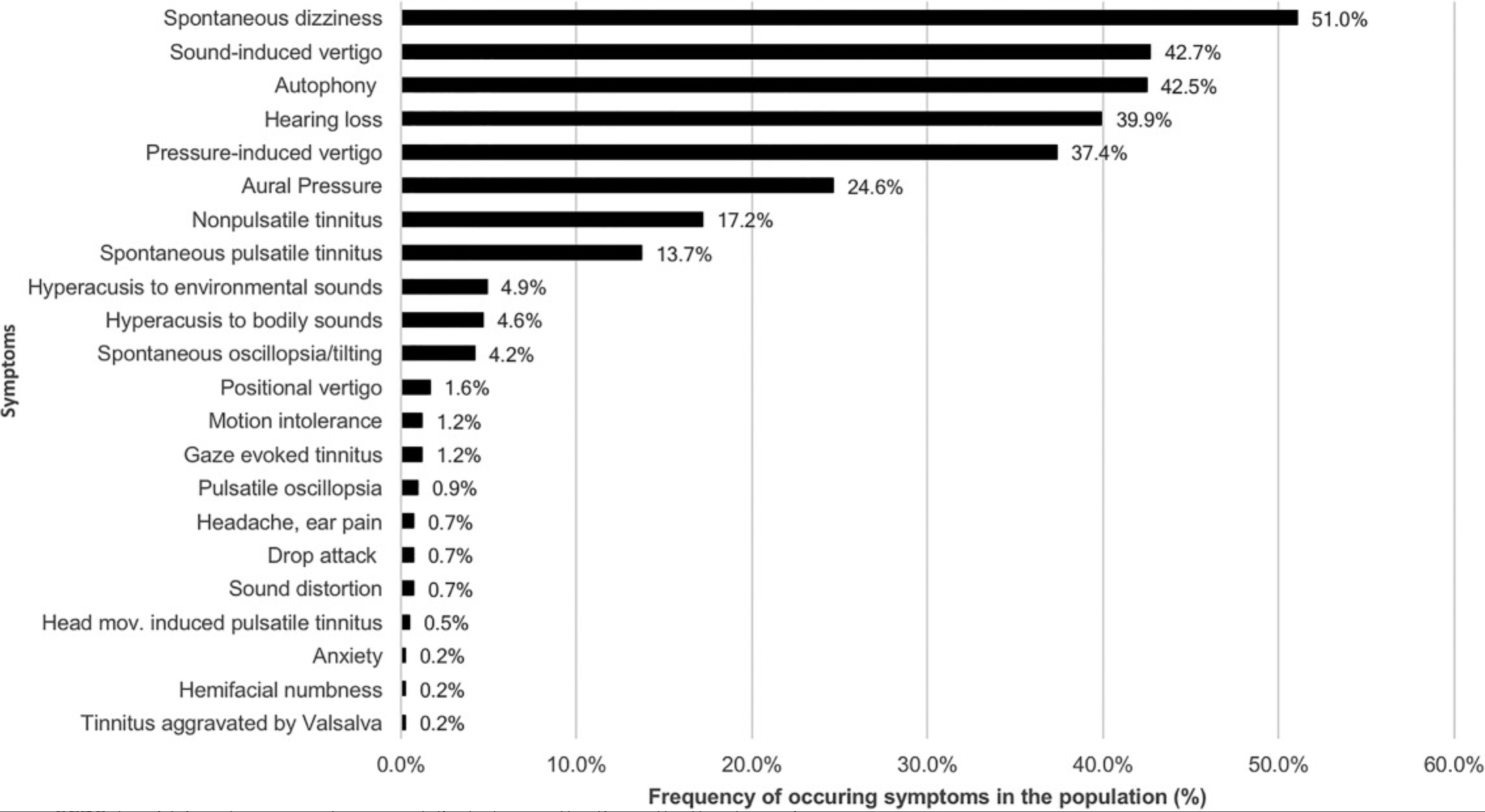
A re-analysis of these data by category of symptom is shown below.
- Vestibular symptoms
- Spontaneous dizziness (51.0%)
- Sound-induced vertigo (42.7%)
- Positional vertigo (1.6%)
- Motion intolerance (1.2%)
- Drop attack (0.7%)
- Hearing loss
- Hearing loss (39.9%)
- Hyperacusis
- Autophony (42.5%)
- Hyperacusis to environmental sounds (4.9%)
- Hyperacusis to bodily sounds (4.6%)
- Tinnitus
- Non-pulsatile tinnitus (17.2%)
- Spontaneous pulsatile tinnitus (13.7%)
- Gaze-evoked tinnitus (1.2%)
- Head-movement-induced pulsatile tinnitus (0.5%)
- Tinnitus aggravated by Valsalva (0.2%)
- Sound distortion (0.7%)
- Other aural symptoms
- Aural pressure (24.6%)
- Ear pain (0.7%)
- Visual symptoms
- Spontaneous oscillopsia/tilting (4.2%)
- Pulsatile oscillopsia (0.9%)
- Other symptoms
- Anxiety (0.2%)
- Hemifacial numbness (0.2%)
In aggregate, the most common symptom categories of superior SCD are vestibular, hearing loss, hyperacusis and tinnitus.
In the author’s clinical experience, a fair proportion of patients complain of “hearing my eyeballs move,” which perhaps corresponds to the “gaze-evoked tinnitus” category in the analysis of Naert and colleagues (Naert, Van de Berg et al. 2018) above. While it is not frequent, we have found this symptom to be sensitive for superior SCD. Some authors have documented patients also reporting that they can “hear” their eye-blinks (Bertholon, Reynard et al. 2018).
Physical examination
On physical examination using a tuning for (usually 128 Hz), Rinne’s test may show that an ear affected by superior SCD may have bone conduction superior to air conduction — in other words, conductive hyperacusis.
Ocular motor examination (especially if done with infrared video oculography) may show oscillatory nystagmus in response to a loud tone (the ocular motor correlate of Tullio’s phenomenon). In some cases there will be a pendular nystagmus (usually with torsional and vertical components) synchronous with the cardiac pulse (Tilikete, Krolak-Salmon et al. 2004, Hain and Cherchi 2008, Milenkovic, Sycha et al. 2021).
When an examiner applies the stem of a vibrating 128 Hz tuning fork to either medial malleolus of a patient with unilateral superior semicircular canal dehiscence, the patient may “hear” the tone in the affected ear. This finding is similar to autophony, except that the vibrations in the body are occurring from bone oscillation driven by an externally applied source (the tuning fork). This finding was the basis for “ankle audiometry” (Verrecchia, Freden Jansson et al. 2023).
Tests
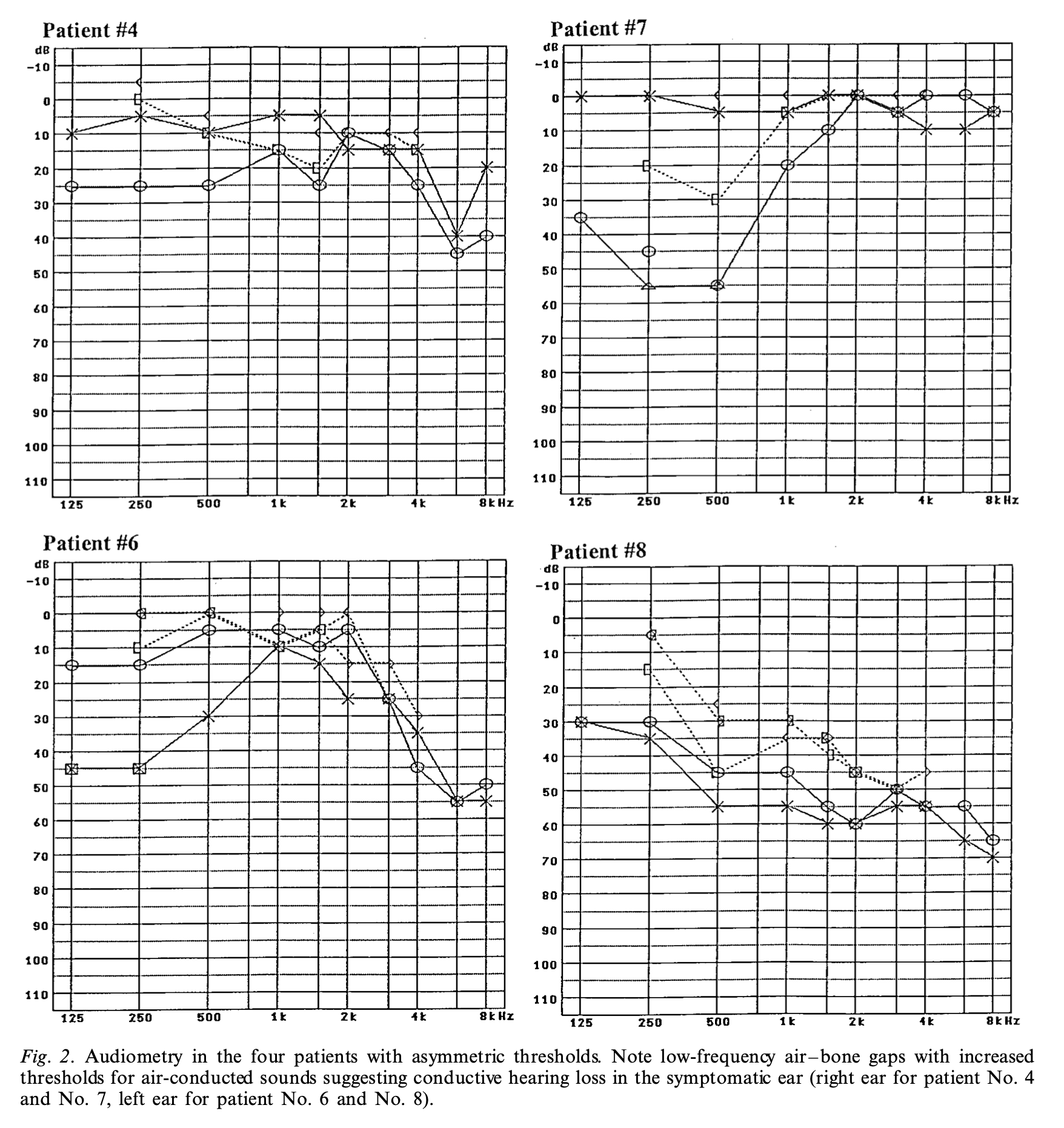
In SSCD audiometry usually shows an air-bone gap, and sometimes shows conductive hyperacusis.
The Figure below from Brantberg and colleagues (Brantberg, Bergenius et al. 2001) shows audiograms from four patients with SSCD. Each audiogram demonstrates a significant air-bone gap in the affected ear.
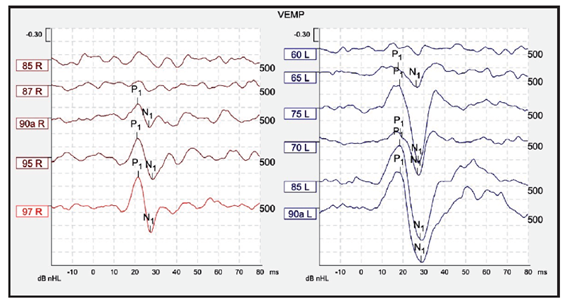
On the side affected by SSCD, vestibular evoked myogenic potentials (whether cervical or ocular) often show a combination of an abnormally large amplitude response and a low threshold. Interestingly, the degree of abnormality on vestibular evoked myogenic potentials does not appear to correspond to the degree of symptom severity (Noij, Wong et al. 2018).
The Figure below is from a case of left-sided SSCD reported by Mercado and colleagues (Mercado, Fernández et al. 2017) in which cervical vestibular evoked myogenic potentials on the affected side (left) show unusually large amplitude and unusually low thresholds.
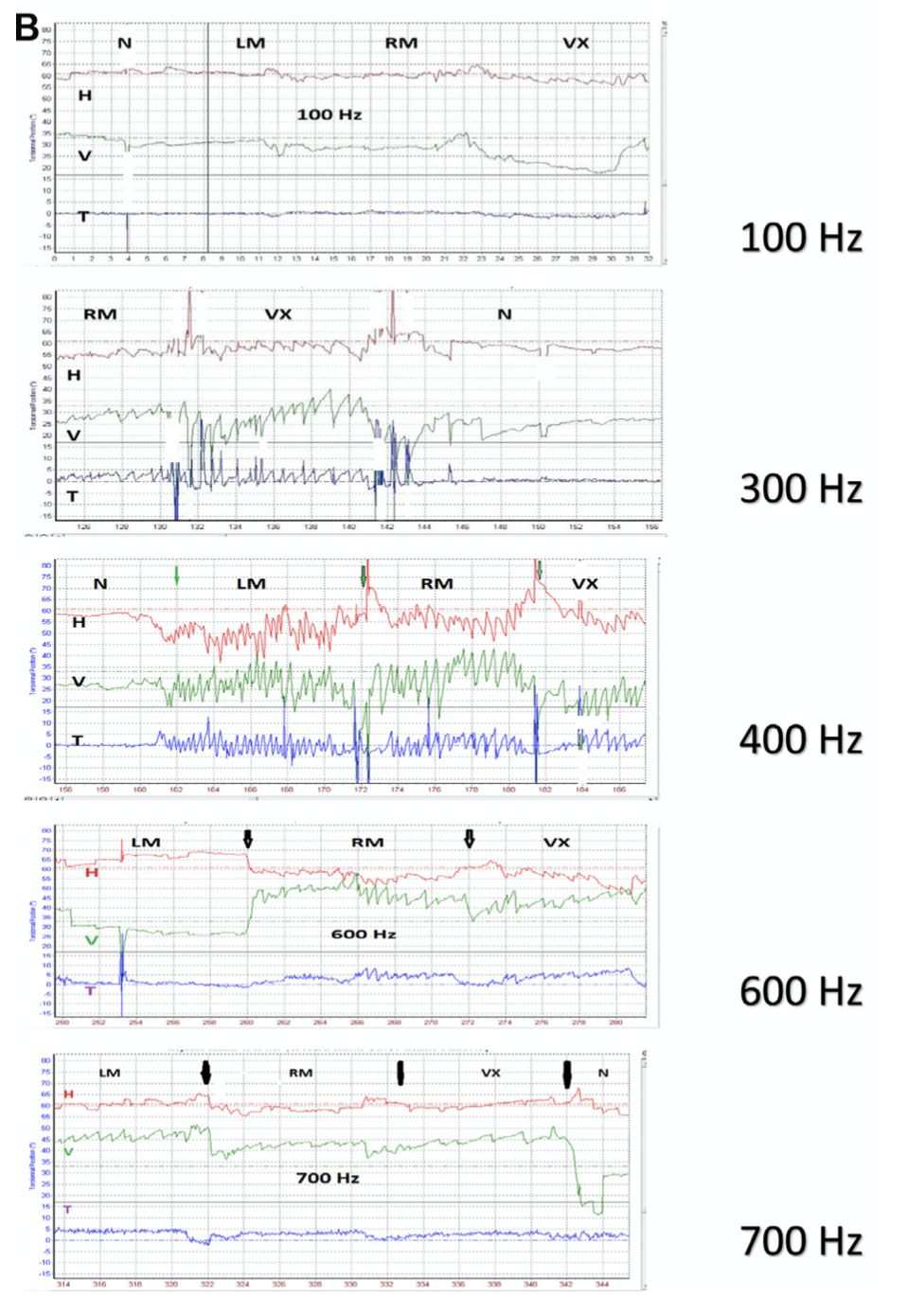
In some cases ocular motor testing may identify rapidly oscillating pendular nystagmus at a specific frequency of stimulation (White, Hughes et al. 2007, Manzari, Modugno et al. 2008, Dumas, Lion et al. 2014, Dumas, Tan et al. 2019).
In the Figure below from Dumas and colleagues (Dumas, Tan et al. 2019) a skull vibration frequency of 400 Hz brings out the most prominent nystagmus.
Imaging
The best studied imaging modality to evaluate for SSCD is a temporal bone CT without contrast. The collimation cuts should be 0.6 mm or better. Depending on the radiology service’s experience, when there is suspicion for superior canal involvement, it may be helpful to protocol the study with reconstructions in Stenver’s and Pöschl’s projections.
Pöschl’s projection (Pöschl 1943), named after Max Pöschl (1909-1990), is also referred to as the axiopetrosal plane or axial pyramidal plane, because it is perpendicular to the petrous pyramid. The plane of this projection runs from anterolateral to posteromedial, which is in the same plane as the superior semicircular canal.
Stenver’s projection (Stenvers 1916, 1917), named after Hendrik Willem Stenvers (1889-1973). The plane of this projection runs from anteromedial to posterolateral. Stenver’s projection acquires images through the semicircular canal “end on.”
These planes can be imagined by thinking of the so-called RALP-LARP planes. “RALP” refers to the fact that planes of the Right Anterior semicircular canal and the Left Posterior canal are parallel. “LARP” refers to the fact that the Left Anterior semicircular canal and the Right Posterior semicircular canal are parallel. These are depicted in the Figure below from Aw and colleagues (Aw, Todd et al. 2010).
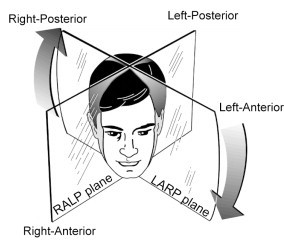
For the right superior semicircular canal, Stenver’s view corresponds to a projection through the LARP plane, while Pöschl’s view corresponds to a projection through the RALP plane.
For the left superior semicircular canal, Stenver’s view corresponds to a projection through the RALP plane, while Pöschl’s view corresponds to a projection through the LARP plane.
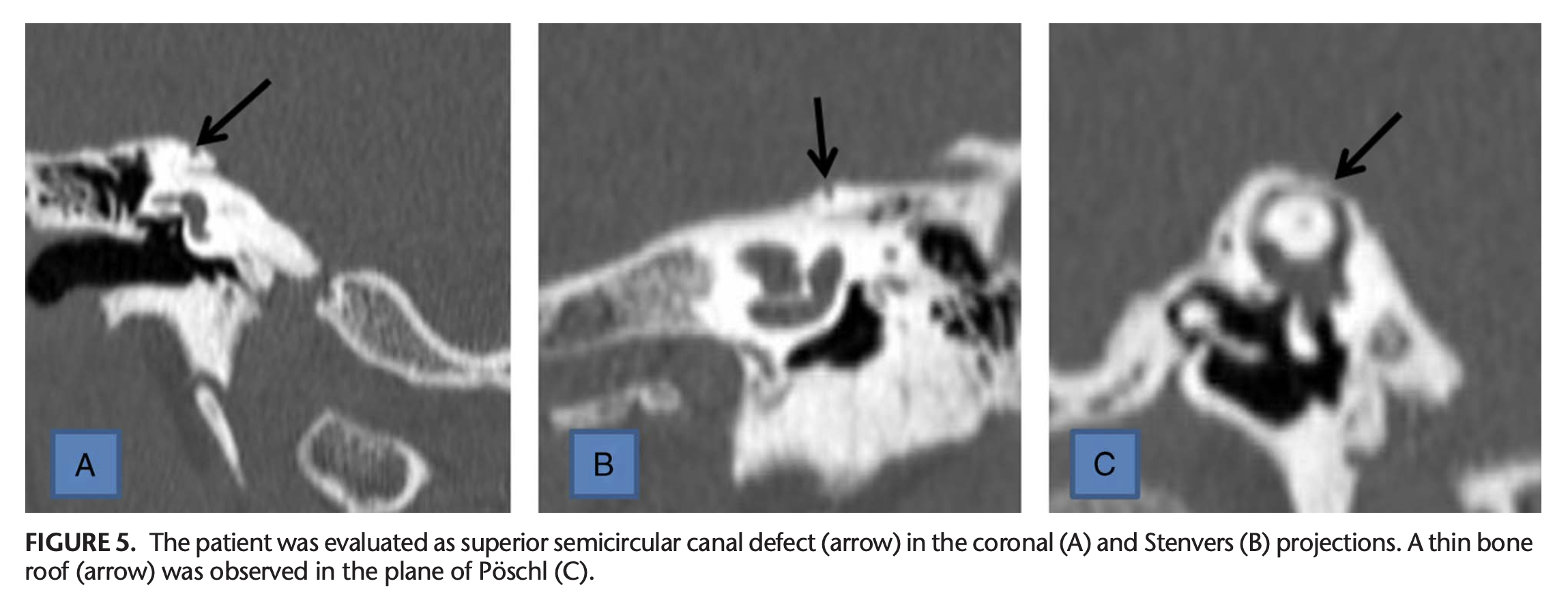
Thus, the coronal plane, Stenver’s plane and Pöschl’s plane provide three different views of the superior semicircular canal. The Figure below, from Duman and Dogan (Duman and Dogan 2020), juxtaposes three panels containing each view taken of a patient with superior SCD.
Performing a temporal bone CT entails exposure to a significant amount of ionizing radiation, so there is interest in other imaging modalities that could avoid this risk. MRI does not image bone well, but studies are beginning to explore whether high resolution MRI of the internal auditory canals has utility in the diagnosis of SSCD (Beyazal Celiker, Ozgur et al. 2018).
While imaging is a helpful tool for securing a diagnosis of SCD, the diagnosis does not rely on radiographic findings alone. As mentioned earlier, there is an incomplete overlap between (1) the group of individuals with radiographic SCD, and (2) the group of individuals with symptoms compatible with SCD.
A retrospective study (Berning, Arani et al. 2019) of temporal bone CTs done in 500 consecutive patients with no auditory or vestibular symptoms found that 10 (2%) had radiographic SCD, whereas of 110 patients whose symptoms were compatible with SCD only 15 (13.6%) had radiographic SCD.
Another study observed that, “When a diagnosis of superior canal dehiscence syndrome (SCDS) was made solely on CT scans, 80% of cases assessed were false positive” (Masaki 2011). The study retrospectively analyzed CT scans of 82 patients (164 ears), identified that 5 (3%) of 164 ears had radiographic superior SCD, and of those 5 ears, only 1 (0.5%) additionally had a clinical history compatible with superior SCD. The study author concluded that, “This stresses the importance of diagnosing SCDS on the basis of both CT findings and clinical symptoms.”
Diagnosis
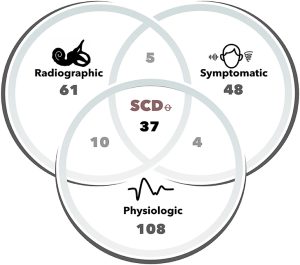
We would go a step further, and add that, when possible, the diagnosis should be supported not only by history and imaging, but also by audiologic and otovestibular testing (such as from audiometry and vestibular evoked myogenic potentials). In other words, the greatest confidence can be achieved when the clinical history, audiologic and otovestibular testing, and imaging all support the diagnosis. The Venn diagram in the Figure below from Tran and colleagues (Tran, Swanson et al. 2020) schematically depicts the intersection of these types of data; the specific numbers are from their data set.
Treatment
Depending on whether a patient’s symptoms from SSCD are tolerable, one reasonable management strategy is simply watchful waiting.
If a patient’s symptoms interfere significantly with function in daily activities, then intervention may be warranted. Treatment of SSCD is surgical. The most common surgical approaches are re-roofing the superior canal, and plugging the superior canal.
Re-roofing the superior canal is an invasive procedure (Peterson, Lazar et al. 2008) in which the otolaryngologist usually works in tandem with a neurosurgeon. The neurosurgeon often performs a temporal craniotomy and retracts the temporal lobe in order to expose the middle cranial fossa and thus the superior surface of the temporal bone, after which the otolaryngologist locates the dehiscence and “patches” it, usually with a synthetic substance.
Plugging the superior canal can be performed via a trans-mastoid surgical approach (Brantberg, Bergenius et al. 2001), and is thus less invasive. While plugging the superior canal may eliminate some symptoms from SSCD, it also effectively ablates the function of the superior canal, which can lead to some degree of chronic unsteadiness.
Even if the surgery is initially successful, it is possible for the plugged superior canal to become “un-plugged,” or for the re-roofed dehiscence to re-dehisce.
Prognosis
A multi-center study of 27 patients found that, “Patients with larger superior canal dehiscences show significantly more vestibulocochlear symptoms/signs… compared with smaller ones” (Pfammatter, Darrouzet et al. 2010).
Untreated, the clinical course of superior SCD is variable. We have had patients whose symptoms have remained stable for decades, while other patients may deteriorate over months to years.
Variants: posterior semicircular canal dehiscence (PSCD)
Posterior SCD is rare. One series of 23 patients with PSCD reported that, “86% presented with vertigo, 9% with hearing loss or tinnitus” (Krombach, DiMartino et al. 2003). Some surgeons treat PSCD with canal plugging (Mikulec and Poe 2006).
Variants: lateral semicircular canal dehiscence (LSCD)
Lateral SCD is rare. Yaniv and Hacking (Yaniv and Hacking 1987) reported two cases in which each patient had combined round window perilymphatic fistula and lateral SCD. Yaniv and Traub (Yaniv and Traub 1988) included those patients into a larger series of 15 patients with suspected perilymphatic fistulae reported that two patients had both a round window fistula and lateral SCD, and another two patients had isolated lateral SCD.
Differential diagnosis
The main differential diagnosis is perilymphatic fistula (PLF). Symptoms such as episodic disequilibrium, hearing loss and tinnitus certainly overlap with other diagnoses, such as Ménière’s disease, but in SCD at least some of the symptoms will tend to have a component of pressure sensitivity.
References
Aw ST, Todd MJ, Michael Halmagyi G (2010) Chapter 12 – Head impulse testing: angular vestibulo-ocular reflex (VOR). Handbook of Clinical Neurophysiology, vol 9. Elsevier, pp 150-164
Berning AW, Arani K, Branstetter BFt (2019) Prevalence of Superior Semicircular Canal Dehiscence on High-Resolution CT Imaging in Patients without Vestibular or Auditory Abnormalities. AJNR Am J Neuroradiol 40: 709-712. doi: 10.3174/ajnr.A5999
Bertholon P, Reynard P, Lelonge Y, Peyron R, Vassal F, Karkas A (2018) Hearing eyeball and/or eyelid movements on the side of a unilateral superior semicircular canal dehiscence. Eur Arch Otorhinolaryngol 275: 629-635. doi: 10.1007/s00405-017-4781-7
Beyazal Celiker F, Ozgur A, Celiker M, Beyazal M, Turan A, Terzi S, Inecikli MF (2018) The Efficacy of Magnetic Resonance Imaging for the Diagnosis of Superior Semicircular Canal Dehiscence. J Int Adv Otol 14: 68-71. doi: 10.5152/iao.2017.4103
Brantberg K, Bergenius J, Mendel L, Witt H, Tribukait A, Ygge J (2001) Symptoms, findings and treatment in patients with dehiscence of the superior semicircular canal. Acta Otolaryngol 121: 68-75. doi: 10.1080/000164801300006308
Carey JP, Minor LB, Nager GT (2000) Dehiscence or thinning of bone overlying the superior semicircular canal in a temporal bone survey. Arch Otolaryngol Head Neck Surg 126: 137-47.
Duman IS, Dogan SN (2020) Contribution of Reformatted Multislice Temporal Computed Tomography Images in the Planes of Stenvers and Poschl to the Diagnosis of Superior Semicircular Canal Dehiscence. J Comput Assist Tomogr 44: 53-58. doi: 10.1097/RCT.0000000000000957
Dumas G, Lion A, Karkas A, Perrin P, Perottino F, Schmerber S (2014) Skull vibration-induced nystagmus test in unilateral superior canal dehiscence and otosclerosis: a vestibular Weber test. Acta Otolaryngol 134: 588-600. doi: 10.3109/00016489.2014.888591
Dumas G, Tan H, Dumas L, Perrin P, Lion A, Schmerber S (2019) Skull vibration induced nystagmus in patients with superior semicircular canal dehiscence. Eur Ann Otorhinolaryngol Head Neck Dis. doi: 10.1016/j.anorl.2019.04.008
Hain TC, Cherchi M (2008) Pulse-synchronous torsional pendular nystagmus in unilateral superior canal dehiscence. Neurology 70: 1217-8. doi: 10.1212/01.wnl.0000307752.25133.8f
Krombach GA, DiMartino E, Schmitz-Rode T, Prescher A, Haage P, Kinzel S, Gunther RW (2003) Posterior semicircular canal dehiscence: a morphologic cause of vertigo similar to superior semicircular canal dehiscence. Eur Radiol 13: 1444-50. doi: 10.1007/s00330-003-1828-5
Manzari L, Modugno GC, Brandolini C, Pirodda A (2008) Bone vibration-induced nystagmus is useful in diagnosing superior semicircular canal dehiscence. Audiol Neurootol 13: 379-87. doi: 10.1159/000148201
Masaki Y (2011) The prevalence of superior canal dehiscence syndrome as assessed by temporal bone computed tomography imaging. Acta Otolaryngol 131: 258-62. doi: 10.3109/00016489.2010.526145
McCrary HC, Babajanian E, Patel N, Yang S, Kircher M, Carlson ML, Gurgel RK (2021) Superior Semicircular Canal Dehiscence Syndrome Following Head Trauma: A Multi-institutional Review. Laryngoscope 131: E2810-E2818. doi: 10.1002/lary.29751
Mercado VM, Fernández FA, Hernandez CB, Pino CU, Novoa IC, Herrera PC (2017) Dehiscence syndrome superior semicircular canal: A case of dehiscence syndrome of the superior semicircular. Journal of Otolaryngology-ENT Research 6: 175-178. doi: 10.15406/joentr.2017.06.00181
Mikulec AA, Poe DS (2006) Operative management of a posterior semicircular canal dehiscence. Laryngoscope 116: 375-8. doi: 10.1097/01.mlg.0000200358.93385.5c
Milenkovic I, Sycha T, Berger-Sieczkowski E, Rommer P, Czerny C, Wiest G (2021) Pulse-synchronous torsional nystagmus. Pract Neurol 21: 445-447. doi: 10.1136/practneurol-2021-003027
Minor LB, Solomon D, Zinreich JS, Zee DS (1998) Sound- and/or pressure-induced vertigo due to bone dehiscence of the superior semicircular canal. Arch Otolaryngol Head Neck Surg 124: 249-58. doi: 10.1001/archotol.124.3.249
Naert L, Van de Berg R, Van de Heyning P, Bisdorff A, Sharon JD, Ward BK, Van Rompaey V (2018) Aggregating the symptoms of superior semicircular canal dehiscence syndrome. Laryngoscope 128: 1932-1938. doi: 10.1002/lary.27062
Noij KS, Wong K, Duarte MJ, Masud S, Dewyer NA, Herrmann BS, Guinan JJ, Jr., Kozin ED, Jung DH, Rauch SD, Lee DJ (2018) Audiometric and cVEMP Thresholds Show Little Correlation With Symptoms in Superior Semicircular Canal Dehiscence Syndrome. Otol Neurotol 39: 1153-1162. doi: 10.1097/MAO.0000000000001910
Ota I, Sakagami M, Kitahara T (2021) The Third Mobile Window Effects in Otology/Neurotology. J Int Adv Otol 17: 156-161. doi: 10.5152/JIAO.2021.8632
Peterson EC, Lazar DA, Nemecek AN, Duckert L, Rostomily R (2008) Superior semicircular canal dehiscence syndrome: Successful treatment with repair of the middle fossa floor: technical case report. Neurosurgery 63: E1207-8; discussion E1208. doi: 10.1227/01.NEU.0000335179.02759.A8
Pfammatter A, Darrouzet V, Gartner M, Somers T, Van Dinther J, Trabalzini F, Ayache D, Linder T (2010) A superior semicircular canal dehiscence syndrome multicenter study: is there an association between size and symptoms? Otol Neurotol 31: 447-54. doi: 10.1097/MAO.0b013e3181d27740
Pöschl M (1943) Der tomographische Querschnitt durch das Felsenbein [The tomographic cross-section through the petrous bone]. Forschrtschritte auf der Gebiete des Röntgenstrahlen [Advances in Radiology] 68: 174-179.
Stenvers HW (1916) The clinical significance of radiographs of the orbital region. Archives of Radiology and Electrotherapy 20: 411-417.
Stenvers HW (1917) Roentgenology of the os petrosum. Archives of Radiology and Electrotherapy 22: 97-112.
Tilikete C, Krolak-Salmon P, Truy E, Vighetto A (2004) Pulse-synchronous eye oscillations revealing bone superior canal dehiscence. Ann Neurol 56: 556-560. doi: PMID: 15455401
Tran ED, Swanson A, Sharon JD, Vaisbuch Y, Blevins NH, Fitzgerald MB, Steenerson KK (2020) Ocular Vestibular-Evoked Myogenic Potential Amplitudes Elicited at 4 kHz Optimize Detection of Superior Semicircular Canal Dehiscence. Front Neurol 11: 879. doi: 10.3389/fneur.2020.00879
Verrecchia L, Freden Jansson KJ, Westin M, Velikoselskii A, Reinfeldt S, Hakansson B (2023) Ankle Audiometry: A Clinical Test for the Enhanced Hearing Sensitivity for Body Sounds in Superior Canal Dehiscence Syndrome. Audiol Neurootol 28: 219-229. doi: 10.1159/000528407
White JA, Hughes GB, Ruggieri PN (2007) Vibration-Induced Nystagmus as an Office Procedure for the Diagnosis of Superior Semicircular Canal Dehiscence. Otol Neurotol 28: 911-916. doi: 10.1097/mao.0b013e31812f7222
Yaniv E, Hacking DP (1987) Combined perilymphatic fistulas of the round window and lateral semicircular canal. A report of 2 cases. S Afr Med J 71: 391-2.
Yaniv E, Traub P (1988) Traumatic perilymphatic fistulae of the lateral semicircular canal. J Laryngol Otol 102: 521-3. doi: 10.1017/s0022215100105535
![]()