By Marcello Cherchi, MD PhD
For patients
Here are answers to some of the most common questions about computerized dynamic posturography (CDP).
|
What is this test? |
Computerized dynamic posturography (CDP) is a balance test. |
|
What is this test looking for? |
This test checks how vision, touch, and inner ear function contribute to your equilibrium.. |
|
Is this test experimental or investigational? |
This test is approved by the Food and Drug Administration. It is neither experimental nor investigational. |
|
What happens during this test? |
During this test the audiologist or technician will have you stand in a phone-booth-sized device. You will wear a security harness do you do not fall. During the test there are some times that you will be blindfolded, some times that the walls appear to wiggle, and some times that the floor wiggles. |
|
Is this test uncomfortable? |
This test is not uncomfortable. |
|
How long does this test take? |
The test takes about 30 minutes. |
|
Do I have to prepare for this test? |
Try to avoid taking medications such as meclizine (Bonine®) and dimenhydrinate (Dramamine®) for 24 hours prior to taking this test. There are otherwise no specific preparations for this test. |
|
Are there any special instructions for what to do after the test? |
There are no special instructions for what to do after this test. |
For clinicians
Overview
Computerized dynamic posturography (CDP) tests whether somatosensory deficits, visual deficits, otovestibular deficits, or some combination thereof, is contributing to a person’s disequilibrium. It is not useful for identifying other sources of disequilibrium (such as cardiovascular disease, migraine, basal ganglionic disorders.) Some physical therapists advocate its use for monitoring a patient’s progress over their course of treatment.
§1: Introduction
“Computerized dynamic posturography (CDP),” also called “moving platform posturography,” is based on the work of Louis Nashner (Nashner 1970, 1971), that he subsequently developed with other colleagues (Nashner et al. 1982). CDP is intended to discriminate among several general categories of imbalance, including those resulting from somatosensory, visual and vestibular deficits. CDP is not a purely vestibular test, because it assesses a composite response. It is useful to conceptualize CDP as a method for quantifying overall balance, and for identifying some patterns of imbalance in different testing conditions.
The CDP machine is an expensive device (Chieffe et al. 2023), and some investigators have criticized its diagnostic potential (Dobie 1997; Evans and Krebs 1999; Kingma et al. 2011; O’Neill et al. 1998).
§2: Underlying biomechanics and physiology, and their terminology
Several terms are helpful for understanding CDP:
- Base of support (BOS) = the area contained within the perimeter of contact between the surface and the two feet. The BOS is approximately square when the feet are placed comfortably apart while standing still.
- Center of gravity (COG) = the point within the mass of a system of particles at which, for many physics purposes, the system behaves as if its mass were concentrated.
- Limits of stability (LOS) = the maximum possible COG sway angle as a function of sway direction from the center position (where center position = any position along the vertical line above the center of the BOS).
In a healthy person, the center of gravity (COG) remains immediately above the center of the base of support (BOS) despite changes in body configuration, as illustrated in the Figure below from Nashner (Nashner 1997c).
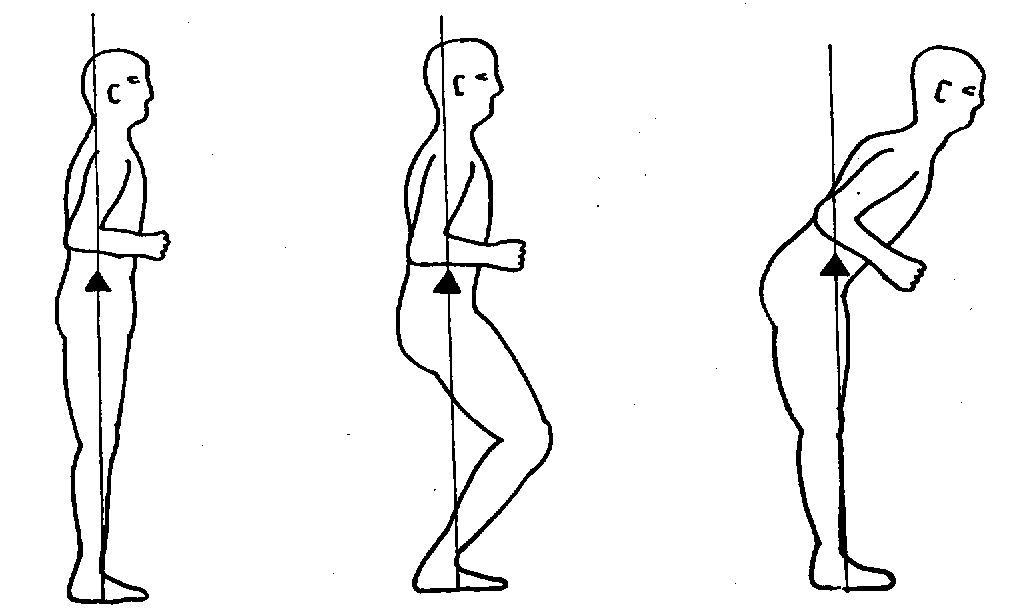
A healthy person standing in a neutral position (whose only points of contact are the feet with the support surface) always has some degree of sway, because:
“It is impossible to maintain the COG motionless, because in-place standing is an inherently unstable task requiring periodic corrections to overcome the destabilizing influence of gravity” (Nashner 1997c).
Two other terms pertain to this sway:
- Limits of sway is the maximum spontaneous COG sway angle (Nashner 1997c) when the patient is standing in a neutral position. (This is different from the limits of stability.)
- COG alignment is the point at the center of the area contained within the limits of sway perimeter (Nashner 1997c). (Note that this is different from the COG.)
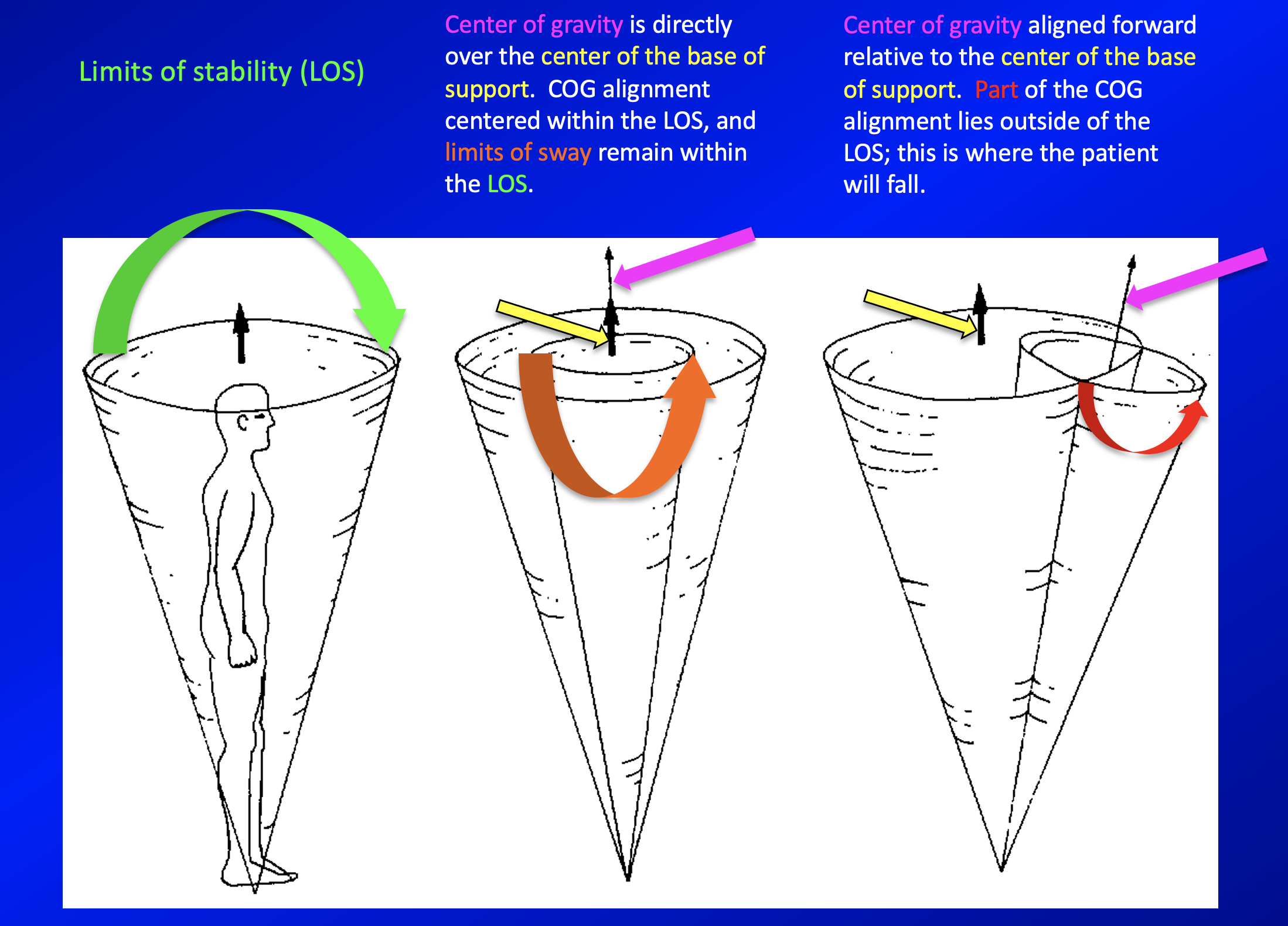
The limits of sway and COG alignment affect balance differently (Nashner 1997c). Specifically:
-
- When COG is aligned directly above the center of the BOS, then the limits of sway can be as large as the LOS (limits of stability) before balance is lost.
- In contrast, a person whose COG alignment is offset (i.e., is not directly over the center of the BOS) is not as stable, even though the LOS (limits of stability) is similar. This is because in a person with the offset COG alignment, smaller sway angles (in the direction of offset) will move the COG beyond the LOS perimeter.
These consequences are illustrated in the Figure below, adapted from Nashner (Nashner 1997c).
The vestibular system integrates multiple sensory inputs and produces multiple outputs, as summarized by the schematic in the Figure below, from Davies (Davies 2004).
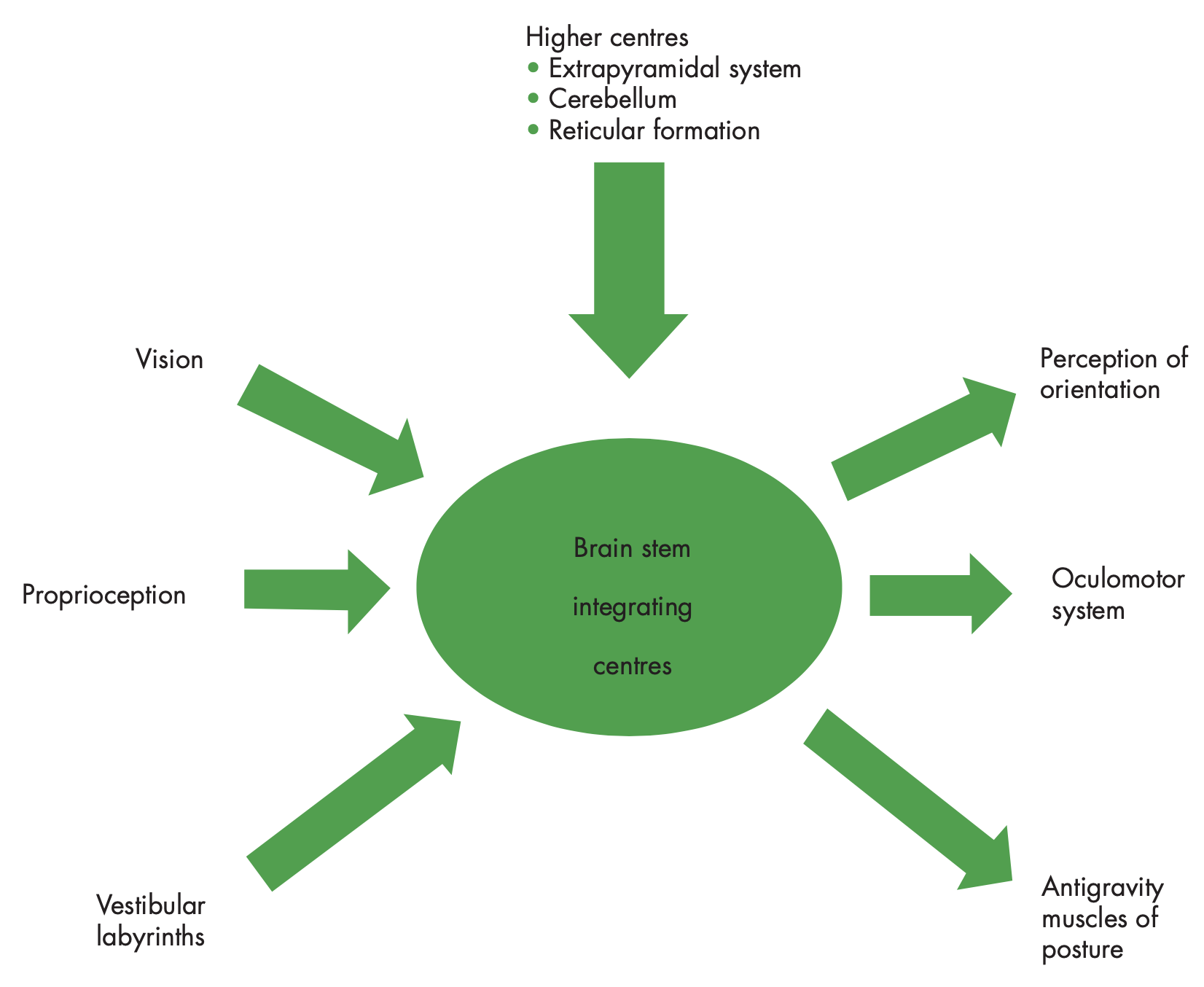
CDP makes the reasonable simplifying assumption that there are 3 basic sensory input modalities relevant to balance:
- Somatosensory input is derived from contact forces and motions between the feet and the support surface (Nashner 1997c). Somatosensory inputs dominate balance when the support surface is fixed, regardless of the status of visual and vestibular inputs (Nashner 1997a).
- Visual input. This plays a significant role when the support surface is unstable (Nashner 1997c).
- Vestibular input. When functionally useful somatosensory and visual inputs are available, vestibular inputs play a minor role in controlling COG position. However, it becomes critical when both somatosensory and visual inputs are misleading or unavailable (Nashner 1997c).
§3: Equipment needed
Computerized dynamic posturography is a phone-booth-sized device in which the patient stands. The patient is strapped in with a harness to prevent falls. The support surface is composed of two mobile force plates (one for each foot) which can detect anterior pressure (when the patient is leaning forward) and posterior pressure (when the patient is leaning backward), and the force plates themselves can tilt anteriorly or posteriorly. The visual surround can also move, appearing to tilt anteriorly or posteriorly. The test measures the degree and direction of sway, and whether a patient falls (although the fall is broken by the harness).
The computerized dynamic posturography device is one of the most expensive pieces of equipment in a well-outfitted otoneurology clinic. The FDA-approved version of computerized dynamic posturography was originally produced under the name EquiTest® by a company called NeuroCom®; this was eventually taken over by a company called Bertec®.
§4: How to perform the test
Intuitively one would expect that medications with vestibular suppressant effects could influence the results of CDP. This has not been evaluated thoroughly, though one study reported that dimenhydrinate did not influence CDP results (Gill et al. 2000). Nevertheless, our general practice is to request that patients refrain from taking meclizine or dimenhydrinate for 24 hours prior to the test.
CDP comprises several sections. The sections of particular clinical relevance include:
- Sensory organization test (SOT). This assesses a patient’s performance on successively more difficult balance tasks and looks for trends.
- Motor control test (MCT). This test includes protocols that induce brief perturbations (both shear and tilt) in the support surface to elicit particular compensatory postural adjustments. This is sometimes coupled with surface EMG recordings.
§4.1: How to perform the sensory organization test
The sensory organization test (SOT) is a series of six different conditions, each of which selectively perturbs either visual input, or proprioceptive input, or both. Vestibular sensation cannot be selectively perturbed with this test. The CDP machine measures:
-
- Equilibrium score. This is “a nondimensional percentage which compares the patient’s peak amplitude of AP [anterior-posterior] sway to the theoretical AP limits of stability” (Nashner 1997a).
- Falls.
Before enumerating the six testing conditions of SOT we must discuss sway referencing. Sway referencing involves “tilting of the support surface and/or the visual surround about an axis co-linear with the ankle joints to directly follow the patient’s COG sway in the AP direction” (Nashner 1997a). This means that the visual surround and the platform’s tilt can vary.
- “Sway referencing” of the platform means that as the patient sways in the anterior-posterior plane (putting more pressure on the toes or heels), the platform tilts in that same direction, effectively reducing the usefulness of proprioceptive input.
- “Sway referencing” of the visual surround means that as the patient sways in the anterior-posterior plane (putting more pressure on the toes or heels), the visual environment tilts in that same direction, effectively reducing the usefulness of visual input.
Sway referencing tends to have different effects in health and disease. Specifically:
- “Healthy subjects ignore a sway-referenced sensory input that is functionally inaccurate and maintain balance using other sensory inputs” (Nashner 1997a).
- Diseased subjects may fail to “downweight” such sway-referenced (misleading) sensory inputs.
We now move on to discussing the six conditions of sensory organization testing. These are summarized in the Figure below, adapted from Nashner (Nashner 1997a).
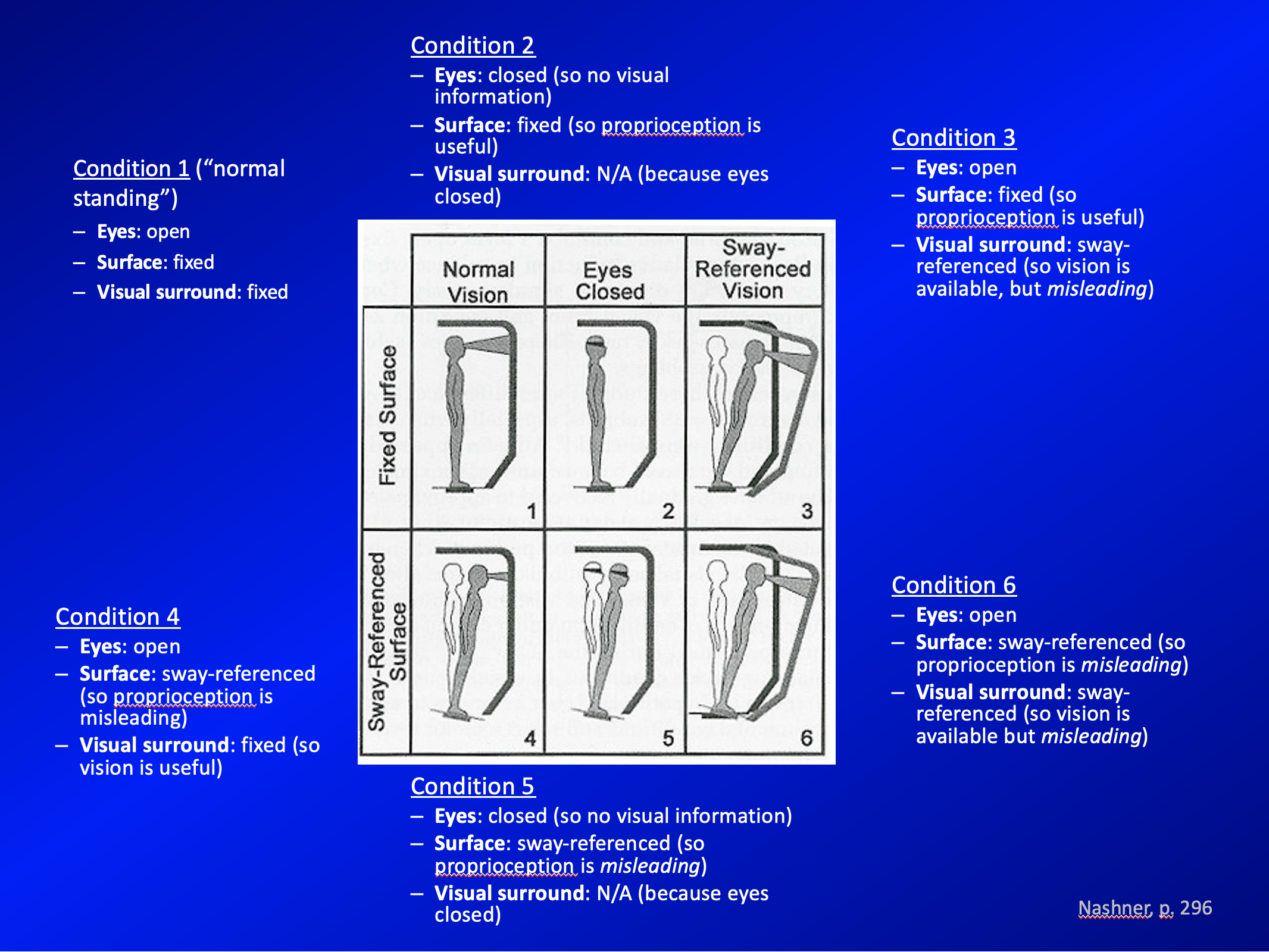
Here is a summary of sensory organization testing in tabular format:
|
Visual condition |
||||
|
Vision fixed. This is good because vision is useful. |
Eyes closed. This is somewhat bad because vision cannot be used. |
Vision sway-referenced. This is really bad because vision is misleading. |
||
|
Support condition |
Support fixed. This is good, because proprioception is useful. |
Condition 1: • Vision fixed. • Support fixed. |
Condition 2: • Vision absent. • Support fixed. |
Condition 3: • Vision sway-referenced. • Support fixed. |
|
Support sway-referenced. This is really bad because proprioception is misleading. |
Condition 4: • Vision fixed. • Support sway-referenced. |
Condition 5: • Vision absent. • Support sway-referenced. |
Condition 6: • Vision sway-referenced. • Support sway-referenced. |
|
The SOT conditions 1 – 6 become progressively more difficult in that sensory information gradually goes from present, to absent, to misleading.
By condition 6, the only sensory modality that provides useful information is the vestibular system. Obviously, none of the sensory conditions selectively impairs vestibular input.
Condition 4 (sway-referenced support) is “more difficult” than condition 3 (sway-referenced vision) because somatosensory inputs dominate balance when the support surface is fixed, regardless of the status of visual and vestibular inputs (Nashner 1997a).
§4.2: How to perform motor control testing and response adaptation testing
The next two tests, motor control testing and response adaptation testing, measure output — postural reactions — in response to different mechanical perturbations. In motor control testing the stimulus is a shearing force applied at the soles of the feet in the anterior-posterior directions. In response adaptation testing the stimulus is a torque co-axial with the ankles.
Illustrations of the stimuli for motor control testing and response adaptation testing are shown in the Figure below, from Nashner (Nashner 1976).
§4.2.1: How to perform motor control testing
The motor control testing component of CDP involves anterior/posterior translation of the support surface by varying distances. Several parameters are measured:
- Weight symmetry, which quantifies the relative distribution of weight on each leg. In healthy individuals the weight should be approximately evenly distributed between the two legs.
- Latency, which quantifies the elapsed time between the stimulus onset and the initiation of the patient’s motor response.
- Amplitude scaling, which measures the strength of response in each leg in each of three translation distances.
§4.2.2: How to perform response adaptation testing
Response adaptation testing involves rotation of the support surface co-axial with the ankle joints, delivering a torquing force at a rotational velocity of 8 deg/sec (Nashner 1997a). The patient is subjected to five trials of the “toes up” rotation and five trials of the “toes down” rotation. In this protocol, computerized dynamic posturography measures the “sway energy,” which Nashner defines as:
“Sway energy is used as a nonspecific but quantitative measure of the magnitude of COG [center of gravity] sway during the recovery period following a support surface rotation. Sway energy is a weighted sum of the root mean square COG sway velocity and sway acceleration measured over the 2.5-second interval immediately following rotation” (Nashner 1997a).
§5: What this test assesses
Computerized dynamic posturography measures motor responses (sway, and whether a patient falls) in a variety of conditions in which visual input and somatosensory (proprioceptive) input are selectively perturbed in various combinations. In simple terms, the results give information about postural stability and postural responses in the various testing conditions.
§6: How to interpret the test results
In the sub-sections below we discuss the parts of CDP with the greatest clinical relevance.
There is some suggestion that serial CDP “can be used to follow the progress of the patient over time,” and “track progress during treatment” (Nashner 1997a), and some physical therapy practices will repeat CDP evaluations over several follow-up visits to monitor progress (Alfieri et al. 2012; Badke et al. 2005; Gonzalez Eslait et al. 2023; Ledin et al. 1990a; Li et al. 2010). This practice is somewhat controversial.
§6.1: How to interpret sensory organization testing
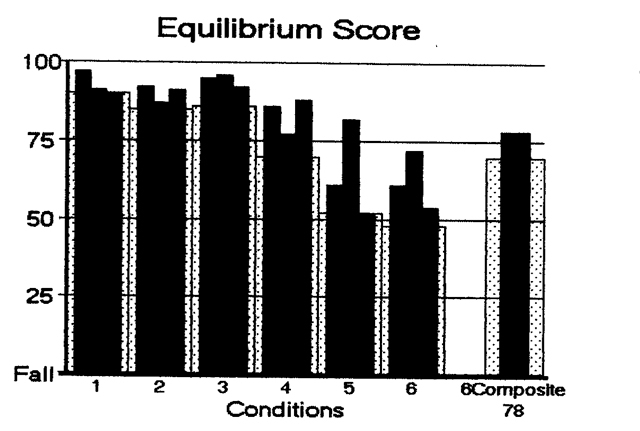
On sensory organization testing (SOT), the performance on each condition (equilibrium score averaged over 3 trials) should approximately scale with the level of difficulty of that condition. An example of this is shown in the Figure below, from https://dizziness-and-balance.com/testing/posturography.html (accessed 5/5/23).
In contrast, inconsistent performance may be compatible with non-physiologic causes of disequilibrium. An example is shown in the Figure below, from https://dizziness-and-balance.com/testing/posturography.html (accessed 5/5/23).
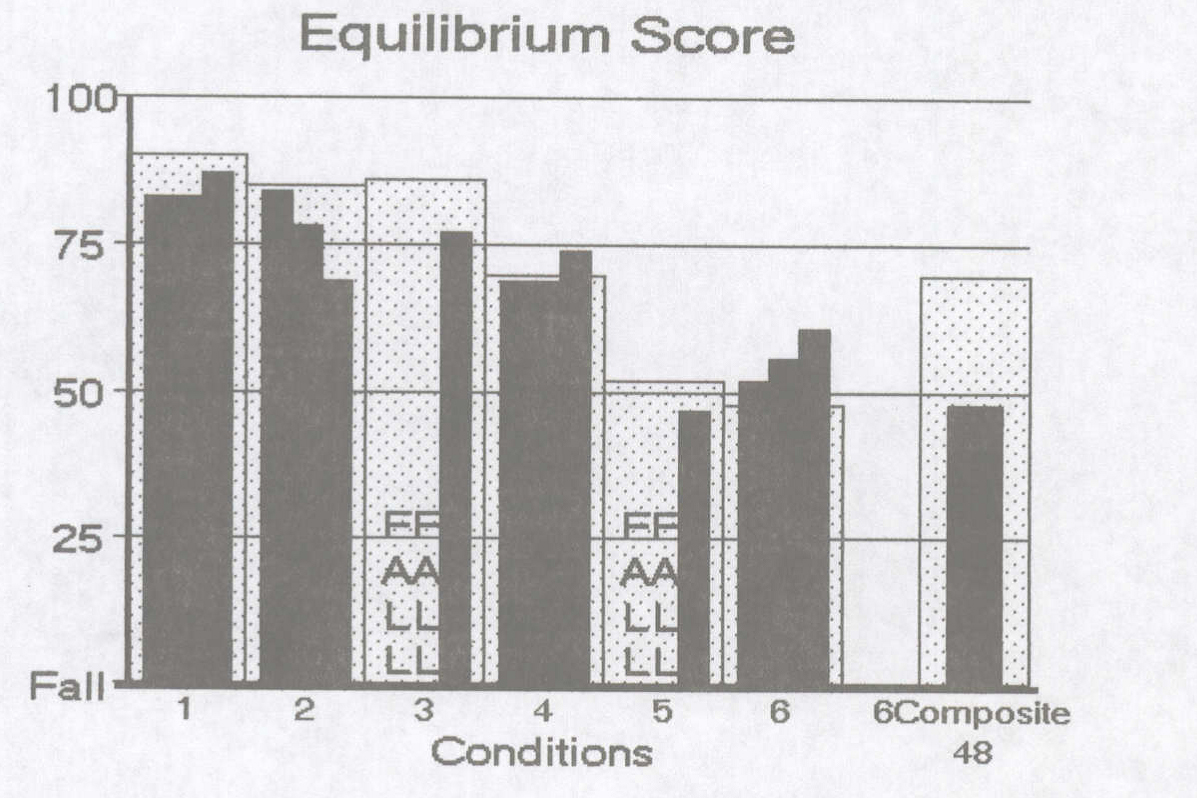
In an individual whose disequilibrium is due to non-physiologic causes, sensory organization testing may show that they perform more poorly than expected on easy conditions, and better than expected on harder conditions. In addition, the person’s performance for a given condition will vary when repeated. As Nashner says:
“When the patient is suspected of exaggerating symptoms for secondary gain, the best approach is to repeat the test and look for inconsistent results. When inconsistent CDP results are obtained, the finding can be further supported by repeating the test components in a random order” (Nashner 1997a).
A number of studies have explored CDP results in patients believed to have non-physiologic causes of disequilibrium (Artuso et al. 2004; Cevette et al. 1995; Gianoli et al. 2000; Goebel et al. 1997; Herdman et al. 2011; Jacob et al. 1997; Krempl and Dobie 1998; Larrosa et al. 2012; Larrosa et al. 2013; Mallinson and Longridge 2005; Morgan et al. 2002; Uimonen et al. 1995).
§6.2: How to interpret sensory analysis
Results from the six conditions of sensory organization testing can be used to calculate a “sensory analysis,” which is supposed to reflect the main deficit(s) accounting for the disequilibrium. The algorithm is as shown in the Table below, from Nashner (Nashner 1997a).
|
Ratio name |
Numerator test condition(s) |
Denominator test condition(s) |
Ratio |
Question the ratio tries to answer |
Significance of low score. |
|
“SOM” (somato-sensory) |
Condition 2: • Vision absent. • Support fixed. |
Condition 1: • Vision fixed. • Support fixed. |
2 / 1 |
Does sway increase when visual cues are removed (even though proprioceptive cues remain constant)? |
Patient makes poor use of somatosensory references. |
|
“VIS” (visual) |
Condition 4: • Vision fixed. • Support sway-referenced. |
Condition 1: • Vision fixed. • Support fixed. |
4 / 1 |
Does sway increase when somatosensory cues are inaccurate (even though visual cues remain constant)? |
Patient makes poor use of visual references. |
|
“VEST” (vestibular) |
Condition 5: • Vision absent. • Support sway-referenced. |
Condition 1: • Vision fixed. • Support fixed. |
5 / 1 |
Does sway increase when visual cues are removed and somatosensory cues are inaccurate (even though vestibular stimuli remain constant)? |
Patient makes poor use of vestibular cues, or vestibular cues are unavailable. |
|
“PREF” (visual preference) |
Condition 3: • Vision sway-referenced. • Support fixed. Condition 6: • Vision sway-referenced. • Support sway referenced. |
Condition 2: • Vision absent. • Support fixed. Condition 5: • Vision absent. • Support sway-referenced. |
(3+6) / (2+5) |
Does sway increase when visual cues are inaccurate (compared to visual cues simply being absent)? |
Patient relies on visual cues even when they are inaccurate. “The pathologic basis for vision preference is unknown” (Nashner 1997b). |
In our experience the “VEST” (vestibular) result from sensory analysis often correlates with vestibular deficits identified on other otoneurological testing, whereas the “SOM” (somatosensory) and “VIS” (visual) results do not appear to have clear correlation with, say, peripheral neuropathy and poor vision.
§6.3: How to interpret strategy analysis
The strategy analysis component of CDP draws on data from sensory organization testing to help distinguish “hip-dominant” from “ankle-dominant” balance strategies. The significance of this is that, “Normal subjects use ankle movements when the COG [center of gravity] is well within the LOS [limits of stability], and will increase their use of hip movements as the COG approaches the LOS” (Nashner 1997a, b).
The ankle-dominant and hip-dominant strategies are illustrated in the Figure below, adapted from Nashner (Nashner 1997c).
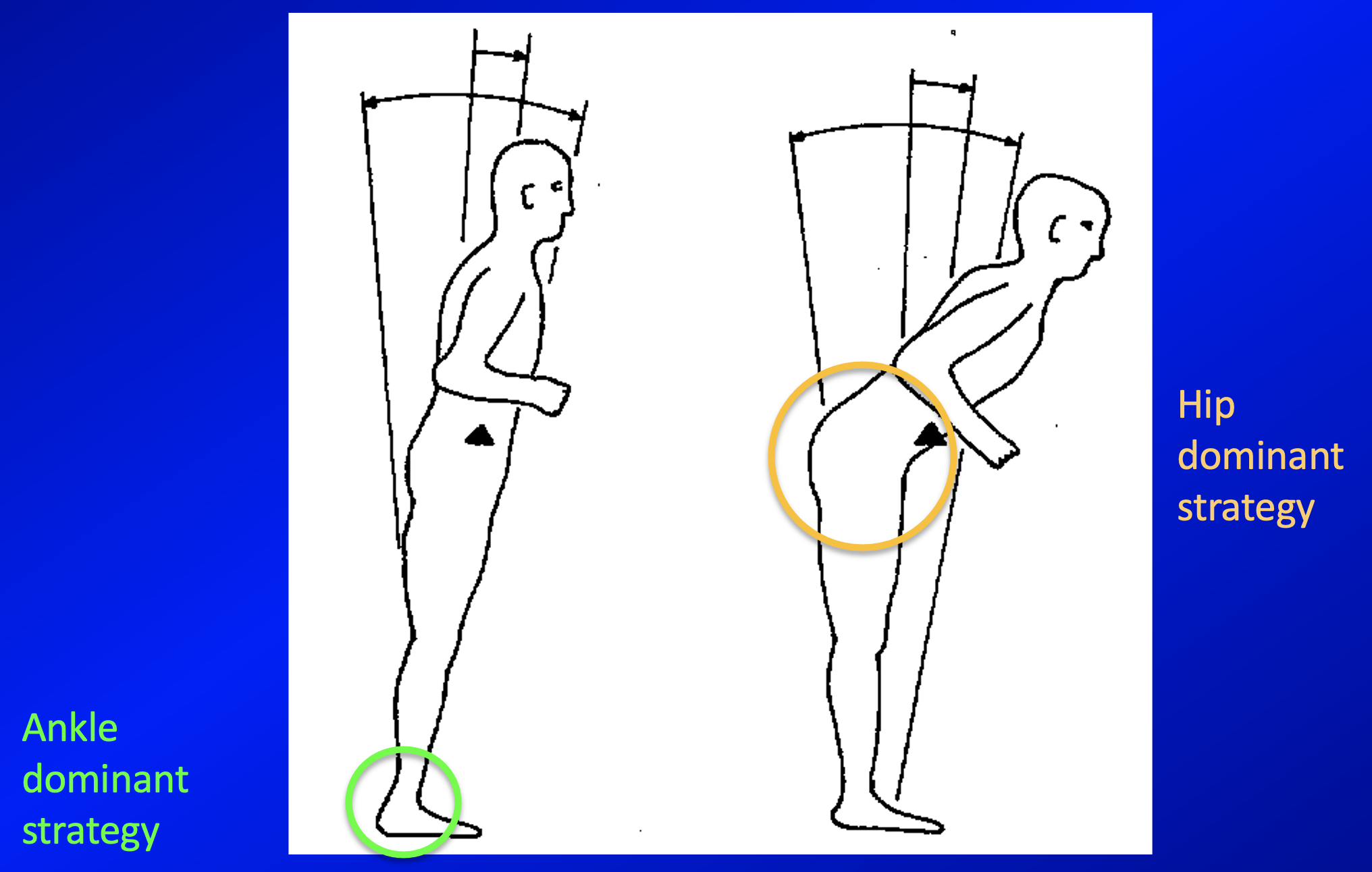
Nashner (Nashner 1997c) makes the following general observations regarding ankle-dominant versus hip-dominant strategies:
- Patients with impaired vestibular function (e.g., bilateral vestibular loss from gentamicin ototoxicity) prefer ankle movements, avoid hip movements under all conditions, and tend to fix the position of the head relative to the trunk (because they are trying to minimize the types of movement whose monitoring would require an intact vestibular system, and they instead rely more on proprioceptive input from the ankles).
- Patients with impaired somatosensory input (e.g., severe diabetic peripheral neuropathy) prefer hip movements and avoid ankle movements under all support surface conditions (because they are trying to minimize the types of movement whose monitoring would require intact proprioception, and they instead rely more on vestibular input).
In other words, patients with otologic vestibular disorders (such as bilateral vestibular weakness) tend to exhibit an ankle-dominant strategy, while patients with impaired somatosensory input (such as peripheral neuropathy) tend to exhibit a hip-dominant strategy.
§6.4: How to interpret center of gravity alignment
The center of gravity alignment component of CDP draws on data from sensory organization testing to plot the anterior-posterior and lateral movement of the COG (center of gravity) over time. Nashner (Nashner 1997a) comments that:
“Clinically normal subjects align the COG very near to the center of the base of support under all six sensory conditions [from sensory organization testing]. Even though sway amplitudes increase significantly under conditions 5 and 6, the average COG positions remain very nearly centered. Because offset positions in all directions are considered abnormal, alignment values beyond ±2 SD in lateral and AP [anterior-posterior] directions are considered abnormal” (Nashner 1997a).
§6.5: How to interpret motor control testing
The results from motor control testing (MCT) component of CDP may help identify several types of deficits.
Delayed latencies of postural responses can occur in somatosensory deficits (such as peripheral neuropathy) and musculoskeletal (orthopedic) problems.
Hypermetric amplitude scaling of postural responses (essentially, dysmetria) can occur in some cerebellar diseases.
§6.6: How to interpret response adaptation testing
A healthy individual’s sway energy (as defined earlier) will progressively decrease over the course of a sequence of the five “toes up” or “toes down” rotational stimuli co-axial with the ankles. In essence, the individual’s stability improves as their postural responses adapt to the successive exposure to the same perturbing stimulus. Nashner comments that:
“Asymptomatic normal individuals show progressive reductions in sway energy over the course of five repeated exposures to toes-up or toes-down rotations… While the sway energy scores of younger and older subjects are similar during the first toes-up and toes-down trials, improvements in stability are significantly greater in the younger subjects by the fourth and fifth trials. Poorer adaptation in the elderly may be caused by a slowing of central adaptation, musculoskeletal factors which increase mechanical stiffness at the ankles, or a combination of these factors” (Nashner 1997a).
An example of results from response adaptation testing is shown in the Figure below.
§7: Limitations
Patients with significantly limited mobility will be unable to participate in computerized dynamic posturography.
§8: Contraindications
There are no firm contraindications to CDP.
§9: Pitfalls
CDP has limitations in what it can diagnose (Furman 1994). Bear in mind that CDP is “designed around” the visual, proprioceptive and vestibular systems, and analysis of its data relies on the simplifying assumption that these are the only factors involved. Therefore, CDP does a poor job of identifying (much less characterizing) balance problems due to disorders of other systems, such as cardiovascular (e.g., orthostatic hypotension) or neurological diseases (e.g., cerebellar disorders, extrapyramidal disorders).
§10: When is the test indicated
CDP is reasonable to consider in a patient complaining of disequilibrium. As with any test, its diagnostic utility tends to be greater when interpreted in the context of other data about a patient.
§11: Diseases that may be diagnosed by this test
In our experience, CDP shows utility in providing corroborative evidence for vestibular disease. We do not find it to be as helpful in supporting the presence of visual contributors or somatosensory contributors to disequilibrium.
Posturography test results can be greatly altered by “nonphysiologic” influences such as poor motivation, anxiety, and deliberate exaggeration of symptoms (Nashner 1997a). These types of influences usually lead to inconsistencies on the sensory organization test, and can sometimes be detected, or at least suspected (Nashner 1997b). Cevette and colleagues (Cevette et al. 1995) developed a linear discriminant algorithm to provide an alternative analysis for aphysiologic performance.
Bearing in mind those caveats, CDP abnormalities have been described in association with a broad range of diseases, or over the course of their natural history, or during their treatment. Some of these diagnoses are listed below. The reader will note that many of these diseases do not clearly fall under the categories of deficits for which CDP was designed to detect.
- Alcohol, both acute intoxication (Goebel et al. 1995; Ledin and Odkvist 1991) and effects of chronic alcoholism (Ahmad et al. 2002)
- Benign paroxysmal positional vertigo (Cohen and Kimball 2004)
- CANVAS (Mila de la Roca-Morales et al. 2018)
- Cerebellar lesions (Baloh et al. 1998)
- Cervicogenic vertigo (Alund et al. 1991; Alund et al. 1993)
- Chronic subjective dizziness (Morisod et al. 2018)
- Diabetes mellitus, both with neuropathy (Emam et al. 2009) and without neuropathy (Di Nardo et al. 1999)
- Head injury (Kisilevski et al. 2001)
- HIV infection (Dellepiane et al. 2005)
- Huntington’s disease (Fekete et al. 2012; Medina et al. 2013; Reyes et al. 2018; Salomonczyk et al. 2010)
- Intermittent claudication (Mockford et al. 2011; Vanicek et al. 2013)
- Ménière’s disease (Daneshi et al. 2009; Domenech-Vadillo et al. 2010; Hong et al. 2013)
- Migraine with migraine associated vertigo (Hong et al. 2013)
- Motion sickness (Shahal et al. 1999; Tal et al. 2010)
- Multiple sclerosis (Brichetto et al. 2015; Fritz et al. 2015; Grassi et al. 2017; Hebert and Manago 2017; Jackson et al. 1995; Nelson et al. 1995)
- Myotonic dystrophy (Scarano et al. 2022)
- Neuropathy (Di Nardo et al. 1999; Ledin et al. 1990b; Vrethem et al. 1991)
- Normal pressure hydrocephalus (Grassi et al. 2017)
- Parkinson’s disease (Bloem et al. 1998; Ebersbach et al. 2008; Ebersbach and Gunkel 2011; Halmi et al. 2019; Johnson et al. 2013; Lee et al. 2012; Nonnekes et al. 2013; Rossi-Izquierdo et al. 2014; Rossi-Izquierdo et al. 2009; Souza et al. 2019; Visser et al. 2010)
- Perilymphatic fistula (Black et al. 1990)
- Psoriatic arthritis (Amor-Dorado et al. 2017)
- Spinocerebellar ataxia (Herdman et al. 2011)
- Traumatic brain injury (Buster et al. 2016; Cesar et al. 2021)
- Tullio’s phenomenon (Ben-David et al. 1997)
- Unilateral vestibular weakness (Didilescu et al. 2015; Hong et al. 2013; Mbongo et al. 2005; Mishra et al. 2009; Picciotti et al. 2005; Schwesig et al. 2014)
- Vestibular schwannoma (Bergson and Sataloff 2005; Gouveris et al. 2007a; Gouveris et al. 2007b; Levine et al. 1993; Nam et al. 2018)
- Visual impairment (Shlamkovitch et al. 2017)
§12: Additional notes
There have been various attempts at modifying CDP, generally by making testing conditions more challenging. These have included:
- Head shaking (Honaker et al. 2009; Honaker et al. 2016; Mishra et al. 2009; Pang et al. 2011)
- Head tilt (Chandra and Shepard 1996; Jackson et al. 1991)
- Pedal anesthesia (Stal et al. 2003)
- Visual manipulation with virtual reality (Cesaroni et al. 2019; Ghiringhelli and Gananca 2011; Keshner et al. 2022; Rosiak et al. 2022; Song et al. 2014; Tossavainen et al. 2002; Tossavainen et al. 2003; Tossavainen et al. 2001; Tossavainen et al. 2006; Trueblood et al. 2018)
None of these modifications is currently in wide clinical use.
References
Ahmad S, Rohrbaugh JW, Anokhin AP, Sirevaag EJ, Goebel JA (2002) Effects of lifetime ethanol consumption on postural control: a computerized dynamic posturography study. J Vestib Res 12: 53-64.
Alfieri FM, Riberto M, Abril-Carreres A, Boldo-Alcaine M, Rusca-Castellet E, Garreta-Figuera R, Battistella LR (2012) Effectiveness of an exercise program on postural control in frail older adults. Clin Interv Aging 7: 593-8. doi: 10.2147/CIA.S36027
Alund M, Larsson SE, Ledin T, Odkvist L, Moller C (1991) Dynamic posturography in cervical vertigo. Acta Otolaryngol Suppl 481: 601-2. doi: 10.3109/00016489109131481
Alund M, Ledin T, Odkvist L, Larsson SE (1993) Dynamic posturography among patients with common neck disorders. A study of 15 cases with suspected cervical vertigo. J Vestib Res 3: 383-9.
Amor-Dorado JC, Barreira-Fernandez MP, Llorca J, Gonzalez-Gay MA (2017) Oculographic, Clinical Test of Sensory Integration and Balance and Computerized Dynamic Posturography Findings in Patients With Psoriatic Arthritis. Otol Neurotol 38: 448-453. doi: 10.1097/MAO.0000000000001296
Artuso A, Garozzo A, Contucci AM, Frenguelli A, Di Girolamo S (2004) Role of dynamic posturography (Equitest) in the identification of feigned balance disturbances. Acta Otorhinolaryngol Ital 24: 8-12.
Badke MB, Miedaner JA, Shea TA, Grove CR, Pyle GM (2005) Effects of vestibular and balance rehabilitation on sensory organization and dizziness handicap. Ann Otol Rhinol Laryngol 114: 48-54. doi: 10.1177/000348940511400109
Baloh RW, Jacobson KM, Beykirch K, Honrubia V (1998) Static and dynamic posturography in patients with vestibular and cerebellar lesions. Arch Neurol 55: 649-54. doi: 10.1001/archneur.55.5.649
Ben-David J, Teszler CB, Duchman H, Podoshin L (1997) Evaluation of Tullio Phenomenon by Computerized Dynamic Posturography. Int Tinnitus J 3: 105-112.
Bergson E, Sataloff RT (2005) Preoperative computerized dynamic posturography as a prognostic indicator of balance function in patients with acoustic neuroma. Ear Nose Throat J 84: 154-6.
Black FO, Lilly DJ, Peterka RJ, Shupert C, Hemenway WG, Pesznecker SC (1990) The dynamic posturographic pressure test for the presumptive diagnosis of perilymph fistulas. Neurol Clin 8: 361-74.
Bloem BR, Beckley DJ, van Hilten BJ, Roos RA (1998) Clinimetrics of postural instability in Parkinson’s disease. J Neurol 245: 669-73. doi: 10.1007/s004150050265
Brichetto G, Piccardo E, Pedulla L, Battaglia MA, Tacchino A (2015) Tailored balance exercises on people with multiple sclerosis: A pilot randomized, controlled study. Mult Scler 21: 1055-63. doi: 10.1177/1352458514557985
Buster TW, Chernyavskiy P, Harms NR, Kaste EG, Burnfield JM (2016) Computerized dynamic posturography detects balance deficits in individuals with a history of chronic severe traumatic brain injury. Brain Inj 30: 1249-55. doi: 10.1080/02699052.2016.1183822
Cesar GM, Buster TW, Burnfield JM (2021) Test-retest reliability and minimal detectable change of the computerized dynamic posturography PROPRIO for adults with chronic traumatic brain injury. Disabil Rehabil 43: 2038-2044. doi: 10.1080/09638288.2019.1688872
Cesaroni S, Silva AMD, Gananca MM, Caovilla HH (2019) Postural control at posturography with virtual reality in the intercritical period of vestibular migraine. Braz J Otorhinolaryngol. doi: 10.1016/j.bjorl.2019.06.015
Cevette MJ, Puetz B, Marion MS, Wertz ML, Muenter MD (1995) Aphysiologic performance on dynamic posturography. Otolaryngol Head Neck Surg 112: 676-88. doi: 10.1016/S0194-59989570175-3
Chandra NS, Shepard NT (1996) Clinical utility of lateral head tilt posturography. Am J Otol 17: 271-7.
Chieffe DJ, Zuniga SA, Marmor S, Adams ME (2023) Nationwide Utilization of Computerized Dynamic Posturography in an Era of Deimplementation. Otolaryngol Head Neck Surg. doi: 10.1002/ohn.333
Cohen HS, Kimball KT (2004) Treatment variations on the Epley maneuver for benign paroxysmal positional vertigo. Am J Otolaryngol 25: 33-7. doi: 10.1016/j.amjoto.2003.09.010
Daneshi A, Bozorgzadeh N, Asghari A, Jome HE, Mirhaj P, Nojoumi M (2009) Dynamic posturography for staging of patients with Meniere’s disease. J Laryngol Otol 123: 863-7. doi: 10.1017/S0022215109004423
Davies R (2004) Bedside neuro-otological examination and interpretation of commonly used investigations. J Neurol Neurosurg Psychiatry 75 Suppl 4: iv32-44. doi: 10.1136/jnnp.2004.054478
Dellepiane M, Medicina MC, Mora R, Salami A (2005) Static and dynamic posturography in patients with asymptomatic HIV-1 infection and AIDS. Acta Otorhinolaryngol Ital 25: 353-8.
Di Nardo W, Ghirlanda G, Cercone S, Pitocco D, Soponara C, Cosenza A, Paludetti G, Di Leo MA, Galli I (1999) The use of dynamic posturography to detect neurosensorial disorder in IDDM without clinical neuropathy. J Diabetes Complications 13: 79-85. doi: 10.1016/s1056-8727(99)00032-x
Didilescu AA, Didilescu A, Enache R, Musat G, Sarafoleanu C (2015) The importance of computerized dynamic posturography in vestibular rehabilitation of patients with unilateral peripheral vestibular deficiency. Romanian Journal of Rhinology 5: 173-178. doi: 10.1515/rjr-2015-0020
Dobie RA (1997) Does computerized dynamic posturography help us care for our patients? Am J Otol 18: 108-12.
Domenech-Vadillo E, Montes-Jovellar L, Rey-Martinez J, Perez-Fernandez N (2010) Normal and vestibular patterns in dynamic posturography in patients with Meniere’s disease. Acta Otorrinolaringol Esp 61: 34-40. doi: 10.1016/j.otorri.2009.08.003
Ebersbach G, Edler D, Kaufhold O, Wissel J (2008) Whole body vibration versus conventional physiotherapy to improve balance and gait in Parkinson’s disease. Arch Phys Med Rehabil 89: 399-403. doi: 10.1016/j.apmr.2007.09.031
Ebersbach G, Gunkel M (2011) Posturography reflects clinical imbalance in Parkinson’s disease. Mov Disord 26: 241-6. doi: 10.1002/mds.23189
Emam AA, Gad AM, Ahmed MM, Assal HS, Mousa SG (2009) Quantitative assessment of posture stability using computerised dynamic posturography in type 2 diabetic patients with neuropathy and its relation to glycaemic control. Singapore Med J 50: 614-8.
Evans MK, Krebs DE (1999) Posturography does not test vestibulospinal function. Otolaryngol Head Neck Surg 120: 164-73. doi: 10.1016/S0194-5998(99)70401-8
Fekete R, Davidson A, Ondo WG, Cohen HS (2012) Effect of tetrabenazine on computerized dynamic posturography in Huntington disease patients. Parkinsonism Relat Disord 18: 896-8. doi: 10.1016/j.parkreldis.2012.04.029
Fritz NE, Newsome SD, Eloyan A, Marasigan RE, Calabresi PA, Zackowski KM (2015) Longitudinal relationships among posturography and gait measures in multiple sclerosis. Neurology 84: 2048-56. doi: 10.1212/WNL.0000000000001580
Furman JM (1994) Posturography: uses and limitations. Baillieres Clin Neurol 3: 501-13.
Ghiringhelli R, Gananca CF (2011) Posturography with virtual reality stimulations in normal young adults with no balance complaints. J Soc Bras Fonoaudiol 23: 264-70.
Gianoli G, McWilliams S, Soileau J, Belafsky P (2000) Posturographic performance in patients with the potential for secondary gain. Otolaryngol Head Neck Surg 122: 11-8. doi: 10.1016/S0194-5998(00)70137-9
Gill C, Mallinson AI, Longridge NS (2000) Effects of dimenhydrinate on computerized dynamic posturography. J Otolaryngol 29: 337-9.
Goebel JA, Dunham DN, Rohrbaugh JW, Fischel D, Stewart PA (1995) Dose-related effects of alcohol on dynamic posturography and oculomotor measures. Acta Otolaryngol Suppl 520 Pt 1: 212-5. doi: 10.3109/00016489509125232
Goebel JA, Sataloff RT, Hanson JM, Nashner LM, Hirshout DS, Sokolow CC (1997) Posturographic evidence of nonorganic sway patterns in normal subjects, patients, and suspected malingerers. Otolaryngol Head Neck Surg 117: 293-302. doi: 10.1016/S0194-5998(97)70116-5
Gonzalez Eslait FJ, Escudero Trivino PA, Giraldo Vergara YV, Morales Garcia MA, Lucero Gutierrez VF (2023) Implementation outcomes of a sensory integration therapy program with computerized dynamic posturography in patients with balance and sensory dysfunction. J Otol 18: 26-32. doi: 10.1016/j.joto.2022.12.001
Gouveris H, Helling K, Victor A, Mann W (2007a) Comparison of electronystagmography results with dynamic posturography findings in patients with vestibular schwannoma. Acta Otolaryngol 127: 839-42. doi: 10.1080/00016480601075357
Gouveris H, Stripf T, Victor A, Mann W (2007b) Dynamic posturography findings predict balance status in vestibular schwannoma patients. Otol Neurotol 28: 372-5. doi: 10.1097/01.scs.0000246736.92095.34
Grassi L, Rossi S, Studer V, Vasco G, Motta C, Patane F, Castelli E, Rossi S, Cappa P (2017) Quantification of postural stability in minimally disabled multiple sclerosis patients by means of dynamic posturography: an observational study. J Neuroeng Rehabil 14: 4. doi: 10.1186/s12984-016-0216-8
Halmi Z, Dinya E, Mally J (2019) Destroyed non-dopaminergic pathways in the early stage of Parkinson’s disease assessed by posturography. Brain Res Bull 152: 45-51. doi: 10.1016/j.brainresbull.2019.07.001
Hebert JR, Manago MM (2017) Reliability and Validity of the Computerized Dynamic Posturography Sensory Organization Test in People with Multiple Sclerosis. Int J MS Care 19: 151-157. doi: 10.7224/1537-2073.2016-027
Herdman SJ, Hall CD, Eggers R, Sampson S, Goodier S, Filson B (2011) Misclassification of patients with spinocerebellar ataxia as having psychogenic postural instability based on computerized dynamic posturography. Front Neurol 2: 21. doi: 10.3389/fneur.2011.00021
Honaker JA, Converse CM, Shepard NT (2009) Modified head shake computerized dynamic posturography. Am J Audiol 18: 108-13. doi: 10.1044/1059-0889(2009/09-0012)
Honaker JA, Janky KL, Patterson JN, Shepard NT (2016) Modified head shake sensory organization test: Sensitivity and specificity. Gait Posture 49: 67-72. doi: 10.1016/j.gaitpost.2016.06.024
Hong HR, Shim DB, Kim TS, Shim BS, Ahn JH, Chung JW, Yoon TH, Park HJ (2013) Results of caloric and sensory organization testing of dynamic posturography in migrainous vertigo: comparison with Meniere’s disease and vestibular neuritis. Acta Otolaryngol 133: 1236-41. doi: 10.3109/00016489.2013.820343
Horak FB, Nashner LM (1986) Central programming of postural movements: adaptation to altered support-surface configurations. J Neurophysiol 55: 1369-81. doi: 10.1152/jn.1986.55.6.1369
Jackson RT, Epstein CM, Boyette JE (1991) Enhancement of posturography testing with head tilt and energy measurements. Am J Otol 12: 420-5.
Jackson RT, Epstein CM, De l’Aune WR (1995) Abnormalities in posturography and estimations of visual vertical and horizontal in multiple sclerosis. Am J Otol 16: 88-93.
Jacob RG, Furman JM, Durrant JD, Turner SM (1997) Surface dependence: a balance control strategy in panic disorder with agoraphobia. Psychosom Med 59: 323-30. doi: 10.1097/00006842-199705000-00016
Johnson L, James I, Rodrigues J, Stell R, Thickbroom G, Mastaglia F (2013) Clinical and posturographic correlates of falling in Parkinson’s disease. Mov Disord 28: 1250-6. doi: 10.1002/mds.25449
Keshner EA, Mallinson AI, Longridge NS, Sinno S, Petersen H, Perrin P (2022) Evolution of postural control assessment: From dynamic posturography to virtual reality. Front Neurol 13: 1054346. doi: 10.3389/fneur.2022.1054346
Kingma H, Gauchard GC, de Waele C, van Nechel C, Bisdorff A, Yelnik A, Magnusson M, Perrin PP (2011) Stocktaking on the development of posturography for clinical use. J Vestib Res 21: 117-25. doi: 10.3233/VES-2011-0397
Kisilevski V, Podoshin L, Ben-David J, Soustiel JF, Teszler CB, Hafner H, Chistyakov A (2001) Results of otovestibular tests in mild head injuries. Int Tinnitus J 7: 118-21.
Krempl GA, Dobie RA (1998) Evaluation of posturography in the detection of malingering subjects. Am J Otol 19: 619-27.
Larrosa F, Dura MJ, Cordon A, Hernandez A, Garcia-Ibanez L (2012) [Prevalence of aphysiologic performance on dynamic posturography in work-related patients]. Acta Otorrinolaringol Esp 63: 187-93. doi: 10.1016/j.otorri.2011.11.002
Larrosa F, Dura MJ, Menacho J, Gonzalez-Sabate L, Cordon A, Hernandez A, Garcia-Ibanez L (2013) Aphysiologic performance on dynamic posturography in work-related patients. Eur Arch Otorhinolaryngol 270: 93-7. doi: 10.1007/s00405-012-1930-x
Ledin T, Kronhed AC, Moller C, Moller M, Odkvist LM, Olsson B (1990a) Effects of balance training in elderly evaluated by clinical tests and dynamic posturography. J Vestib Res 1: 129-38.
Ledin T, Odkvist LM (1991) Effect of alcohol measured by dynamic posturography. Acta Otolaryngol Suppl 481: 576-81. doi: 10.3109/00016489109131475
Ledin T, Odkvist LM, Vrethem M, Moller C (1990b) Dynamic posturography in assessment of polyneuropathic disease. J Vestib Res 1: 123-8.
Lee JM, Koh SB, Chae SW, Seo WK, Kwon DY, Kim JH, Oh K, Baik JS, Park KW (2012) Postural instability and cognitive dysfunction in early Parkinson’s disease. Can J Neurol Sci 39: 473-82. doi: 10.1017/s0317167100013986
Levine SC, Muckle RP, Anderson JH (1993) Evaluation of patients with acoustic neuroma with dynamic posturography. Otolaryngol Head Neck Surg 109: 392-8. doi: 10.1177/019459989310900303
Li H, Waite GN, Moga MM, Lam P, Geib RW (2010) Balance improvements after a week-long tai chi workshop as determined by dynamic posturography – biomed 2010. Biomed Sci Instrum 46: 172-7.
Mallinson AI, Longridge NS (2005) A new set of criteria for evaluating malingering in work-related vestibular injury. Otol Neurotol 26: 686-90.
Mbongo F, Patko T, Vidal PP, Vibert N, Tran Ba Huy P, de Waele C (2005) Postural control in patients with unilateral vestibular lesions is more impaired in the roll than in the pitch plane: a static and dynamic posturography study. Audiol Neurootol 10: 291-302. doi: 10.1159/000086081
Medina LD, Pirogovsky E, Salomonczyk D, Goldstein J, Panzera R, Gluhm S, Simmons R, Corey-Bloom J, Gilbert PE (2013) Postural limits of stability in premanifest and manifest Huntington’s disease. J Huntingtons Dis 2: 177-84. doi: 10.3233/JHD-130048
Mila de la Roca-Morales AM, Andreo-Marroig JF, Santos-Perez S, Soto-Varela A (2018) Instability in Patients with CANVAS: Can Computerized Dynamic Posturography Help in Diagnosis? J Int Adv Otol 14: 130-134. doi: 10.5152/iao.2018.4335
Mishra A, Davis S, Speers R, Shepard NT (2009) Head shake computerized dynamic posturography in peripheral vestibular lesions. Am J Audiol 18: 53-9. doi: 10.1044/1059-0889(2009/06-0024)
Mockford KA, Mazari FA, Jordan AR, Vanicek N, Chetter IC, Coughlin PA (2011) Computerized dynamic posturography in the objective assessment of balance in patients with intermittent claudication. Ann Vasc Surg 25: 182-90. doi: 10.1016/j.avsg.2010.07.021
Morgan SS, Beck WG, Dobie RA (2002) Can posturography identify informed malingerers? Otol Neurotol 23: 214-7.
Morisod B, Mermod M, Maire R (2018) Posturographic pattern of patients with chronic subjective dizziness before and after vestibular rehabilitation. J Vestib Res 27: 305-311. doi: 10.3233/VES-170628
Nam GS, Jung CM, Kim JH, Son EJ (2018) Relationship of Vertigo and Postural Instability in Patients With Vestibular Schwannoma. Clin Exp Otorhinolaryngol 11: 102-108. doi: 10.21053/ceo.2017.01277
Nashner LM (1970) Sensory Feedback in Human Posture Control. Dissertation, Massachusetts Institute of Technology
Nashner LM (1971) A model describing vestibular detection of body sway motion. Acta Otolaryngol 72: 429-36. doi: 10.3109/00016487109122504
Nashner LM (1976) Adapting reflexes controlling the human posture. Exp Brain Res 26: 59-72. doi: 10.1007/BF00235249
Nashner LM (1997a) Computerized dynamic posturography. In: Jacobson GP, Newman CW, Kartush JM (eds) Handbook of Balance Function Testing. Delmar, New York, pp 280-307
Nashner LM (1997b) Computerized dynamic posturography: clinical applications. In: Jacobson GP, Newman CW, Kartush JM (eds) Handbook of Balance Function Testing. Delmar, New York, pp 308-334
Nashner LM (1997c) Practical biomechanics and physiology of balance. In: Jacobson GP, Newman CW, Kartush JM (eds) Handbook of Balance Function Testing. Delmar, New York, pp 261-279
Nashner LM, Black FO, Wall C, 3rd (1982) Adaptation to altered support and visual conditions during stance: patients with vestibular deficits. J Neurosci 2: 536-44. doi: 10.1523/JNEUROSCI.02-05-00536.1982
Nelson SR, Di Fabio RP, Anderson JH (1995) Vestibular and sensory interaction deficits assessed by dynamic platform posturography in patients with multiple sclerosis. Ann Otol Rhinol Laryngol 104: 62-8. doi: 10.1177/000348949510400110
Nonnekes J, de Kam D, Geurts AC, Weerdesteyn V, Bloem BR (2013) Unraveling the mechanisms underlying postural instability in Parkinson’s disease using dynamic posturography. Expert Rev Neurother 13: 1303-8. doi: 10.1586/14737175.2013.839231
O’Neill DE, Gill-Body KM, Krebs DE (1998) Posturography changes do not predict functional performance changes. Am J Otol 19: 797-803.
Pang MY, Lam FM, Wong GH, Au IH, Chow DL (2011) Balance performance in head-shake computerized dynamic posturography: aging effects and test-retest reliability. Phys Ther 91: 246-53. doi: 10.2522/ptj.20100221
Picciotti PM, Fiorita A, Di Nardo W, Quaranta N, Paludetti G, Maurizi M (2005) VEMPs and dynamic posturography after intratympanic gentamycin in Meniere’s disease. J Vestib Res 15: 161-8.
Reyes A, Salomonczyk D, Teo WP, Medina LD, Bartlett D, Pirogovsky-Turk E, Zaenker P, Bloom JC, Simmons RW, Ziman M, Gilbert PE, Cruickshank T (2018) Computerised Dynamic Posturography in Premanifest and Manifest individuals with Huntington’s Disease. Sci Rep 8: 14615. doi: 10.1038/s41598-018-32924-y
Rosiak O, Puzio A, Kaminska D, Zwolinski G, Jozefowicz-Korczynska M (2022) Virtual Reality-A Supplement to Posturography or a Novel Balance Assessment Tool? Sensors (Basel) 22. doi: 10.3390/s22207904
Rossi-Izquierdo M, Basta D, Rubio-Rodriguez JP, Santos-Perez S, Ernst A, Sesar-Ignacio A, Alberte-Woodward M, Guijarro-Del Amo M, Estany-Gestal A, San Roman-Rodriguez E, Faraldo-Garcia A, Zubizarreta-Gutierrez A, Soto-Varela A (2014) Is posturography able to identify fallers in patients with Parkinson’s disease? Gait Posture 40: 53-7. doi: 10.1016/j.gaitpost.2014.02.003
Rossi-Izquierdo M, Soto-Varela A, Santos-Perez S, Sesar-Ignacio A, Labella-Caballero T, Rossi-Izquierdo M, Soto-Varela A, Santos-Perez S, Sesar-Ignacio A, Labella-Caballero T (2009) Vestibular rehabilitation with computerised dynamic posturography in patients with Parkinson’s disease: improving balance impairment. Disabil Rehabil 31: 1907-16. doi: 10.1080/09638280902846384
Salomonczyk D, Panzera R, Pirogovosky E, Goldstein J, Corey-Bloom J, Simmons R, Gilbert PE (2010) Impaired postural stability as a marker of premanifest Huntington’s disease. Mov Disord 25: 2428-33. doi: 10.1002/mds.23309
Scarano S, Sansone VA, Ferrari Aggradi CR, Carraro E, Tesio L, Amadei M, Rota V, Zanolini A, Caronni A (2022) Balance impairment in myotonic dystrophy type 1: Dynamic posturography suggests the coexistence of a proprioceptive and vestibular deficit. Front Hum Neurosci 16: 925299. doi: 10.3389/fnhum.2022.925299
Schwesig R, Fischer D, Becker S, Lauenroth A (2014) Intraobserver reliability of posturography in patients with vestibular neuritis. Somatosens Mot Res 31: 28-34. doi: 10.3109/08990220.2013.822364
Shahal B, Nachum Z, Spitzer O, Ben-David J, Duchman H, Podoshin L, Shupak A (1999) Computerized dynamic posturography and seasickness susceptibility. Laryngoscope 109: 1996-2000. doi: 10.1097/00005537-199912000-00019
Shlamkovitch N, Eviatar E, Gavriel H (2017) Computerized dynamic posturography for evaluating dizziness in patients with scarred orbital content. Ear Nose Throat J 96: E10-E12. doi: 10.1177/014556131709600803
Song YB, Chun MH, Kim W, Lee SJ, Yi JH, Park DH (2014) The effect of virtual reality and tetra-ataxiometric posturography programs on stroke patients with impaired standing balance. Ann Rehabil Med 38: 160-6. doi: 10.5535/arm.2014.38.2.160
Souza CO, Voos MC, Barbosa AF, Chen J, Francato DCV, Milosevic M, Popovic M, Fonoff ET, Chien HF, Barbosa ER (2019) Relationship Between Posturography, Clinical Balance and Executive Function in Parkinson s Disease. J Mot Behav 51: 212-221. doi: 10.1080/00222895.2018.1458279
Stal F, Fransson PA, Magnusson M, Karlberg M (2003) Effects of hypothermic anesthesia of the feet on vibration-induced body sway and adaptation. J Vestib Res 13: 39-52.
Tal D, Bar R, Nachum Z, Gil A, Shupak A (2010) Postural dynamics and habituation to seasickness. Neurosci Lett 479: 134-7. doi: 10.1016/j.neulet.2010.05.044
Tossavainen T, Juhola M, Ilmari P, Aalto H, Toppila E (2002) Virtual reality stimuli for force platform posturography. Stud Health Technol Inform 90: 78-82.
Tossavainen T, Juhola M, Pyykko I, Aalto H, Toppila E (2003) Development of virtual reality stimuli for force platform posturography. Int J Med Inform 70: 277-83.
Tossavainen T, Juhola M, Pyykko I, Toppila E, Aalto H, Honkavaara P (2001) Towards virtual reality stimulation in force platform posturography. Stud Health Technol Inform 84: 854-7.
Tossavainen T, Toppila E, Pyykko I, Forsman PM, Juhola M, Starck J (2006) Virtual reality in posturography. IEEE Trans Inf Technol Biomed 10: 282-92.
Trueblood PR, Rivera M, Lopez C, Bentley C, Wubenhorst N (2018) Age-based normative data for a computerized dynamic posturography system that uses a virtual visual surround environment. Acta Otolaryngol 138: 597-602. doi: 10.1080/00016489.2018.1429653
Uimonen S, Laitakari K, Kiukaanniemi H, Sorri M (1995) Does posturography differentiate malingerers from vertiginous patients? J Vestib Res 5: 117-24.
Vanicek N, King SA, Gohil R, Chetter IC, Coughlin PA (2013) Computerized dynamic posturography for postural control assessment in patients with intermittent claudication. J Vis Exp: e51077. doi: 10.3791/51077
Visser JE, Oude Nijhuis LB, Janssen L, Bastiaanse CM, Borm GF, Duysens J, Bloem BR (2010) Dynamic posturography in Parkinson’s disease: diagnostic utility of the “first trial effect”. Neuroscience 168: 387-94. doi: 10.1016/j.neuroscience.2010.03.068
Vrethem M, Ledin T, Ernerudh J, Odkvist L, Holmgren H, Moller C (1991) Correlation between dynamic posturography, clinical investigation, and neurography in patients with polyneuropathy. ORL J Otorhinolaryngol Relat Spec 53: 294-8. doi: 10.1159/000276232
![]()